Abstract
Objective
This study aims to demonstrate a real-world preventive response to the external combined occipital and trigeminal neurostimulation (eCOT-NS) device at baseline and at 3-month follow-up in patients treated for headache.
Methods
By using a prospective, observational, longitudinal approach for this study, we assessed the preventive treatment of headache in patients from a subspeciality clinic over 3 months. Participants with any headache disorder managed by the Lewis Headache Center who were prescribed eCOT-NS as part of standard of care were enrolled from July 2022 to June 2024. While using the eCOT-NS device, participants were assessed at baseline and at 3-month follow-up for moderate to severe headache days (MMD). Secondary measures included monthly headache days (MHD), average headache severity and Migraine Disability Assessment (MIDAS). Responder rate (in terms of reduction in MMD), subjective response, and adverse events were also reported.
Results
Seventeen participants completed the trial with mean age 50.9 years (range, 27–71 years) and 88% (15/17) female. At baseline, 94% (16/17) of participants had chronic migraine, and one participant (6%) had isolated occipital neuralgia. There was no statistically significant decrease (95% confidence interval) in MMD (P = .071) from a baseline median of 11.7 (3rd and 1st quartiles: 10, 28.3) to 6 (3rd and 1st quartiles: 3, 5) days at 3-month follow-up. Increased adherence was not associated with MMD, but was associated with more improved MHD (median β: 3.25, 95% CI: 0.689, 6.707). Of the secondary outcomes, only MHD were significantly reduced from a median of 30 (3rd and 1st quartiles: 27, 30) to 23 (3rd and 1st quartiles: 15, 23) [P-value .002]. The other outcomes did not show significant changes from baseline (P-values for Severity: 0.124, MIDAS: 0.277). Of the 17 participants, 47% (8/17) were ≥30% responders, 41% (7/17) were ≥50% responders, 23% (4/17) were ≥75% responders, and 11% (2/17) were 100% responders. Subjectively, 29% of participants (5/17) reported no benefit, 11% (2/17) reported inconsistent benefit, 41% (7/17) reported partial benefit, and 17% (3/17) reported complete benefit. No severe adverse outcomes occurred, but 23% of participants (4/17) reported worsening headache.
Conclusion
Use of the eCOT-NS device as a preventive treatment may reduce MHD, but larger confirmatory studies are needed. Therefore, if a patient can afford it, the eCOT-NS device could be tried preventively in patients with chronic migraine given its low side effect profile.
Trial Registration
None
This is a visual representation of the abstract.
Introduction
Migraine has many new exciting treatments, including non-pharmacologic options like neuromodulation devices, to help reduce the significant associated disability.1,2 The current neuromodulation devices for migraine include noninvasive transcutaneous supraorbital nerve stimulation, noninvasive vagal nerve stimulation, remote electrical neuromodulation, single pulse transmagnetic stimulation, and, most recently, the external combined occipital and trigeminal neurostimulation (eCOT-NS) devices. The eCOT-NS device, called Relivion™, received Breakthrough Device designation by the US Food and Drug Administration for acute treatment of migraine in February 2021. 3 This wearable at-home device provides stimulation through three adaptive channels to both the occipital and trigeminal neuralgia. 4 The device has embedded artificial intelligence and can be monitored through a smartphone app called Relivion MG Patient App, which includes a diary that tracks patient adherence.
A pivotal randomized, double-blinded, placebo-controlled study, RIME (NCT03631550), evaluated 131 patients with migraine for response to acute treatment using the eCOT-NS device. 5 Of the 50 patients given treatment, 46% (23/50) achieved 2-hour pain freedom and 60% (30/50) achieved 2-hour pain relief. Of the 59 patients given sham treatment, 12% (7/59) achieved 2-hour pain freedom and 37% (22/59) achieved 2-hour pain relief. No serious adverse events were reported. This device has not been studied for preventive treatment of migraine; however, similar devices, like the noninvasive transcutaneous supraorbital nerve stimulator, have been approved for preventive use for treating episodic migraine. 6 Neither device has been studied in chronic migraine or other headache disorders.
This prospective, observational, longitudinal study compares baseline response with a 3-month follow-up response for preventive treatment in patients with headache. The clinical indication for the device was kept open to understand its use across a range of headache conditions, including migraine. The study aims to demonstrate real-world responses to this device in clinical populations that experience frequent headache.
