Abstract
Optical coherence tomography has improved the ophthalmic clinical examination due to its ability to more accurately characterize pathology of the eye. Optical coherence tomography is now an integral imaging modality for many adult diseases of the retina and is a ubiquitous part of a retinal practice. However, although its use in adults is well recognized, the adoption of optical coherence tomography in pediatric patients may be undervalued as macular diseases in children are relatively uncommon. Recent technological advancements have made optical coherence tomography scans quicker and more precise, generating normative data and leading to more widespread use in children. In this review article, we discuss how pediatric optical coherence tomography data can be used to understand normal development, as well as diagnose and monitor retinal disease. In particular, we highlight clinical optical coherence tomography findings of several pediatric retinal conditions, including retinopathy of prematurity, congenital infections, retinal dystrophies, and phakomatoses such as neurofibromatosis.
Introduction
The use of optical coherence tomography (OCT) in clinical practice has increased exponentially over the past 10 years due to technological advancements and its widespread availability. In adults, OCT is integral to diagnosing and monitoring macular conditions such as age-related macular degeneration, diabetic retinopathy, retinal vein occlusion, and pachychoroid spectrum disorder.1–3 Although OCT offers many advantages over clinical examination, such as higher sensitivity and localization of pathology to individual retinal layers, its value in the pediatric population may be underrecognized. Toddlers as young as 3 years old can often cooperate for desktop OCT, and handheld devices are available for use in infancy and under anesthesia. The development of faster image acquisition with spectral-domain OCT, along with eye-tracking technology, has made OCT even more feasible in children.4,5 While most pediatric retinal diseases primarily affect the periphery (e.g. retinopathy of prematurity, familial exudative vitreoretinopathy, Coat’s disease), OCT has increased our understanding of macular pathology in peripheral retinal diseases. In this review, we discuss the clinical relevance of OCT in pediatric patients.
Retinopathy of prematurity
Retinopathy of prematurity (ROP) is one of the most common retinal diseases in children, occurring in preterm neonates and increasing in incidence around the world due to advancements in premature neonatal care. 6 With the increased prevalence of ROP, there has been more interest and opportunity to use OCT to better understand macular development in prematurity. In addition, with the options of primary laser, anti–vascular endothelial growth factor (anti-VEGF), and combination treatment, OCT now serves as an important tool for comparing the advantages and disadvantages of treatment options.
One of the first papers using OCT in preterm infants imaged subjects from India with gestational age averaging 31–32 weeks and followed patients serially until 52 weeks. 7 The study demonstrated cystoid spaces and greater central foveal thickness in infants who developed ROP. Another paper by Vajozovic and colleagues 8 used the presence of the ellipsoid zone (EZ) at the foveal center, distance from Bruch’s membrane to the EZ, and radial distance from foveal center to EZ as measures of retinal maturity and found that photoreceptor inner and outer segment development is delayed in preterm infants from 23 to 31 weeks of gestational age compared to full-term infants. Longitudinal studies in preterm infants have also characterized the maturation of the fovea, demonstrating centrifugal migration of the inner retina with outer retina thickening. 9
Lee and colleagues 10 reported 61% of 23 neonates imaged had the findings of cystoid spaces on OCT during ROP screening. The authors suggest this may be from elevated VEGF levels and could be used as a marker of additional ocular or systemic disease. The same group followed 13 children with cystoid spaces during ROP screening longitudinally and found that children with this finding had worse visual acuity and either sensorimotor deficits or poor neurodevelopment at 2 years of age. 11 Thus, the subclinical findings of cystoid spaces on OCT in infancy seem to predict both visual and neurodevelopmental outcomes for preterm infants (see Figure 1 for an example of OCT images in ROP).

OCT image of an 11 year-old female who was born at 24 weeks of gestational age. She developed type I retinopathy of prematurity in infancy that was treated with laser therapy. The foveal cross section demonstrates increased central foveal thickness and retained inner retinal layers.
