Abstract
Background:
Given that unilateral branch retinal vein occlusion (BRVO) and glaucoma share common systemic vascular risk factors, the fellow eyes of patients with BRVO may be at increased risk of glaucoma.
Objectives:
To analyze the radial peripapillary capillary density (RPCD) in eyes with unilateral BRVO and their unaffected fellow eyes using optical coherence tomography angiography (OCTA).
Design:
Cross-sectional, prospective study.
Methods:
The study included 120 eyes of 80 patients: 40 affected eyes of BRVO, 40 fellow eyes of BRVO, and 40 control eyes. The RPCD, retinal nerve fiber layer thickness (RNFLT) were analyzed using OCTA.
Results:
RPCDs in the whole image, peripapillary region, all the hemispheres, and quadrants were statistically lower in the affected eyes than in both the fellow and control eyes (p < 0.05 for all). RPCD values in the whole image and the peripapillary region were significantly lower in the fellow eyes than in the control eyes (p = 0.013, and p = 0.021, respectively). RNFLTs in the peripapillary region, inferior hemisphere and inferior quadrant were significantly lower in the affected eyes than in the control eyes (p < 0.05 for all). No significant differences were detected between the fellow eyes and the control eyes in term of RNFLT values in any regions (p > 0.05 for all).
Conclusion:
Lower RPCD values despite similar RNFLT values were observed in the fellow eyes of patients with unilateral BRVO compared with healthy controls. These results may indicate the shared vascular mechanisms and risk factors that account for the development of BRVO and glaucoma.
Keywords
Introduction
Retinal vein occlusion (RVO) is the second-most common sight-threatening retinal vascular disease after diabetic retinopathy. 1 Venous occlusion may occur either in the central retinal vein (central retinal vein occlusion; CRVO) or in a branch of retinal vein (branch retinal vein occlusion; BRVO). 2 It is already known that RVO is commonly associated with underlying systemic diseases such as arteriosclerosis, uncontrolled hypertension, diabetes mellitus, hypercholesterolemia, and older age. 3 The relationship between RVO and glaucoma has long been recognized. 4 It has been suggested that rather than a cause and effect relationship, the relationship between the two is a reflection of underlying systemic vascular diseases such as atherosclerosis, hypertension, and diabetes mellitus. 5 Stewart and Clearkin emphasized that vascular dysregulation and autoregulatory dysfunction as extended consequences of insulin resistance may play a pivotal role in the pathogenesis of both RVO and glaucoma. 6 The fact that RVO and glaucoma have common systemic risk factors has focused the attention of researchers on the unaffected fellow eyes of patients with RVO, with the consideration that there may be early glaucomatous changes in the fellow eye. Kim et al. 7 found a decreased retinal nerve fiber layer (RNFL) to be an early sign of glaucoma in the fellow eyes of patients with RVO compared to control eyes. Optical coherence tomography angiography (OCTA) is a new non-invasive imaging tool that can be used to visualize the radial peripapillary capillary (RPC) layer which is the most superficial capillary layer nourishing the RNFL surrounding the optic nerve head (ONH). 8 Lower radial peripapillary capillary density (RPCD) in patients with glaucoma and suspected glaucoma compared to healthy controls have been reported in previous studies.9,10 A recent study by Shin et al. 11 revealed that the peripapillary microvascular density in the fellow eyes of unilateral RVO was decreased compared with control eyes. In addition, in a recent study, Ozcaliskan and Ozcan 12 also demonstrated microvascular changes in both macular and peripapillary regions of the unaffected fellow eyes of patients with unilateral RVO. In those studies, patients were not grouped as BRVO or CRVO. The relationship of glaucoma with CRVO is known to be stronger than its relationship with BRVO. 13 Hence decreased RPCD in the fellow eye in aforementioned studies may be due to the inclusion of both CRVO and BRVO patients.
