Abstract
Background
Monoclonal antibodies targeting the calcitonin gene-related peptide (CGRP) pathway represent a paradigm shift in migraine prevention, with generally favorable safety profiles. However, CGRP's widespread distribution and immunoregulatory functions raise theoretical concerns about potential off-target effects.
Case presentation
A 41-year-old woman with chronic migraine developed interstitial pneumonia seven weeks after initiating galcanezumab therapy. Extensive diagnostic workup excluded infectious, autoimmune, and implant-related causes. Bronchoalveolar lavage revealed marked lymphocytosis (73%) with inverted CD4/CD8 ratio. Complete radiological resolution occurred three months after drug discontinuation.
Discussion
To our knowledge this is the first documented case of interstitial lung disease associated with galcanezumab therapy. Despite methodological limitations, the temporal relationship, the distinctive immunological pattern, and resolution after cessation suggest a causal association. This finding emphasizes the need for vigilance regarding rare but potentially serious respiratory adverse events with anti-CGRP therapies.
This is a visual representation of the abstract.
Introduction
Migraine stands out as one of the most debilitating medical conditions, representing a major global health challenge due to its increasing burden on societies and healthcare systems. 1 Therapies targeting the calcitonin gene-related peptide (CGRP) pathway marked a paradigm shift in migraine prevention,2,3 demonstrating robust efficacy and favorable safety profiles in clinical trials and real-world studies. 4
Beyond its role as a central neuromodulator, 5 CGRP emerged as an archetypical neuroimmune connector involved in host surveillance and immune modulation, 6 exerting broad immune-modulatory effects through multiple cellular mechanisms involving dendritic cells, macrophages, and T-cell differentiation. 7 Pharmacological CGRP blockade could theoretically disrupt this immunological balance, potentially shifting it toward proinflammatory responses. While respiratory adverse events have not been specifically prominent in anti-CGRP safety analyses, 8 emerging case reports documented various inflammatory complications across multiple organ systems, suggesting potential for disrupted CGRP-mediated immune regulation.9–11
We report the first case of interstitial pneumonia temporally associated with galcanezumab treatment. Following comprehensive exclusion of alternative etiologies, we discuss the potential implications of CGRP blockade in pulmonary immune homeostasis.
Case presentation
A 41-year-old female textile expert with tertiary education presented with long-standing migraine. She had no history of tobacco use, pulmonary disease, or autoimmune conditions. Her medical history was otherwise unremarkable, except for bilateral breast augmentation in 2015 and two uncomplicated pregnancies.
Clinical presentation fulfilled International Classification of Headache Disorders (ICHD-3) criteria for chronic migraine and medication overuse headache. 12 Attacks consisted of severe frontotemporal pulsating pain (average intensity 8/10 on a numerical rating scale) associated with nausea, vomiting, phonophobia, photophobia, ictal allodynia, and need to rest. Previous prophylactic therapies included topiramate (100 mg/day), pizotifen (0.5 mg/day), flunarizine (5 mg/day), and amitriptyline (12 mg/day). All prophylactic medications were discontinued due to lack of efficacy except for amitriptyline, which was discontinued due to significant daytime somnolence. Among traditional preventive therapies, beta-blockers were contraindicated due to low blood pressure levels. She reported 18 monthly migraine days with severe functional disability (Headache Impact Test-6: 62; Migraine Disability Assessment Scale: 114). This study is reported in accordance with the CARE guidelines for case reports.
