Abstract
Ranibizumab has proven its efficacy in various retinal diseases and particularly in neovascular age-related macular degeneration (nAMD). The number of injections and the frequent follow-up visits has been burdensome to patients particularly during the COVID era. Ranibizumab port delivery system (RPDS) seems to be a boon in prolonging the action of the drug without the need for frequent injections and follow-up visits. This review article highlights the dosage, adverse effects, and visual outcome associated with various trials of RPDS. For this article, we conducted a PubMed search and review of literature on nAMD, the incidence of AMD, anti-vascular endothelial growth factor (anti-VEGF) agents, RPDS, phase-1 trial of RPDS, phase-2 (LADDER trial) of RPDS, phase-3 (ARCHWAY trial) of RPDS, PORTAL trial of RPDS, results of phase-1 trial of RPDS, results of phase-2 (LADDER) trial of RPDS, and results of phase-3 (ARCHWAY) trial of RPDS.
Keywords
Introduction
Age-related macular degeneration (AMD) is one of the leading causes of visual impairment and blindness among the elderly. AMD affects about 10–13% of people aged above 65 years of age in North America, Europe, Australia, and Asia.1–3 Meta-analysis of incidence and prevalence in European population shows that about 67 million people are blind with AMD in the aging population in European Union, and this is expected to increase by 15% by 2050. 4 According to United Nations about 20–25 million people are affected worldwide with AMD, and this is expected to increase in the next 30 years with an increasing aging population. According to the World Health Organization, about 8 million have severe visual impairment due to AMD, and this excludes countries from which data are not available or scarce. 5 A comparative study of prevalence and risk factors for AMD in Singapore and India showed that prevalence was similar in both groups, but Singapore-residing Indians had more vascular risk factors and early onset of AMD. 6 Global Burden of Disease study 2010 states that glaucoma and AMD account for nearly 10.7% of vision loss years lived with a disability which is expected to increase further because of aging population. 7 Studies have shown that vision impairment and blindness associated with AMD has profoundly impacted quality of life affecting both mental and emotional health.8–10
Anti-vascular endothelial growth factor agents have almost become a norm for treating patients with nAMD. The introduction of anti-VEGF agents revolutionized the treatment of nAMD in terms of its efficacy and fewer adverse effects. A Danish study and an Israeli study showed an overall reduction in legal blindness following the use of anti-VEGF agents in nAMD.11,12
Ranibizumab is an anti-VEGF injection which has shown promising results in various retinal diseases particularly neovascular AMD (nAMD). MARINA and ANCHOR trials have shown that monthly intravitreal injections of ranibizumab improve visual acuity and prevent further vision loss in patients with nAMD. There have been disappointing results with other modalities of treatment like photodynamic therapy and laser photocoagulation prior to the introduction of anti-VEGF agents for various retinal diseases.13–16
Various clinical trials have shown tremendous improvement with anti-VEGF use. But the challenge is to translate the same amount of vision gain that we see in clinical trials to the real world. This requires compliance from patients in terms of continuous monitoring and follow-up which is quite taxing in the current scenario. This has adversely affected both visual and anatomical outcomes as these patients require prolonged VEGF suppression. Pre-filled syringes containing a dosage of 0.5 and 0.3 mg, and monthly, pro-re-nata (PRN) or treat and extend regimen did not bring down the number of injections, resulting in non-compliance of patients.17–19 Introduction of ranibizumab port delivery system (RPDS) could reduce the need for above.
Method of literature search
The method of literature search was PubMed search. The search was pertaining to nAMD, the incidence of AMD, anti-VEGF agents, RPDS, phase-1 trial of RPDS, phase-2 (LADDER trial) of RPDS, phase-3 (ARCHWAY trial) of RPDS, PORTAL trial of RPDS, results of phase-1 trial of RPDS, results of phase-2 (LADDER) trial of RPDS, and results of phase-3 (ARCHWAY) trial of RPDS.
