Abstract
KSI-301 is a new intravitreal anti-vascular endothelial growth factor (VEGF) antibody biopolymer conjugate under investigation for the treatment of age-related macular degeneration (AMD), diabetic macular oedema (DME) and retinal vein occlusion (RVO). Preclinical and early clinical trials so far have shown promising results in retinal vascular diseases. When using anti-VEGF agents for treatment of retinal disorders, the frequency of injections and follow-up visits has increased the treatment burden, greatly affecting the treatment outcome. There are new anti-VEGF agents in the horizon with extended duration of action, durability, safety profile and efficacy, which seem to address the above issues. PubMed search and Medline search were performed on newer anti-VEGF agents, KSI-301, antibody biopolymer conjugate in retina, KODIAK KSI-301, DAZZLE study, GLEAM study, GLIMMER study, GLOW study and BEACON study. This review article showcases the biophysical properties and ongoing trials related to KSI-301. Moreover, we discuss the efficacy and safety profile of KSI-301 on the basis of the results of available trials.
Introduction and background
In 2019, the global prevalence of diabetes was estimated as 9.3%. This figure is expected to increase by 10.2% (578 million) by 2030 and 10.9% (700 million) by 2045. 1 National diabetes statistics report 2020 states that the prevalence of diagnosed diabetes in the Unites States was 8.2% of all ages (crude estimate for 2018). The incidence of diagnosed diabetes among adults aged 18 years or older was 6.9 per 1000 persons. The incidence was higher among those who were aged between 45–64 years and those above 65 years of age. 2 Without appropriate interventions, there is projected increased incidence of 26.6 million, prevalence of 570.9 million and deaths of 1.5 million for 2025. The years of healthy life lost due to disability for 2025 is expected to be 79.3 million. 3 About 7% of the UK population has diabetes, and nearly 1 million people are believed to have undiagnosed diabetes mellitus type 2. 4 The age-standardized disability-adjusted years rate (a measure of overall disease burden, defined by the number of years lost to ill-health, early death or disability and was developed to compare the overall health and life expectancy of different countries) increased by 39.6% (32.1–46.7) from 1990 to 2016 in India. This was considered the highest increase among the major non-communicable diseases. 5 A cross-sectional study in India showed the prevalence of diabetic retinopathy (DR) to be 21.7%. The prevalence was found to be more in males, diabetes duration above 5 years, age > 40 years, insulin users, those with diabetic nephropathy and vascular accidents. 6 Another study showed the prevalence of DR in urban India and in an urban population in south India to be 17.6% and 22.4% respectively.7,8 Studies have shown the prevalence of age-related macular degeneration (AMD) and particularly geographic atrophy to be higher among Europeans than among Asians and Africans. The number of people with AMD globally is expected to reach 288 million by 2040. 9 The number of people affected by AMD in the European Union is approximately 67 million which is expected to increase by 15% in 2050. 10
Vascular endothelial growth factor (VEGF) and angiopoietin have been proven to be specific and critical for blood vessel formation.11,12 The VEGF family consists of VEGF-A, VEGF-B, VEGF-C, VEGF-D and placental growth factor (PIGF).13–15 Hypoxia is a major factor that is known to cause VEGF transcription which when instigated by oxidative stress leads to angiogenesis and increased vascular permeability.16,17 Among the VEGF, VEGF-A has been shown to be strongly associated with angiogenesis and is elevated in patients with neovascular AMD (nAMD), 18 diabetic macular oedema (DME) and retinal vein occlusion (RVO). The VEGF-mediated actions are brought about by binding with its receptors VEGFR1 and VEGFR2, the predominant one being VEGFR2. Hence, the current therapies are targeted at inhibiting VEGF-mediated actions through these receptors.15,19
Intravitreal bevacizumab (Avastin) was manufactured by Genentech/Roche and has not been approved for intraocular use or for intravitreal injection by the US Food and Drug Administration (FDA). 20 However, its off-label use has been proven effective in AMD through CATT and IVAN trials.21,22 The efficacy of intravitreal ranibizumab (Lucentis, Genentech, and Novartis) was proven by RISE and RIDE trials in DME, 23 ANCHOR and MARINA trials in nAMD24,25 and BRAVO and CRUISE trials in RVO.26,27 The efficacy of intravitreal aflibercept (Eylea, Bayer/Regeneron) has already been established through VIEW 1 and VIEW 2 trials in nAMD,28,29 VIVID and VISTA trials in DME 30 and COPERNICUS and GALILEO trials in RVO.31,32 The frequent dosage and follow-up associated with these injections increased the treatment burden greatly affecting the treatment outcome. Hence the need for a safe and effective drug which has a long duration of action is warranted. KSI-301 is a new investigational anti-VEGF antibody biopolymer conjugate (ABC) which may be poised to address these issues with its safety, efficacy and remarkable biological durability of action.