The aim of this study was to compare the microcirculation in the peripapillary area of eyes with unilateral BRVO and their unaffected fellow eyes, with the eyes of age- and gender-matched control subjects.
Materials and methods
This prospective, cross-sectional study was conducted in the retina department of a tertiary care hospital from June 2019 to May 2020. The study received approval from the Ethics Committee of the Ankara Diskapi Training and Research Hospital (report number: 2019-63/09) and conformed to the tenets of the Declaration of Helsinki. Written informed consent was obtained from each patient.
The study consisted of 120 eyes of 80 patients in three groups: Group 1 included 40 eyes with BRVO (affected eye), Group 2 included 40 unaffected fellow eyes, and Group 3 included the 40 right eyes of 40 age- and gender-matched healthy control subjects. Healthy controls were selected from the patients without any systemic disease and who came to the general ophthalmology outpatient clinic for refraction examination.
BRVO was diagnosed by the same retina specialists (CUA, YSG, MC) based on the characteristic clinical features of flame-shape, dot and blot hemorrhage, soft and hard exudates, retinal oedema, dilated-tortuous vein, vascular sheath and venous collaterals at the site of vascular occlusion according to dilated fundus examination. Fundus flourescein angiography (FFA) was performed to evaluate the retinal vasculature including the extent of nonperfusion area, macular ischemia, macular oedema, and leakage either at the first visit or within the 2 months follow-up depending on the intensity of retinal hemorrhages. Spectral domain optical coherence tomography (SD-OCT) was performed to determine whether macular oedema was present or not. Only patients with unilateral, non-ischemic, and major BRVO who had a minimum follow-up period of at least 6 months and/or who had no macular oedema and anti-vascular endothelial growth factor treatment in the last 6 months were included in this study. Non-ischemic BRVO was defined as BRVO with non-perfused areas smaller than 5-disc diameters in FFA, whereas major BRVO was defined as the occlusion of a retinal vein that drains one of the retinal quadrants. 14
A detailed history, which included pre-existing medical, ocular, systemic, and family history was obtained as self-reported by the patient.
The study exclusion criteria were as follows: opaque media (e.g. cataract and band keratopathy) that precluded the fundus examination or OCTA measurements, the presence of vitreoretinal disorders other than BRVO in the affected eye and any vitreoretinal disorders (e.g. vascular abnormalities) in the fellow eye, a history of any vitreoretinal surgery and/or argon laser photocoagulation in affected or fellow eyes, a history of phacoemulsification surgery within 12 months prior to study enrollment, diagnosed with glaucoma and/or a glaucomatous ONH (cup-to-disc (C/D) ratio > 0.6, vertical cup asymmetry > 0.2, and neuroretinal rim loss or notching), a history of anti-glaucomatous drug usage, age < 40 or >80 years, a refraction error above −2/+2 diopters, axial length (AL) of <22 mm or >24 mm, a drug and/or alcohol addiction or any known systemic disorders other than hypertension such as diabetes mellitus, cerebrovascular disease, or migraine.
All participants underwent a series of ocular examinations including best-corrected visual acuity (BCVA) using the Snellen chart, Goldmann applanation tonometry, central corneal thickness (CCT) using Pachymeter, gonioscopy with Goldmann-three mirror in patients with suspected narrow anterior chamber angle, AL measurements, slit-lamp biomicroscopy, dilated fundus examination using a 78 D lens, FFA, and SD-OCT.
Diastolic and systolic blood pressure values of all patients taken by a nurse just before OCTA measurement were recorded.
OCTA imaging of the optic disc was performed using an AngioVue OCTA device (RTVue XR Avanti, version 2017.1.0.151; Optovue, Inc., Fremont, CA, USA) after pupillary dilatation in a dark room. All the OCTA image measurements were taken by the same clinician (CUA), paying attention to the signal strength index (SSI) ⩾ 7. To quality of the scans was assessed before analysis by one experienced independent grader masked to the participants’ clinical information. Poor quality images were excluded defined as those with signal strength < 7, the presence of one or more blink artifacts, poor fixation resulting in motion or doubling artifacts, and segmentation errors.