Ranibizumab port delivery system
Insertion procedure
RPDS consists of a refillable sustained release reservoir of ranibizumab which delivers the drug into the vitreous cavity for treatment of nAMD. It is of the size of a grain of rice.20–26
Implant insertion is performed under local anesthesia in operation theater under complete aseptic precautions. In the original procedure, a conjunctival peritomy was performed in the supero-temporal quadrant. Then a full-thickness stab incision was made using a 3.2 mm slit knife, 4 mm posterior to limbus through the sclera and pars plana, and drug is inserted through this. The above procedure had complication of vitreous hemorrhage (VH) post-operatively. To avert this, the technique was optimized, which included layer by layer incision through the sclera with laser ablation (532 nm) and additional diathermy of exposed pars plana. The implant was then inserted after making an incision through ablated pars plana using a 3.2 mm slit knife. The final step involved careful suturing of conjunctiva and tenon’s capsule in both original and optimized procedures to provide good visualization of implant flange.
Refill procedure
Patients were followed up monthly and as per defined criteria, refill was performed as an office-based procedure under topical anesthesia. Port delivery system (PDS) refill needle was inserted through the conjunctiva into implant septum in a perpendicular fashion. Refill consists of injecting 0.1 mL of specified ranibizumab formulation using a dual-lumen refill needle into the implant. This facilitates withdrawal of remaining ranibizumab in implant and getting replaced by a new drug. Total fluid exchange of old drug with new drug is achieved using the above method.
Thus, the implant has four components – an extra scleral flange to secure PDS in the sclera, a self-sealing septum for replenishing drug, a reservoir for storing drug and a porous metal releaser for passively allowing the drug to get released into the vitreous cavity via a concentration gradient. This passive diffusion allows controlled and continuous delivery of the drug into the vitreous cavity slowly over a period of time. It is a permanent and long-acting delivery system. Current PDS has a customized formulation of ranibizumab which is different from the dosage of the drug that is used for intravitreal injections.20–22 Pre-filled syringes of ranibizumab in dosage of 0.5 mg and 0.3 mg were approved by Food and Drug Administration (FDA) earlier.
Phase-1 trial
This was conducted by ForSight vision 4.21,23,24,27,28 This was an open-label prospective study. This included 20 treatment-naïve patients with nAMD who had a 250 µg ranibizumab pre-filled device on Day 0. Patients were followed up every month for a year. This was followed by observation which extended the study to 36 months. The device was refilled with 250 µg into the implant for sustained release and 250 µg bolus totaling to 500 µg based on the predetermined visual acuity and OCT retreatment criteria. Refill was however stopped at 12 months.
The primary outcome was to look for safety (incidence and severity of adverse events (AEs)), problems related to implantation, refill, and explantation, and secondary outcome was to look for improvement in best corrected visual acuity (BCVA) and anatomical outcome.
For the primary outcome, 77 study-related AEs were reported.
Conjunctival hyperemia in 95% of patients, VH in 25% and hyphema in 20%.
There were four serious AEs associated with device placement:
One endophthalmitis – it was successfully treated following which the patient had a refill and went on to gain three letters of BCVA from baseline at 12 months.
One traumatic cataract – it was treated with phacoemulsification and IOL implantation after which the patient gained 29 letters from baseline at 12 months.
Two cases of persistent VH – out of two cases of VH, one gained four letters from baseline at 12 months and the other hand movements at 12 months.
Mean refill for the entire 20 patients was 4.8, but with the exclusion of two patients with persistent VH, it was 4.2 (95% confidence interval). The median time to first refill was 3 (2–3) months.
Table 1 gives details of mild and severe adverse events in phase-1 trial
Mild and severe adverse events in Phase-1 trial.

As far as the secondary outcome was concerned,
Approximate improvement in BCVA from baseline was 10 Early Treatment Diabetic Retinopathy Study (ETDRS) letters at 12 months for 20 patients (95% confidence interval).
Omitting two patients with persistent VH, improvement in BCVA was 15 letters in 12 months.
Ten (50%) out of the 20 patients improved by at least three lines.
Two (10%) patients lost at least three lines.
A parallel decrease in mean central retinal thickness from baseline was also observed at 12 months.
Mean serum ranibizumab concentration at 8 weeks for 10 patients was maintained at a plateau.