On 5 March 2024, according to local regulatory guidelines, treatment with galcanezumab was initiated with a loading dose of 240 mg administered subcutaneously, followed by monthly maintenance injections of 120 mg. Within two months, the patient achieved complete migraine remission and marked improvement in overall well-being. However, approximately seven weeks after initiating galcanezumab therapy (late April 2024), she developed non-productive dry cough and localized right sub-mammary chest pain without fever or dyspnea. Empiric antibiotics, including amoxicillin-clavulanate (875 + 125 mg twice daily for seven days) and azithromycin (500 mg once daily for six days), and a tapering course of methylprednisolone starting at 16 mg daily, provided no clinical improvement. Initial chest radiograph (mid-June 2024, eight weeks post-symptom onset) was normal. Due to persistent symptoms, emergency evaluation revealed normal vital signs and laboratory results. Chest computed tomography (CT) showed subtle ground-glass opacities in the antero-basal region of the right lower lobe (Figure 1(A)). Transthoracic ultrasonography identified stress fractures of the 10th and 7th ribs, attributed to persistent coughing without trauma history. Rheumatological and pneumological assessment in late June 2024 raised suspicion of an interstitial process. Galcanezumab was discontinued by mutual physician–patient agreement. One week later, bronchoscopy with bronchoalveolar lavage (BAL) was performed. Comprehensive microbiological testing was negative for all pathogens including bacteria, mycobacteria, fungi, extensive viral panel, and atypical organisms. Cytological analysis revealed marked lymphocytosis (72.9% of total cells) with T-lymphocyte predominance (CD3 + 79.9%). Immunophenotyping demonstrated striking CD8 + predominance (36.6% of lymphocytes) over CD4 + cells (9.5% of lymphocytes), yielding an inverted CD4/CD8 ratio of 0.6 (reference range: 1.5–2.5) (Table 1). Cytopathology showed alveolar macrophages, ciliated epithelial cells, numerous lymphocytes, neutrophils, and eosinophils. Extensive serological and autoimmune workup were unremarkable, including complete blood count, inflammatory markers, immunoglobulins, and comprehensive autoimmune panels (ANA, ENA, anti-dsDNA, rheumatoid factor, anti-CCP, ANCA, myositis-specific, and scleroderma-associated antibodies). Infectious disease