A 4.5 × 4.5 mm (400 × 400 pixels) rectangle scan centered on the ONH area was obtained. The device automatically attaches two ONH-centered concentric circles with diameters of 2 mm (inner) and 4 mm (outer) (ring width: 1 mm), shown in Figure 1. The RPCD was evaluated between these rings in the peripapillary region, in four quadrants (superior, inferior, nasal, and temporal) and in two equal hemispheres (superior and inferior) using the OCTA density assessment tool, shown in Figure 1. The RPCDs inside the disc and in the whole image were also evaluated. Large retinal vessel–related flow signals were removed by the recent Angio Disc Vue OCT software update (Phase 7). The segmentation was between the inner limiting membrane and the RNFL. The peripapillary RNFL thicknesses (RNFLTs) were also evaluated in four quadrants (superior, inferior, nasal, and temporal) and in two equal hemispheres (superior and inferior), as for the corresponding areas of the RPCD, via the ONH analysis of the Angio Disc Vue. The C/D area ratio, C/D vertical ratio, C/D horizontal ratio, rim area (mm2), disc area (mm2), and cup volume (mm3) were also measured via a scanning laser ophthalmoscopy (SLO) analysis of the ONH (Figure 1).
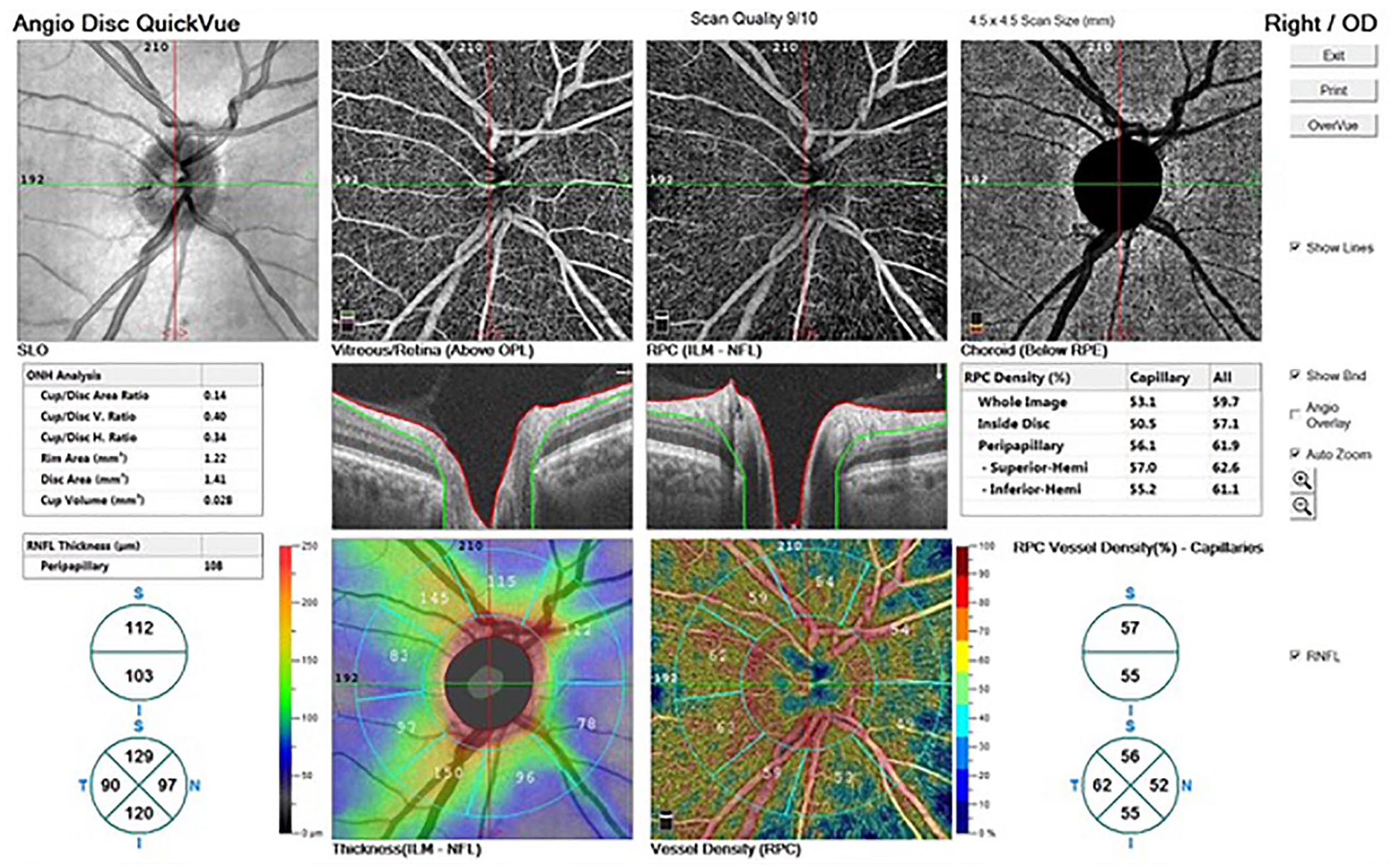
The radial peripapillary capillary vessel density (RPVD) and retinal nerve fiber layer (RNFL) thickness (μm) were automatically measured in a 1-mm width ring between 2- and 4-mm concentric circles with the optic nerve head (ONH) centered in four equal quadrants and two equal hemispheres. A 4.5 mm × 4.5 mm rectangle scan was obtained, and the segmentation was between the inner limiting membrane and the RNFL. The cup/disc area ratio, the cup/disc vertical ratio, the cup/disc horizontal ratio, rim area (mm2), disc area (mm2), and cup volume (mm3) were automatically calculated via the Angio Disc Quick Vue analysis of the ONH.
