Abstract
Background
Post-craniotomy headache (PCH) is a common, often debilitating complication with limited treatment options and unclear pathophysiology. While botulinum toxin A (BoNT-A) is effective for various headache disorders, its use in PCH is underexplored. This systematic review examines case reports and series on BoNT-A's efficacy and safety for PCH.
Methods
A systematic search of PubMed, Scopus, and Web of Science was conducted in February 2025 using relevant keywords. Case reports and series on BoNT-A treatment for PCH were included, while unrelated studies, reviews, and incomplete abstracts were excluded. Data on patient characteristics, treatment protocols, efficacy, and adverse events were extracted.
Results
Five case series published up to 2025 report on 15 patients from France, Canada, and the United States. Each study enrolled only three or four patients, all with persistent PCH unresponsive to standard analgesics. BoNT-A regimens differed widely, ranging from 15 to 165 U, and included single versus repeated sessions, as well as injection sites such as the temporalis muscle, incision margins, and cranial suture lines, highlighting the lack of a standardized protocol. Eleven patients achieved 75–100% pain relief within 10–15 days, with therapeutic effects persisting for several weeks to over 5 years. Many also demonstrated improvements in daily functioning and a reduction in analgesic consumption. No serious adverse events were reported, supporting BoNT-A as a safe and promising treatment for PCH.
Conclusion
BoNT-A is a well-tolerated and effective option for patients with refractory PCH, offering substantial pain relief and functional improvement. However, given the reliance on small-scale studies, larger clinical trials are needed to confirm its efficacy and establish standardized treatment protocols.
This is a visual representation of the abstract.
Introduction
A craniotomy is a fundamental neurosurgical procedure that involves the temporary removal of a portion of the skull to access intracranial structures for the treatment of various conditions, including brain tumors, aneurysms, arteriovenous malformations, subdural empyemas, subdural hematomas, and intracerebral hematomas. 1 Despite advancements in surgical techniques, post-craniotomy headache (PCH) remains a prevalent complication, affecting up to 60 to 90% of patients and presenting notable diagnostic and therapeutic challenges.2–5 According to the International Classification of Headache Disorders, 3rd edition (ICHD-3), PCH is classified as “Acute headache attributable to craniotomy” and “Persistent headache attributable to craniotomy.” Acute PCH refers to a headache that arises within a few days following craniotomy and resolves within three months. In contrast, chronic PCH persists beyond this period. 6
Despite advancements in neurosurgical techniques, optimal management of PCH remains uncertain. 7 Its pathophysiology is unclear, though proposed mechanisms include nerve injury, muscle trauma, dural adhesions, and scar formation. Theories suggest PCH may result from peri-cranial muscle adherence to the dura, aseptic meningitis induced by bone drilling, or neuroma formation and nerve entrapment within surgical scars.8,9 Abortive treatments such as opioids, ordinary analgesics, non-hormonal anti-inflammatory drugs, and triptans can be administered. There have been reports of improvements using sodium divalproex, verapamil, and local anesthetics. 8
Recently, botulinum toxin type A (BoNT-A) has emerged as a promising therapy for PCH due to its demonstrated efficacy in treating PCHs and pain. 10 BoNT-A exerts its analgesic effects through neuromuscular blockade and inhibition of pain-mediating neurotransmitters, making it a potential candidate for managing PCH. 7
In a retrospective analysis of 102 patients, Gee et al. found that 38% of individuals who underwent supratentorial craniotomy for cerebral bleeding, intractable epilepsy, or tumors experienced postoperative headaches. 11 Conventional treatment options for PCH include nonsteroidal anti-inflammatory drugs, anticonvulsants, muscle relaxants, and tricyclic antidepressants. However, these treatments often provide incomplete relief or are associated with adverse effects, underscoring the need for alternative therapeutic approaches.
This systematic review aims to evaluate published case reports and case series to assess the therapeutic potential of BoNT-A in managing PCH. By analyzing patient outcomes, treatment protocols, and safety profiles, this review seeks to provide insight into the efficacy and limitations of BoNT-A as a treatment option for PCH.
Methods
Search strategy
A systematic literature search was conducted across PubMed, Scopus, and Web of Science databases to identify relevant studies. The search strategy included the keywords “post-craniotomy headache,” “botulinum toxin A,” “BoNT-A,” “case report,” and “case series.” To ensure a comprehensive review, references from selected studies were manually screened for additional relevant reports. We conducted this systematic review and meta-analysis based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 12
Eligibility criteria
Studies were included if they were case reports or case series reporting the use of BoNT-A specifically for PCH. Eligible studies were required to provide detailed information on injection sites, dosages, patient outcomes, and follow-up assessments evaluating treatment efficacy. Exclusion criteria encompassed studies involving headaches unrelated to craniotomy, review articles, editorials, conference abstracts lacking full data, and studies without clearly defined outcome measures necessary for meaningful analysis.