In school-aged children born preterm, desktop OCT angiography (OCT-A) has demonstrated macular immaturity similar to what has been detected in infancy as well as decreased foveal avascular zone (FAZ) size. 12 Smaller FAZ and increased central foveal thickness have been correlated to worse best-corrected visual acuity. 13 OCT and OCT-A have also been applied in studies on school-aged children investigating the use of anti-VEGF medications during infancy, and these studies have found reduced refractive error and more normally developed macula compared with laser treatment during infancy.14–16
Congenital infections
Toxoplasmosis
Toxoplasmosis is a parasitic infection known to cause birth defects with characteristic chorioretinal scarring. OCT images can demonstrate activity by detecting subretinal fluid with preservation of the retinal pigment epithelium (RPE), high reflection in the area of retinitis, and shadowing of the choroid. 17 These findings are helpful in the diagnosis and monitoring of subtle recurrence of ocular toxoplasmosis, particularly in judging the severity of ocular infection. More commonly, recurrence is associated with vitritis which can be detected clinically without the need for OCT. 18
Zika virus
A more recently recognized congenital infection, Zika virus, gained interest across multiple disciplines of medicine due to its role in causing significant birth defects in pregnant women who have not been previously exposed to the infection. 19 Chorioretinal scarring in Zika virus overlaps with toxoplasmosis, with one distinction being that RPE changes in toxoplasmosis are usually confined to the lesion and in Zika virus can be seen outside of chorioretinal scarring. 20 One study analyzed OCT images of infants with clinically confirmed congenital Zika syndrome (CZS) and found discontinuation of the ellipsoid zone and hyperreflectivity underlying the RPE in 100% of patients. 21 Slightly less common findings were retinal thinning (89%) and choroidal thinning (78%). Another study found similar OCT findings in eight children with CZS and eight children with cobalamin deficiency, and the authors speculated that the pathogenesis of both diseases might be similar. 22
Rubella
Rubella is another congenital infection with known OCT findings. One prospective study analyzed OCT images of 13 children (24 eyes) with diagnosed congenital rubella syndrome and found these patients were significantly more likely to have U-shaped foveal pits (61.1%), an absent or stippled inner segment–outer segment junction (70%), shaded outer segment–RPE junction (100%), and absent RPE (85%). 23 A multimodal case report found similar outer retinal changes associated with congenital rubella. 24
Inherited retinal dystrophies and diseases
OCT has been routinely used to diagnose and follow inherited retinal dystrophies. As there are many types of retinal dystrophies, here we focus on a select few with particularly salient OCT findings.
Retinitis pigmentosa
