Abstract
Multimodality ophthalmic imaging systems aim to enhance the contrast, resolution, and functionality of existing technologies to improve disease diagnostics and therapeutic guidance. These systems include advanced acquisition and post-processing methods using optical coherence tomography (OCT), combined scanning laser ophthalmoscopy and OCT systems, adaptive optics, surgical guidance, and photoacoustic technologies. Here, we provide an overview of these ophthalmic imaging systems and their clinical and basic science applications.
Keywords
Introduction
Since the first demonstration of fundus photography, technological advancements have allowed for increasingly high-resolution and high-contrast imaging of the anterior and posterior eye. Development of scanning laser ophthalmoscopy (SLO) in 1980 allowed for en face retinal imaging with significantly reduced light exposure compared with conventional indirect ophthalmoscopy, with recent advancements focused on providing an ultra-wide field of view (FOV).1–3 The introduction of optical coherence tomography (OCT) in 1991 enabled depth-resolved volumetric imaging, further facilitating ophthalmic disease diagnosis by providing access to subsurface features. 4 Adaptive optics (AO) was first introduced for fundoscopy in 1997 and has since been applied to both OCT and SLO to achieve cellular-resolution ophthalmic imaging by correcting aberrations of the eye. 5 Recent advances have focused on supplementing structural contrast with functional modalities, such as OCT-based quantification of the metabolic and biomechanical properties of ocular tissue and multimodality systems such as OCT-SLO, surgical microscope–integrated OCT, and multimodal photoacoustic microscopy (PAM). Here, we provide an overview of these ophthalmic imaging systems and their clinical and basic science applications (see Table 1).
Summary of multimodal ophthalmic imaging technologies.

OCT, optical coherence tomography.
OCT-derived contrast mechanisms
OCT is the clinical standard for diagnosing and monitoring ophthalmic diseases and enables subsurface visualization of tissue scattering. A combination of system modifications and post-processing algorithms can be applied to achieve a variety of complementary imaging modes that probe additional contrast mechanisms. Examples of these multimodal OCT techniques include optical coherence tomographic angiography (OCTA), polarization-sensitive OCT (PS-OCT), optical coherence elastography (OCE), phase-decorrelation OCT (PhD-OCT), photothermal OCT (PT-OCT), and pump-probe OCT (PP-OCT).
Optical coherence tomographic angiography
Changes to retinal vascular density and perfusion accompany many ophthalmic diseases, including glaucoma, age-related macular degeneration (AMD), and diabetic retinopathy (DR). 6 Fluorescein angiography (FA) and indocyanine green angiography (ICGA) have traditionally been used to detect vascular leakage, neovascularization, and occlusions. 7 However, these techniques require injection of exogenous contrast and lack depth selectivity for specific retinal layers. 8 Several different OCT acquisition and post-processing methods, collectively termed OCTA, have emerged as non-invasive alternatives for volumetric imaging of retinal vascular perfusion. Doppler OCT uses phase differences between repeated OCT acquisitions to estimate Doppler frequency shift and the relative velocity of flowing scatterers within perfused vessels. 9 However, phase-based angiography techniques are highly susceptible to bulk motion, which reduces sensitivity to blood flow. Power-Doppler methods, including optical micro-angiography (OMAG),10,11 phase variance, 12 and Doppler variance angiography, 13 trade off flow velocity resolution for reduced sensitivity to bulk motion. These methods have demonstrated benefits for high resolution in vivo imaging of retinal and choroidal perfusion. Intensity-based angiography methods, such as split-spectrum amplitude-decorrelation angiography (SSADA) 14 and speckle decorrelation, 15 use intensity changes between repeated OCT cross sections as surrogate measures of blood flow and have been translated for clinical imaging of changes in optic disk perfusion in glaucoma and choroidal neovascularization in AMD patients.16–19 Angiography methods that take advantage of both the amplitude and phase of the OCT signal further enhance robustness against phase instability and include complex differential variance (CDV)20,21 and eigenvalue decomposition. 22 SSADA and OMAG have been integrated into commercially available systems (Optovue AngioVue and Zeiss AngioPlex, respectively), enabling high-quality clinical imaging and quantification of retinal perfusion.23,24
Handheld OCTA
Despite commercialization of OCTA, the majority of these systems require patients be imaged upright, thus precluding imaging in supine or bedridden patients. To address these limitations, several handheld OCT probes have been developed for point-of-care imaging,25,26 including in pediatric patients. 27 Commercially available handheld OCT devices, such as the iVue (Optovue, Inc, Fremont, CA, USA) and Envisu C2300 (Leica Microsystems, GmbH, Wetzlar, DE), have relatively slow imaging speeds that limit sampling density and OCTA sensitivity. Research prototypes optimized for OCTA imaging28–30 increase speeds by an order of magnitude compared with current-generation commercial systems by using 100–400 kHz swept sources. Clinical imaging of retinopathy of prematurity (ROP) patients using these systems has provided in vivo images of the foveal avascular zone and retinal capillary complex in neonates (Figure 1(a)–(d)).31,32

(a)–(d) Handheld OCTA of ROP at the (a) optic nerve head, (b) peripapillary region, (c) perifoveal region, and (d) margin of the fovea. Visible-light OCT in a rodent model showing (e) OCTA projection with delineation of arteries (red) and veins (green), and circumpapillary (f) retinal structure and (g) Doppler blood flow cross section.
