Abstract
Background and Aim
Eptinezumab is a quarterly intravenous infusion monoclonal antibody (mAb) with high affinity for calcitonin gene related peptide (CGRP) and a long-lasting mechanism of action. In our retrospective observational study, we sought to investigate the effectiveness and safety of eptinezumab both in mAbs-naïve and mAbs-non responders’ patients.
Methods
We collected clinical data from electronic charts of 30 migraineurs who underwent six months of eptinezumab treatment; 18 patients had never undergone mAbs-prophylaxis (G1) while 12 had been non-responders to erenumab/galcanezumab (G2). Monthly Headache Days (MHDs), MIDAS, HIT6 and ASC scores were recorded before eptinezumab (T0) and thereafter every three months. Patients were considered responders whenever MHDs reduction was >50% at six months (T2).
Results
Overall, 25 out of 30 patients (15 in G1 and 10 in G2) were eptinezumab responders (83.3%). Headache frequency (MHDs) at T0 was 12.4 in G1 and 17.0 in G2, while at T2 it was 7.1 in G1 and 8.0 in G2 (p-value < 0.0001). Significant reductions in MIDAS, HIT6 and ASC scores were also observed. No safety concerns were registered.
Conclusions
Based upon our retrospective observational study, eptinezumab might be effective and safe also for those migraineurs who resulted being non-responders to other anti-CGRP(R) mAbs.
This is a visual representation of the abstract.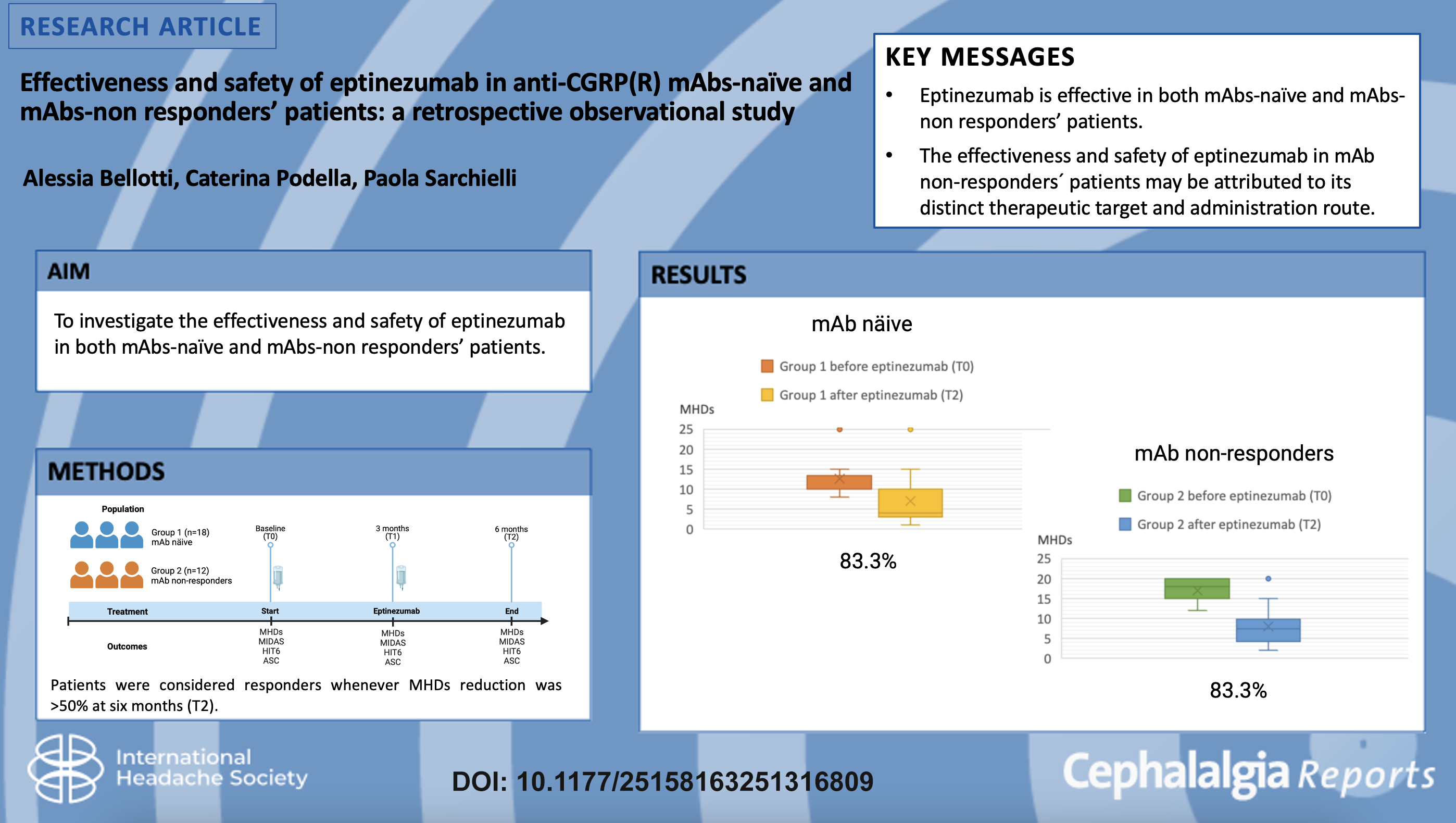
Introduction
Recently, anti-Calcitonin gene-related peptide (CGRP) monoclonal antibodies (mAbs) and antagonists have positively impacted migraine preventive therapy. The theoretical base of mAbs use as migraine preventive drugs is its ability to block CGRP alone or its receptors, which are expressed both in central and peripheral trigeminal neurons.1,2 This neuropeptide acts as a powerful vasodilator and is involved in the initiation and maintenance of peripheral and central sensitization, leading to head pain. Furthermore, the interaction of CGRP with its receptors on mast cells in the meninges, glial cells or astrocytes in the trigeminal ganglion, and the central sites of trigeminal pathway stimulates the production of several pro-inflammatory cytokines which contribute to trigeminal sensitization.3–5