Retinitis pigmentosa (RP) is one of the most common retinal dystrophies, comprising a group of heterogeneous disorders leading to the progressive degeneration of retinal photoreceptors and typically presenting with peripheral vision loss and difficulty seeing at night. 25 Without a known family history, RP can be difficult to diagnose early on. Classic clinical findings include RPE mottling, optic nerve pallor, and attenuated vasculature, although most patients do not manifest these early on. Electroretinography (ERG) is useful to diagnose and characterize the disease, which may require examination under anesthesia for patients below 7 years old. Therefore, OCT offers a more practical way to monitor disease in these patients, as it can often be performed without an examination under anesthesia for patients above 3 years of age. One study found the ratio between foveal outer segment pigment epithelial thickness and photoreceptor outer segment length, measured with OCT, could be used as a surrogate to monitor central retina disease in RP, which is useful to predict visual outcomes in these patients. 26 OCT is also used to monitor more obvious structural changes in the retina such as outer nuclear layer (ONL) thinning and thinning of the outer segment, with no associated changes in the inner nuclear layer or ganglion cell layer of the inner retina.22,27,28 In addition, OCT is used to detect and monitor cystoid macular edema (CME) associated with RP, which can be treated with carbonic anhydrase eye drops with variable outcomes (see Figure 2 for example). 29
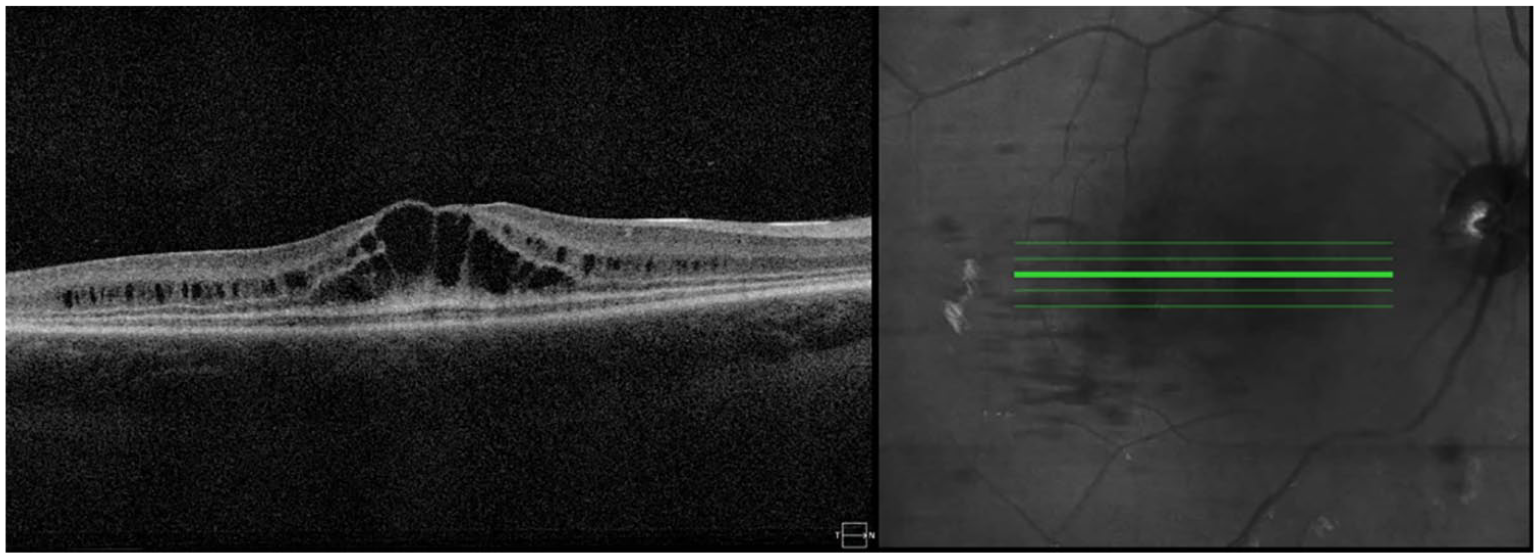
OCT image of a 9-year-old female diagnosed with retinitis pigmentosa. The image demonstrates significant cystoid macular edema in the outer plexiform layer.
Stargardt disease
Stargardt disease is a recessively inherited macular dystrophy that leads to progressive central loss of vision. The underlying cause is the accumulation of lipofuscin in the RPE, which is toxic to photoreceptors. 30 Classic clinical findings include pisciform yellow flecks, pigment mottling, and a dark choroid on fluorescein angiogram, although these are not always evident in early stages of the disease and may be missed upon initial clinical presentation. 31 OCT offers an additional clinical test to analyze the ultrastructure of the retina and is particularly useful in detecting lipofuscin accumulation and changes in the photoreceptor layer in early disease (see Figure 3 for an example). 32 A recent study found that fundus autofluorescence underestimated the choriocapillaris layer loss compared to OCT, suggesting that RPE damage may occur before the choriocapillaris (CC) layer and demonstrating how OCT might be used to monitor early disease. 33 Typical OCT findings in late Stargardt disease include bull’s-eye atrophy, loss of inner and outer photoreceptor segment layers, and variable thinning of other retinal layers. Thus, OCT provides useful information in the diagnosis and staging of Stargardt disease and has also provided fundamental insights into the pathophysiology of the disease.

OCT image of a 62-year-old male with confirmed Stargardt disease and preserved central vision. The image demonstrates bull’s-eye maculopathy with widespread lipofuscin accumulation in the RPE (red arrows).