Visible-light OCTA
Ophthalmic OCT has conventionally been performed using near-infrared wavelengths because of the availability of light sources and benefits in reduced scattering and increased penetration depths. Commercial availability of broadband supercontinuum light sources has enabled OCT imaging at visible wavelengths, which provides access to more endogenous and exogenous molecular contrast mechanisms.33–38 When combined with OCTA maps of retinal vascular perfusion, visible-light OCT can provide complementary contrast for quantifying blood oxygenation as a surrogate for quantifying metabolic changes associated with DR and AMD (Figure 1(e)–(g)).36,38–45 In vivo visible-light OCT retinal imaging has been demonstrated, and development of robust clinical systems is ongoing.36,46–48
Clinical utility and challenges
Ongoing clinical OCTA research focuses on imaging in AMD, DR, and diabetic macular edema (DME) and vascular response to therapy or surgery.49–52 Wide-field OCTA systems have also been developed and are capable of imaging a 100° FOV, which benefits imaging of the peripheral retina.53,54 Despite significant advantages over FA and ICGA, the clinical utility of OCTA is limited by several key factors. Engineering advancements in OCTA algorithms require physician education for proper interpretation of angiograms. 55 OCTA algorithms also require repeated imaging and dense sampling over small FOVs, making them susceptible to bulk motion during long acquisitions. Handheld OCTA imaging is further complicated by combined patient and photographer motion, both of which degrade vascular contrast and resolution, distort anatomic features, and preclude robust quantitative measurements.31,56 Additional image artifacts, such as shadowing from large retinal vessels, axially smeared vessel cross sections as a result of scattering, and out-of-focus OCTA projections, can significantly impact vessel segmentation algorithms and quantitative analysis of retinal vascularity.57–60 Motion-compensation methods, such as novel scanning or eye-tracking technologies, are actively being studied to overcome these limitations.61–64 Furthermore, vessel enhancement techniques, such as the complex continuous wavelet transform and multiple en face registration and averaging, can be applied to improve the accuracy of vessel segmentation.65–68 Despite these methods, various commercial systems employ different post-processing and segmentation algorithms, making interpretation by clinicians challenging. 7 While visible-light OCT provides complementary endogenous functional contrast, clinical translation of the technology is challenging because visible wavelengths are distracting and make fixation extremely difficult. 48 In addition, visible-light OCT images routinely require dense averaging to overcome low image quality that results from higher laser noise and significantly lower maximum permission exposure compared with near-infrared sources. 69
Polarization-sensitive OCT
PS-OCT measures changes in polarization states between incident and back-reflected light, which can provide additional tissue-specific contrast. 70 The amplitude and relative phase delay between orthogonal polarization states can be used to compute parameters such as tissue birefringence and degree of polarization uniformity.71–77 These methods enable quantitative in vivo imaging of changes in tissue properties resulting from disease progression and treatment.78,79
Anterior segment
PS-OCT is particularly well suited for corneal imaging because the collagen stroma exhibits strong birefringence. 80 Previous works have shown that corneal birefringence is related to fibril orientation in the lamellae, and quantitative metrics, such as phase retardation and slow axis orientation, have been used to distinguish healthy from diseased corneas.81,82 Keratoconus, which is characterized by thinning and distortion of the central cornea, exhibits increases in phase retardation near the rim of corneal thinning and changes in slow axis orientation resulting from rearrangement of stromal collagen. 83 PS-OCT can also enhance visualization of the trabecular meshwork, ciliary body, and iris, which can be used to study the progression of diseases such as glaucoma.84–86 Excess buildup of aqueous humor because of glaucoma can require surgical intervention, commonly trabeculectomy, which allows for aqueous flow into the subconjunctival space. Post-operative PS-OCT has characterized intra-bleb fibrosis and associated complications to benefit clinical management after trabeculectomy.87,88 Improvements in OCT speed and resolution have benefited PS-OCT by enabling quantitative measurement of corneal layer thicknesses and enhanced visualization of Bowman’s membrane and the sub-basal nerve plexus.89–92