Methods
Methodology
This prospective, observational, longitudinal study observed the use of the eCOT-NS device for the preventive treatment of headache. The primary hypothesis is that using the eCOT-NS device will lead to a significant reduction in monthly moderate to severe headache days (MMD) among patients with migraine or other headache disorder after 3 months of preventive use. Participants were instructed to use the device nightly for 20 minutes. Participants were assessed at baseline for demographics, MMD, monthly headache days (MHD), average headache severity, Migraine Disability Assessment (MIDAS), depression using Patient Health questionnaire-9 (PHQ-9), anxiety using the Generalized Anxiety Disorder 7-item scale (GAD-7), and insomnia using the Epworth sleepiness scale (ESS). The participants were monitored over the first 3 months of device use then reassessed for any changes. Assessments were conducted based on patient recall and did not require a headache diary.
Inclusion and exclusion criteria
The inclusion criteria for this study required adults with a headache disorder, who were under the care of a provider at the Lewis Headache Clinic and were prescribed the eCOT-NS device as part of their standard treatment of care. These patients prescribed eCOT-NS were asked about interest in participation then screened by the study coordination for inclusion and exclusion criteria. The reasons for the decision to prescribe the eCOT-NS device was not tracked but as a standard in the Lewis Headache Clinic, the device is used as either an adjuvant treatment or as a non-pharmacologic treatment option. As standard practice, patients are provided with both preventive and acute treatment instructions. Participants were required to read, write, speak, and understand the English language unless a medical translator was consistently available to provide adequate interpretation. The exclusion criteria included pediatric patients, non-English speaking or reading patients without a medical translator, patients with cognitive impairment preventing their ability to respond reliably, and other medical comorbidities at the investigators’ discretion. Headache diagnoses were kept broad to allow for all headache conditions in the outcome analysis. Participants underwent written informed consent. Enrollment started July 2022 and closed June 2024. This data has not been previously published or presented.
Primary outcome
The primary outcome explored from baseline to the 3-month follow-up were changes in moderate to severe headache days (MMD), which in the setting of migraine is considered equivalent to monthly migraine days but due to the mixed headache population was defined more broadly. This treatment effect was based on modified intention-to-treatment analysis and, therefore, included all participants with completed 3-month data regardless of treatment adherence.
Secondary outcomes
The secondary outcomes were exploratory. Change in MHD, headache severity, MIDAS, PHQ-9, GAD-7, and ESS were assessed. Responders rate in terms of reduction in MMD of 30%, 50%, 75%, and 100% at 3-month follow-up were reported as well as participant's subjective opinion on the reliability of the treatment and its adverse effects.
Data acquisition
Before the eCOT-NS device was started, participants attended a baseline visit where they completed several forms including informed consent, demographics, headache history, and questionnaires such as the MIDAS, PHQ-9, GAD-7, and ESS. Demographics included categorization into chronic migraine versus episodic migraine according to the International Classification of Headache Disorders edition 3 (ICHD-3) as well as into resistant versus refractory migraine as defined by the European Headache Federation.7,8 For classification according to the EHF definitions, retrospective chart review was done to pull all prior medication trials according to the appropriate medication classifications noting that calcium channel blockers (flunarizine and cinnarizine) are not available in the United States, so memantine taken from the 2021 American Headache Society Consensus Statement was used as a category instead.8,9 Outcomes were reassessed at the 3-month follow-up, and all data was collected in a Redcap database. Completed data was exported to an Excel file for data analysis. Data analysis was conducted on patients with baseline and 3-month data regardless of treatment adherence.
Statistical analysis
Each variable of interest was evaluated via Shapiro–Wilk test for normality. Our study used mean values and standard deviation for continuous variables to present descriptive statistics of the final cohort on demographics, headache history, and patient questionnaires. For primary and secondary outcome variables, the median and interquartile ranges were used to describe the data. Given the limited sample size and heterogeneous sampling, we leveraged a two-tailed Wilcoxon signed-rank test to evaluate if there was a reduction in MMD from baseline to 3 months with an alpha value of .05. 10
For each continuous secondary outcome (i.e., MHD, severity, MIDAS, ESS, PHQ, and GAD), a two-tailed Wilcoxon signed-rank test was applied to evaluate if there was a symptom reduction from baseline to 3 months. A Bonferroni correction was applied to the estimated P-values control the family-wise error rate, setting the significance level at P < .008 (0.05/6).