Statistical analysis was performed with SPSS (Statistical Package for Social Sciences) version 22.0 for Windows software (SPSS Inc., Chicago, IL, USA). A prior power analysis using the PASS 11 calculation (power and sample size, version 11) showed that at about 40 eyes for each group should be enrolled to reach a power equal to at least 80% in this study. Conformity of the data to normal distribution was analyzed with the Kolmogorov–Smirnov test. Conformity of the data to homogeneity of distribution was analyzed with Levene test of homogeneity of variance. The categorical variables between the groups were analyzed using the chi-square test. Descriptive statistics were stated as mean ± standard deviation (SD) values. One-way analysis of variance was used for the analysis of the differences between the three groups for data with homogeneous distribution, and Tukey’s test was used as a post hoc test for pairwise comparisons. The Welch analysis of variance was used for data without homogeneous distribution and Games–Howell was used as the post hoc test for pairwise comparisons. A value of p < 0.05 was considered statistically significant.
Results
The demographic and clinical characteristics of all the study participants are shown in Table 1. There were no statistically significant differences between the groups in terms of age, gender, intraocular pressure (IOP), CCT, and AL measurements (p > 0.05 for all). BCVA was significantly decreased in the affected eyes compared to the fellow eyes and the control eyes (p < 0.001, and p < 0.001, respectively). The SSI of OCTA images was 8.24 ± 0.07 (range: 7–9), 8.11 ± 0.08 (range: 7–9), and 8.31 ± 0.054 (range: 7–9) for Groups 1, 2, and 3, respectively. No statistically significant differences were detected between the groups in terms of SSI (p = 0.541).
The demographical and clinical characteristics of all patients.

BCVA, best corrected visual acuity; BP, blood pressure; IOP, intraocular pressure; µm, micrometer.
Analysis of variance (ANOVA) (comparison among three groups).
Chi-square test.