Best disease
Best vitelliform macular dystrophy (BVMD) is one of the most common macular dystrophies in children, caused by an autosomal dominant mutation in the BEST1 gene. 34 Classically, the disease causes a yellow ‘egg yolk’ appearance in the macula with RPE involvement early on and often progresses to retinal and RPE atrophy. Although visual prognosis is typically good, the condition can lead to significant vision loss due to atrophy or choroidal neovascularization (CNV). OCT is used in the diagnosis and staging of Best disease, with typical findings demonstrating outer retinal layer disruption and the presence of vitelliform material underneath the retina (see Figure 4 for OCT images of the stages of BVMD), 35 although one paper suggests the separation of the outer retinal layers occurs between the RPE and Bruch’s membrane. 36 OCT can also detect the central RPE atrophy and CNV that are associated with later clinical stages of the disease. 37 Some studies have demonstrated benefits using anti-VEGF therapies to treat CNV in BVMD, which can also be monitored for treatment response with OCT. 38 In addition, OCT can detect macular holes, which can result from late stages of BVMD.35,39

Characteristic imaging of the stages of Best vitelliform macular dystrophy. From left to right are fundus photographs, blue-light fundus autofluorescence, and OCT images. From top to bottom are subclinical (a, stage I), vitelliform (b, stage II), pseudohypopyon (c, stage III), vitelliruptive (d, stage IV), and macular atrophy (e, stage V) stages. Beginning in stage II, OCT demonstrates vitelliform material (yellow arrow), with a focal area of underlying subretinal fluid (red arrow). Stages III and IV demonstrate continued subretinal fluid and the presence of hyperreflective material, with advanced disease (stage V) demonstrating macular atrophy and the loss of outer retinal layers.
Coats disease
Coats disease is an idiopathic eye disease that is found almost exclusively in males, with findings of unilateral, peripheral telangiectatic vessels. 40 OCT findings include epiretinal membrane (ERM), vitreomacular traction, CME, subretinal/intraretinal fluid or exudate, central macular thickness, and subfoveal choroidal thickness (see Figure 5 for an example of characteristic OCT findings of Coats disease). 41 Ong and colleagues 42 have used OCT to demonstrate that retinal exudates and CME appear later in the disease course, often resulting in fibrosis and poor visual outcomes. A separate study also found that compact hyperreflective structures corresponding to fibrosis and outer retinal atrophy were associated with poor post-treatment visual acuity (VA), whereas bright hyperreflective areas corresponding to exudates and foveal subretinal thickness were associated with improved post-treatment VA. 43 Recently, OCT-A has also been used clinically in diagnosing and monitoring Coats disease. OCT-A was found to be equally sensitive in detecting medium-to-large vascular abnormalities compared to fluorescein angiography (FA) in a series of nine patients but could not detect small telangiectasias seen in FA. 44 OCT and OCT-A have also been used to analyze and monitor treatment in Coats disease, such as one study which found anti-VEGF injections combined with laser therapy led to telangiectasia regression and exudate resolution. 45 A limitation of OCT-A in Coats disease is that field of view is confined to the macula and vascular pathology requiring treatment is located in the periphery.

OCT image of an 11-year-old male with Coats disease with history of cryotherapy, laser therapy, and multiple anti-VEGF treatments. The image demonstrates subfoveal fibrosis and scar.
Ocular albinism
Ocular albinism (OA) stems from various genetic mutations in the tyrosine/melanin biochemical cascade and leads to permanent pigmentary deficiencies and vision loss in children. 46 OCT can be used to demonstrate characteristic foveal hypoplasia resulting from abnormal RPE development, which resembles the immature retina in ROP.46,47 In addition to the loss of a foveal pit, high-reflectivity bands across the fovea, presence of inner retinal layers typically absent at the foveal center, and absence of the typically thickened photoreceptor foveal nuclear layer have been described. 48 These OCT findings can help rule out other differential diagnoses and be used to evaluate future visual acuity as this mostly depends on the foveal hypoplasia which does not appear to improve with time. 49 Nystagmus, commonly associated with OA, may limit the ability to acquire OCT images.
