Abstract
Background
Autoimmune/inflammatory syndrome induced by adjuvants (ASIA) encompasses a set of immune rare diseases triggered by adjuvants, like silicone. Although headache is typical in these cases, the literature lacks information about its characteristics and pathophysiology in ASIA.
Objective
Describe and discuss a case of ASIA with headache as the main symptom, approaching its immunopathological and clinical aspects.
Case
A 45-year-old woman with silicone prosthesis presented an intense headache, accompanied by chronic fatigue, sleep and skin disturbances, paresthesia, and generalized body aches. She had sought medical care over the past 2 years, with no underlying identified until then. The fulfillment of ASIA diagnostic criteria and subsequent prosthesis explantation resulted in symptom resolution.
Conclusion
Additional research is required to understand the semiotic patterns linked to ASIA syndrome-induced headaches. A dysregulation in histaminergic GPCR and GRK levels is hypothesized by the authors to be involved in the main mechanisms responsible for leading the headache nociceptive storm induced in ASIA.
Keywords
Introduction
ASIA syndrome (Autoimmune/inflammatory Syndrome Induced by Adjuvants) refers to a group of immune-mediated diseases that can affect genetically predisposed individuals exposed to immunoadjuvant compounds, 1 which include silicone, aluminum, squalene, pristane, methacrylate, and hyaluronic acid, often found in mineral oils, reconstructive/aesthetic prostheses, orthopedic and dental implants, intrauterine devices, tattoo inks, and certain vaccines. 2 Such substances promote hyperactivation of the innate and adaptive immune system, resulting in a high titer of antibodies, increased local immunogenic reactivity, and the release of inflammatory chemokines. The symptoms of ASIA can differ between individuals and the type of molecule that triggers the disorder. 3 Typical manifestations include arthralgias, myalgias, pyrexia, cognitive decline, sleep disorders, fibromyalgia, chronic fatigue syndrome, and neurological symptoms. 1 Cardiological symptoms like orthostatic intolerance and tachycardia may also develop. Current literature states these manifestations begin on average 13.4 (2.92) years after exposure to the agent. 4 The diagnostic criteria were proposed 1 and are described in Table 1.
Schoenfeld's major and minor criteria for the diagnosis of ASIA syndrome.

ASIA: autoimmune/inflammatory syndrome induced by adjuvants.
A growing collection of evidence has been mediating a statistical correlation between silicone, a siloxane polymer highly used in breast prostheses, and induced immunological dysfunctions with the development of autoimmunity.5,6 However, conflicting studies and opinions of experts persist regarding the causal relationship and pathophysiological mechanisms of silicone in precipitating these immune conditions.6–8 In this scenario, the current scientific evidence is insufficient to conclusively determine the safety of silicone breast implants (SBI) and their precise association with the adverse effects commonly presented. 7
We present a case of a patient who developed refractory headache, chronic fatigue syndrome, erythematous and pruritic plaques, and generalized weakness as a result of ASIA due to SBI, with prosthesis removal resulting in symptom resolution. This article provides a relevant description of the semiological and therapeutic patterns of ASIA-induced headaches and correlates them with recent findings in the literature that may justify the presence of persistent cephalalgia in this case. This article was approved by the Ethics Committee of the Federal University of Parana and the study's patient provided written informed consent. Additionally, this article was prepared following the Case Reports (CARE) Guidelines. 9
Patient information
A 45-year-old woman systems analyst sought medical attention due to a severe holocranial nocturnal headache (Visual Analogue Scale/VAS-10). In the beginning, the crises woke the patient up during sleep, lasting a few days, and were accompanied by tinnitus and visual disturbances, with asymptomatic periods, being initially characterized as an infrequent episodic headache, likely episodic migraine. The cephalalgia progressed, becoming chronic and daily, refractory to prophylactic and acute migraine medications, changing semiological patterns and with the emergence of associated symptoms. Therefore, it was reclassified as a secondary headache 10 (Table 2).
Semiological manifestations of headache in ASIA syndrome.