KSI-301
KSI-301 (KODIAK SCIENCES INC. Nasdaq: KOD, Palo Alto, California) is a new investigational intravitreal anti-VEGF. ABC that is under trial for the management of nAMD, DME and macular oedema due to RVO. The ABC platform comprises a humanized IgG1 antibody with inert immune effector function and a biopolymer which is an optically clear, high molecular weight phosphorylcholine polymer covalently bound by single-site specific linkage. Its design optimized both size and molar dose to increase intraocular durability.33–36
The molecular weight of KSI-301 is 950 kDa and that of biopolymer is 800 kDa. In rabbit models, KSI-301 has been shown to bind VEGF-A with high affinity (KD 6.75 pM), higher than its cognate receptors VEGFR1 and VEGFR2, as demonstrated by Surface Plasmon Resonance (SPR) and Kinetic Exclusion Assay (KinExA) thereby preventing signalling and inhibition of proangiogenic and propermeability activities. It has been shown to have high bioavailability in both retina and choroid/retinal pigment epithelium (RPE). The ocular tissue half-life has been demonstrated to be more than 10.5 days in retina and more than 12.5 days in choroid in rabbit models. It blocks all isoforms of VEGF-A and is given as an intravitreal injection.37,38
Table 1 compares the biochemical properties of various anti-VEGF agents. 39
Compares the biochemical properties of various anti-VEGF agents. 39

kDa, kilo Dalton, equivalent values are shown as (approximate) fold difference relative to aflibercept; NA, not available – low affinity of bevacizumab precludes a useful comparison; VEGF, vascular endothelial growth factor.
Method of literature search
PubMed search and Medline search were performed on newer anti-VEGF agents, KSI-301, ABC in retina, KODIAK KSI-301, DAZZLE study, GLEAM study, GLIMMER study, GLOW study and BEACON study.
Phase-1a study
This was a single ascending dose escalation (1.25, 2.5 and 5 mg) study, in which nine patients, three in each dosage group with severe previously treated DME, were enrolled.36,38 The primary outcome was the safety profile at week 2, and secondary outcome was improvement in best corrected visual acuity (BCVA) and anatomical outcome over a period of 12 weeks. Rapid improvements in BCVA and anatomy were observed as early as 1 week after the injection. There were no drug-related adverse events, intraocular inflammation or dose-limiting toxicities. There was sustained median BCVA improvement of nine eye chart letters and median optical coherence tomography (OCT) improvement of central subfield thickness (CST) of 121 µ at 12 weeks after a single dose, pooled across all three dose levels. Dosage of 5 mg was chosen for the ongoing trials.
Thus, the primary endpoint was met which paved way to the Phase-1b trial.
Phase-1b study
This was an open-label, randomized multicenter exploratory study to evaluate the bioactivity, ocular and systemic safety, tolerability, and pharmacokinetics of KSI-301 in treatment-naïve patients with nAMD, RVO and DME using two doses of KSI-301-2.5 mg and 5 mg and were followed over 72 weeks.35,39–44 They were randomized to receive 3 monthly loading doses of either 2.5 mg or 5 mg of KSI-301 and then evaluated monthly. Retreatment was disease-specific and based on retreatment protocol. The primary outcome was safety profile (systemic and ocular adverse events) and the secondary outcome was mean change in BCVA and mean change in CST using OCT from baseline during the time period of 72 weeks.
Retreatment criteria
NAMD
Increase in CST of > 75 µm with a decrease in BCVA of ⩾ 5 letters compared to week 12 or decrease in BCVA of > 5 letters when compared to day 1, due to worsening of wet AMD activity or decrease in BCVA of ⩾ 10 letters compared to the best prior BCVA, due to worsening of nAMD activity or 6 months have elapsed since the last retreatment.