Eptinezumab is currently the most recently approved mAb for the prevention of migraine which has been reported to be highly effective in the prevention of migraine attacks, with good tolerability and safety.6–8 Eptinezumab is a quarterly intravenous infusion humanized recombinant IgG1 (immunoglobulin IgG1) antibody with high affinity (>100,000-fold vs related neuropeptides such as amylin, calcitonin, adrenomedullin and intermediin) that binds to the α- and β- forms of the human ligand of CGRP gene; it prevents the activation of CGRP receptors and, therefore, the cascade of pathophysiological events underlying migraine attacks. 9 Eptinezumab has a particularly rapid and long-lasting mechanism of action. Specifically, due to its molecular conformation, eptinezumab binds CGRP with a ‘latch-and-lock’ system that prevents dissociation of the neuropeptide.10,11 In this way, although the half-life of the drug is 27 days, its efficacy is sustained over a much longer period. Moreover, given its intravenous administration, eptinezumab is able to reach its plasma peak by the end of the infusion (Cmax in 30 min) and thus can be effective immediately. 11 Finally, the rapid effect from eptinezumab contrasts with the longer median values (4–14 days) of subcutaneous anti-CGRP (SC).7,12
Regarding its pharmacokinetic and pharmacodynamic characteristics, we sought to investigate effectiveness and safety of eptinezumab both in mAbs-naïve and mAbs-non responders’ patients. Therefore, our primary objective was to evaluate the reduction in headache frequency and severity after therapy with eptinezumab in both groups; the secondary objective was to evaluate any side effects in these patient populations.
Materials and methods
Study design and population
This retrospective observational study was carried out at the Headache Centre of the Dya Swiss Institute SA in Lugano, Switzerland. The patients’ clinical data were recorded on the Medionline.ch system, in compliance with the current law on privacy. All patients signed an informed consent authorizing the use of anonymized data in line with data protection regulations (GDPR EU2016/679).