Explantation of implant
At 12 months, six out of explanted devices were functionally similar to unimplanted devices when refilled with ranibizumab. Fourteen patients whose device was not explanted during the 12 months period transitioned to intravitreal injections during the observation period.21,23,27,28
Thus, the phase-1 trial proved its safety and efficacy.
Phase 2 trial (LADDER)
Genetech conducted a randomized, multicenter, active treatment-controlled phase-2 trial (GX28228) to assess the safety and efficacy of RPDS for nAMD patients.20,21,24,25,29
Inclusion criteria
Patients who had earlier responded to two or more anti-VEGF injections and had shown improvement in anatomical and visual outcomes.
Patients who had shown improvement earlier but had new choroidal neovascular lesions.
BCVA between 20/20 and 20/200.
Exclusion criteria
Patients who had received anti-VEGF injections other than ranibizumab within 1 month prior to randomization.
Post-vitrectomy surgery in the study eye.
Post-glaucoma filtration surgery in the study eye.
Post-tube shunt implantation in the study eye.
Micro-invasive glaucoma surgery in the study eye.
Two hundred and twenty (220) treatment responsive patients with newly diagnosed nAMD (within 9 months) were randomized to 3:3:3:2 of PDS filled with 10, 40, and 100 mg/mL dosage or 0.5 mg of monthly intravitreal ranibizumab injection. This was the primary analysis. The total duration of the study was 38 months with a mean time of 22.1 and 21.7 months for the PDS arm and 0.5 mg intravitreal ranibizumab group, respectively. About 91.1% in the PDS group and 87.8% in the intravitreal ranibizumab group completed the study. Around 96.6% of patients shifted to the PORTAL study.
The primary endpoint was time for implant refill based on the criteria given below.
Time for the first implant refill was defined as the time from insertion of the implant to first refill based on protocol-defined refill criteria.
Time for the first actual refill was defined as the time from insertion of the implant to first refill exchange procedure.
Time from the first refill to second refill was defined as the time from first actual refill to second based on protocol-defined criteria in patients who had at least one actual refill.
Implant refill criteria: Patients were assessed on a monthly basis after implant procedure, and the refill was performed based on the following NMD disease activity:
Increase in central foveal thickness (CFT) of 75 µm or more using spectral-domain optical coherence tomography (OCT) at present visit compared with average CFT over last two available measurements.
Increase in CFT of 100 µm or more from lowest CFT measurement in the study.
Decrease in BCVA of five letters or more at present visit compared to average BCVA over the last two available measurements.
Decrease of 10 letters or more from best recorded BCVA on the study.
Presence of new macular haemorrhage.
Median time for first implant refill in 10, 40, and 100 mg/mL arms was 8.7, 13, and 15.8 months, respectively. There was statistically significant difference in median time for refill between 10 and 100 mg/mL PDS arm, and 10 and 40 mg/mL PDS arm but not between 40 and 100 mg/mL PDS arm. Patients who did not require implant refill for 6 months or more were 63.5%, 71.3%, and 79.8%, respectively, in 10, 40, and 100 mg/mL PDS arm. Median time to first implant refill was almost similar in 100 mg/mL PDS arm and 40 mg/mL PDS arm (15 and 13 months, respectively).
Median time to first refill increased by approximately 5% in PDS 100 mg/mL group in comparison to the primary analysis. Percentage of patients who received an actual refill during LADDER trial included 65.5%, 54.8%, and 52.5% in 10, 40, and 100 mg/mL groups, respectively. Median time to first actual refill was 6.4, 5.6, and 8.8 months in 10, 40, and 100 mg/mL groups, respectively. Median time from first to second refill in patients who have received at least one actual refill was 3.3, 5.9, and 8.8 months, respectively in 10, 40, and 100 mg/mL groups.
Rescue criteria using intravitreal ranibizumab 0.5 mg
All PDS patients 1–2 months after VH were given intravitreal ranibizumab 0.5 mg if
There was BCVA loss due to VH.
Macula could not be assessed due to VH.
OCT could not be performed due to VH.