Data extraction
Relevant data were systematically extracted from each study, including patient demographics (age, gender, surgical history), craniotomy details (type, underlying pathology), headache characteristics (onset, duration, severity), and BoNT-A treatment parameters (injection sites, dosages, number of sessions). Efficacy outcomes were assessed based on pain relief, headache frequency, and quality-of-life improvements. Additionally, adverse events and overall safety profiles were documented to evaluate potential risks associated with BoNT-A therapy.
Quality assessment
To ensure the reliability and accuracy of the extracted data, two authors independently performed the quality assessment (QA) of the included studies using the Joanna Briggs Institute (JBI) critical appraisal checklist for case reports and case series. 13 Any discrepancies were resolved through discussion. The QA process aimed to minimize bias and enhance the validity of the findings, providing a robust basis for assessing BoNT-A's role in PCH management.
Results
Study selection
A total of 15 records were identified through database searches. After removing duplicates and screening titles and abstracts, eight full-text articles were assessed for eligibility. Four case series met the inclusion criteria and were included in the final analysis. The selection process is shown in the PRISMA 2020 flow diagram in Figure 1.

PRISMA 2020 flow diagram. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Study characteristics
This review includes five case series examining the use of BoNT-A for chronic PCH, involving a total of 15 patients. The studies were conducted in France, Canada, the United States, and Japan between 2015 and 2024. Sample sizes ranged from three to four patients per study, with patient ages spanning from 36.3 to 64.7 years. Detailed characteristics, including demographics, surgical indications, headache onset, follow-up duration, and clinical presentation, are summarized in Table 1.
Summary of included case series on BoNT-A for post-craniotomy headache.
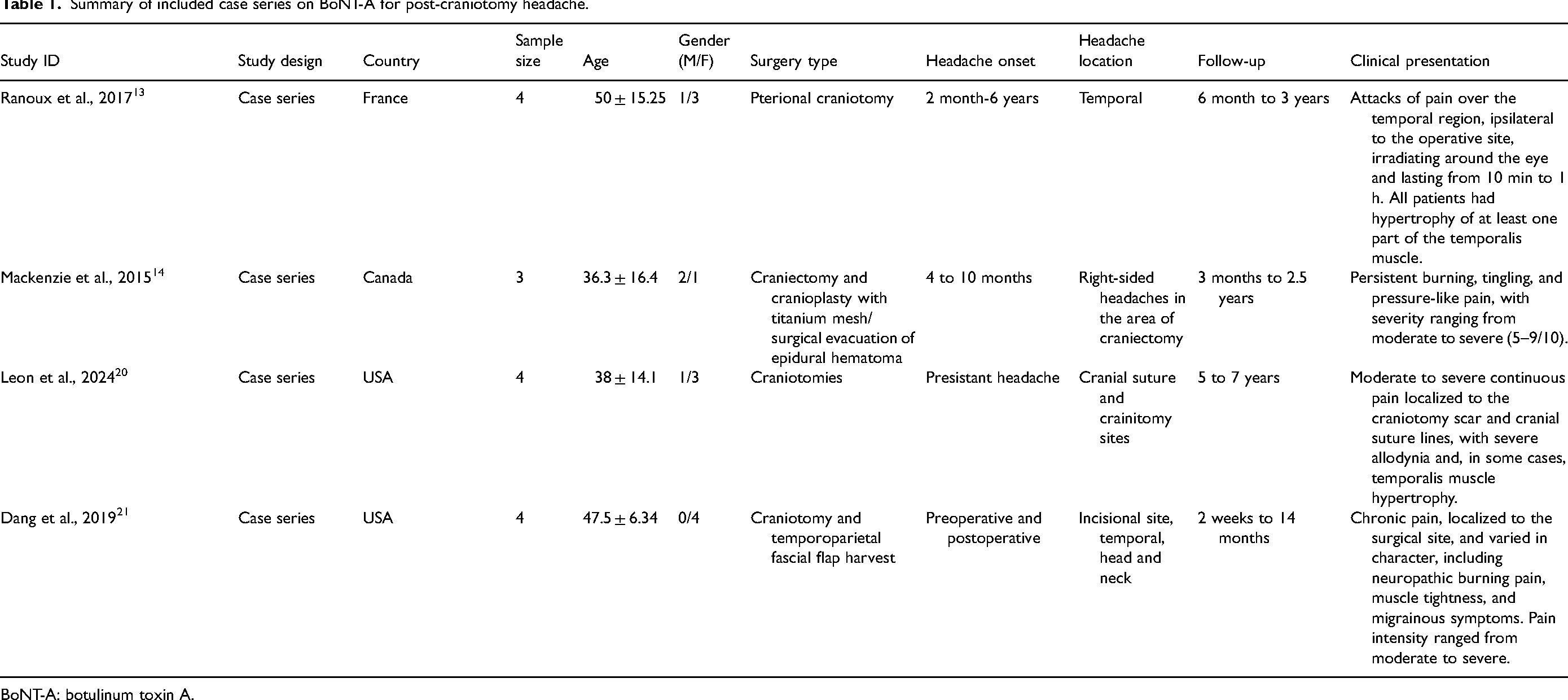
BoNT-A: botulinum toxin A.