Eptinezumab was prescribed to patients according to the criteria for prescribing anti-CGRP(R) monoclonal antibodies established by the Swissmedic (the Swiss authority for the approval and control of medicinal products and medical devices), the European Headache Federation and the American Headache Society. 13
The inclusion criteria were the following:
Adult age (>18 years) Diagnosis of episodic or chronic migraine by experienced neurologists according to the criteria of the International Classification of Headache Disorders (ICHD-III) at least one year prior Frequency of migraine ≥8 days per month over the three months prior to eptinezumab prescription Non-responder to ≥2 previous migraine prophylaxes treatments at full dosages and for at least 3 months In the case of receiving previous subcutaneous anti-CGRP(R) mAbs, ≥4 months of wash out (i.e. at least 4 months since the last subcutaneous antibody administration) was necessary In the case of receiving previous therapy with onabotulinum toxin-A, ≥6 months of wash out (i.e. at least six months since the last botulinum toxin inoculation) was necessary Uncontrolled arterial hypertension, cardio- and cerebrovascular diseases (myocardial infarction, carotid dissection, ischemic stroke, etc.) Ongoing liver or kidney failure
Before stating eptinezumab treatment, patients were also screened for the following exclusion criteria:
The first dose of eptinezumab was given at the lowest effective dosage of 100 mg, and subsequently, if well tolerated, dosage was increased to 300 mg. Patients were allowed to continue their previous migraine prophylaxis, but the simultaneous administration of onabotulinum toxin-A was not allowed. Patients with chronic migraine and/or drug overuse did not undergo a detoxification cycle before starting eptinezumab therapy.
Clinical data from 30 patients who received eptinezumab therapy were collected from their electronic charts and transcribed into a retrospective database. We evaluated different variables regarding general demographic and anamnestic characteristics: clinical history of headache, previous prophylactic therapies used, response and side effects to this therapy, time and dosage of eptinezumab and the response to. Concerning general demographic and anamnestic characteristics, gender, age, current therapy, and comorbidities were recorded. The assessment of headache history included age of onset, cumulative headache years, frequency of attacks (expressed in headache monthly days – MHDs), quality of pain (pulsatile or not), pain intensity, associated symptoms (photo- and phonophobia, nausea and vomiting) and ictal allodynia. Diagnosis was based upon current International Headache Classification (ICHD3). As for attack treatment is concerned, previous and current use of triptans, NSAIDs and combined analgesics and their efficacies were recorded, as well as their monthly intake rates and the presence of pharmacological overuse (>= 10 doses/month of triptans or combination analgesics, >= 15 doses for nonsteroidal anti-inflammatory drugs or analgesics/month) referred by the patient over the three months before starting eptinezumab. For ineffective or non-tolerated migraine prophylaxes, both the number and types of drugs prescribed as well as their duration were specified. Additionally, with regard to previous treatment with onabotulinum toxin-A, the total dose injected, and the number of cycles were also recorded.
The outpatient re-evaluations, were performed according to the following timelines:
T0: last outpatient follow-up visit before starting eptinezumab prophylaxis, which was followed by the first 100 mg eptinezumab infusion within a week T1: follow-up visit at 3 months; in the absence of referred side effects, the second infusion was prescribed at 300 mg to estimate the effectiveness of full-dose therapy T2: follow-up visit at 6 months of treatment.
Information available from electronic records regarding outpatient re-evaluation included: vital parameters (blood pressure, heart rate, oxygen saturation, weight), headache frequency and intensity, frequency of aura, symptomatic drugs used as needed and their efficacy (yes/no), scores of migraine related disability and severity such as MIDAS
14
(Migraine disability assessment is a specific questionnaire for migraineurs to assess the disability caused by headache) and HIT-6
15
(Headache Impact Test 6 is a short 6-item questionnaire used to assess the impact and severity of migraine), scores of allodynia as a measure of central sensitization such as ASC
16
(Allodynia Symptom Checklist, a 12-question questionnaire on the frequency of presentation of various symptoms of allodynia).
Eptinezumab infusions were carried out by specialized nursing staff; eptinezumab was diluted before administration and infused for 30 min. The same nursing staff who administered the infusion also monitored the patients during the entire procedure.
To investigate for any differences in response to eptinezumab we classified the study patients were classified into two groups:
Group 1 (G1): mAbs-naïve patients (n = 18) Group 2 (G2): patients who had already received anti-CGRP(R) mAbs prophilaxes (namely erenumab/ galcanezumab) (n = 12).