Progressive worsening of BCVA and CFT or both over two successive visits because of nAMD disease activity that did not satisfy criteria for refill procedure.
Lack of clinical efficacy – loss of BCVA of 15 or more letters from best-recorded BCVA after two successive implant refills with a separation of 1 month because of nAMD disease activity unless there was an improvement of five letters or more of BCVA that would warrant an implant refill. Increase of 150 µm or more of CFT from lowest recorded CFT after two successive implant refills with a separation of 1 month unless there was a reduction of 75 µm or more of CFT from the last refill that would warrant an implant refill.
Rescue therapy of 0.5 mg intravitreal injection was given if clinical efficacy criteria were not met, or there was no response to PDS refill. Around 22.4%, 4.8%, and 1.7% of 10, 40, and 100 mg/mL of PDS wing, respectively, required rescue therapy.
Table 2 gives details of primary outcome in 10, 40, and 100 mg/mL PDS groups in LADDER trial.
Primary outcome in 10,40 and 100 mg/mL groups in LADDER trial.

PDS, port delivery system.
Secondary outcomes were.
BCVA.
At 9 months, adjusted mean BCVA from baseline in 1, 40, and 100 mg/mL and 0.5 mg of monthly intravitreal ranibizumab injection was –3.2 ETDRS letters, –0.5 ETDRS letters and +5.0 ETDRS and +3.9 ETDRS letters, respectively. At 9 months, there was a difference of +1.1 ETDRS letters in 100 mg/mL arm of RPDS and 0.5 mg of intravitreal ranibizumab group. Thus, improvement in BCVA from baseline was comparable in both groups. At 22 months, observed mean BCVA from baseline in 10, 40, 100 mg/mL and 0.5 mg intravitreal group was –4.6, –2.3, +2.9, and +2.7, respectively. Ability to maintain baseline BCVA over time – loss of <5 ETDRS letters from baseline – 57.7%, 80.0%, 87.5%, and 88.9% in the 10, 40, 100 mg/mL and intravitreal ranibizumab group, respectively, maintained baseline BCVA at 22 months and which was also based on dose response.
Anatomical outcom
At 9 months, adjusted mean CFT change from baseline at 9 months excluding pigment epithelial detachment (PED) height was +54.4, –0.5, –1.7, and –6.3 µm in 10, 40, and 100 mg/mL PDS arms and monthly 0.5 mg intravitreal ranibizumab, respectively. At 9 months, adjusted mean CFT from baseline including PED height was +57.4, +22.2, +11.1, and –29.3 µm in 10, 40, and 100 mg/mL PDS arms and monthly 0.5 mg intravitreal ranibizumab, respectively. At 22 months, observed mean CFT from baseline was similar in 100 mg/mL and 0.5 mg intravitreal group. At 22 months, the observed mean CFT change from baseline from ILM – RPE was –0.7, –20. 9, –4.0, and –10.9 µm in 10, 40, and 100 mg/mL and 0.5 mg intravitreal ranibizumab group, respectively. At 22 months, the observed mean CFT change from baseline including PED was consistent with the primary analysis. Macular atrophy – 14.5%, 11.5%, 13.6%, and 7.3%, respectively in 10, 40, and 100 mg/mL, and 0.5 mg intravitreal group, respectively, after an average of 3.5 months and an average of 2.9 anti-VEGF injections. At last study visit, it was 38.6%, 40.0%, 40.4%, and 45.7%, respectively.
Implant functionality and drug exposure.
The implant was explanted using PDS explant tool in operation theater under local anesthesia under complete aseptic precautions. It was explanted between 8 and 500 days after implantation with a median time of 274 days.
Six implants were removed because clinical efficacy criteria were not met:
Four due to AE.
Two due to physician’s decision.
The explanted implant was functional showing measurable levels in aqueous and vitreous. Later, the plan was modified to give a rescue injection of intravitreal ranibizumab 0.5 mg along with a compulsory refill with 100 mg/mL formulation at the next monthly visit. There was no clogging or any other reason that might account for the lack of meeting clinical efficacy criteria. This enabled patients to retain implant and get a refill based on whether implant refill criteria were met or not.