To evaluate whether adherence level was associated with differential response to the eCOT-NS device, we calculated the change in MHD (ΔMHD) and in MMD (ΔMMD) from baseline to 3-month follow-up for each participant. Adherence was stratified into a high adherence group (most days and daily) and a low adherence group (infrequent and 1–2 times a week). We then compared the distribution of these change scores between adherence groups (high vs. low) using the Mann–Whitney U test. This test was only applied to MHD and MMD, as these were the primary headache frequency outcomes of interest. Other secondary measures (e.g., severity, MIDAS, PHQ-9) were not analyzed in this manner due to limited power and exploratory intent.
Headache diagnoses were kept broad for all headache conditions in the outcome analysis. Power analysis was not done because the endpoints were primarily exploratory. There was no missing data to account for.
MATLAB 2024B: Statistics and Machine Learning Toolbox and R were used in estimation of descriptive statistics and in statistical tests.11,12
Ethics
The study was reviewed and approved by the St Joseph's Hospital and Medical Center Institutional Review Board (23-500-224-00-10). Intramural funding was received.
Results
Enrollment
Of the 58 patients prescribed the eCOT-NS device and screened for enrollment, 21 were enrolled. Of the 37 patients not enrolled, 15 were lost to follow-up, 8 were unable to access the device due to cost and lack of insurance coverage, 4 decided against participation, 3 decided to start a different treatment, 3 started on the device before the baseline visit, 1 never received the device, 1 had an implanted cardiac monitor, 1 had a metal plate, and 1 had a speech impediment that impacted the patient's ability to participate. Of the 21 enrolled patients, 3 withdrew from the study, and 1 patient was lost to follow-up. Of the 3 withdrawals, 1 patient decided against continuing in the study after using the device 3 times, and 2 patients reported various adverse effects (worsened headache, vertigo, nausea, and disrupted sleep). Figure 1 demonstrates the recruitment process of the 17 participants who completed the trial (“Completers-only” data). Enrollment was stopped at 21 participants because multiple patients declined to participate due to the cost of the device and interim response rates assessed prior to formal analysis did not justify continued enrollment.