Regarding Group 2, data were collected on any previous efficacy of mAbs, the duration of treatment and any referred side effects.
The reason why these patients were not initially treated with eptinezumab but with the other mAbs was the availability of these drugs in Switzerland before eptinezumab. Whenever there was an initial therapeutic benefit and either a subsequent loss of efficacy and/or development of side effects, a switch to another antibody with a similar mechanism of action was allowed (e.g. from erenumab to galcanezumab, or from these two antibodies to eptinezumab).
Statistical analysis
The pool of patients (n = 30) who underwent eptinezumab therapy fulfilled the sample size estimate calculated for a 95% confidence interval, with a margin of error of 5%. The calculation of the sample size was based on current data on the response rate to monoclonal antibody therapy in migraine patients of about 70% from the first month of treatment.6,17
The T0 attack frequency was defined based on the number of attacks per month over the three months prior to the start of eptinezumab treatment. The average attack frequency was also recorded at each time point (every 3 months, at follow up visits for anti-CGRP MAbs prescription). Patients who had a decrease in the attack frequency ≥50% were considered “responders”. MIDAS, HIT6, and ASC scores were also recorded at the same time points over follow-up.
Categorical variables were expressed as numbers and percentages, while continuous variables as a mean ± standard deviation (SD).
For the quantitative data, the Kolmogorov-Smirnov test was used to determine whether the data were normally distributed. Depending on data distribution, the ANOVA for repeated measures or the Friedman test were used for the longitudinal analysis. For the comparison between the two groups, the T-test and the Wilcoxon Mann–Whitney test were used. Post hoc analysis was performed with the Turkey or Kruskal–Wallis tests. Qualitative data were analyzed using the Chi-square test. The limit of statistical significance was set at P < 0.05.
Results
Between January 2023 and April 2024, we collected data from 30 migraineurs on eptinezumab prophylaxis. All of the patients had at least two infusions (6 months).
Demographic and clinical variables are reported in Table 1. Changes in clinical variables after therapy are summarized in Table 2.
Demographic and clinical variables at baseline.

Clinical variables at follow-up visits (T1, T2).

The mean number of headache days per month (Monthly Headache Days - MHDs) of all patients at T0 was 14.23 (SD 4.8), while at T1 it reduced to 8.1 (SD 5.3) and at T2 it further reduced to 7.48 (SD 5.6) with a two-tailed p-value of less than 0.0001. The number of responders already after the first 100 mg eptinezumab infusion was 23 out of 30 patients (76.6%), while after increasing therapy to 300 mg we reached 25 out of 30 patients (83.3%). This difference was not statistically significant, nor was the further reduction in MHDs from T1 to T2. MIDAS was also reduced from T0 to T2: from 77.9 at T0 (SD 24.6) to 19.8 at T3 (SD 18.9), with a two-tailed p-value of 0.0003, considered statistically significant. HIT-6 declined from 66.4 (SD 6.2) at T0 to 48.5 at T2 (SD 11), with a two-tailed p-value of 0.02 considered statistically significant. The ASC decreased as well from 8.5 (SD 4.5) at T0 to 4.8 (SD 4.1) at T2 with a two-tailed p-value of 0.0181 considered statistically significant. Hand in hand with the reduction in MHDs, the intake of symptomatic drugs was also reduced. Whereas 40% of patients suffered from symptomatic drug overuse (>15 symptomatic drugs doses per month, including triptans, acetaminophen and NSAIDs) before eptinezumab treatment, after the first infusion, drug overuse persisted in only 8% of cases.
Regarding any reported side effects, only one patient (3%) complained of discomfort associated with venous access placement, however the patient continued the therapy for six months. Despite regular monitoring of blood pressure values at follow-up visits, we found no pressure increases; no patients reported constipation.
One patient had to stop treatment after 6 months due to pregnancy. All non-responder patients (n = 5) discontinued eptinezumab after six months of treatment. Their efficacy data were therefore included in statistical analysis until T2, that is, as long as they continued treatment. Three of these patients belonged to Group 1 and had never been prescribed other monoclonal antibodies; the other two patients belonged to Group 2 and had already been deemed non-responders to both erenumab and galcanezumab.