Associated symptoms included generalized muscle weakness, cold intolerance, sweating, mental slowness, body aches, sudden and transient hemiparesis (2 days) recurrent several times associated with ipsilateral allodynia and pain. Headache and neurological symptoms were always more pronounced when the patient presented dermatological manifestations such as erythematous-pruritic plaques and papules, diffusely spread across the upper and lower limbs, abdomen, chest, and face (Figure 1). The symptoms presented paroxysmal periods of exacerbation alternating with milder ones. The patient denies alcohol consumption, smoking, or use of illicit drugs. The extensive list of medications utilized should be noted. As prophylactic agents for headache: amitriptyline, propranolol, metoprolol, topiramate, flunarizine, botulinum neurotoxin type A two applications, anesthetic blocks of peripheral nerves without clinical response. Medications for potential preventive pain treatment were individually administered following a principle of a minimum duration of 3 months, with dosages escalated until deemed efficacious or until compromising adverse effects were experienced. Acute treatment used by the patient included: sumatriptan, naratriptan, rizatriptan, dihydroergotamine derivatives, simple painkillers, anti-inflammatories, and codeine with no results. Medications for managing acute cephalalgia treatment were also individually prescribed during 4–5 daily attacks, with replacement by a different medication, from the same class or not, in cases of ineffectiveness.

Dermatological manifestations during acute exacerbations in ASIA syndrome. Confluent lesions in patches interspersed with blistering, pruritic, associated with significant discoloration (erythematous) located in (A) anterior chest, (B) facial, (C) upper limbs, and (D) abdomen. The recurrent symptoms, along with the normal results of neurological investigations, led the patient to stop seeking emergency services, as she was often mistakenly diagnosed with a psychiatric illness. ASIA: autoimmune/inflammatory syndrome induced by adjuvants.
Regarding the pre-event cephalalgia status, the patient did not experience any notable headaches before the onset of the condition and also had no family history of migraines. Past medical history revealed breast augmentation for the first time in 2000, involving the use of a silicone-filled prosthesis with a smooth outer shell. In 2013, the SBI was changed, maintaining the same characteristics as the previous one, with the subsequent complication of seroma of the left breast. In 2017, the patient was diagnosed with right ductal carcinoma in situ and underwent a new silicone implant replacement for reconstructive purpose after surgical treatment, this time with a textured surface. In the same year, the symptoms of ASIA began. The patient had sought for several medical care since then, mistakenly receiving a diagnosis of factitious syndrome.
Physical, neurological examination, and complementary exams—such as Cerebral Magnetic Resonance Angiography Arterial and Venous phases—were within normal limits. The brain magnetic resonance imaging did not exhibit any nonspecific white matter lesions or signs of idiopathic intracranial hypertension. Mini-Mental State Examination score—30 points. Evaluation by three psychiatrists did not find criteria for active diseases. Tilt Test Examination demonstrated Vasovagal Dysautonomia associated with postural orthostatic tachycardia syndrome (POTS). No altered immunoglobulins IgE and IgG levels were detected. Bone marrow aspiration puncture and bone marrow immunophenotyping by flow cytometry were normal. The hypothesis of systemic mastocytosis was abandoned after a normal tryptase measurement. The clinical picture deteriorated over time, with a reduction in the time interval between episodes of worsening pain, generalized weakness, difficulty in maintaining daily activities, cognitive decline, and unintentional weight loss.
The explantation of the SBI brought significant improvement. Five days after the procedure, she began to experience relief from her symptoms. Medical reassessment conducted 40 days post-explant found the patient had no headaches, a body weight gain of 6 kg, normal speech, and no changes in sensitivity, gait, or coordination. The clinical presentation did not suggest POTS, with the heart rate measurements in supine, upright sitting, or standing positions showing no greater changes than 30 beats per minute. Moreover, there was a significant improvement in the symptoms of depression. Reassessment 12 and 24 months after the breast explantation revealed that the patient was completely asymptomatic.
Discussion
The patient described in this report satisfies the Shoenfeld criteria for the diagnosis of ASIA. 1 The adjuvant action in SBI often causes adverse effects such as paresthesia, night sweats, morning stiffness, xerostomia and xerophthalmia, cognitive deficits, depression, fatigue, myalgia and arthralgia, alopecia, skin rashes, sleep disorders, tachycardia, hearing abnormalities, allergic skin reactions, depression, hair loss, urinary and intestinal symptoms, consistent with the findings in this case.2,11 Hyperstimulation of the immune system associated with silicone can also be a stimulus for autonomic imbalances, inducing the appearance of chronic fatigue syndrome, fibromyalgia, small fiber neuropathy, and POTS, among others. 12 Although headache is common in these cases, 2 literature lacks information on its description and pathophysiology. To date, we have not found any material addressing the characterization of headaches, according to their frequency, pattern, evolution, and associated manifestations, in individuals with ASIA.