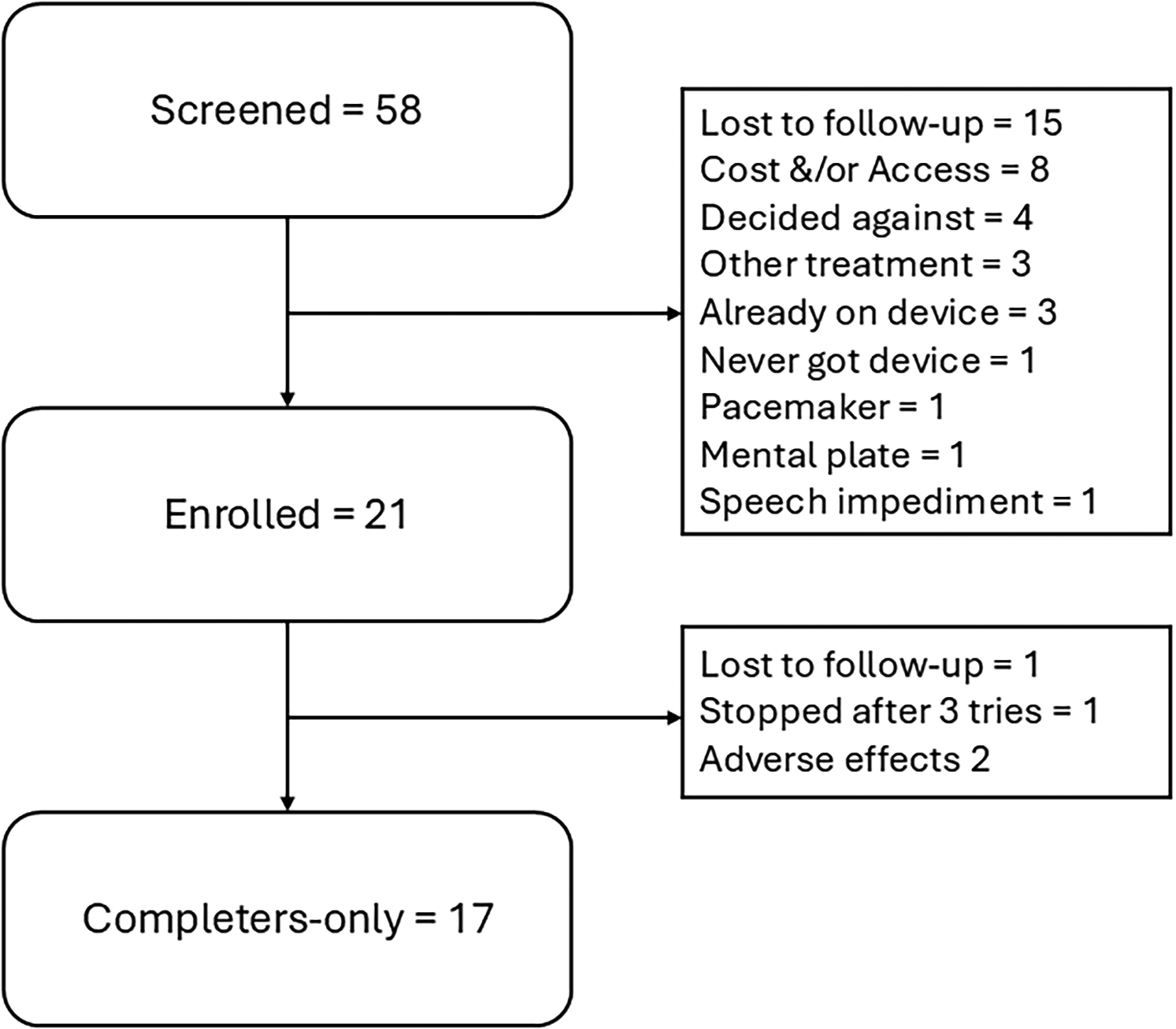
Flow diagram of participant progress through the study. This diagram depicts the progression of participants through each stage of our prospective observational study. It includes the number of individuals screened approached, those who were enrolled, and those who were included in the final analysis. Reasons for non-participation at each stage are also provided.
Completers-only demographics
The average age of the 17 completers-only participants with completed data sets was 50.9 years (range, 27–71), and 88% (15/17) female. Of these 17 participants, 94% (16/17) had a diagnosis of chronic migraine, but 23% of participants (4/17) reported a potential alternative diagnosis. One patient (6%) had chronic migraine and reported a possible diagnosis of paroxysmal hemicrania versus trigeminal neuralgia, and 18% (3/17) had new daily persistent headache presenting with migrainous features consistent with a chronic migraine phenotype. Eight participants (47%) reported migraine with aura, 18% (3/17) reported concurrent diagnoses of cervicogenic headache, 23% (4/17) reported occipital neuralgia, and 12% (2/17) reported medication overuse headache. Of the 16 patients with chronic migraine, 75% (12/16) met criteria for resistant migraine and 19% (3/16) met criteria for refractory migraine. One patient (6%) without chronic migraine reported isolated occipital neuralgia, and was prescribed the device as an alternative to pharmaceutical treatment or nerve blocks as a non-invasive occipital nerve stimulation option. There were also 35% (6/17) who reported occipital allodynia and 29% (5/17) who reported supraorbital allodynia at baseline. See Table 1 for patient demographics.
Demographics of completers-only study participants (n = 17).

Angiotensin pathways blockers included candesartan or lisinopril.
Antidepressants included amitriptyline and venlafaxine.
Anti-seizure drugs included topiramate and divalproex/valproate.
Beta blockers included propranolol, metoprolol, atenolol, nadolol, or timolol.
CGRP (calcitonin gene-related peptide)-directed medications included atogepant, erenumab, eptinezumab, fremanezumab, galcanezumab, or rimegepant every other day.