Group 1 and Group 2 patients were similar in terms of their clinical characteristics: average age, gender, number of previously prescribed prophylaxes. The two groups however did differ in monthly headache days (mean MHDs 12.4 in G1 vs. 17 in G2, p-value 0.01). Considering the previous conventional prophylactic therapies, Group 1 patients had already been prescribed on average 3 prophylactic drugs (from 2 to 5), including calcium channel blockers (flunarizine), antiepileptics (topiramate), tricyclics (amitriptyline), beta-blockers (propranolol) and botulinum toxin (onabotulinum toxin A) at full doses and for at least three months. In Group 2, the average number of previous prophylaxes was 3.5 (from 3 to 5). Erenumab was administered to all patients in Group 2 for a period of 3 months (one patient), 6 months (6 patients), 1 year (3 patients) and 2 years (2 patients). Patients who had been prescribed erenumab for 3 and 6 months did not achieve clinically significant efficacy (reduction in MHDs by about 25–30%) while other patients had initial benefit (reduction in MHDs >50%) followed by a loss of efficacy (increase in frequency of attacks). For this reason, three patients had also been prescribed galcanezumab for up to 1 year; however, none of the three patients achieved complete benefit (reduction in MHDs < 50%). The side effects reported by patients receiving erenumab and galcanezumab therapy were constipation (one patient) and local reactions at the site of drug inoculation (one patient).
After eptinezumab treatment, both groups had an improvement, with statistically significant differences in MHDs before (T0) and after six months of therapy (at T2): the two-tailed p-values in the paired t-tests were less than 0.0001 in both Group 1 and Group 2 (Figure 1). The response rate after the first 100 mg eptinezumab infusion was 83.3% in Group 1 (15 of 18 patients) and 66.6 percent in Group 2 (8 of 12 patients), but after the increase to 300 mg both groups achieved a response rate of 83.3%. The initial difference after only 3 months of therapy was not significant (p = 0.30). Furthermore, no differences in headache frequency between Group 1 and Group 2 emerged after six months of eptinezumab therapy, showing a greater reduction in MHDs in Group 2 than in Group 1 (−9.5 MHDs in Group 2; −5.6 MHDs in Group 1).
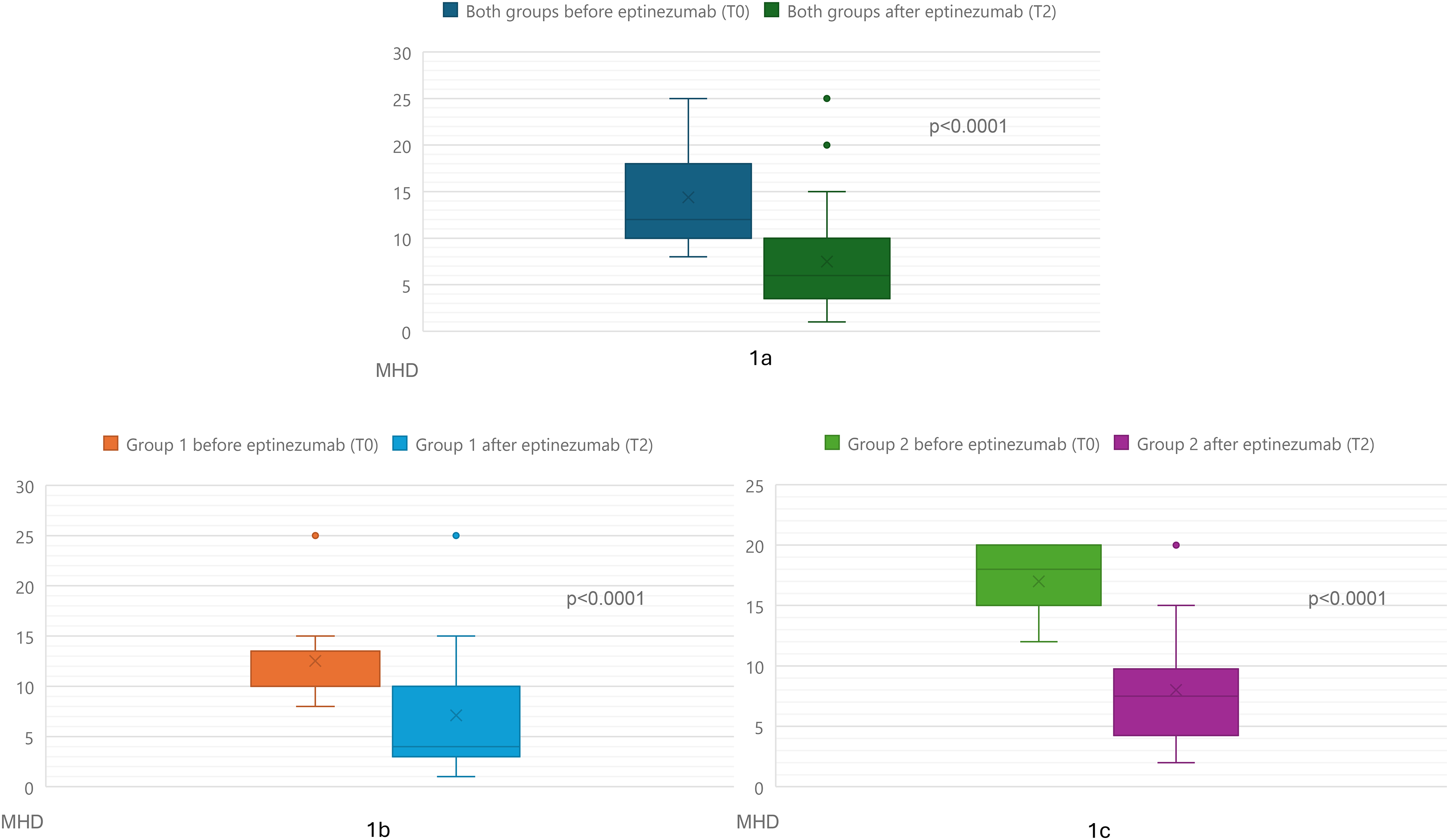
Box plots of monthly headache days before and after eptinezumab treatment. In the figure above we can see the overall reduction in MHDs in all 30 patients after 6 months of therapy. In the figures below we see the distinction between group 1 and group 2.
Discussion
According to current published literature, eptinezumab has been demonstrated to be effective for the treatment of episodic and chronic migraine patients, allowing for a statistically significant reduction in MHDs from the first infusion. 7 Data regarding the efficacy of eptinezumab in patients who had two to four previous unsuccessful oral prophylaxes are also currently available confirming its efficacy in migraine patients with several prior preventive treatment failures.7,18,19 In agreement with this, our study findings suggest that eptinezumab is effective despite an average of 3 previous treatment failures, with an overall response rate of 83.3%.