Mean number of injections during the 9-month period
10 mg/mL RPDS – 3.7
40 mg/mL RPDS – 2.6
100 mg/mL RPDS – 2.4
Mean total number of treatments with monthly intravitreal ranibizumab – 16.8
Lack of meeting clinical efficacy criteria during the 9-month period
10 mg/mL RPDS – 22.4%
40 mg/mL RPDS – 4.8%
100 mg/mL RPDS – 1.7%
Explantation of implants by the end of the study
Total – 15.
10 mg/mL implant group – 9: six due to lack of clinical efficacy, two due to AE, and one due to physician’s decision.
40 mg/mL implant group – 4: two due to lack of clinical efficacy and two due to AE.
100 mg/mL implant group - 2 – one each due to lack of clinical efficacy and AE.
Total patients not meeting clinical efficacy criteria in PDS implant group – 18% – 12 (20.7%) in 10 mg/mL PDS group, 5 (8.1%) in the 40 mg/mL PDS group and 1 (1.7%) in 100 mg/mL PDS group, respectively.
Mean number of ranibizumab treatments during entire duration
10 mg/mL RPDS – 4.1.
40 mg/mL RPDS – 3.0.
100 mg/mL RPDS – 2.9.
Mean total number of treatments with monthly intravitreal ranibizumab – 21. 9
Table 3 gives details of secondary outcome in 10, 40, and 100 mg/mL PDS groups and 0.5 mg intravitreal ranibizumab group in LADDER trial.
Secondary outcome in 10, 40 and 100 mg/mL PDS groups and 0.5 mg intravitreal ranibizumab group in LADDER trial.

BCVA, best corrected visual acuity; CFT, central foveal thickness; ETDRS, Early Treatment Diabetic Retinopathy Study; PDS, port delivery system; PED, pigment epithelial detachment.
a. Adverse events.
Serious 8.9% of patients in PDS arm and none in intravitreal ranibizumab group during the 9-month period.
VH – 7 patients – 50% initially had VH within the first month but after optimizing the procedure with pars plana ablation it occurred in 4.5% of RPDS patients.
Endophthalmitis – three patents – within 1 month in one patient and other two after months of implantation – explantation was done, culture was negative in all of them, BCVA returned to baseline.
Rhegmatogenous retinal detachment (RD) – four patients – one soon after implantation and three later – two out of four RD patients underwent pneumatic retinopexy with retention of implant and remaining two underwent scleral buckle surgery along with explantation of implant.20,21,24
Cataract – 13 patients in PDS group, two in monthly intravitreal group.
Conjunctival bleb – five patients within 1-month duration and one after 1 month.
During the full study period
Ocular AE – post-operative – 89.9 % in PDS treated group after post-operative period – 69.2% in PDS group and 63.4% in the monthly intravitreal ranibizumab group.
Serious ocular AE – 9.5% only in the PDS group and none in the intravitreal group.
Serious AE of VH – 7 (3.9%) – six during post-operative period before optimization of procedure, one after optimization, all of them resolved by the end of the study.
AE – any grade + serious – VH – 19 (10.6%) – PDS group – 11 before optimization and 8 after optimization of the procedure. Except for one, the rest recovered within 120 days.
Cataract – 14 new patients compared with primary results and 5 new in the intravitreal ranibizumab group.
Conjunctival bleb and hyphema – one.
PDS treated patients throughout the study period – 87.7% of AE, 62.6% – conjunctival hemorrhage, 22.3% – conjunctival hyperemia, 15.6% – pain, pain in 5% most frequently reported during refill – exchange procedure.
Systemic AE – five deaths – one in 10 mg/mL, two in 40 mg/mL, one in 100 mg/mL, and one in intravitreal ranibizumab group.
Table 4 gives details of adverse events throughout the study period in both PDS group and monthly 0.5 mg intravitreal ranibizumab group in LADDER trial.
Adverse events in both PDS group and monthly 0.5 mg intravitreal ranibizumab group in LADDER trial.

AE, adverse events; PDS, port delivery system; VH, vitreous hemorrhage.
b. Additional assessments.