Posterior segment
PS-OCT in the posterior segment has traditionally measured retinal nerve fiber layer (RNFL) birefringence and thickness.81,93–95 Similar to the cornea, bundles of parallel cylindrical axons in the RNFL exhibit high birefringence, and decreases in RNFL birefringence can potentially be used as a surrogate measure for ganglion cell and axonal atrophy in glaucoma. 96 Similar changes in phase retardation and birefringence resulting from RNFL thinning have been observed in diabetic eyes (Figure 2).97,98 PS-OCT characterization of the macula has shown that the retinal pigment epithelium (RPE) is highly depolarizing, scrambling the polarization of backscattered light from the RPE and choroid.99,100 Studies have quantified the degree of polarization uniformity (DOPU) from PS-OCT images and used it to aid RPE segmentation and thickness mapping, which can benefit quantitative monitoring of drusen and geographic atrophy in AMD.71,101–105 PS-OCT has also been used to detect and segment subretinal fibrosis, which is common in neovascular AMD and difficult to differentiate from other hyperreflective tissues.106,107

Comparison of (a), (d), (g) RNFL thickness, (b), (e), (h) phase retardation, and (c), (f), (i) birefringence between healthy, diabetic, and glaucoma subjects. Reduced retardance is seen in both diabetic and glaucoma subjects compared with healthy subjects. In addition, there is significantly reduced birefringence in diabetic patients.
Clinical utility and challenges
The majority of PS-OCT research has focused on quantifying changes in tissue properties resulting from ophthalmic diseases. Polarization-sensitive OCTA has also been demonstrated for ophthalmic imaging using Jones matrix-based CDV. 21 The use of polarization-based angiography has advantages over conventional OCTA by improving vessel contrast and increasing image signal; four OCTA images (two incident and two detected polarization states) can be generated and averaged for increased SNR. 108 Despite the utility of PS-OCT in differentiating birefringent and depolarizing tissues in the eye, there are no commercially available PS-OCT systems routinely used in clinical ophthalmology. Challenges in clinical translation include increased cost and system complexity over conventional OCT systems. Many PS-OCT systems use free-space optics, which are sensitive to misalignment, 109 whereas fiber-based PS-OCT systems are more robust but are limited by birefringence and polarization mode dispersion of fiber-optic components. 110 One limitation of using depolarization maps for RPE segmentation is distinguishing it from the choroidal stroma. However, by combining PS-OCT with OCTA, the vessel-rich choroidal stroma can be separated from the vessel-free RPE and can be used to evaluate damage in the RPE layer due to serous RPE detachments.111,112
OCT elastography
OCE uses OCT imaging to detect micron-scale displacements caused by an external mechanical stimulus to extract biomechanical properties of tissue.113,114 PhD-OCT is an alternate method for measuring tissue biomechanics that uses the decorrelation of scattered light from Brownian motion as a surrogate measure of tissue viscosity.115–117 Initial OCE demonstrations used OCT speckle tracking of axial displacements from a static loading force to quantify tissue strain and derived Young’s modulus from the linear stress–strain relationship.118,119 OCE can also be used to measure Young’s and shear moduli by combining dynamic loading forces, such as steady-state harmonic loading and transient excitation sources, with advanced wave propagation models. 120 These dynamic OCE methods have been used to non-invasively measure biomechanical properties of the human cornea in vivo, showing the potential for clinical translation and utility. 121
Cornea
Corneal elasticity can be a useful direct measure for diagnosing keratoconus and corneal ectasia and monitoring corneal collagen crosslinking (CXL) treatment.122–125 The corneal elastic modulus changes with increased intraocular pressure (IOP) and can be used as an indirect measure for diagnosing glaucoma. 126 OCE has shown increased corneal stiffness in in vivo rabbit eyes after CXL treatments with an air puff as the external mechanical stimulus (Figure 3(a) and (b)).127–129 OCE studies using air-coupled ultrasound excitation, which is non-contact and more applicable for clinical translation, have successfully shown quantitative 4-dimensional (4D) visualization of corneal stiffness.130–133 More recent OCE demonstrations have used acoustic radiation force (ARF) loading with an ultrasound transducer, which has a faster response time as compared with air puff loading. 134 Additional OCE stimuli include passive mechanical perturbations caused by the heartbeat 135 or using a pulsed laser to induce mechanical waves, 136 but these methods have yet to be demonstrated in vivo. PhD-OCT has successfully identified changes in corneal biomechanics after CXL in vivo without the need for external stimuli. 117