Patients underwent craniotomy or craniectomy for various indications, including traumatic brain injury, tumor resection, and other neurosurgical procedures. Despite differences in underlying conditions and surgical approaches, all patients developed persistent PCH unresponsive to conventional pain management strategies such as oral analgesics and conservative treatments.
Headache onset varied, ranging from the immediate postoperative period to several years after surgery. Common headache locations included the surgical incision site, the temporal region, and cranial sutures. Follow-up durations ranged from 2 weeks to 7 years, reflecting the diverse monitoring periods across the studies.
The clinical presentation, intervention strategies, and treatment outcomes of each study are reported in Table 2. BoNT-A was administered in varying doses and injection sites. Across all studies using BoNT-A, patients experienced notable improvements in headache intensity, frequency, and duration, with no adverse effects reported.
Inclusion criteria, BoNT-A treatment protocols, and patient outcomes.

BoNT-A: botulinum toxin A.
Individual case data reveal that all patients experienced moderate to severe daily headaches, often measured using Visual Analog Scale scores ranging from 3 to 10. BoNT-A treatment parameters, including dosage and injection sites, varied across cases, most commonly involving peri-incisional areas and the temporalis muscle. Reported clinical responses were generally favorable, with pain relief ranging from 50% to 100%. However, limited information in two reports restricts broader interpretation of outcomes, as detailed in Table 3.
Diagnosis, headache characteristics, treatment parameters, and clinical response.

Visual Analog Scale.
Injection protocols
The injection protocols varied across all studies, with clear differences in the location was injection, the amount used, and the frequency of patient administration. Injections were mainly done around the temporalis muscle, incision site, and cranial suture lines, focusing on areas where post-surgical pain and muscle tightness were most severe. The dosage varied a lot, ranging from as little as 15 units to as much as 165 units per session, depending on how bad the symptoms were, how big the affected area was, and the doctor's treatment approach.
As for how often patients get injections. Only one patient received just a single injection, while others had multiple sessions, spaced anywhere from 2 weeks to 6 months apart. One study noted that one patient received only a single injection, while another reported ongoing treatment every few months, with therapy continuing for years in certain cases. This inconsistency in dosing and frequency makes it clear that standardized guidelines are needed to figure out the best way to use BoNT-A for long-term relief.
Intervention and efficacy
Across all the case series, BoNT-A consistently provided pain relief, reinforcing its potential as a treatment for PCH. Patients started noticing improvement within 10 to 15 days after the injection, with a significant drop in headache severity; many experienced a 75–100% reduction in pain intensity. Beyond just reducing pain, the frequency of headaches also declined, allowing patients to function better in their daily lives and improving their overall quality of life.
However, the duration of pain relief varied considerably, ranging from as little as a few weeks to sustained benefits lasting over five years. Many had between two to seven pain-free days per week, while others improved significantly over long periods. This range of responses suggests that while BoNT-A is highly effective for 11 patients, individual factors like the root cause of PCH, symptom severity, and the specifics of the injection protocol likely influence the results.
Beyond just easing pain, BoNT-A had a broader impact on patients’ lives. Several studies highlighted a decreased reliance on pain medications post-treatment, suggesting that BoNT-A could serve as a long-term alternative to conventional analgesics. The reduction in chronic headache symptoms also contributed to improvements in daily activities, work productivity, and even emotional well-being, making it a promising option for managing PCH.
Safety outcomes
None of the studies reported serious adverse effects. Patients did not experience systemic complications, infections, or long-term side effects. Overall, the treatment demonstrated a strong safety profile, supporting its potential as a low-risk option for managing persistent PCHs.
Quality assessment
The included studies were assessed using the JBI Checklist for Case Series as reported in Table 4. All studies met key criteria for reliable diagnosis and outcome reporting. However, the consecutive inclusion of participants was unclear across all studies, and site demographic details were missing in Ranoux et al. (2017) and Mackenzie et al. (2015).15,18 Additionally, statistical analysis was not applicable in any of the studies, reflecting their descriptive nature. Despite these limitations, all studies were included, demonstrating a consistent trend of BoNT-A's effectiveness and reinforcing the need for future controlled trials.