DME and RVO
Increase in CST of > 75 µm with a decrease in BCVA of ⩾ 5 letters compared to week 12 or prior visit or decrease in BCVA of ⩾ 10 letters compared to the best prior BCVA, due to worsening of DME or RVO disease activity.
Investigators can retreat at their discretion if there is significant disease activity that does not meet the above criteria in all subjects.
At 12 weeks, the number of patients included were 17, 8 and 10 in the nAMD, DME and RVO group, respectively, there were no systemic or ocular adverse events during this time period.
At 16 weeks, the number of patients included were 35 in each group, there were no systemic or ocular adverse events during this time period.
After three loading doses, 80% of patients in nAMD group were able to extend for 4 months or longer before the first retreatment, 82% of patients in DME group were able to extend for 3 months or longer, and few even extended to 6 months or longer before the first retreatment. Diabetic retinopathy (DR) was either maintained or improved with no worsening to PDR, the baseline CST in RVO group was 250 µ greater than that in the nAMD or DME groups. Fifty six percentage of patients were able to extend beyond 3 months after the last loading dose.
At 20 weeks, the number of patients included were 25, 15 and 15, respectively, in the nAMD, DME and RVO group, there were no systemic or ocular adverse events during this time period.
After the last loading dose, 92% in the nAMD eyes extended to 3 months or longer without retreatment, 72% in the DME eyes extended to 4 months or longer, Half of RVO eyes extended to 3 months or longer.
At 24 weeks, 39 the number of patients included were 31, 19 and 30 in the nAMD, DME and RVO group, respectively, no intraocular inflammation or ocular serious adverse events noted, no drug-related adverse events or drug-related serious adverse events noted.
55% have achieved a 6 month interval before a mandated retreatment in nAMD group, 64% achieved 6 months or longer without retreatment in the DME group, 53% achieved 4 months or longer without retreatment in the RVO group.
At 44 weeks,43,44 The number of patients included were 31 in the nAMD group, 27 without retinal pigment epithelial detachment (PED), 18 in the DME group and 33 in the RVO group, 68% achieved 6 month treatment interval atleast once during follow up, 45% have not required a single retreatment yet, 71% have achieved ⩾ 4 month treatment interval atleast once during follow up in the RVO group, Mean injections were 1.32, 0.61 and 1.33 which were administered 8 months following the loading phase in nAMD, DME and RVO respectively.
Safety profile Five hundred and forty six injections in 130 subjects have been given, 29 serious adverse events in 16 subjects were noted but none was drug-related. One serious adverse event in the study eye showed worsening of DME and was not drug related. Second was intraocular inflammation (trace to 1+ cells) which resolved completely. There were no immunogenicity or anti-drug antibody that were reported.
The mean improvement in BCVA letters and mean reduction in CST at 12, 16, 20, 24 and 44 weeks are plotted in a two-line graph as shown in Figures 1 and 2.

Shows mean improvement in best corrected visual acuity (BCVA) letters at 12, 16, 20, 24 and 44 weeks from baseline. At 24 weeks, 84% achieved 4 months or longer without retreatment in wet AMD group and 76% achieved 4 months or longer in the DME group.