OCE imaging of the (a), (b), cornea and (c), (d) retina. (a) Structural OCT and (b) OCE elastogram cross sections of in vivo rabbit cornea pre-, post-, and 1 week after CXL treatment (top to bottom, respectively). (c) Structural OCT and (d) OCE elastogram cross sections of ex vivo porcine retina showing differences in retinal layer stiffness.
Lens
OCE can offer a quantitative, non-invasive method for early detection and monitoring of changes in lens biomechanics associated with cataracts and aging. 137 ARF-OCE has shown higher Young’s modulus in cataract lenses compared with healthy lenses in ex vivo rabbit eyes and significant increases in lens stiffness with aging in vivo. 138 OCE imaging has also been combined with Brillouin microscopy, which measures material stiffness using differences in Brillouin light scattering, to show a correlation between Young’s and Brillouin moduli and provide a more complete mapping of lens stiffness in ex vivo porcine eyes. 139 Finally, OCE has also been used to investigate changes in lens elasticity as a function of IOP. 140
Retina
Cellular changes in AMD can alter the elasticity of retinal tissue, making OCE a potential technology for early disease diagnosis. 141 ARF-OCE studies have shown distinct elasticity differences in retinal layers in in vivo rabbit and ex vivo porcine models (Figure 3(c) and (d)).142,143 Decreased retinal stiffness observed in in vivo rabbit AMD eyes was hypothesized to result from lymphocyte infiltration, but initial results did not show statistical significance. 143 OCE studies have also shown that increased optic nerve head Young’s modulus and posterior scleral stiffness are correlated with increasing IOP, which suggests that OCE can also be used to monitor progression of glaucoma.144,145
Clinical utility and challenges
Both OCE and PhD-OCT technologies are in the early stages of clinical translation, and current research is focused on improving imaging speed and phase stability. 146 Repeated images are used to compute differential phase in OCE, but this increases total imaging time and is susceptible to motion artifacts. Recent advances in swept-source OCT technology may overcome these limitations by enabling OCE imaging at megahertz rates. 147 Currently, there are no commercial OCE systems available for clinical use. The broad clinical adoption of OCT in ophthalmology may benefit clinical translation of multimodal OCE technologies pending successful identification of optimal mechanical stimulus mechanisms and clinical applications.
Molecular contrast methods
OCT sensitivity to changes in optical pathlength can be increased by leveraging phase information and used to probe additional mechanisms of contrast. As an example, light absorption by endogenous or exogenous contrast agents induces local temperature gradients and index of refraction changes. PT-OCT uses these index changes as a complementary contrast mechanism for detecting functional cellular and subcellular changes that could enable earlier disease detection. 148 PT-OCT has been demonstrated using exogenous gold nanoparticles and endogenous melanin in the RPE and choroid (Figure 4(a) and (b)) in in vivo and ex vivo animal models.148–151 Most recently, PT-OCT using indocyanine green (ICG) was demonstrated as a method for distinguishing the inner limiting membrane, which is of interest for macular surgery applications. 152 PP-OCT is another technique for adding molecular contrast to OCT imaging. PP-OCT excites ground-state electrons using a modulated excitation (pump) and detects the transient absorption at a lower energy wavelength (probe) to identify specific molecular compounds such as methylene blue, hemoglobin, or melanin.153,154 PP-OCT imaging in ex vivo porcine iris has demonstrated sensitivity to melanin, which may be a useful diagnostic tool for ocular melanoma (Figure 4(c) and (d)). 155

Molecular contrast imaging. In vivo PT-OCT comparing (a) pigmented to (b) albino mouse retina with computed melanin concentration (green) overlaid on OCT cross sections. PP-OCT image of ex vivo porcine iris showing the (c) relative concentration of melanin and (d) corresponding OCT cross section.