Of the 17 participants, 53% (9/17) had depression, 35% (6/17) had anxiety, 6% (1/17) had a stroke, 12% (2/17) had postural orthostatic tachycardia syndrome, 6% (1/17) had hypermobile Ehlers–Danlos syndrome, 12% (2/17) had chronic pain, 12% (2/17) had a history of concussion, 59% (10/17) had neck pain, and 12% (2/17) used opioids. Some of the participants had prior use of neuromodulation devices. Of the 17 participants, 18% (3/17) had used an external trigeminal nerve stimulator, 12% (2/17) had used noninvasive vagal nerve stimulation, 6% (1/17) had used remote electrical neuromodulation, 12% (2/17) had used single pulse transmagnetic stimulation, 6% (1/17) had used an implanted occipital nerve stimulator, and 6% (1/17) had used an implanted spinal cord stimulator. No participants had an active implanted device such as a deep brain stimulator or pacemaker.
The minority of participants (24% [4/17]) reported having tried an occipital nerve block, a trigeminal nerve block, or both. The 16 participants with chronic migraine were treatment resistant having tried an average of 5 (range, 2–7) migraine preventive categories. Specifically, 44% (7/16) tried an angiotensin pathway blocker (e.g., candesartan or lisinopril), 69% (11/16) had tried an antidepressant, 88% (14/16) tried an antiseizure drug (topiramate or divalproex/valproate), 81% (13/16) tried a beta-blocker (e.g., propranolol, metoprolol, nadolol, atenolol, or timolol), 81% (13/16) tried a calcitonin gene-related peptide monoclonal antibody or preventive gepant (e.g., atogepant, erenumab, eptinezumab, fremanezumab, galcanezumab, or rimegepant every other day), 44% (7/16) tried memantine, and 75% (12/16) tried onabotulinumtoxinA. The one patient with occipital neuralgia had only tried over-the-counter analgesia.
When asked about device adherence, only 8 participants reported using it daily, 4 participants reported using it most days, 4 participants reported using it 1–2 days per week, and 1 reported using it less than once a week. Two of the 17 participants (12%) reported using the device purely as a preventive treatment, and 88% (15/17) used it for a combination of preventive and acute treatment. None of the participants used it purely for acute treatment.
Primary outcome (completers-only data)
There was no statistically significant decrease in MMD from baseline to follow-up (estimated P-value .071) baseline median of 11.7 (3rd and 1st quartiles: 10, 28.3) to 6 (3rd and 1st quartiles: 3, 5) days at 3-month follow-up (Figure 2). The power of our statistical analysis for the primary outcome was 95% assuming an alpha value of .05. See Figure 2.

Change in MHD, MMD, headache severity, and MIDAS from baseline to 3 months (n = 17). MHD = monthly headache days, MMD = monthly moderate-to-severe headache days, MIDAS = migraine disability assessment.
Secondary outcomes (completers-only data)
Of the secondary outcomes, only monthly headache days had a significant reduction from a median of 30 to 23 (P-value: .002). See Figure 2. The other outcomes did not show significant changes from baseline (P-values for Severity: .124, MIDAS: 0.277, PHQ-9: 0.103, GAD-7: 0.939, ESS: 0.399) detailed in Table 3.
There were 12 participants with high adherence and 5 participants with low adherence. Increased adherence was not associated with the primary outcome of MMD (U: 30, P-value: .960), but improvement in MHD was more likely in those with high adherence (U: 10, P-value: .039). See Figure 3.