In their study, Watad et al. point to a prevalence of females (89%) and a mean age of 43 years among patients diagnosed with ASIA. 3 The reduction or disappearance of symptoms after implant removal, as reported in this case, constitutes a major indicator for diagnosing ASIA. Current literature documents that 75% of individuals experienced relief from silicone-associated complaints after the explant procedure. 2 The precise recovery period and symptom amelioration following explantation are not firmly established and may fluctuate based on the specific surgical procedure, disease attributes, length of time of implantation, and the type of adjuvant material utilized. 2 In line with our patient, Habib et al. present a case series in which women diagnosed with breast implant illness exhibited significant recovery 1 week after the prosthesis removal. 13
There is still a bipolar debate surrounding SBI and their real association with reported complaints.7,8 The time to diagnose ASIA is slow and challenging, lacking clinical management strategies and postoperative monitoring. 14 Due to non-specificity, clinical symptoms often do not meet the diagnostic criteria for well-defined immune-mediated rheumatic diseases, just as it is not irregular for laboratory tests not to detect significant abnormalities, directing to inefficient treatment of psychosomatic illnesses. 12 In addition, the overlap of factors such as diet, sex, chemical agents, microbiome condition, hormones, and the patient's psychosocial profile can lead to the emergence of correction biases and apparent causality. 7
The pathophysiological mechanism in ASIA has not yet been fully elucidated and comprises a complex interaction between genetic, environmental, immunological, and hormonal triggers. Current literature suggests that persistent exposure to silicone induces the activation of Th1/Th17 lymphocytes, promoting increased levels of IgE and IgG1, greater release of interleukins IL-1 and IL-6, and suppression of regulatory T cells. 15 In their observational study, Halpert et al. found a reduction in the levels of anti-β1 adrenergic, anti-angiotensin II type 1, and anti-endothelin receptors in women with SBI when compared with healthy women. 16 Borba et al. highlighted the presence of autoantibodies that act against the adrenergic and muscarinic G-protein receptors of the autonomic nervous system and the presence of fine fiber lesions in patient biopsy with SBI as a phenomenon that might serve as a common explanation for the manifestations of dysautonomia reported. 11
Hypothesis
Here we make a bold hypothesis that ASIA-induced headaches may be correlated with elevated histamine levels and subsequent activation of G-protein-coupled receptors (GPCR), as well as compromised functioning of G-protein receptor kinases (GRK) (Figure 2). Further research is necessary to substantiate this theory.