Clinical utility and challenges
PT-OCT and PP-OCT are promising multimodal techniques that supplement OCT contrast, but neither have been demonstrated in in vivo human applications nor have commercialized clinical systems. Tissue heating and high excitation power levels are two safety concerns that need to be addressed. Acquisition speed is an additional limiting factor because of the need to critically sample the modulation frequency of the pulsed light source and slow heating response of absorbers. 152
OCT-SLO
The inherent orthogonal priority acquisition orientations between OCT and SLO provide complementary depth-resolved and en face information that makes combined OCT-SLO an ideal multimodality technology. These systems can leverage fundus features for motion-tracking and volumetric imaging of anatomic structures and pathologies.
Tracking SLO
SLO has been used extensively for aiming and motion compensation of ophthalmic OCT.156–159 Tracking SLO has been particularly critical for OCTA, which requires dense sampling over small FOVs and is, therefore, difficult to target ROIs and highly susceptible to saccadic and bulk motion artifacts. 62 Many current generation combined OCT-SLO systems use shared optics and scanners to relay light into the eye to reduce overall system complexity and provide pixel-level co-registration between corresponding OCT and SLO images.160–163 SLO motion-tracking precision is related to both spatial and temporal resolution, and development of line-scanning SLO (LSLO) has overcome frame-rate limitations of conventional point-scanning SLO systems at the expense of full confocality, allowing imaging of fast dynamics up to several hundred frames per second.163–166
Handheld OCT-SLO
Multimodal OCT-SLO systems have traditionally been integrated with slit lamps that required patients to sit upright during imaging. The development of compact OCT-SLO handheld probes has benefited ophthalmic disease diagnostics in pediatric and bedridden patients.27,167 When combined with a white-light supercontinuum light source, additional spectroscopic information can be extracted, including color SLO images that can be used to detect early signs of fundus discoloration in AMD. 168 Novel extended source LSLO illumination and detection schemes, such as those using spectral encoding, have replaced complex free-space relays from source to detector with fiber optics, and handheld prototypes using these technologies have recently been used for motion-corrected in vivo ophthalmic OCT and OCTA.30,169,170
Adaptive optics
Lateral resolution in ophthalmic imaging is fundamentally limited by wavefront aberrations of the eye. AO overcomes this limitation by actively compensating for these aberrations using a deformable mirror, enabling cellular-resolution in vivo imaging.5,171 Combined AO-SLO and AO-OCT systems are capable of imaging individual photoreceptors with up to two to three times higher transverse resolution than conventional SLO and OCT.172–175 When combined with hardware- and software-based retinal-tracking technologies, 176 AO systems can be used to quantify retinal photoreceptor distributions across large FOVs and study cellular changes associated with retinal disease progression (Figure 5(a)–(f)).103,177–207 In addition to structural imaging, AO systems have also demonstrated benefits for functional imaging, including visualizing perfusion in the retinal microvasculature and phase dynamics from photoreceptor photostimulation.208–210

In vivo human (a)–(c) AO-OCT and (d)–(f) AO-SLO showing photoreceptors at (a), (d) the fovea, (b), (e) 5° TR, and (c), (f) 10° TR. Scale bar = 50 µm. (g)–(i) oSLO fluorescence projections and (j)–(l) corresponding OCTA projections showing murine retinal perfusion. Scale bar = 200 µm.