Neurocutaneous syndromes
Neurocutaneous syndromes, also known as phakomatoses, are a group of disorders that have multiple systemic effects of the nervous, skeletal, and integumentary systems and also have characteristic ocular findings. 50 OCT is useful in detecting several unique findings of neurofibromatosis type 1 (NF1), a neurocutaneous syndrome, including monitoring optic nerve gliomas (OPGs) and choroidal abnormalities. Several studies have found that NF1 patients with OPGs demonstrate retinal nerve fiber layer (RNFL) and macular thinning on OCT.51,52 Authors have suggested these findings might be used as a screening tool for children with NF1 to reduce the need for magnetic resonance images (MRIs) in these young patients or could be used to monitor the progression of existing OPGs. Viola and colleagues 53 describe choroidal abnormalities, such as choroidal nodules and choroidal nevi, in NF1, and the authors suggest these findings could be used as an additional criterion for NF1 diagnosis. Neurofibromatosis type 2 (NF2), a related but less common condition, has almost pathognomonic OCT findings of scrolled or flame-shaped ERMs and retinal hamartomas. 54 Scrolled ERMs on OCT have been described before systemic findings in NF2, and children presenting with these eye findings on OCT should have neuroimaging performed (see Figure 6). 55

OCT of an 11-year-old male with mosaic neurofibromatosis 2, confirmed with genetic testing. The OCT image demonstrates scrolled or flame-shaped epiretinal membrane with a retinal astrocytic hamartoma in the inner retina.
Tuberous sclerosis
Tuberous sclerosis complex (TSC) is a separate neurocutaneous syndrome and is mostly associated with retinal astrocytic hamartoma (RAH), occurring in 40–50% of TSC patients. 56 TSC with RAH has been characterized using OCT and demonstrates an initially flat lesion in the RNFL that transforms into a full-thickness nodular mass, sometimes with calcified areas. 57 A separate study used OCT to analyze RAHs of 86 eyes in 43 patients with confirmed TSC and found four types of lesions which might correlate with systemic manifestations of TSC. 58 For instance, type III lesions, which demonstrate a ‘gently sloping, slightly irregular surface’ with an ‘optically dense, disorganized mass replacing the retinal architecture’ and classic mulberry-like calcifications, were associated with subependymal giant cell astrocytoma. The study found decreasing prevalence with lesion severity, with type I lesions being most common (47%) and type IV least common (14%). Another study used the same classification system and was able to identify RAHs not found on fundoscopy using OCT. 59 The authors suggest that the detailed information provided by OCT might be used to diagnose and stage RAHs, including those not found with other imaging modalities, but also as a proxy marker of treatment response for chemotherapy in these tumors. However, they advocate for the use of OCT in addition to infrared imaging to elucidate the most clinically relevant information. Thus, it may be important for TSC patients to have regular follow-up with OCT imaging to follow both ocular and systemic manifestations of their condition.
Incontinentia pigmenti
Incontinentia pimenti (IP) is an X-linked neurocutaneous disorder with diverse ophthalmic manifestations, including progressive peripheral retinal hemorrhage and neovascularization, macular and peripheral tischemia, and retinal detachment. 60 Although many of these peripheral findings have been described using FA and fundoscopy, the use of OCT has become more common in monitoring and diagnosing the condition because it can show inner retinal thinning and outer plexiform layer irregularities. 61 In addition, one study has demonstrated retinal thickening due to a dense ERM, accompanied by vitreomacular traction in the area of retinal thickening. 62 However, these studies failed to establish RPE abnormalities, and the authors suggest that the neuronal retinal changes in IP are the result of vascular defects rather than an abnormality in the RPE, challenging previously proposed mechanisms.62,63 In addition to these many vascular abnormalities in IP, OCT-A has been used to demonstrate decreased vascular density and areas of decreased blood flow in superficial and deep retinal plexuses. 61 However, flow was not obstructed in the choriocapillaris or choroid layer. In addition, OCT has been used with FA to stratify pediatric IP patients into five disease stages, allowing for better disease monitoring and possibly improved prognosis determination. 64 Future work might use integrated OCT and OCT-A to similarly stage IP disease to evaluate risk of disease progression.
