Abstract
Background
Mexiletine is used as a preventive medication for refractory chronic migraine (rCM) and to prolong migraine improvement following intravenous lidocaine infusion. However, published evidence supporting its use in migraine remains limited. This retrospective study describes the dosing, treatment duration, and adverse effects of mexiletine in a group of patients with rCM following lidocaine infusion.
Methods
Electronic medical records were reviewed at the Jefferson Headache Center. Baseline pain intensity and monthly headache days were collected at baseline and up to 6 office visits within 1-year post-discharge. The duration of mexiletine treatment was evaluated for up to 40 months post-discharge.
Results
The study included 329 rCM patients who were prescribed a daily dose of 450 mg. At the last follow-up, the median daily dose was 600 mg (450 mg–750 mg). Mexiletine was discontinued in 36.4% (120/329) of patients due to side effects and in 11.2% (37/329) due to insufficient treatment response.
Conclusion
In our population, 37% (104/329) remained on mexiletine for over 500 days, indicating a favorable safety profile. While mexiletine can be safely administered in outpatient settings, its use should be reserved for patients with the most refractory cases unless stronger evidence supporting its efficacy in migraine becomes available.
Introduction
Refractory chronic migraine (rCM) presents a significant challenge in clinical management due to its resistance to standard treatments and its significant impact on quality of life. rCM is a severe form of chronic migraine that persists despite adequate trials of preventive medication from at least 2 drug classes. Individuals with rCM often endure daily or near-daily pain and other disabling symptoms.
Inpatient hospitalization, including the use of continuous lidocaine infusion,1–4 is among the treatment options for patients who fail to improve with outpatient care. Sustained migraine improvement post-hospitalization remains a challenge. 3 Lidocaine, an amide anesthetic, blocks voltage-gated sodium channels (VGSCs) involved in nociception. Although not fully understood, lidocaine may reduce pain through anti-inflammatory mechanisms, decreasing hyperalgesia and allodynia via attenuation of neutrophils and inflammatory cytokines. Proposed central mechanisms of analgesia include inhibiting glutamate receptors via N-Methyl-D-aspartic acid receptor and glycine receptor potentiation. Once intravenous (IV) lidocaine is discontinued, it is rapidly eliminated in approximately 15 min.5,6
Intravenous lidocaine is contraindicated in patients with certain cardiac arrhythmias, including Wolff-Parkinson-White and sinoatrial, atrioventricular, or intraventricular heart block. Continuous ECG monitoring (telemetry) and daily basic metabolic panels for electrolyte monitoring are recommended during infusion. 7 Mexiletine, contraindicated in patients with untreated second or third-degree heart block, cardiogenic shock, or hypersensitivity to the drug, also warrants cardiology consultation for patients with any history or symptoms of cardiac arrhythmia before use of either mexiletine or IV lidocaine. 8
Mexiletine, a class Ib sodium channel antagonist, is used following lidocaine infusion to prolong its therapeutic effects, given its similar mechanism to lidocaine. Orally administered, mexiletine is predominantly metabolized by the liver (primarily CYP2D6 enzyme) with peak plasma levels in 2–3 h and a half-life of 9–12 h. 8 Previous research has demonstrated its analgesic benefits in neuropathic disorders such as diabetic neuropathy and peripheral nerve injury.9,10 Side effects of mexiletine are commonly reported and include nausea, tremor, and dizziness. In a study by Carroll et al., patients discontinued mexiletine for neuropathic pain after a median of 43 days due to side effects, though dosing details were not provided. 11
Since 2005, Thomas Jefferson University Hospital has discharged patients who respond to lidocaine infusion on daily oral mexiletine as a migraine preventive. The rationale is that, given its similarity to lidocaine, mexiletine may extend the migraine improvement initiated by lidocaine. However, published evidence for its use in headache disorders remains limited despite its common use at our institution. 12 The prescribed starting dosage of 150 mg three times daily is derived from its application in cardiac literature. 8 Supporting evidence includes a retrospective case series by Marmura et al., in which nine patients with chronic daily headache reported significant improvement with mexiletine. All patients reported that mexiletine was more effective than multiple previously failed preventives; five had received lidocaine infusion prior to mexiletine initiation. Daily doses ranged from 600 to 1500 mg, with seven patients tolerating side effects without discontinuing the medication. 12
Given this gap in literature, this retrospective study is the first to describe dosing, serum concentrations, treatment duration, and adverse effects of mexiletine in a group of rCM patients following lidocaine infusion. Secondary aims include identifying associations between outcomes and mexiletine dose or serum level.