Ranibizumab measurements – minimum of 15 pg/mL in serum for 15 months or more with 100 mg/mL of ranibizumab in the PDS implant group.
Anti-drug antibody – 0–10.5% at the start of study, 3.5–13.6% during study, and 6.9–15.3% at the end of study.
Thus, phase-2 trial met its primary criteria paving way to phase-3 trial.
Phase-3 trial (ARCHWAY)
This was a randomized, multicenter open-label active comparator trial (GR40548 – Genentech/Roche) comparing efficacy, safety, and pharmacokinetics of RPDS with 0.5 mg of intravitreal injection of ranibizumab in patients with nAMD.30–33 Two hundred and forty-eight patients belonged to the PDS arm and had refill at 24 weeks and 167 patients were in intravitreal ranibizumab arm who received a monthly dose of 0.5 mg of intravitreal injection for 6 months. The primary endpoint was change in BCVA from baseline averaged over 36–40 weeks. This trial also planned to give onus to the surgical technique of conjunctival and tenons dissection and hemostasis to prevent immediate or long-term complications.
Results
About 98.4% of patients went 6 months without the need for any additional treatment and the outcome was non-inferior and equivalent to 0.5 mg ranibizumab intravitreal injections.
Patients had a mean BCVA of 74.8 letters and had already received anti-VEGF injections (mean of 5) prior to this. There was a gain of 0.2 letters in the PDS arm compared to 0.5 letter gain in the intravitreal ranibizumab arm from baseline averaged over 36–40 weeks. The percentage of patients who gained vision in the RPDS group was 57.8% while that in the intravitreal group were 58.9%. There was also transient surgery-related reduction of BCVA that came back to baseline by week 12 in the PDS arm and was similar to intravitreal ranibizumab thereafter.
The change in center point thickness was +5.4 µm in the PDS arm at week 36 compared with +2.6 µm in the ranibizumab arm.
The total mean number of treatments of implantation, refill, and supplemental treatment was 2 in the PDS arm compared with 10.7 in the ranibizumab arm through week 40.
Table 5 gives results of ARCHWAY trial in the PDS group and monthly 0.5 mg intravitreal ranibizumab group.
Results of ARCHWAY trial in the PDS group and monthly 0.5 mg intravitreal ranibizumab group.
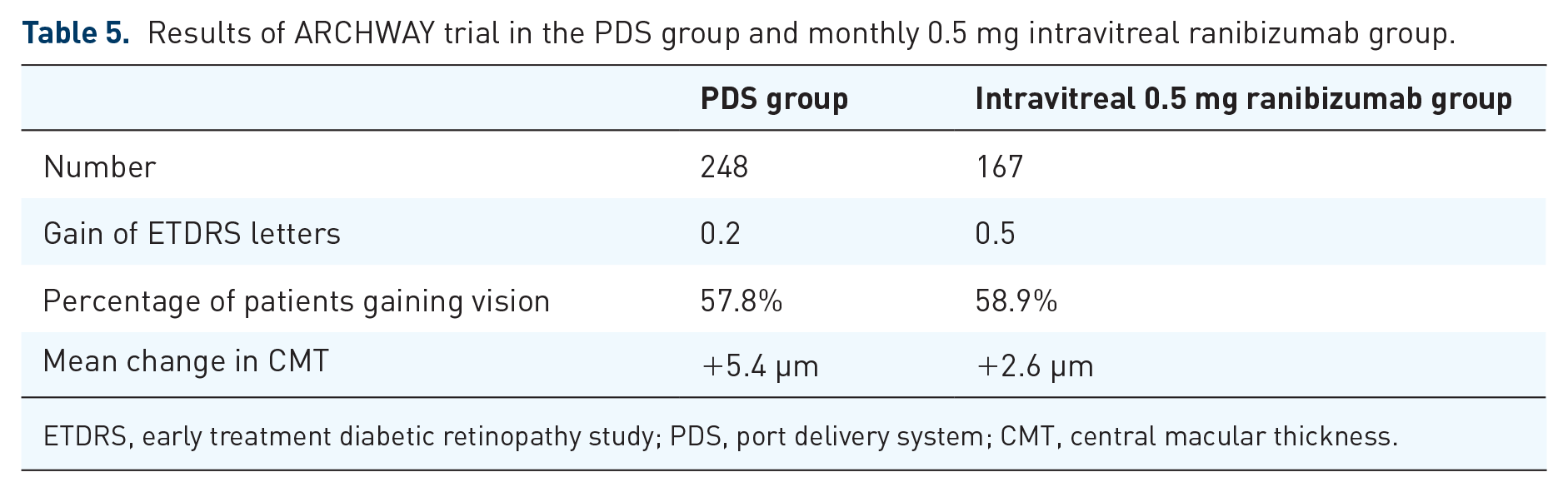
ETDRS, early treatment diabetic retinopathy study; PDS, port delivery system; CMT, central macular thickness.