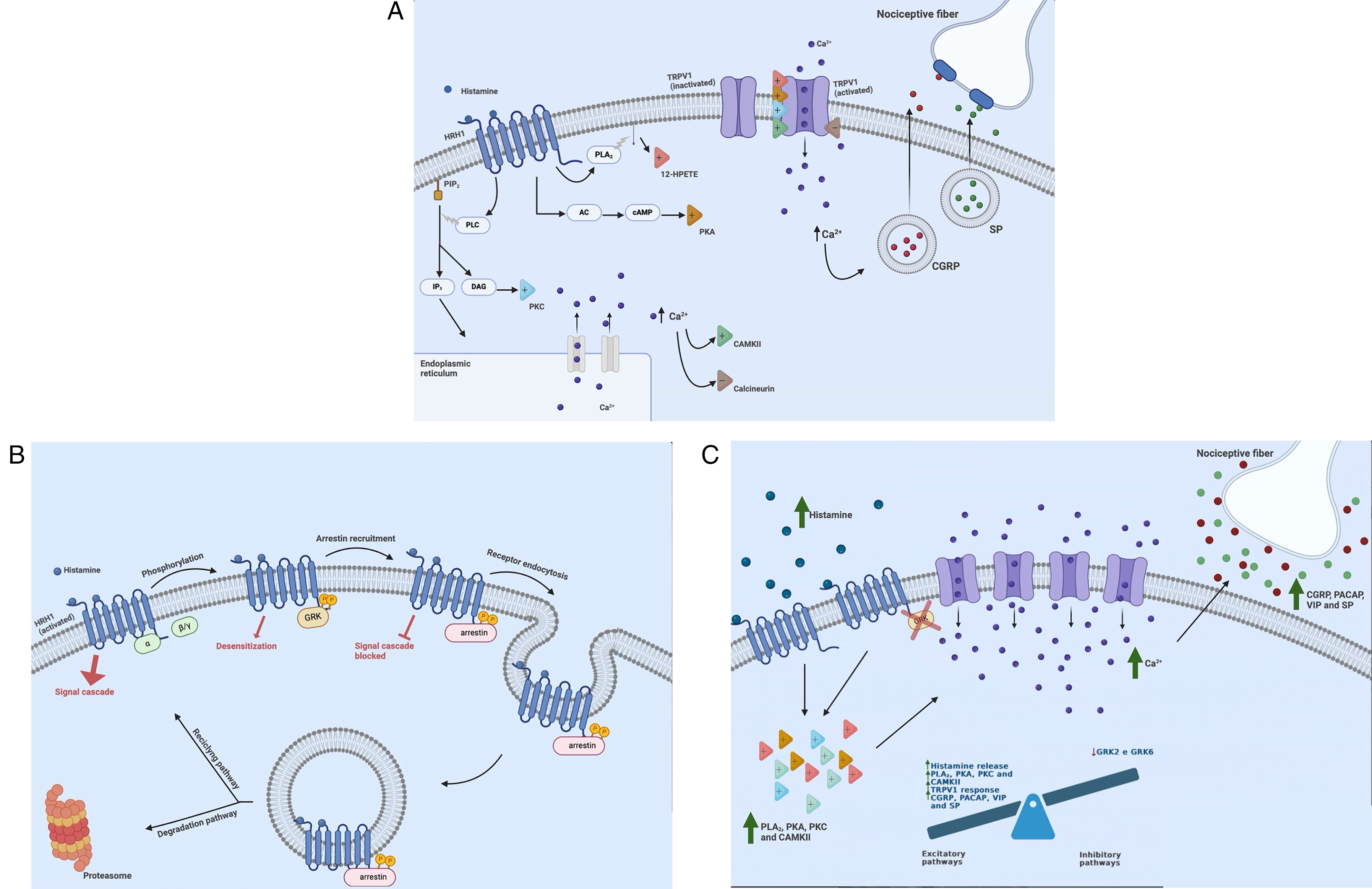
(A) Activation of histaminergic GPCRs and molecular signaling triggered. Upon stimulation, histamine receptors stimulate five main pathways, four excitatory (+) and one inhibitory (−), that modulate the activity of the TRPV1. The excitatory pathways operate via: (1) PLA2 enzyme, whose arachidonate products formed, particularly 12-HPETE, can act as agonists in capsaicin receptors; (2) PKA, whose signaling pathway involving the activation of adenylate cyclase and the subsequent increase in cAMP levels leads to phosphorylation of specific residues and interaction with other pathways; (3) PKC, phosphorylating peptide formed by association with DAG recently hydrolyzed by phospholipase C; and (4) CAMKII, through phosphorylation of specific residues and regulation of intracellular calcium levels. The inhibitory pathway operates via (5) calcineurin, whose dephosphorylated action plays a negative regulation role. The activation of TRPV1s, permeable ion channels, and the subsequent increase in intracellular calcium triggers the exocytosis of vesicles containing CGRP and SP. These neuropeptides are then released into the extracellular space and surrounding tissues, and act on nociceptive fibers, leading to sensitization that promotes pain, inflammation, and allodynia. GPCR: G-protein-coupled receptor; TRPV1: transient receptor potential vanilloid member 1; PLA2: phospholipase A2; 12-HPETE: 12-hydroxyeicosatetraenoic acid; PKA: protein kinase A; cAMP: cyclic adenosine monophosphate; PKC: protein kinase C; DAG: 1,2-diacyl-sn-glycerols; CAMKII: Ca2+/calmodulin-stimulated protein kinase II; CGRP: calcitonin gene-related peptide; SP: substance P. (B) Regulation mechanism of histaminergic GPCRs by GRKs and arrestins. The GRKs play a crucial role in regulating the response of GPCRs. Upon activation, GRKs specifically phosphorylate the activated GPCRs, leading to the inhibition of further G-protein coupling and subsequent intracellular signaling, a process known as desensitization. Facilitated by GRKs, arrestins then bind to the phosphorylated domains of the GPCRs and promote receptor internalization by recruiting a diverse set of endocytic machinery. Following internalization, the receptor may undergo recycling, returning to the plasma membrane, or degradation via the proteasomal system. GRK: G-protein receptor kinase; GPCR: G-protein-coupled receptor. (C) Pathophysiological hypothesis involving GPCRs and GRKs in ASIA syndrome-induced headaches. In ASIA syndrome cephalalgic patients, we hypothesize deregulation at the cellular level in nociceptive signaling. This deregulation is believed to be caused by hyperstimulation of excitatory pathways involving GPCRs and their downstream signaling, as well as impairment of inhibitory pathways mediated by GRKs. GPCR: G-protein-coupled receptor; GRK: G-protein receptor kinase.