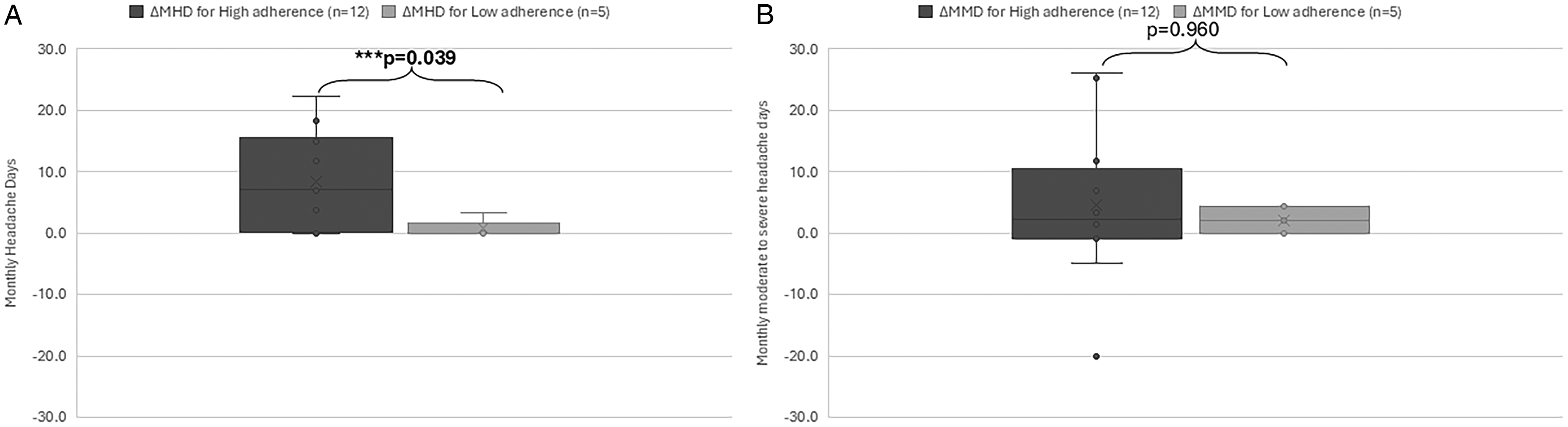
A. Box plot comparing the change in MHD for those with high versus low adherence. B. Boxplot comparing the change in MMD for those with high versus low adherence. MHD = monthly headache days, MMD = monthly moderate-to-severe headache days.
The response rates for the reduction in MMD from baseline were 47% (8/17) of participants at 30% response, 41% (7/17) at 50% response, 23% (4/17) at 75% response, and 12% (2/17) at 100% response.
When asked how beneficial the device was, 5 participants reported no benefit, 2 reported inconsistent benefits, 7 reported partial benefits, and 3 reported receiving complete benefit. The 3 participants who reported complete benefit also had an underlying diagnosis of chronic migraine. A patient with isolated occipital neuralgia reported experiencing partial benefits and liked the device but would not continue due to cost. The 3 patients with refractory migraine reported no benefit (2/3) or inconsistent benefit (1/3); however, the 12 patients with resistant migraine reported complete benefit in 3 and partial benefit in 6. Table 2 provides additional details on participant diagnoses and response.
Description of subjective patient response (completers-only, n = 17) for each diagnostic category.

Patient with isolated occipital neuralgia reported no response.
Median and IQR values for baseline and 3-month follow-up secondary outcomes for completers-only (n = 17).

IQR = interquartile range, MHD = monthly headache days, MIDAS = Migraine Disability Assessment, PHQ-9 = Patient Health Questionnaire, GAD-7 = Generalized Anxiety Disorder 7-item, ESS = Epworth sleepiness scale.
No serious adverse effects were reported. Four participants reported worsening headache with the device, including 1 patient who did not continue using the device after 1 month. Another patient reported that soaking the pads made her head cold. Note that two of the participants who withdrew reported adverse effects including worsened headache, vertigo, nausea, and disrupted sleep.
Discussion
The eCOT-NS device has not been studied for preventive use for migraine, but many neuromodulation devices for migraine are used acutely and preventively. 2 This study's objective was to assess preventive responses in a complex and treatment-resistant group of patients experiencing headache from a subspecialty headache clinic. Looking at the primary analysis of these data, MMD exhibited no statistically significant reduction from baseline to 3 months. The secondary analysis showed that MHD significantly improved from baseline to 3 months; however, the other secondary headache outcomes (severity and MIDAS) showed no significant change from baseline. The participants’ mood and sleep conditions also did not significantly change over the 3 months, though future study indications for the eCOT-NS include depression, anxiety, and insomnia. 13 Some participants reported having an excellent response and found the device very beneficial, but most reported no benefit, inconsistent benefit, or only partial benefit. Furthermore, 2 participants with chronic migraine did report a complete resolution of MMD despite having ongoing headache days, and almost half of participants reported at least a 30% responder rate. Treatment resistance did not prevent a response as 9 patients with resistant migraine reported a partial or complete response; however, none of the 3 patients with refractory migraine reported a partial or complete response. Overall, the eCOT-NS device was well-tolerated except for 4 participants who experienced worsening headache and an additional participant who withdrew early in the study due to a similar complaint.