Sturge–Weber syndrome
Sturge–Weber syndrome (SWS), or encephalotrigeminal angiomatosis, is another neurocutaneous disorder that has significant ocular manifestations. Unlike many of the other phakomatoses, SWS occurs sporadically (i.e. not genetically inherited) and is classically associated with hemangiomas and a significantly increased risk of glaucoma (prevalence 30–70%). 65 Less common manifestations include retinal tortuosity and retinal detachment. 66 Several articles have described SWS OCT findings of diffuse choroidal hemangiomas with increased choroidal thickness (see Figure 7).67,68 In addition, focal defects in the RPE were described, as well as RNFL thinning. A separate study also found increased choroidal thickness and noted unilateral thickening in affected eyes for patients without bilateral manifestations. 69 OCT has been used to monitor choroidal thickness in SWS after photodynamic therapy (PDT) and showed a choroidal thickness reduction of 251 to 83 µm during follow-up. 70 Notably, the choroidal hemangiomas and increased choroidal thickness in SWS are difficult to treat and do not typically respond to conventional therapy, placing patients at increased risk of complications including retinal hemorrhage, retinal detachment, and choroidal effusion. Thus, other therapies such as PDT, anti-VEGF, and radiotherapy are commonly used in treatment, and OCT offers a noninvasive approach to monitor the response to these treatments in SWS.
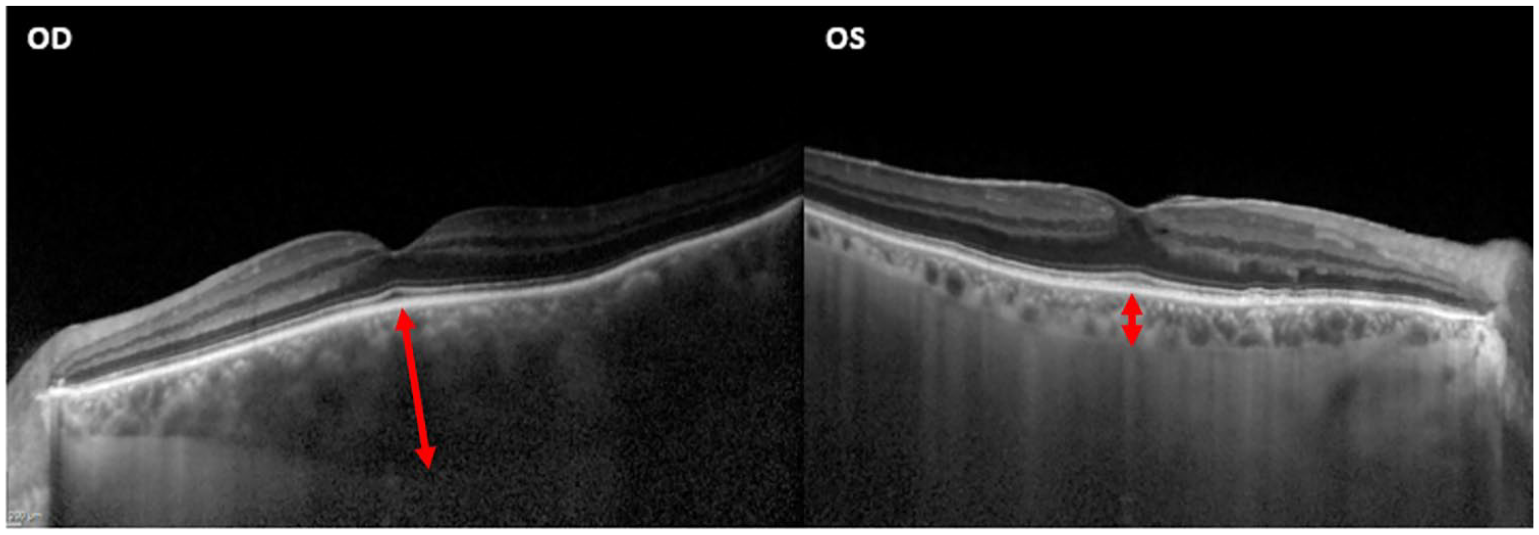
OCT image of a patient with Sturge–Weber syndrome with unilateral diffuse choroidal hemangioma. The images demonstrate a choroidal hemangioma with choroidal thickening of the right eye (700 µm) with normal choroid of the left eye (200 µm). Red arrows demonstrate the increased choroidal thickness OD.