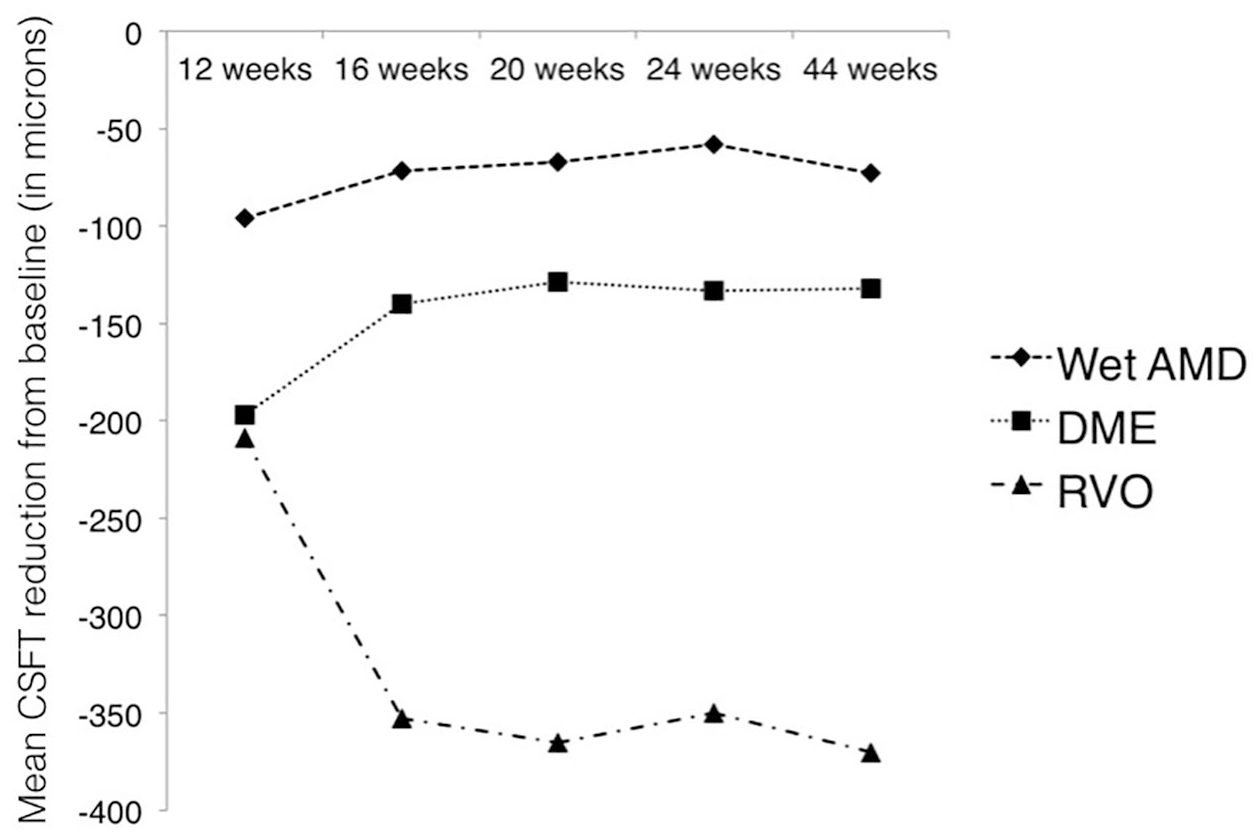
Shows mean reduction in central subfield thickness (CSFT) in microns at 12, 16, 20, 24 and 44 weeks from baseline. At 44 weeks, 82% achieved 4 months or longer without retreatment in the wet AMD group, 76% achieved 4 months or longer in the DME group and 56% achieved 4 months or longer in the RVO group.
Table 2 – Benchmarking: KSI-301 Phase 1b wet AMD, DME and RVO data ‘Generation 2.0’ compared to aflibercept.35,45,46
AMD, age-related macular degeneration; DME, diabetic macular oedema; RVO, retinal vein occlusion.
Mean interval – average interval per patient over 2 years, mean maximum interval – the average of the longest interval achieved per patient at any point during follow-up, mean first interval – First retreatment.
Preclinical studies
KSI-301 is a humanized anti-VEGF monoclonal antibody conjugated to high molecular weight phosphorylcholine-based biopolymer. Affinity of KSI-301 to VEGF was studied using SPR and KinExA. KSI-301-mediated inhibition of VEGF receptor was tested using competitive enzyme-linked immunosorbent assay (ELISA) and compared with existing anti-VEGF therapeutic molecules. Its effects on VEGF-mediated angiogenic functions were tested in human retinal microvascular endothelial cells (HRMVEC) proliferation assay and vascular sprouting assay which uses a bead-based three-dimensional model. The results showed that KSI-301 binds VEGF with single digit picomolar (PM) affinity (a measure of KD which is the equilibrium dissociation constant between antibody and its antigen and are inversely related – lower the KD value, the higher the affinity of the antibody to its antigen). This high affinity in turn blocks VEGF interaction with its cognate receptor with maximal inhibition relative to other molecules and similar half maximal inhibitory concentration compared to aflibercept and ranibizumab. When compared to aflibercept- or bevacizumab-treated cells, maximal inhibition of KSI-301-treated cells showed significant improvement. Also, KSI-301 reduced the number and length of vascular sprouts in the co-culture assay similar to ranibizumab and aflibercept. Thus KSI-301 has been shown to be effective in downstreaming VEGF-mediated effects in diabetes and AMD. 37 Structurally, the biopolymer conjugation prevents the antibody from getting affected by non-specific interactions or PH changes in any chemical environment. It causes preferential changes in the protein colloidal properties while maintaining the IgG structure and function unaffected. 47 Evaluation of seven doses of 5 mg KSI-301 administered at four weekly interval in cynomolgus monkeys over 26-week dosing period showed that repeated bilateral intravitreal administration of maximal feasible dose (5 mg) of KSI-301 was well tolerated and showed a clear margin of safety both ocular- and systemic-wise after repeated administration. 48 In the rabbit ocular pharmacokinetic model, the ocular tissue half-life of KSI-301 was found to be greater than 10 days in retina and greater than 12.5 days in choroid as compared to 3–4 days for ranibizumab and 4–5 days for aflibercept. 38 KSI-301 had a flatter (better) ocular pharmacokinetic (PK) curve which means that it has the potential for extended durability and a flexible retreatment window and hence an increasing concentration advantage over time. This was estimated by data obtained from rabbit model for ranibizumab, which in turn was adjusted arithmetically to human doses and dosing intervals.49–51