screening (HIV, hepatitis B and C, EBV) was negative. Follow-up rheumatological examination in August 2024 revealed no evidence of systemic autoimmune disease. Breast MRI with contrast in September 2024 demonstrated intact bilateral implants without rupture or inflammatory changes, thus excluding implant-related pathology. Progressive symptomatic improvement began within one month of galcanezumab discontinuation. Follow-up chest CT three months after discontinuation demonstrated complete resolution of ground-glass opacities (Figure 1(B)). The complete timeline of clinical events is summarized in Table 2.
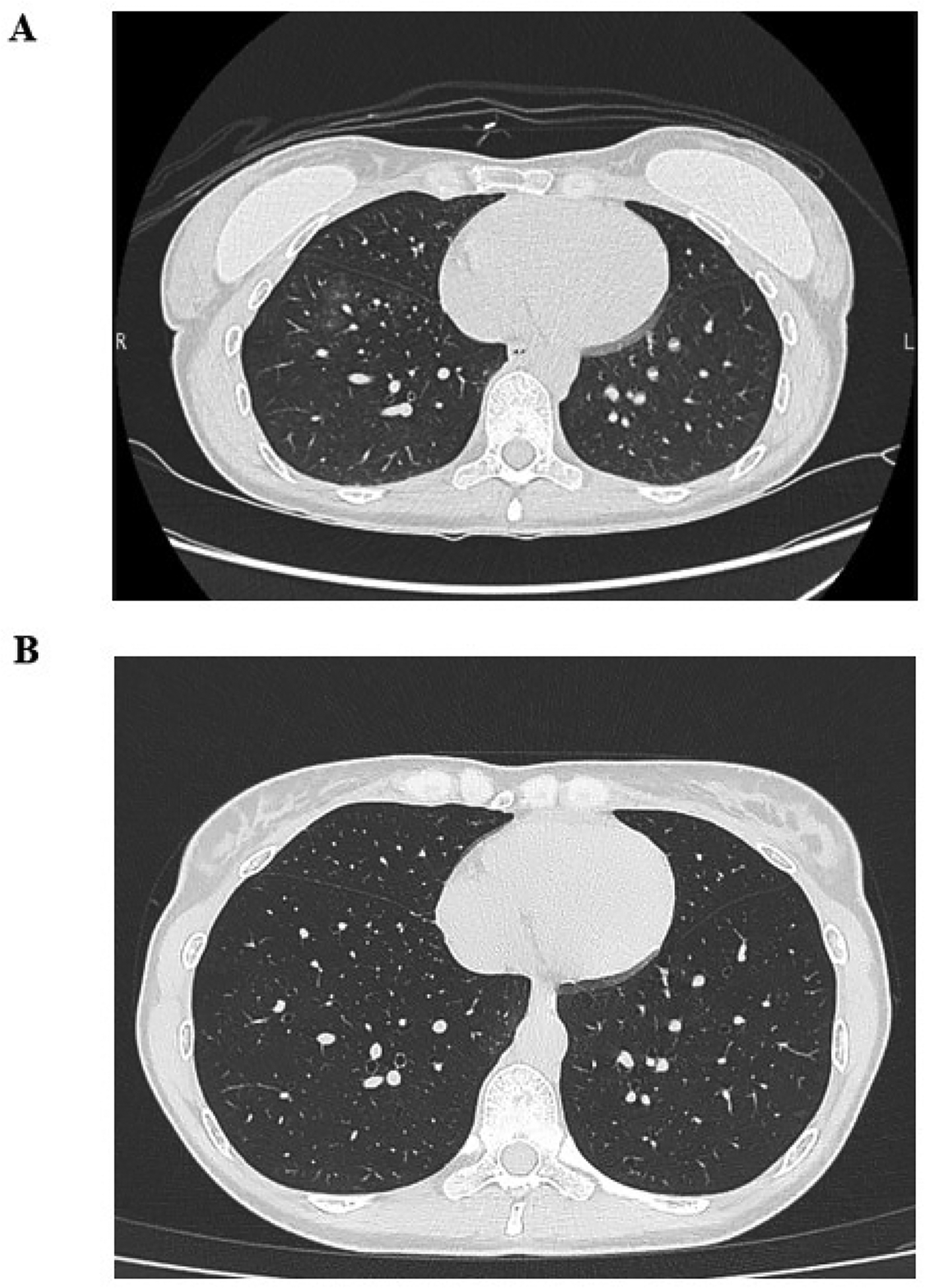
Radiological evolution following galcanezumab discontinuation. High-resolution chest CT images at the level of the lower lobes. (A) Initial scan performed eight weeks after symptom onset, demonstrating subtle ground-glass opacities in the antero-basal region of the right lower lobe. (B) Follow-up imaging at three months demonstrating complete radiological resolution with normalized lung parenchyma.
Bronchoalveolar lavage cellular analysis and lymphocyte immunophenotyping.

Timeline of clinical events.

BAL: bronchoalveolar lavage; CT: computed tomography; MRI: magnetic resonance imaging; SC: subcutaneous.
Based on comprehensive clinical-radiological correlation, temporal relationship, systematic exclusion of alternatives, characteristic BAL immunophenotype, and favorable outcome after drug withdrawal, a diagnosis of probable galcanezumab-associated interstitial lung disease was made.
Discussion
This case describes a temporal association between galcanezumab administration and interstitial pneumonia in a patient with chronic migraine. Diagnosis of drug-induced interstitial lung disease (DI-ILD) relies on temporal association, exclusion of alternatives, characteristic findings, and response to drug withdrawal. 13 The temporal relationship is compelling, with symptoms emerging seven weeks post-initiation, consistent with drug-induced immune sensitization (typical latency: 2–12 weeks). 13 Extensive investigations excluded alternative etiologies. Specifically, exhaustive microbiological testing ruled out infectious causes, comprehensive autoimmune panels argued against underlying autoimmune disease, and breast MRI excluded implant-related pathology. Rib stress fractures were secondary to chronic coughing rather than causative.