Emerging data on the use of eptinezumab in patients with previous failure with other anti-CGRP(R) mAbs are also encouraging therapeutic switching in this class of therapies: in particular, recent studies highlight how eptinezumab was effective even in patients who had already failed up to three mAbs, with a response rate that decreases as treatment failures increase (none: 78.6%, one: 45.0%, two: 32.1%, three: 23.5%).20,21
Another recent study regarding the switch to eptinezumab after double failure with erenumab and another subcutaneous antibody similarly highlights its effectiveness, with a ≥30% reduction in migraine days per month in 29.7% of patients after six months of treatment. 22
Other findings regarding subcutaneous monoclonal antibodies (erenumab, galcanezumab, fremanezumab) show that patients who switched among anti-CGRP(R) mAbs during the course of their treatment still benefited overall, but treatment response in patients who switched twice was markedly lower compared to the no switch and one switch subgroup. 23
Compared with subcutaneous anti-CGRP(R) mAbs, due to its pharmacokinetic and pharmacodynamic properties, eptinezumab might be more suitable for a drug switch attempt whenever other drugs of the same class fail. 21 To this regard, our study findings point to an effectiveness of eptinezumab in terms of MHDs reduction of >50% following a lack or a loss of efficacy from erenumab/galcanezumab. In fact, on our sample of migraine patients, the response rate to eptinezumab after six months was 83.3% also in patients who had already undergone therapy with other anti-CGRP(R) mAbs but with only a partial response (reduction of MHDs <50%). It is our opinion that the effectiveness of eptinezumab in these patients could be explained by the different mode of administration (intravenous) and its different pharmacokinetics and pharmacodynamics properties: eptinezumab has a more rapid action than other subcutaneous antibodies, a rapid binding and slow dissociation with CGRP, as reported in past in vitro studies.11,12,24 Specifically, at equivalent circulating concentrations, eptinezumab binds to CGRP twice as fast as others mAbs, and possesses a high affinity for CGRP alpha and beta ligands (binding affinity constant 4 × 10–12 and 3 × 10–12 molar, respectively).11,12,24 Furthermore, eptinezumab effectiveness in our group of patients could also be explained by other factors: 1) all of the 12 migraineurs in G2 had already undergone treatment with erenumab, which is an anti-receptor of CGRP, while eptinezumab binds directly to the neuropeptide; thus, these drugs have similar but partially different mechanisms of action; 2) 42% of the G2 patients had an initial good response to other antibodies for a long period, but they lost efficacy over time (42% of patients after one year and 16% of patients after 2 years of treatment); this might suggest possible receptor remodulation or an increase in CGRP in response to its receptor blockade, that would need to be evaluated with ad-hoc clinical studies.
For these same reasons, the efficacy data of eptinezumab will also need to be evaluated over the long term to identify any loss of efficacy.