The RPCD measurements of the groups are summarized in Table 2. A statistically significant difference was determined in respect of all the RPCD values except inside disc (p < 0.05 for all). When these parameters were analyzed between the groups, the pairwise comparisons revealed that all the RPCD values except inside disc were statistically lower in the affected eye than in the fellow eyes and the control eyes (p < 0.05 for all). The RPCD values in the whole image and peripapillary region were determined to be significantly lower in the fellow eyes than in the control eyes (p = 0.013, and p = 0.021, respectively).
Radial peripapillary capillary vessel density measurements of all patients.

Hemi, hemisphere; Quad, quadrant; RPCD, radial peripapillary capillary density; SD, standard deviation.
G1&G2: difference between group 1 and group 2 (pairwise comparison). G1&G3: difference between group 1 and group 3 (pairwise comparison). G2&G3: difference between group 2 and group 3 (pairwise comparison).
Data in bold faces are significant values.
Analysis of variance (ANOVA) (comparison among three groups).
Tukey’s post-test (pairwise comparison).
The RNFLT measurements of all the patients are shown in Table 3. There were statistically significant differences among the three groups in respect of the peripapillary, inferior hemisphere, and inferior quadrant RNFLT values (p = 0.012, p = 0.004, and p = 0.000, respectively). When these parameters were analyzed between the groups, the pairwise comparisons revealed that the RNFLT values in peripapillary, inferior hemisphere, and inferior quadrant were statistically significantly lower in the affected eyes than in the control eyes (p = 0.009, p = 0.005, and p = 0.001, respectively). There were no significant differences between the fellow eyes and the control eyes in terms of the RNFLTs in any region (p > 0.05 for all).
Retinal nerve fiber layer thickness measurements of all patients.

Hem, hemisphere; µm, micrometer; Quad, quadrant; RNFLT; retinal nerve fiber thickness; SD, standard deviation.
G1&G2: Difference between group 1 and group 2 (pairwise comparison). G1&G3: Difference between group 1 and group 3 (pairwise comparison). G2&G3: Difference between group 2 and group 3 (pairwise comparison).
Data in bold faces are significant values.
Welch analysis (comparison among three groups).
Games–Howell Test (pairwise comparison).
The SLO parameters of the ONH are shown in Table 4. There were no significant differences among the three groups with regard to C/D area, C/D vertical ratio, C/D horizontal ratio, rim area, disc area, and cup volume (p > 0.05 for all).
Scanning laser ophthalmoscopy parameters of optic nerve head.

C/D, Cup/Disc; SD, standard deviation.
Analysis of variance (ANOVA) (comparison among three groups).
Discussion
OCTA is a new imaging tool that can quantitatively evaluate microvascular changes in the retina. 15 Wider foveal avascular zone, lower macular microvascular density, decreased peripapillary microvasculature, and lower perfusion of choriocapillaris in the fellow eyes of patients with unilateral RVO compared with healthy eyes have been detected in previous OCTA studies.11,12,16,17 All these changes are based on the fact that underlying systemic vascular diseases may affect the retinal microcirculation of both eyes. The aim of this study was to evaluate RPCD in the unaffected fellow eyes of patients with BRVO specifically, as well as the affected eyes and compare them with age- and gender-matched healthy individuals.