Methods
This study received an exemption from the Institutional Review Board of Thomas Jefferson University (#21E.1014).
Inclusion criteria
The Epic electronic medical record system was queried to identify patients admitted to the Jefferson Headache Unit in Philadelphia, PA for their first inpatient lidocaine infusion for rCM between 4/1/2017 through 4/1/2020.
Exclusion criteria
Patients without at least one post-discharge follow-up visit were excluded from the study.
Treatment protocol during lidocaine admission
During elective inpatient admission, pre-existing preventive medications were generally continued, and lidocaine infusion was started at 1 mg/min, titrated up to a maximum of 4 mg/min based on tolerability, pain response, and daily serum lidocaine levels. Lidocaine infusion with cardiac monitoring was continued through almost the entirety of the admission unless poorly tolerated. Patients also received other intravenous medications and procedures, including scheduled dihydroergotamine, ketorolac, methylprednisolone, magnesium, neuroleptics, and nerve block/trigger point injections. Patients were discharged with instructions to start mexiletine 150 mg three times daily after discharge and to obtain a serum mexiletine level one week afterward.
Data was extracted between January and February 2023 from hospitalization records and from self-submitted questionnaires completed by patients before each office visit, with clinic notes used if questionnaires were unavailable. For patients re-admitted within a year, subsequent office visits were excluded to avoid confounding from repeat hospitalizations. Collected variables included details of mexiletine treatment, such as dosage, frequency, and side effects reported during these follow-up visits, as well as serum mexiletine levels within 1 year post-discharge. All patients were instructed to obtain a serum mexiletine level one week after discharge. The timing of subsequent serum mexiletine lab measurements varied subject to subject, depending on individual providers’ practice, dose adjustments, or onset of side effects. For patients without a precise discontinuation date, the date of last documented mexiletine use was recorded instead. Additional data included monthly headache days (MHDs) and average pain intensity (11-point NRS) reported at hospitalization and across up to six office visits within 1 year post-discharge if a patient remained on mexiletine during these visits. Data were stored in a HIPAA-compliant web-based REDCap electronic data capture tool hosted at Thomas Jefferson University.
Statistical analysis
For summary statistics, the mean ± standard deviation, median, minimum, and maximum were calculated for continuous variables, while frequency distributions were presented for discrete variables as in Table 1. Pain intensity and MHDs of study subjects are displayed across follow-up visits in boxplots. The duration of mexiletine treatment was defined as the number of days from the discharge date to the date of discontinuation and treated as a survival variable (up to 40 months post-discharge). Subjects still on mexiletine treatment at the last follow-up date during data extraction are considered censored.
Description of the patients that started mexiletine (n =329).

The effect of individual variables on the duration of treatment was evaluated using Kaplan-Meier curves with the log-rank test and the Cox proportional hazards regression. A multivariate Cox model was applied to determine if a variable's univariate effect remained significant after adjusting for other factors.
Pain intensity and MHDs at follow-up visit 1 were compared to their respective values at admission using the Wilcox signed rank test. For the longitudinal analysis, pain intensity across various follow-up visits was modeled in relation to mexiletine dosage and serum levels using linear mixed-effects models. Data was analyzed using R version 4.3.3.
Results
Screening
A total of 373 patients met the inclusion criteria. At discharge, 91% (341/373) of patients were prescribed mexiletine as a new preventive medication, while 9% (32/373) were not. The most common reasons for not prescribing mexiletine at discharge included inadequate treatment response to lidocaine (n = 14) and intolerance to lidocaine (n = 7), with other reasons being patient refusal, pre-existing polypharmacy, previous unsuccessful trials of mexiletine, and transaminitis. At discharge, patients were maintained on their existing home preventives, including anticonvulsants (41% of patients), antidepressants (60%), antihypertensives (35%), CGRP (Calcitonin Gene-Related Peptide) inhibitors (40%), and OnabotulinumtoxinA (44%) (Table 1). Our standard practice is to adjust preventive medications one at a time, adding mexiletine individually to monitor its effects.
3% (12/373) of patients never started the prescribed medication at discharge, citing reasons such as lack of instruction, concern for side effects, or inaccessibility. In total, 329 patients initiated mexiletine (Figure 1).

Screening and continuation groups.