In our study, 5 patients resulted in being unresponsive to eptinezumab treatment (2 after failure of both erenumab and galcanezumab). Our result on the efficacy of eptinezumab in only one of the three patients who had failed both erenumab and galcanezumab (33.3%), seems in line with the data reported in the study regarding switch after double failure with other mAbs (responder rate of 29.7%). 22 As for anti CGRP mAbs failure, current data in literature suggest that CGRP might not be the only molecule involved in the genesis of migraine. Patients who did not respond to either subcutaneous antibodies or eptinezumab need to be further studied not only to identify any predictors of ineffectiveness, but also to identify any mediators of migraine pain so to develop additional personalized therapies.25,26
No serious AEs were reported by our patients, nor did any patient discontinue treatment due to adverse events. This finding is consistent with data from RCT 27 and open label trials. 28
The major limitations of our study include its small sample size and the retrospective analysis of clinical data. In fact, since the data were collected retrospectively, the neurologist of our Headache Center initially decided to switch to eptinezumab independently and not based on strict a priori criteria. Similarly, the decision to start with eptinezumab at 100 mg and subsequently increase to eptinezumab 300 mg was dictated by caution and the desire to verify efficacy, rather than by pre-established criteria. On the other side, one of the strengths of our study is that we were able to obtain data on the efficacy and safety of eptinezumab therapy in patients who had already been prescribed other mAbs. Presently, most regulatory agencies throughout European countries do not reimburse the cost of eptinezumab in patients who had already been previously prescribed with other mAbs.
Conclusion
Based upon our study results, eptinezumab seems effective and safe for all migraineurs, regardless of the number of previous failed prophylaxis, the presence of pharmacological overuse or the high frequency of attacks. Eptinezumab seems effective also for those migraineurs who resulted being non- responders to other anti-CGRP(R) mAbs, especially when we change the mAbs site of action and for patients who had shown, at least initially, a good response to the mAbs-therapy, which however was lost over time.
This data provides further evidence of the need to allow in all countries the refund for the switch from one antibody to another, in order to find the best personalized therapy for the patient.
Article highlights
Our study findings highlight the effectiveness of eptinezumab in reducing MHDs by at least 50% despite previous failures with several preventive treatments.
Eptinezumab seems effective and safe even in patients who have lost efficacy and failed subcutaneous mAbs therapy such as erenumab, probably due to the change in therapeutic target and route of administration.
Patients who did not respond to either subcutaneous mAbs or eptinezumab need to be studied to identify any other possible mediators of migraine pain.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and patient consent
All patients signed informed consent authorizing the use of anonymized data in line with data protection regulation (GDPR EU2016/679).
Funding
The authors received no financial support for the research and authorshipof this article. Publication and revision costs were supported by Prof. Paola Sarchielli's institutional funds.