The BAL immunophenotype provides crucial diagnostic evidence. Lymphocytosis exceeding 70% with an inverted CD4/CD8 ratio of 0.6 (CD8 + predominance) is highly characteristic of hypersensitivity pneumonitis and DI-ILD. 14 The striking contrast between marked alveolar lymphocytosis and normal serum inflammatory markers underscores the compartmentalized nature of this immune response. Moreover, this pattern, reflecting CD8 + cytotoxic T-lymphocyte expansion, suggests a cell-mediated immune response against pulmonary interstitial antigens, supporting a drug-induced mechanism rather than infectious or sterile inflammatory processes. 15
Clinical course following discontinuation further supports causality. Improvement emerged exclusively following galcanezumab cessation, with progressive symptomatic resolution within one month and complete radiological clearance at three months. This favorable trajectory occurred without intensive immunosuppression beyond empirically initiated methylprednisolone. Notably, this contrasted sharply with the absence of response during prior antibiotic and corticosteroid trials, a pattern consistent with drug-induced pulmonary reactions. Biological plausibility derives from CGRP's established role as a neuroimmune mediator. Beyond its sensory function, the peptide exerts broad immunoregulatory effects on dendritic cells and macrophages, which are critical for maintaining pulmonary immune homeostasis.6,16,17 Consequently, pharmacological blockade of CGRP signaling might compromise this delicate equilibrium, potentially enhancing inflammasome activation and tipping the balance toward pro-inflammatory responses in susceptible individuals. Supporting this mechanistic framework, recent case series have documented various inflammatory complications following anti-CGRP therapy across multiple organ systems, including autoimmune hepatitis, Susac's syndrome, drug reaction with eosinophilia and systemic symptoms, granulomatosis with polyangiitis, psoriasis exacerbations, 11 suggesting that CGRP blockade may precipitate immune-mediated conditions through disruption of immunoregulation. Although large-scale pharmacovigilance analyses have not identified prominent respiratory safety signals, these emerging reports warrant consideration of rare, organ-specific reactions.8,18 Whether such reactions represent a galcanezumab-specific phenomenon, a potential class effect of anti-CGRP monoclonal antibodies, or idiosyncratic responses remain undetermined. Nevertheless, the occurrence of immune-mediated events across different anti-CGRP agents suggests that a class effect cannot be ruled out.
Several limitations warrant consideration. The monolateral focal distribution (right lower lobe) is atypical for a systemic immunopathogenic mechanism. Possible explanations include regional heterogeneity in CGRP distribution, as CGRP-containing nerve fibers and pulmonary neuroendocrine cells are non-homogeneously distributed throughout the respiratory tract, 19 a two-hit hypothesis requiring a local secondary trigger, or early-phase imaging of potentially progressive disease arrested by timely discontinuation. A confounding factor arises from empiric corticosteroid and antibiotic administration preceding diagnostic workup. Namely, methylprednisolone and multiple antibiotics create interpretive difficulty. Although the temporal sequence suggests that improvement followed galcanezumab cessation, distinguishing between these potential contributions remains challenging. Additionally, delayed imaging (eight weeks after symptom onset) prevented assessment of initial disease extent, and absence of baseline CT precluded detection of pre-existing abnormalities. Absence of histopathological confirmation through lung biopsy represents another important limitation.
The insidious presentation with non-productive cough and localized pain, initially attributed to bronchitis and then to rib stress fractures, highlights that galcanezumab-induced pulmonary reactions may present subtly. Normal chest radiograph provided false reassurance, with ground-glass opacities apparent only on CT. This underscores the importance of maintaining suspicion for pulmonary complications in patients receiving anti-CGRP therapies who develop respiratory symptoms, pursuing chest CT when radiography is unrevealing and symptoms persist, also considering bronchoscopy with BAL when interstitial lung disease is suspected.
Conclusion
To our knowledge, this represents the first case of reported galcanezumab therapy-associated interstitial pneumonia. Although the safety profile of anti-CGRP monoclonal antibodies is favorable, the rarity of the reported adverse event may explain its absence from previous studies, also considering the challenges in diagnosis and the potential for misattribution to more common respiratory conditions.
Clinical implications
Clinicians should be aware of potential respiratory complications with anti-CGRP monoclonal antibodies, particularly in patients who develop persistent respiratory symptoms.
Bronchoalveolar lavage with lymphocyte immunophenotyping can provide valuable diagnostic information when drug-induced interstitial lung disease is suspected in patients receiving anti-CGRP therapy.
Early recognition and drug discontinuation may be sufficient for resolution without intensive immunosuppression.
Footnotes
Consent to participate
Informed consent was obtained from the patient to utilize her medical records for the purpose of this report. All information has been appropriately anonymized.
Consent for publishing
Yes.
Author contributions
All authors critically reviewed the manuscript, agreed to be fully accountable for ensuring the integrity and accuracy of the work, and read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data needed to evaluate the conclusions are present in the paper. Additional data related to this paper may be requested from the corresponding author, upon reasonable request.