Of these 329 patients, 14% (47/329) discontinued before the first follow-up visit, and 67% (221/329) discontinued mexiletine within 40 months post-discharge. At the time of the last office visit within this period, 108 patients were still taking mexiletine. However, 28 patients were lost to follow-up in the final year (months 28–40). In total, 80 patients continued long-term mexiletine, defined as still taking it at their last visit within the 28–40 month range post-discharge (Figure 1).
Demographics
Of the 329 patients who started mexiletine, the mean age was 45, and 80% (264/329) were female. 90% (296/329) patients were white, 6% (19/329) were African American, 2% (6/329) were Asian, and 1% (2/329) were Hispanic. Patients had a mean migraine history of 16 years, and the mean duration of inpatient hospitalization was 5.0 days. The mean interval from discharge to the first follow-up was 28.4 ± 30 days (Table 1).
Dosing
All patients discharged with mexiletine were started on total daily dose of mexiletine 450 mg (150 mg three times daily). Figure 2 depicts the total daily doses reported by patients at follow-up visits within 1 year post-discharge. Among the 80 patients on long-term mexiletine, the mean and median last daily doses were 602 mg and 600 mg, respectively (range: 150 mg–1350 mg), typically reached by the fourth office visit. The most common last dose frequency was three times daily (n = 45), followed by twice daily (n = 31). For patients discontinuing due to side effects, the mean last dose was 441 mg (range:150–900 mg, n = 120), while for those stopping due to inadequate treatment response, the mean last dose was 503 mg (range:150–1000 mg, n = 37). Multiple reasons for discontinuation could be cited.

Mexiletine total daily dose at follow-up visits within 1 year of hospitalization.
Between discharge and the first follow-up visit, the mean ± SD serum mexiletine level was 0.69 ± 0.45 (range: 0.1–3.0 mcg/mL). The mean mexiletine level was highest between the third and fourth office visits at 1.36 ± 0.9 mcg/mL (range: 0.2–3.2 mcg/mL) (Figure 3). For reference, the standard therapeutic range of mexiletine plasma levels is 0.8 to 2 mcg/mL.(13)

Mexiletine serum levels were collected before follow-up visits within 1 year after discharge.
Because dosages were typically adjusted during follow-up visits, there was little variability in dosages and serum mexiletine levels when grouped by post-discharge time interval. Therefore, dose adjustments and serum values are displayed at the time point of the follow-up visit number (x-axis of Figures 2 and 3).
Effectiveness/longitudinal pain intensities
Pain intensity and MHDs at the first follow-up visit showed a statistically significant reduction from admission (p < 0.00001, Wilcox signed rank test). On admission, the mean ± SD baseline pain intensity was 6.6 ± 1.6, decreasing to 1.43 ± 2.0 at discharge. The mean ± SD at office visits 1, 2, 3, 4, 5, and 6 was 5.1 ± 2.2, 5.6 ± 2.0, 5.5 ± 2.3, 6.0 ± 2.3, 5.5 ± 1.9, and 5.7 ± 1.98, respectively (Figure 4). Regarding MHDs, the mean ± SD at admission was 26.9 ± 3.2. While the median and third quartile MHDs remained at 28 across all time points, changes were observed in the first quartile and mean values. MHDs dropped to 14 and 21.6 ± 9.3 at the first visit, 14 and 21.3 ± 8.7 at the second visit, and 18 and 22.3 ± 8.3 at the third visit. Values increased slightly in later visits as the sample size diminished (Figure 5).

Box plot of pain intensity at admission, discharge, and office visits within 1-year post-discharge.

Monthly headache days reported at admission and office visits within 1 year.
A longitudinal analysis using random intercept linear mixed-effects models across up to six office visits within 1 year post-discharge revealed no significant association between changes in post-discharge pain intensity (subtracted by the admission average pain intensity) and the dosages at visits (p = 0.45) or mexiletine level before office visit 1 (p = 0.69), after adjusting for age, sex, and migraine. For MHDs, higher mexiletine doses were associated with more MHDs (subtracted by baseline) (p = 0.02). Each increase in the mexiletine level is associated with 4.85 more headache days (p = 0.007).