Fluorescence SLO
The combination of SLO and fluorescence contrast is uniquely suited for high-specificity functional imaging in clinical ophthalmology and basic science. Traditional clinical applications have included fundus autofluorescence to detect endogenous fluorophores and exogenous fluorescein or ICG to visualize retinal vascular perfusion and leakage. 211 In animal models, combined OCT and fluorescence SLO systems enabled longitudinal imaging of retinal structure and characterization of function and cell populations in transgenic models expressing fluorescent proteins in serum albumin, microglia, and photoreceptors.212,213 Animal models also provide an opportunity to study the effects of retinal injury, including laser-induced choroidal neovascularization, retinal vascular occlusion, laser lesioning, and image-guided intraocular drug-delivery.214–217 One major limitation of fluorescence SLO is its limited depth-sectioning and volumetric imaging capability. Oblique SLO (oSLO) addresses this limitation by using obliquely oriented excitation and detection paths to acquire fluorescence cross sections and volumes (Figure 5(g)–(l)). 218 Fluorescence SLO can also be combined with AO to improve depth-sectioning and lateral resolution to enhance visualization of individual photoreceptors, ganglion cells, and RPE cells in animal models.219–224
Clinical utility and challenges
Combined OCT-SLO technology has been integrated into commercial systems, including the Heidelberg Spectralis, Optos Silverstone, and Zeiss PLEX Elite, for active motion-tracking and high-resolution en face and cross-sectional imaging of the fundus. While several AO systems, such as the Imagine Eyes rtx1 and Physical Sciences compact AO retinal imager (CAORI), are commercially available, broad adoption of the technology is limited because of system complexity, the need for long imaging and post-processing times, and lack of standardized processing and analysis algorithms. 225 Sensorless AO systems have been demonstrated in research prototypes and eliminate the need for physical wavefront sensors, which significantly reduces system complexity.226–230 These technologies have been recently integrated into handheld probes for AO imaging in supine and pediatric patients, which may further motivate broad clinical adoption.231,232 Fluorescence angiography and fundus autofluorescence are well established in clinical ophthalmology. More recently, autofluorescence lifetimes have been used as a surrogate for retinal metabolism that provides additional contrast for identifying ophthalmic and systemic diseases.233–235
Multimodal surgical visualization
Integration of OCT with ophthalmic surgical microscopy can provide additional high-resolution depth-resolved volumetric visualization of tissue microstructures that benefits surgical decision-making. 236 Initial demonstrations of intraoperative OCT (iOCT) were performed with handheld probes during surgery,237–239 and the technology has since advanced to microscope-integrated systems that allow for OCT imaging concurrent with ophthalmic surgery.240,241 Recent iOCT research has focused on improving speed and ergonomics, such as automated instrument-tracking and enhanced visualization. The combination of high-speed swept-source OCT systems with real-time volumetric rendering methods has allowed for 4D visualization of surgical dynamics (Figure 6(b)).242–244 OCTA imaging has also been explored as a method for integrating vascular contrast to further enhance intraoperative visualization.245,246
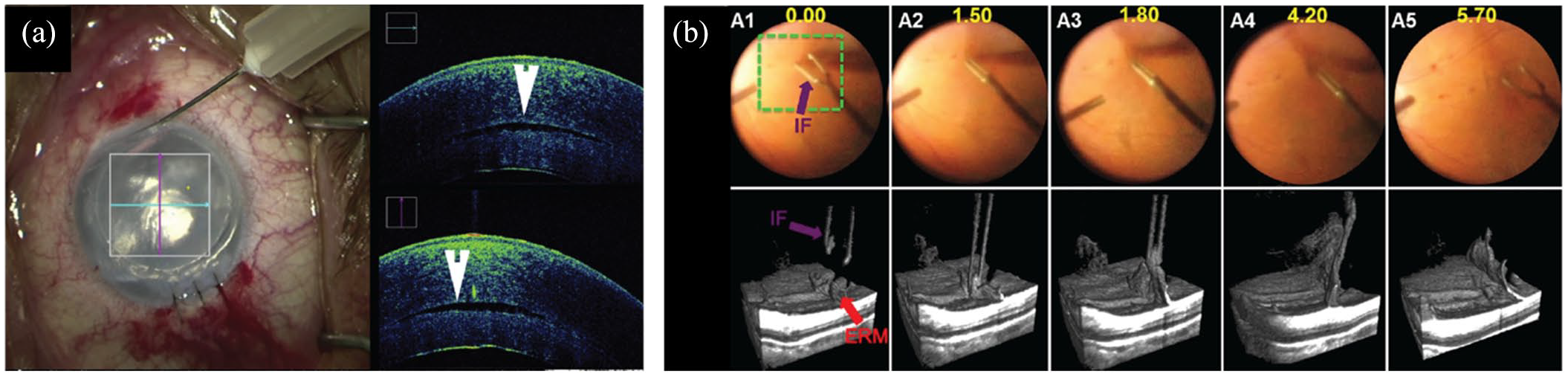
iOCT visualization during (a) anterior and (b) posterior segment surgery. (a) iOCT showing graft placement during corneal transplant showing a persistent fluid interface (arrowhead). (b) 4D iOCT imaging of forces peeling epiretinal membrane.