Adverse events
Endophthalmitis – four, out of which three of them were related to conjunctival retraction. They were effectively treated and BCVA returned to baseline. One had severe and permanent vision loss.
Device dislocation – 1.
VH – 13 – resolved spontaneously.
This trial also emphasized the need for proper closure of conjunctiva and tenons capsule. Surgeons were meticulously trained to reduce long-term complications related to implant device.
Based on the results, so far PDS could reduce treatment number from 12 per year to 2 per year without compromising on efficacy and well tolerated with a favorable risk–benefit profile. Also, a patient-oriented outcome survey revealed that patients chose PDS implant over monthly intravitreal injection.
Long-term results after completion of 96 weeks are eagerly expected.
PORTAL trial
This is a non-randomized, multicenter, open-label extension study (GR40549 – Genentech/Roche) evaluating long-term safety and tolerability of 100 mg/mL PDS with refills administered every 24 weeks for a period of 144 weeks in patients with nAMD who have completed either phase-2 (LADDER) or phase-3 (ARCHWAY) trial. 34 Primary outcome measures include incidence and severity of ocular and systemic AE, incidence, severity, and duration of adverse events of special interest (AESI) and incidence, severity, and duration of PDS-associated ocular AESI during the post-operative period. The secondary outcome is change in BCVA from baseline.
Evaluation of ocular toxicology and biocompatibility of PDS device
To analyze this, the implant along with four ancillary devices (initial refill needle, insertion tool, refill needle, and explant tool were studied). As there were no specific national or international guidelines and standards for evaluating these devices, International conference for harmonization guidelines, International Organization for Standardization (ISO) 10993 and American National Standards Institution (ANSI) Z 80.7 standards were used initially. Then, Center for Devices and Radiological Health Device Classifications were considered. Minipigs were used and phase-3 results showed a favorable risk–benefit profile of the PDS implant.
The biocompatibility of the PDS device was analyzed in rabbit models. As the implant was too large for the rabbit eye, a non-functional surrogate was used. Results of a 6-month study showed that the implant was well tolerated. Both in vitro and in vivo biocompatibility tests showed that PDS implant was non-cytotoxic, non-genotoxic, non-sensitizing, and non-irritating. 35
Long-term stability of anti-VEGF-A under physiologically relevant conditions
Ranibizumab underwent minimal aggregation when placed in phosphate-buffered saline (PBS) at 37° for several months while bevacizumab and aflibercept showed an increased tendency for aggregation. Even after long-term incubation in PBS, ranibizumab showed a very small loss in antigen binding capacity which is necessary for long-term drug stability and in turn its efficacy when placed within a drug delivery system, such as the PDS implant. 36
Discussion
RPDS seems to be promising in meeting the goal of long-term VEGF suppression. Phase-1 trial showed that PDS implant was well tolerated, and there was sustained release of ranibizumab from the implant. There were only four serious adverse events related to the placement of the implant. The approximate change in BCVA was 10 ETDRS letters from baseline at 12 months in all 20 patients. A mean decrease in CFT was also parallelly observed in all 20 patients at 12 months. Thus, the visual and anatomical outcome was comparable to monthly intravitreal ranibizumab group. Mean refill for 20 patients at 12 months was 4.8 and the median time to first refill was 3 months with mean serum concentration of ranibizumab showing plateau levels. Explanted devices and devices which were left for observation were similar to unimplanted devices retaining integrity and tolerability of the device.21,23,24,27,28