Tolerability/discontinuation
Between discharge and the first follow-up visit, 47 patients discontinued mexiletine. Of the 221 patients discontinuing mexiletine within 40 months post-hospitalization, 54% (120/221) cited side effects, and 17% (37/221) cited inadequate treatment response as the reason for discontinuation (Table 2). The most common side effects leading to discontinuation were nausea/vomiting (n = 33), dizziness (n = 27), and tremor (n = 20) (Table 3). Mexiletine was discontinued at the starting dose of 150 mg three times daily in 120 patients. At 100 days post-discharge, 63% (198/329) of patients remained on mexiletine, decreasing to 52% (154/329) at 200 days and 37% (104/329) at 520 days (Figure 6).

Time to mexiletine discontinuation.
Reasons for discontinuation of mexiletine.

*Multiple reasons for discontinuation could be entered.
Reported side effects leading to discontinuation of mexiletine.

*Multiple side effects could be entered per patient.
For the duration of mexiletine treatment, associations with individual variables were examined using univariate Cox proportional models. Any significant variable at alpha =0.05 was further analyzed for its association after adjusting for discontinuation due to side effects.
Higher maximum tolerated lidocaine rates were associated with longer mexiletine use (p = 0.001), according to Cox regression analysis. However, after adjusting for discontinuation due to side effects from mexiletine, this association was no longer statistically significant (p = 0.14). To explore the possible presence of a threshold or cutoff lidocaine rate, maximum tolerated lidocaine rates were cut into a binary variable at the 25th (2.24 mg/min), 50th (2.5 mg/min), and 75th (2.75 mg/min) percentiles. Only the 75th percentile group showed a significant association with longer mexiletine continuation (p = 0.027), suggesting that patients who tolerated rates above 2.75 mg/min had longer continuation rates.
Patients who experienced dizziness during inpatient treatment were more likely to discontinue mexiletine earlier (p = 0.0003), with this trend persisting after adjusting for discontinuation due to mexiletine side effects as a confounder (p = 0.0003) (Figure 7). Specific side effects while on mexiletine, including dizziness (p = 0.004), anxiety (p = 0.04), and sedation (p = 0.01), were associated with shorter treatment duration. In contrast, the duration of mexiletine treatment was not associated with the maximum tolerated lidocaine serum levels (p = 0.52) or with mexiletine serum levels between discharge and the first follow-up visit (p = 0.79). Other side effects experienced during admission, migraine duration, baseline pain intensity/MHD, comorbidities, BMI, or the presence of a comorbid headache diagnosis were not significantly associated with the duration of mexiletine treatment.