This study has several limitations. First, it was a completers-only analysis, including only the 17 participants who completed both baseline and 3-month follow-up assessments. Four enrolled participants were excluded from analysis, including three who withdrew and one who was lost to follow-up. Notably, two of the three withdrawals cited adverse effects (e.g., worsened headache, vertigo, nausea, disrupted sleep), which may have biased the observed tolerability and safety profile. Excluding early discontinuations may also have overestimated treatment efficacy, particularly given the modest sample size. Second, this was not a randomized controlled trial, and the decision to use the device was influenced by participants’ willingness and ability to pay out of pocket, introducing potential selection bias. Third, all participants identified as white, and socioeconomic status was not assessed, limiting generalizability. Finally, although the sample was demographically homogeneous, the cohort was clinically heterogeneous, with a range of overlapping headache phenotypes and degrees of treatment resistance. While this may complicate interpretation of treatment effect, it also reflects the complexity of real-world patients seen in tertiary headache clinics. Due to the small sample size, we did not perform stratified analyses by headache subtype or comorbid diagnoses, although such subgroup analyses may yield important insights in larger cohorts. Additionally, most participants used the eCOT-NS device as an adjuvant rather than as a stand-alone preventive treatment, which introduces potential confounding by concurrent therapies. However, post hoc analysis did suggest that higher adherence to device use was associated with greater reduction in monthly headache days (MHD). Finally, participants were not required to maintain a headache diary, which may have introduced recall bias or imprecise reporting of headache frequency and severity.
An advantage to this study is that it was a prospective trial of the preventive response to the eCOT-NS device with space to be formally studied and reported. This study was also performed in a complex and treatment-resistant population that is not well-represented in migraine clinical trials. The majority of patients had tried at least one prior preventive treatment suggesting prior medication failures should not exclude a trial of the eCOT-NS as a preventive treatment.
Conclusion
Given the above findings, the eCOT-NS device could be used as a preventive treatment for chronic migraine in a complex patient population, but larger confirmatory studies are needed. Use of the eCOT-NS device as a preventive treatment is associated with reduced monthly headache days, but responses in this study were varied. The device may not be affordable, especially given the lack of insurance coverage limiting its usage. However, if a patient can afford it, the eCOT-NS device could be tried preventively in patients with chronic migraine, including those with resistant migraine, given its relatively low side effect profile though some patients did report worse headache in particular.
Clinical implications
Use of the eCOT-NS device as a preventive treatment may reduce monthly headache days, especially in those with high adherence.
Real world use of the eCOT-NS device is limited by device cost and limited coverage by insurance.
A single patient with isolated occipital neuralgia was included and reported no response to use of the eCOT-NS device.
The eCOT-NS has a low side effect profile, but a minority of patients reported worsening headache during use.
Given the low side effect profile, eCOT-NS could be considered as a preventive treatment in patients with chronic migraine if cost is not a significant barrier, but larger confirmatory studies are needed.
Footnotes
Abbreviations
Acknowledgments
We thank the staff of Neuroscience Publications at Barrow Neurological Institute for their assistance with manuscript preparation. We would also like to thank Dr Olivia Kingsford, Laurie Bell, Lauren Harvey, Nicholaus Scarfo, and Karrisa Secora for managing patient enrollment.
Author contributions
Robblee performed the literature search, analyzed the data, wrote the draft, and edited the final manuscript. Stancl enrolled the patients, collected the data, and entered the data. Hutson analyzed the data and edited the manuscript.
Consent to participate
Participants underwent written informed consent.
Consent for publishing
Yes.
Data availability statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Robblee discloses grant support from Barrow Neurological Foundation, investigator support from Eli Lilly and AbbVie, and paid Editorial relationships with MedLink Neurology and Neurodiem. Robblee has received personal compensation for serving on advisory boards for Allergan/AbbVie and Tonix and as a speaker for Impel. Robblee also discloses that a family member has partial ownership of Scottsdale Providence Recovery Center. Stancl has no disclosures.
Hutson discloses prior grant support from the Barrow Neurological Foundation, NIH STTR 1R41NS132627-01 and NIH R01NS129643.
Ethics approval
St Joseph's Hospital and Medical Center Institutional Review Board (23-500-224-00-10).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Barrow Neurological Foundation (grant number OSRA: 22-500-278-30-10; Activity: 455003033744).
Open practices
This study did not involve preregistration. No additional study materials or code are publicly available. Data are available from the corresponding author upon reasonable request.