Phase-2 trial initially had implant procedure–related VH of about 50% which reduced to less than 5% after optimization of the procedure. There was no risk of VH following the optimized procedure in that 25% of patients who were taking anticoagulants. The median time to first refill was 15.8 months in 100 mg/mL PDS group. About 79.8% did not require implant refill for 6 months or more in the same group. Patients in the PDS group received 80% fewer ranibizumab injections compared to monthly intravitreal ranibizumab group. A dose-response showing greatest benefit with 100 mg/mL group was observed. At 9 months both anatomical and visual outcome was comparable in 100 mg/mL PDS group and intravitreal ranibizumab group. At 22 months too, anatomical and visual outcome were comparable in 100 mg/mL PDS group and intravitreal ranibizumab group. By the end of the study, 96.6% moved to PORTAL trial.20,21,25,26,29
Patients who enrolled for the LADDER trial had already received a mean of 2.9 anti-VEGF injections before the study. About 87.5% of patients in 100 mg/mL PDS group maintained a baseline BCVA at 22 months which was comparable to monthly intravitreal ranibizumab group at 88.9%. Maintaining a BCVA of 20/40 over a mean of 2 years akin to monthly intravitreal ranibizumab group clearly shows that the PDS system can reduce treatment burden without compromising clinical benefit.20,21,25,29
Consistent anatomical improvement in CFT being comparable to monthly intravitreal ranibizumab group is an added advantage to BCVA improvement. Inclusion of PRN refill criteria and inclusion of PED height in CFT threshold would be more beneficial in clinical practice. Pharmacokinetic profile with well-maintained pharmacological concentrations of this drug was good enough to maintain BCVA for a long period.20,21,25,29
AEs were observed in the PDS arm and not in the intravitreal arm which warrants a safe approach that would alleviate AE of RD, endophthalmitis or VH or systemic AE. To witness this, PDS has already gone through various stages of modification and optimization to mitigate the risks of the implantation procedure. Ongoing ARCHWAY trial30–33 will work through additional safety aspects of the PDS system. PORTAL trial 34 promises to unravel safety aspects related to RPDS in the study.
There are studies showing detrimental effects of long-term VEGF suppression potentiating the risk of geographic atrophy in patients receiving monthly dosage in comparison to those treated as PRN. Possible explanations were attributed to either natural evolution of AMD, direct toxicity of anti-VEGF agents or anti-VEGF agents induced reduced perfusion and ischemia of choroidal neovascular membrane (CNVM) compromising outer retinal blood supply. This aspect of RPDS however cannot be overlooked while considering prolonged VEGF suppression.25,37,38
The ongoing ARCHWAY trial has already shown positive results in terms of BCVA and change in CFT. Onus on the surgical procedure is sure to reduce the AE associated with the implant procedure. People’s satisfaction survey toward the PDS system has already created waves and has given the PDS system overall a positive outlook for a brighter blind-free future.29–32
Toxicology and biocompatibility analysis of the PDS implant both in vitro and in vivo has shown that it is non-toxic, non-sensitizing, and non-inflammatory. 35 Long-term stability of ranibizumab under physiologic conditions was also favorable for ranibizumab when compared to bevacizumab and aflibercept. 36
PDS implant has been a pioneer in proving the fact that a biological on-biodegradable reservoir that would continuously release the drug for long periods of time can be placed inside the eye.
Conclusion
Based on the results of clinical trials, RPDS seems to be promising in terms of reducing the need for frequent anti-VEGF injections, reducing chances of undertreatment and patient compliance associated with it. The burden of treatment in nAMD patients is expected to decrease further with positive outcomes of ongoing and upcoming trials. We can look forward to its effects in treating macular edema associated with diabetic retinopathy and vein occlusions in the future. This success formula would lead to the investigation of other sustained delivery treatments like durasert, hydrogel anti-VEGF depot, and encapsulated cell therapy implant.
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