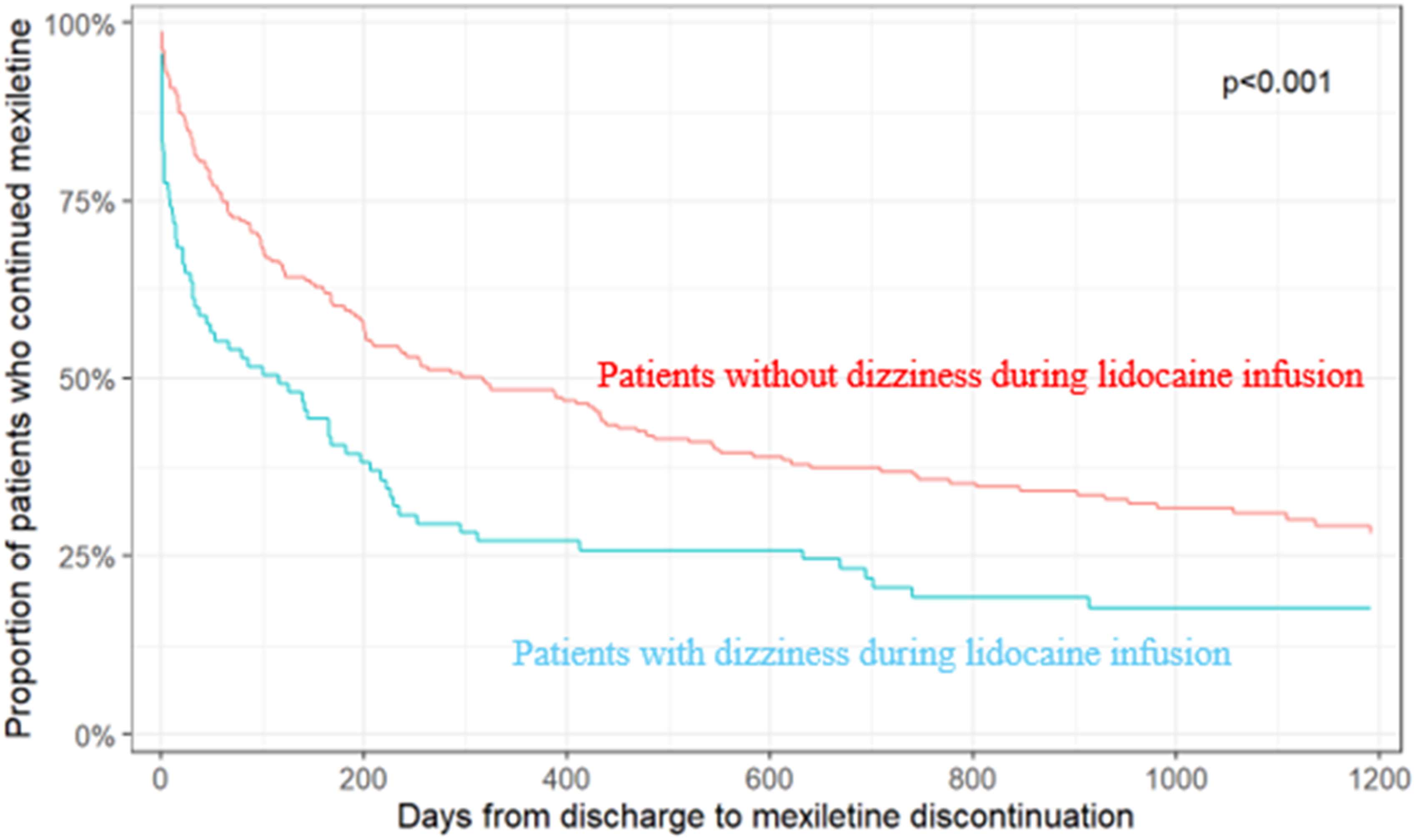
Time to discontinuation of mexiletine for patients who experienced dizziness on lidocaine infusion and patients who did not.
Patients discharged on antiepileptics and CGRP inhibitor preventives had longer durations of mexiletine treatment (p = 0.03 and 0.047, respectively). In contrast, other preventives like antidepressants, OnabotulinumtoxinA, or antihypertensives showed no association with treatment duration.
Discussion
Mexiletine, a medication used in refractory chronic migraine and various other pain disorders, is a standard post-discharge preventive at Thomas Jefferson University following intravenous lidocaine infusion. While continuous lidocaine infusion is a known treatment for rCM, limited evidence exists to guide post-discharge care, including the use of mexiletine. This study, the largest of its kind, details mexiletine use in rCM, including monitoring, dosing, duration of use, and adverse effects.
Regarding dosing, patients remaining on mexiletine long-term were titrated to a median last daily dose of 600 mg, which may be a reasonable target dose in rCM. In Marmura et al.'s case series of nine patients with improved outcomes on mexiletine, effective dosages ranged from 600 mg/day to 1500 mg/day. 12
In evaluating mexiletine's therapeutic effect in rCM, we found no significant association between mexiletine dosage or serum levels and changes in average pain intensity. However, we identified a positive association between mexiletine dose and MHDs, suggesting that patients with more frequent headaches tend to have higher mexiletine doses. We believe that patients with poor outcomes, particularly those experiencing a return of daily headache after hospitalization, are more likely to either increase their medication on their own or contact their provider, resulting in increased dose adjustment. Additionally, this study's ability to evaluate mexiletine's therapeutic benefit is limited by substantial variation in the timing of office visits, serum level measurements, and limited dosage differentiation. Consequently, without a controlled study, the therapeutic effect of mexiletine in treating rCM remains unclear. We recommend that intravenous lidocaine management for rCM be limited to headache specialists, ideally with firsthand experience. Mexiletine can safely be used in the outpatient setting but, without stronger evidence, should be limited to the most refractory of patients.