Clinical utility and challenges
Multiple iOCT systems are commercially available, including the Haag-Streit Surgical iOCT, Zeiss RESCAN 700, and Leica EnFocus. 247 Clinical studies have shown that iOCT can benefit surgical decision-making for donor graft positioning during corneal transplant (Figure 6(a)) and during membrane peeling and retinal detachment repair procedures.248,249 One major challenge in iOCT imaging has been shadowing from optically opaque surgical instruments. However, semi-transparent, OCT-compatible surgical instruments have been explored to overcome these limitations. 250 While preliminary studies have shown the benefits of iOCT for providing real-time surgical feedback, additional improvements in the technology are being developed, and studies demonstrating clinical utility are ongoing.
Photoacoustic imaging
Photoacoustic imaging detects acoustic waves generated by thermoelastic expansion from the absorption of pulsed laser illumination and reconstructs a volume of absorbers.251,252 In ophthalmology, PAM primarily visualizes blood absorption and has advantages over FA and ICGA by not requiring exogenous contrast and OCTA by being sensitive to vascular leakage. PAM also has higher depth penetration compared with OCT and can achieve imaging depths exceeding 10 mm. 253 PAM demonstrations in animal models include visualization of deep retinal and choroidal vasculature, subretinal injections, and neovascularization. 254 Multimodal PAM and OCT imaging provides complementary structural and vascular contrast and has been used to visualize neovascularization in animal models of DR and wet-AMD.255–259 Combined PAM and OCT has also shown advantages for visualizing vascular and structural changes associated with retinal vein occlusion (Figure 7) and choroidal vascular occlusion over FA.260,261 Finally, PAM and OCT can be used to quantify retinal oxygen metabolism rates by combining blood flow measurements from Doppler OCT with hemoglobin oxygen saturation measured using multiwavelength PAM that may benefit early diagnosis of glaucoma, DR, and AMD.39,262

In vivo PAM showing neovascularization in rabbit retina. (a) Color fundus, (b) FA, (c) cross-sectional OCT, (d) PAM maximum amplitude projection, and (e) PAM 3D reconstruction of neovascularization (arrows).
Clinical utility and challenges
Although there are commercial PAM systems such as the Vevo LAZR-X for cardiac and neuroimaging, PAM imaging systems for ophthalmic applications are not currently available. 263 Barriers to clinical translation of PAM include system complexity, slow imaging speeds, and the need for a contact-PAM transducer. Multimodal PAM and OCT requires dedicated laser sources for PAM excitation and OCT imaging, a transducer for PAM detection, and an OCT engine. Methods to share a laser source between PAM and OCT to reduce system complexity have been proposed. However, hemoglobin absorption is low in the near-infrared, which limits PAM sensitivity at OCT wavelengths. 264
Conclusion
Multimodal ophthalmic imaging spans multiple optical and acoustic technologies. Many of these methods, such as OCTA, PS-OCT, OCE, PhD-OCT, PT-OCT, and PP-OCT, aim to add additional contrast to conventional OCT images. The complementary en face and cross-sectional information from combined SLO and OCT systems has benefited motion-tracking and aiming in clinical ophthalmology, and the addition of AO and fluorescence methods has further improved resolution and imaging specificity. Integration of OCT with surgical microscopy has benefited real-time surgical feedback and surgical decision-making. Finally, multimodal photoacoustic imaging systems enable quantitative imaging of blood perfusion and in vivo hemodynamics. Overall, multimodal ophthalmic imaging developments have led to improvements in clinical imaging and novel basic science research with new possibilities for clinical translation and improvements in disease diagnosis and therapeutic monitoring.
Footnotes
Acknowledgements
Morgan J. Ringel and Eric M. Tang contributed equally.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Vanderbilt University, the Vanderbilt Institute for Surgery and Engineering (VISE), and the US National Institutes of Health Grant No. R01-EY030490, R01-EY031769 and T32-EB021937. The content was solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