Ongoing trials of KSI-301
DAZZLE study
DAZZLE study (KODIAK SCIENCES (NCT04049266) is an ongoing Phase-2 prospective, multicenter, double-masked randomized study aimed at evaluating the safety, efficacy, durability, and pharmacokinetics of KSI-301 and aflibercept in treatment-naïve patients with nAMD.35,44,52 This study has already recruited around 550 patients and the baseline randomization will be 1:1 into two arms – 5 mg KSI-301 and 2 mg aflibercept, and at 52 weeks, the aflibercept group will be re-randomized 1:1 to 5 mg KSI-301 and 2 mg aflibercept. KSI-301 will be administered 3, 4 or 5 months after 3 monthly doses and aflibercept will be administered once every 2 months following three monthly doses. The main outcome will be mean change in BCVA by early treatment diabetic retinopathy study (ETDRS) chart at 1 year, and the secondary outcome will be proportion of subjects gaining or losing ⩾ 5, 10 or 15 letters from day 1, mean change in CST, subretinal and intraretinal fluid volume in OCT from day 1, mean change in choroidal neovascularization (CNV) total lesion area and mean chance in area of leakage on fluorescein angiography from baseline at 1 year.
GLEAM study and GLIMMER study
GLEAM Study and GLIMMER study – KODIAK SCIENCES (KS301P104 and KS301P105) are Phase-3 global, multicenter, randomized studies aimed to evaluate the safety, efficacy and durability of KSI-301 in treatment-naïve patients with DME. In each of these studies, patients will be randomized to receive either intravitreal KSI-301 or aflibercept.35,44 Patients will be administered KSI-301 every 8–24 weeks after three loading doses and aflibercept every 8 weeks after five loading doses. The endpoint is change in BCVA from baseline at 1 year, and patients will be treated and followed up for 2 years. The enrolment into each group is expected to be around 450 patients worldwide.
BEACON study
BEACON Study – KODIAK SCIENCES (KS301P103) is a Phase-3 global, multicenter, randomized study to investigate the efficacy, safety and durability of KSI-301 in treatment-naïve patients with RVO-induced macular oedema (both branch and central RVO).35,44 Patients will be randomized to receive either KSI-301 every 8 weeks after two loading doses or aflibercept for the first 6 months as per label. In the next 6 months, patients from both groups will receive treatment based on protocol-specified criteria individually. The primary objective is to look for change in BCVA from baseline at 6 months and patients will be treated and followed up for 1 year.
Table 3 gives the disease activity assessment in Phase-1b, DAZZLE, GLEAM/GLIMMER and BEACON studies.35,44
Gives the disease activity assessment in Phase 1b, DAZZLE, GLEAM/GLIMMER and BEACON studies.

BCVA, best corrected visual acuity; BEACON, study KS301P103; CST, central subfield thickness; DAZZLE, study KSI-CL-102; DME, diabetic macular oedema; DR, diabetic retinopathy; GLEAM and GLIMMER, Studies KS301P104 and KS301P105; RVO, retinal vein occlusion.
GLOW study
GLOW study (KODIAK SCIENCES) is a Phase-3 study which is aimed at evaluating the efficacy, safety and durability of KSI-301 in patients with non-proliferative DR at 1 year.35,44 This study would be recruiting close to 400 patients, and patients would be administered KSI-301 once every 4 or 6 months after two bimonthly doses.