The result of this study demonstrated that the affected eyes had significantly lower RPCD than the fellow eyes and the control eyes in the whole image, peripapillary region, all hemispheres and all quadrants. The RNFLT values in the peripapillary region, inferior hemisphere and inferior quadrant of affected eyes were also significantly lower than those of the control eyes. Sectoral RNFL thinning and reduction in RPCD in eyes with BRVO have previously been emphasized.18,19 Of course, it was not surprising that we also detected decreased RNFL and RPCD in eyes with BRVO in our study. In addition, this study revealed significantly lower RPCD values in the whole image and the peripapillary region of the fellow eyes compared with that of the control eyes. However, there were no significant differences between the fellow eyes and the control eyes in terms of RNFLTs in any region. The strong association between the RPCD reduction and thinning of the RNFL has been previously identified in many studies.20,21 This neurovascular relationship is crucial for the development of glaucoma. 22 A recent systematic review including 43 studies reported that of the OCTA parameters, the whole image RPCD had the highest glaucoma diagnostic capacity. 23 Another recent study demonstrated that peripapillary OCTA vessel density especially in the whole image region has a similar diagnostic capacity to RNFLT in discriminating healthy subjects from those with suspected glaucoma and glaucoma patients. 24 In this study, RPCD was seen to be reduced in the whole image and the peripapillary regions of the fellow eyes of BRVO cases despite mostly similar RNFLT values compared with the age- and gender-matched healthy control subjects. It may be speculated that vascular regression may occur earlier and precede the RNFL thinning and may increase the suspectibility to glaucoma in the fellow eyes. However, a longitudinal study may better document the sequence and compare changes in the vasculature versus structure.
In the recent OCTA study, decreased peripapillary microvascular parameters, GC-IPL thickness, and RNFLT were detected in the fellow eyes of the patients with RVO compared with those of control subjects and peripapillary microvascular parameters were positively correlated with GC-IPL thickness and RNFLT values. However, the researchers were unable to differentiate whether the decreased peripapillary microvascular parameters were due to vascular dysfunction or secondary to ganglion cell damage. 11 The diminished RPCD that was demonstrated probably represents the vascular regression accompanying the RNFL thinning and does not support the primary role of vascular insufficiency. Similarly, Ozcaliskan and Ozcan 12 also found that decreased macular vessel density in the outer ring, the peripapillary flux index, GC-IPL, and RNFL thicknesses in the fellow eyes of patients with RVO. In aforementioned studies, the patients had unilateral RVO and were not separated as BRVO or CRVO while this study included the patients with BRVO only. Although CRVO and BRVO are similar diseases, they tend to have different systemic comorbidities with different pathophysiology. It has been documented that hypertension and atherosclerosis are more common etiological diseases in patients with BRVO than in patients with CRVO.25,26
The relationship between BRVO and increased IOP seen in patients with POAG is already known. 13 However, BRVO has been also reported in patients with NTG. 27 It has been proposed that vascular dysregulation is the major contributing factor that is responsible for glaucomatous damage seen in NTG. 28 This emphasizes that causes other than IOP can lead to BRVO in glaucoma patients and supports the importance of systemic vascular risk factors in the etiology of BRVO.
A strength of this study was the strict participation criteria as only patients with concurrent BRVO and hypertension were included. Another strength of this study was the use of the same device for the analysis of the RNFLT and the RPCD. This provided the opportunity for an accurate correspondence of each sector. However, there are some limitations in this study, primarily the cross-sectional design. A prospective longitudinal study may document a better cause and effect relationship. Another possible limitation of this study was the relatively limited number of patients and that the medical histories collected from all the subjects were self-reported.
In conclusion, the results of this study demonstrated that RPCD was more prominently decreased than RNFLT in the fellow eyes of patients with unilateral BRVO compared to healthy eyes. Such results emphasizes the shared vascular mechanisms and risk factors that account for the development of BRVO and glaucoma. Although significant reduction in RPCD and RNFL thinning and glaucoma are not equivalent entities, not only RNFL thinning but also RPCD reduction especially in particular topographic pattern are reported to be associated with glaucomatous damage in previous studies.9,10 Therefore, it should be kept in mind that the fellow eyes should therefore be more cautiously monitored in terms of glaucoma. Nevertheless, there is a need for further prospective-longitudinal studies with large case series and long-term follow-up be able to monitor the glaucoma course in the fellow eyes of patients with unilateral BRVO.
Footnotes
Acknowledgements
This study was edited by Mrs. Caroline Jane Walker (a native English speaker) for English language.
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