JBI checklist for case series.

JBI: Joanna Briggs Institute.
Discussion
The available evidence suggests that BoNT-A is a promising treatment for persistent PCH. Case series indicate that patients experience significant reductions in headache frequency and severity, aligning with BoNT-A's established mechanisms, including muscle relaxation and inhibition of pain-mediating neurotransmitters. These findings suggest that BoNT-A may be a viable alternative for patients with refractory PCH who do not respond to conventional therapies.
Mechanism of action
BoNT-A exerts its analgesic effects through multiple pathways. It inhibits acetylcholine release at neuromuscular junctions, reducing muscle spasms that may contribute to PCHs. Additionally, it blocks pain-mediating neuropeptides such as substance P and calcitonin gene-related peptide (CGRP), thereby decreasing pain transmission. BoNT-A also modulates central and peripheral sensitization pathways, playing a role in chronic headache conditions. Preclinical research suggests that retrograde axonal transport contributes to its pain-relieving effects, allowing it to influence central pain processing. These mechanisms may explain its long-lasting analgesic benefits in PCH, post-traumatic headache (PTH), and chronic migraine. 14
Clinical applications of BoNT-A
BoNT-A is already well-established as a preventive treatment for chronic migraine, significantly reducing headache days per month and improving patient quality of life. 15 The National Institute for Health and Care Excellence (NICE) guidelines recommend BoNT-A for chronic migraine in patients experiencing headaches on at least 15 days per month, with migraines on at least 8 of those days (NICE, 2012). Treatment involves multiple injections at targeted sites across the head and neck.
Beyond its approved indication,
The included case series support the use of
Collectively, these reports suggest that
Emerging evidence also suggests a potential role for monoclonal antibodies targeting CGRP in the management of PCH. Elevated serum CGRP levels in patients with persistent post-concussion symptoms, with subsequent decreases over time, indicate a possible involvement of CGRP in PTH. However, CGRP plasma concentrations are unlikely to serve as reliable blood-based biomarkers for persistent PTH. Further studies are needed to determine whether CGRP levels could predict the development of persistent PTH.16,17
Taken together,
Safety and limitations
BoNT-A has been extensively evaluated for headache management, with a well-tolerated safety profile. Common adverse effects include neck pain, mild muscle weakness, and injection site discomfort, which are generally transient and self-limiting. However, one study reported a higher incidence of adverse events in BoNT-A-treated patients compared to placebo, though none were classified as severe. 15
Despite its potential, several limitations exist. All studies on BoNT-A for PCH are small case series, limiting the generalizability of findings. There is also considerable variability in injection protocols, including dosage, injection sites, and treatment frequency, making it difficult to establish standardized guidelines. Follow-up durations vary significantly, with only short-term assessments in certain cases, limiting comprehensive evaluation of long-term efficacy and safety. Additionally, all studies lack control groups, making it challenging to attribute symptom improvements solely to BoNT-A treatment.
Future directions
To build on the current evidence, future research should focus on conducting randomized controlled trials to establish BoNT-A's efficacy in PCH management. Standardization of injection protocols, including dosage, frequency, and target sites, is necessary to enhance reproducibility and optimize patient outcomes. Identifying patient subgroups all likely to benefit from BoNT-A based on headache characteristics, surgical history, or other factors could further refine treatment strategies. Additionally, long-term follow-up studies are essential to assess sustained efficacy, recurrence rates, and any delayed adverse effects.
Conclusion
BoNT-A presents a promising and well-tolerated therapeutic option for patients with persistent headaches following craniotomy. Case series suggest that it provides substantial pain relief and improved function in individuals unresponsive to conventional therapies. However, given the reliance on small-scale studies, larger clinical trials are essential to confirm its benefits and establish standardized treatment guidelines. In the broader context of headache management, BoNT-A remains a well-established preventive therapy for chronic migraine, particularly for patients who have not achieved adequate relief with oral prophylactic treatments. Its targeted administration and favorable safety profile make it a valuable addition to headache treatment strategies.
Clinical implications
BoNT-A may offer substantial pain relief for patients with chronic PCH who are unresponsive to conventional therapies.
Injections targeting peri-incisional regions and the temporalis muscle consistently reduced headache frequency, intensity, and duration without adverse effects.
The variability in treatment protocols highlights the need for standardized dosing guidelines to optimize outcomes and improve long-term headache management in neurosurgical patients.
Footnotes
Acknowledgment
The authors extend their gratitude to all individuals who contributed to this work.
Data availability
Data extracted from included studies and the PRISMA checklist are available upon request. No software code or data analytics tools were used in this narrative synthesis.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics declaration
No ethical approval was required for this research.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