Von Hippel–Lindau
Von Hippel–Lindau (VHL) disease represents a phakomatosis with minimal cutaneous findings, although other systemic effects are common, including hemangioblastomas of the retina and brain. Like other retinal tumors, ophthalmoscopy can be used to detect hemangioblastomas in VHL provided they are large enough. OCT has been used to help characterize tumors in VHL and has also demonstrated macular thickening with peripheral or optic nerve hemangioblastomas. 71 Initial presentation of hemangioblastomas in VHL can be subtle, sometimes stemming from small capillary abnormalities, which might be more easily detected using OCT. 72 Thus, OCT serves as a clinical tool to detect tumors earlier than traditional methods. In addition, OCT can be used to detect retinal edema and retinal exudates, which require treatment to hopefully prevent exudative retinal detachment. 73
Retinoblastoma
Retinoblastoma is the most common pediatric ocular tumor and represents 6.1% of all pediatrics cancers under 5 years of age. 74 Most patients with active retinoblastoma are followed by examination under anesthesia for a more complete evaluation and immediate opportunity to treat. Rootman and colleagues 75 reported on the use of handheld OCT to characterize 19 retinoblastoma lesions in 16 patients, of which 5 lesions were small. They highlighted the utility of OCT in detecting small lesions which may be missed with clinical examination alone. 75 Cao and colleagues 76 described three children (7–33 months old) with retinoblastoma located in the macula treated with (systemic versus intra-arterial) chemotherapy where OCT showed tumor regression and resolution of subretinal fluid. Berry and colleagues 77 also described a case report of early detection of retinoblastoma in the fellow eye of a 7-week-old patient with group E retinoblastoma in the primary eye (see Figure 8). Therefore, OCT offers an imaging modality for both early detection of these tumors and treatment response.

OCT images of a 17-week-old male with a germline retinoblastoma mutation. The images (a)–(f) move from the superior aspect of a tumor toward the margin of the optic nerve. They demonstrate the presence of a small retinal tumor not detected by indirect fundoscopy, which exhibits involvement of the outer nuclear layer, outer plexiform layer, and inner nuclear layer, with the OPL and INL draping over the edges of the tumor.
Amblyopia
Amblyopia is one of the most common and preventable causes of permanent visual impairment in children, with the most common types being strabismus and refractive error. Bitirgen and colleagues 78 found that children with both strabismus and hyperopic amblyopia had significantly increased peripapillary choroidal thickness on OCT compared to controls even after controlling for refractive error. Multiple papers have described an increased central macular thickness in amblyopic patients that correlated with the severity of amblyopia.79–81 Authors have proposed that the lack of normal postnatal changes in amblyopia could affect macular maturation, which normally involves centripetal migration of the inner retina leading to the increased overall foveal thickness found in many studies. 80 Yen and colleagues 82 have shown that amblyopia may also lead to increased RNFL thickness, hypothesized to be due to a reduction in normal postnatal ganglion cell apoptosis due to a lack of sharp focus required for normal development. In addition, Borrelli and colleagues 83 recently used OCT-A to describe novel vascular findings in amblyopic children including increased choriocapillaris vessel density and outer parafoveal macular thickness. Although OCT and OCT-A findings in amblyopia are subtle and relatively new, they may be used to understand the mechanism behind visual impairment in these patients and with further research may help shed light on how to use the technology in clinical practice.
Conclusion
In conclusion, OCT has become an integral part of diagnosing and managing pediatric retinal diseases. Children as young as 3 years old can be imaged with a desktop device, and handheld devices can be used in infancy and under anesthesia. OCT has value in both macular diseases such as Best disease and Stargardt disease and primarily peripheral diseases such as ROP and Coats disease.
Footnotes
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by an unrestricted grant from Research to Preventing Blindness given to the Stein Eye Institute.