Histamine plays a crucial role in the inflammatory response to biomaterials. 17 It exposes P1 and P2 epitopes of fibrinogen, promoting enhanced phagocyte adhesion and a widespread inflammatory cascade.17,18 Additionally, the activation of histaminergic GPCRs, particularly types 1 and 2 (HRH1 and HRH2), initiates an intracellular cascade through various pathways, leading to transient receptor potential vanilloid member 1 (TRPV1) activation and sensitization (Figure 2(A)), 19 one of the principal activation mechanisms for the origin of pain, as observed in patients with migraine. 20 HRH1 activation triggers protein kinase C (PKC) via phospholipase C activation and inositol trisphosphate (IP3) formation, while HRH2 activation induces protein kinase A (PKA) via elevated cyclic adenosine monophosphate (cAMP) levels. 21 Moreover, histamine also has the potential to be converted into 4-methyl spinaceamine, a molecule housing a histamine-like aromatic ring. It can thus possess HRH1 agonistic properties, thereby participating in the activation of TRPV1 receptors previously sensitized by PKC.19,21
GRKs and arrestins are pivotal in modulating GPCR signaling cascades. GRKs phosphorylate active GPCRs and subsequently recruit arrestins for receptor desensitization and internalization, comprising a precise cellular regulation mechanism (Figure 2(B)). 22 GRK2 controls the expression of HRH2 and pituitary adenylate cyclase-activating peptide (PACAP), 23 while GRK6 is the primary regulator of GPCRs associated with nociception and pain. 24 Since GRK6 is highly expressed in lymphatic tissue, it plays a crucial role in regulating chemokine receptor signaling and directing leukocyte chemotaxis and extravasation. During acute inflammatory situations, its expression levels decrease in secondary lymphatic organs and peripheral blood lymphocytes. This lowering appears to mainly underlie a post-transcriptional regulatory mechanism and was propagated by pro-inflammatory cytokines. 24
GRK6 knockout studies have demonstrated that its deficiency is associated with hyperalgesia and allodynia. 24 GRK6 also controls the CGPR of purinergic receptors (P2Y1, P2Y12), responsible for platelet aggregation, and indirectly can also be a generator of nociception. Based on the cellular mechanisms in the nervous system and its role in pain perception and inflammation, we hypothesize that deregulation of GPCRs due to excessive histamine stimulation, as well as compromised function of GRKs—specifically GRK2 and 6 by transcriptional and post-transcriptional mechanisms—may contribute to chaotic nociceptive cellular excitability in the context of headaches in ASIA syndrome (Figure 2(C)).
Conclusion
We present a case of ASIA induced by SBI whose main manifestation was refractory headache. The patient presented a throbbing, pulsating, and stabbing pattern, with an association of dysautonomia, photophobia, and nausea. The in-depth analysis of the clinical description of headaches in ASIA reports, as demonstrated in this article, will enhance understanding of semiological patterns and is strongly recommended by the authors. We hypothesize that the hyperstimulation of GPCRs, mainly by histamine, might secondarily lead to an increase in ion channels and relevant neuropeptides associated with inflammation and pain sensation, such as calcitonin gene-related peptide, substance P, PACAP, vasoative intestinal peptide, serotonin, and glutamate, as well as the absence of GPCRs downregulation mechanisms caused by the reduction of GRKs, particularly types 2 and 6, might also be an underlying cause to this genuine nociceptive storm. There is a requirement for further research into the levels of GRKs in individuals affected by this immune-mediated syndrome.
Clinical implications
In ASIA syndrome, we hypothesized that the pathophysiological headache process may be related to a deregulation in GPCR and GRK pathways. This article represents the first attempt to gain a deeper understanding of the headache patterns associated with ASIA. The authors strongly recommend a complete description of the headache semiological picture in future studies.
Limitations and strengths of the study
Certain aspects of this study have limitations. Due to data collection through anamnesis, it is important to consider potential distorting factors associated with patient perception and memory. Just as the type of descriptive study is not suitable for determining cause-and-effect relationships, the sample from a single case does not cover the entirety of individuals with ASIA syndrome. Consequently, the report may not be applicable to all patients with this condition.
However, there are some strengths to this article that should be mentioned. These include the description of a rare condition of autoimmunity, the innovative focus on the semiological pattern of headaches secondary to ASIA, and the low cost of producing scientific information.
Footnotes
Acknowledgments
This article utilized generative AI tools with the specific aim of enhancing scientific writing and improving clarity in the language used. Critical evaluation of the authors was conducted after each AI intervention to ensure text accuracy.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and patient consent
This study was reviewed and approved by the Ethics Committee for Research Involving Human Subjects of Clinical Hospital of the Federal University of Parana (HC/UFPR), and the patient consented to the disclosure of her medical history through a consent form, also reviewed by the committee. The number of approval registered in Plataforma Brasil is 77627724.4.0000.0096.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