In terms of duration of mexiletine treatment, 63% (198/329) of patients remained on mexiletine at 100 days after discharge, while 37% (104/329) at 520 days. Most discontinuations due to side effects occurred at the starting dose of 150 mg three times a day. Thus, prescribers may consider a lower starting dose of mexiletine for patients intolerant of lidocaine infusion rates above 2.75 mg/min or who report dizziness during infusion, as these factors were associated with a shorter duration of mexiletine treatment. This observed association may be influenced by discontinuation due to mexiletine side effects. To better understand these findings, we considered whether genetic variations in enzymatic metabolism or serum drug levels might explain these associations. However, in our experience, we commonly encounter patients with high lidocaine serum levels reporting minimal side effects, while others with low serum levels report intolerable side effects. This variability in tolerability likely extends beyond enzymatic pathways and reflects broader individual sensitivity to medication side effects. Supporting this idea, we observed that patients on antiepileptics—often discontinued for intolerable side effects—tended to stay on mexiletine longer. Research has also shown that patients with a history of side effects are generally more prone to adverse reactions across medications. Additionally, factors like lower quality of life have been associated with a higher incidence of medication side effects. 13
This study had several strengths and limitations. Unlike Schwenk et al., which included patients with prior lidocaine admissions and assessed outcomes only during follow-ups between 25 and 65 days post-discharge, 3 our study exclusively included first-time lidocaine recipients, reducing selection bias. Additionally, with up to 6 office visits within 1 year post-discharge, our study offers the most longitudinal observation on post-lidocaine outcomes.
Later visits, particularly visits 3–6, may reflect a follow-up bias, as patients with worse pain outcomes are likely to attend more frequent follow-ups. Focusing on highly refractory migraine cases at a single institution may limit generalizability, as mexiletine might be more effective in a less refractory population.
This was a retrospective chart review; all available and valid values were included in the analysis. Missing data primarily resulted from uncompleted questionnaires or documentation gaps. Due to medication reconciliations done at office visits, there were no missing values for the mexiletine dose at office 1 or the final dose. Missing values for pain intensity and MHDs at office visit 1 were 7.8% and 6.4%, respectively. If precise discontinuation dates were unavailable, the date of last documented use was substituted, potentially underestimating actual treatment durations.
Medication changes before and during the study period were not controlled, making it challenging to isolate mexiletine's effect, particularly in the post-hospitalization setting. We could not effectively compare post-discharge pain outcomes in patients on mexiletine versus patients not on mexiletine, as only 44 patients were not started on mexiletine, and their treatment responses to lidocaine were different. We speculate that some side effects or withdrawal symptoms from inpatient treatments may be misattributed to mexiletine, as these effects can persist for several days after discontinuation.
In conclusion, among patients who remained on mexiletine at the last follow-up visit during the 40-month survey period, the median final daily dose was 600 mg, which may be a good target dose for rCM patients. Most discontinuations due to side effects usually occurred at the starting daily dose of 450 mg. A lower starting dose should be considered for patients with dizziness or other limiting side effects during lidocaine infusion. Pain intensities and MHDs improved in the year following hospitalization, but controlled studies are needed further to assess the effectiveness of mexiletine in migraine treatment. These data can guide the design for a future prospective controlled research trial to establish the effectiveness of mexiletine as a migraine preventive, ideally comparing lidocaine responders randomized to receive daily mexiletine or no mexiletine.
Clinical implications
Patients on long-term mexiletine were titrated to a median last daily dose of 600 mg, which may be a reasonable target dose for rCM patients.
The presence of dizziness as an adverse effect during lidocaine infusion was associated with early mexiletine discontinuation.
The most reported side effects of mexiletine were nausea, dizziness, and tremors.
Footnotes
Acknowledgements
The authors would like to thank Eric Klepadlo for his assistance with proofreading this paper and Dr. Eric Schwenk, MD, for his assistance with obtaining data. This manuscript was edited with the assistance of ChatGPT and Grammarly, and it was used solely for proofreading purposes and not for the original writing of the paper. Endnote software and ChatGPT were used for citations.
“The principal author takes full responsibility for the data presented in this study, analysis of the data, conclusions, and conduct of the research. The principal author had full access to those data and maintained the right to publish all data independent of any third party.”
Author contributions
Lucia Liao helped with study design, data collection, data analysis, and writing the manuscript; Hsiangkuo Yuan helped with study design and writing the manuscript; Dylan Selbst helped with data collection and writing the manuscript, Emily Casaletto helped with data collection and writing the manuscript, Jiangang Liao helped with data analysis and writing the manuscript, Clinton G Lauritsen helped with study design and writing the manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs: Liao, Selbst, Casaletto, Lauritsen, Liao declare no conflicts of interest.
Within the past 24 months, Dr. Yuan has received institutional support for serving as an investigator from Teva, Abbvie, Parema, and Ipsen; consultant fees from Salvia, Pfizer, Abbvie, and Cerenovus; and royalties from Cambridge University Press and MedLink.
Ethics approval and patient consent
This paper received ethical approval from the Institutional Review Board of Thomas Jefferson University. All study participants provided signed consent to participate.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Jefferson Headache Center.