Future developments of other KSI molecules
KSI-501
Increased levels of interleukin (IL)-6 in the serum and eye has been shown to be associated with disease progression in AMD, DR and macular oedema in RVO and uveitis.35,44 IL-6 is believed to be involved in the upregulation of VEGF expression and VEGF-independent angiogenic functions. This forms the basis for resistance to anti-VEGF in DME. Aqueous levels of IL-6 are superior in predicting the therapeutic response to anti-VEGF in AMD. IL-6 disrupts the outer blood retinal barrier in the pathogenesis of macular oedema.53–59
KSI-501 (KODIAK SCIENCES) is a biconjugate of a dual inhibitor trap antibody fusion (TAF) protein and a phosphorylcholine-based biopolymer (OG2072/KSI-501). It inhibits abnormal angiogenesis and concurrent inflammation. Its anti-inflammatory action is by inhibiting IL-6, and anti-angiogenic action is by inhibiting anti-VEGF. The humanized IgG1 framework sequences and the phosphorylcholine-based biopolymer were similar to that of KSI-301. Preclinical and functional studies show that KSI-501 binds with PM affinity, specifically, simultaneously and independently to its respective receptors (VEGF-A and IL-6). This was assessed using SPR and KinExA. Cell-based assays have shown that the dual inhibitor effectively inhibited lipopolysaccharide-mediated endothelial cell tubule formation and endothelial proliferation in human vascular endothelial cells. This dual inhibition showed a better response than monotherapy targeting either IL-6 or VEGF alone. 60
In 2021, the plan is to submit investigational new drug and initiate Phase-1a/1b trial of KSI-501. In 2022, the plan is to submit data of Phase-1a/1b trial in inflammatory retinal diseases, initiate Phase-3 trials of KSI-501. 35
Discussion
Bevacizumab is not FDA approved for retinal use. Aflibercept and bevacizumab have other applications in medicine, whereas ranibizumab is primarily confined to retinal disease.61,62 The intravitreal half-life of aflibercept in rabbits is 4.7 days. Human and monkey data were not available, hence on mathematical modelling, the estimated intravitreal half-life of aflibercept was 7.1 days. Estimated intraocular half-life of ranibizumab and bevacizumab are 4.75 and 8.25 days, respectively, as determined by their molecular weight. This might contribute to longer duration of action of aflibercept (as long as 2.5 months)63,64 In the study conducted by The Diabetic Retinopathy Clinical Research Network (DRCR), it was shown that significant visual acuity improvement in patients with 20/50 or worse was achieved with aflibercept when compared to ranibizumab and bevacizumab while there was no statistical difference among the three drugs in patients with visual acuity of 20/40 or better. The number of injections and the number of laser treatments required in the aflibercept group was less when compared with that in the other two groups, substantiating its role in the central involving DME. 45 Cumulative data from different studies have shown that the gain in letters was much better with ranibizumab (2.39% with 0.3 mg p < 0.001, 3.56% with 0.5 mg p < 0.001) and aflibercept (2.91% with 0.5 mg p = 0.003, 3.44% with 2 mg p < 0.001) and less with bevacizumab (2.14% with 1.25 mg p = 0.012) in AMD patients. 65 Bevacizumab is more cost-effective. But the long-term cost per patient was lower with aflibercept as the number of injections required were fewer in that group both for DME and AMD.66,67
Conclusion
The high molecular weight biopolymer has been shown to impart better tissue availability, stability, excellent biocompatibility, deeper potency, and fast systemic clearance which in turn are responsible for its durability and an optimal ocular PK. The clinical results (Phase 1a and Phase 1b) of KSI-301 have so far shown excellent safety and greater efficacy in all three retinal diseases – nAMD, DME and RVO. The ongoing Phase-1b trial has also shown extraordinary biological durability with most patients achieving treatment-free intervals of 6 months in nAMD, 6 months or longer in DME and 4 months or longer in RVO. These are interim results and larger trials with long-term follow-up are however required to assess the safety, efficacy and durability of KSI-301 in all three retinal diseases.
Footnotes
Author contributions
Concept, design, literature search, acquisition and analysis of data, manuscript preparation, approval of revisions and manuscript submission was contributed by P.R.C. Literature search, analysis and interpretation of data, manuscript preparation and approval of revisions was contributed by M.V.G.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.

