Abstract
Neuronostatin (NST) is a peptide encoded by the somatostatin gene that serves important physiological functions in diverse tissues. Previous studies have shown that intracerebroventricular administration of NST induces antinociceptive effects and hyperalgesic effects as determined by the tail immersion assay and formalin test, respectively. In the present study, we aimed to evaluate the effects of intrathecal (i.t.) injection of NST on nociception in a model of visceral pain, and determine possible mechanisms of action in mice. NST (1, 3, 6, or 12 nmol) was administered to mice, leading to a dose‐dependent antinociceptive effect as determined by the acetic acid‐induced writhing test in mice. NST (1 nmol) also enhanced the antinociceptive effect of morphine (2.5 and 5 μg/kg) in the spine. Naloxone and β‐funaltrexamine hydrochloride significantly antagonized the antinociceptive effect of NST. The expression of G‐protein‐coupled receptor 107 (GPR107) protein and the phosphorylation of PKA at Thr197 were increased after i.t. administration of NST, suggesting that the μ‐opioid receptor and GPR107/PKA signaling pathway are involved in the analgesic response. In conclusion, i.t. injection of NST may potentially be used as a new approach in the mediation of visceral pain.
1 Introduction
Somatostatin is a peptide secreted by neuroendocrine cells in the gastrointestinal and central nervous system (CNS), and regulates diverse actions by binding to the five different isoforms of the somatostatin receptor [1, 2]. Neuronostatin (NST), also encoded by the somatostatin gene, plays important physiological roles in diverse tissues. It not only inhibits growth hormone and luteinizing hormone secretion in baboon (Papio anubis) primary pituitary cell cultures, but also suppresses food and water intake [3, 4]. In the CNS, NST is primarily expressed in the cortex and hypothalamic paraventricular nucleus, though it also induces c‐fos expression in the cerebellum and hippocampus [4, 5]. NST elevates intracellular Ca2+ concentration in hypothalamic neurons, significantly impairs memory retention, and induces depression‐like effects [5 –8]. Some of the effects of NST are mediated through G‐protein‐coupled receptor 107 (GPR107), as intracerebroventricular (i.c.v.) administration of GPR107 siRNA blocks the effect of NST in the elevation of mean arterial pressure [9]. Therefore, GPR107 represents a promising candidate receptor for NST.
The sensing of pain is complex, and mediated by many components. Nociceptors express transient receptor potentials at the nerve terminals, and sensory afferents transmit this signal to spinal nociceptive neurons, located in the dorsal horn of the spinal cord. This is followed by supraspinal integration and descending regulation of pain perception [10, 11]. The spinal and supraspinal injections of drugs may exert different effects; for example, intrathecal (i.t.) or i.c.v. administration of somatostatin produces analgesic effects as measured by the tail immersion acute test, and in chronic inflammatory pain [12]. I.c.v. injection of angiotensin II has no effect on the nociceptive response in the tail‐pinch test, but i.t. injection of angiotensin II leads to an antinociceptive effect in the tail immersion test [13, 14]. Therefore, it is possible that NST can regulate pain sensing at the spinal level.
Visceral pain is a complex disorder and difficult to manage effectively, as its signals and neurobiological mechanisms differ from the characteristics of somatic sensation [15, 16]. It has been reported that visceral noxious information could be transmitted through different neural pathways at spinal sites [15]. Intestinal afferent nerves convey sensory information of visceral pain to the CNS, and it is known that spinal activity plays an important role in the process [17]. Previous studies have shown that i.c.v. administration of NST induces antinociceptive effects as determined by the tail immersion test, as well as significantly enhances the antinociceptive effect of morphine in mice [18, 19]. In addition, NST can reduce the inflammatory infiltration of acute pancreatitis in rats [20]. However, the effects of NST in visceral pain have not yet been reported at the spinal level.
The present study focused on evaluating the effect of i.t. injection of NST on visceral nociception in the acetic acid‐induced writhing test in mice. Receptor antagonists and protein expression were analyzed to determine potential signaling mechanisms of NST.
2 Material and methods
2.1 Experimental animals
A total of 386 four‐month‐old male Kunming strain Swiss mice weighing between 18–22 g were utilized for this study. Mice were obtained from Lanzhou University Animal Center (China), housed in pathogen‐free facilities (5–6 mice per cage), and maintained in 12:12 h light/dark cycle conditions (lights on from 7: 00 a.m. to 7: 00 p.m.) at 22 ± 2 ℃ in a pathogenfree room. The animals were given access to food and water ad libitum, and were allowed to become accustomed to the laboratory environment for one week prior to the initiation of experiments.
Mice were randomly divided into two major groups, the behavioral test (n = 350) and Western blot (n = 36) groups. The behavioral test group (Group 1) was divided into subgroups were as follows: n = 12 for pain tests in every group; n = 11 for antagonist and GPR107 siRNA experiments; n = 10 for locomotor activity assays; n = 12 for morphine experiments; and n = 14 for naloxone tests, respectively. The Western blot group (Group 2) was divided into subgroups as follows: n = 6 in each experimental group.
All of the experimental manipulations were completed following the guideline recommendations for the care and use of experimental animals provided by Northwest Normal University Ethical Committee for Human and Animal Experimentation, as well as the guidelines from the China Council on Animal Care and the Guide for the Care and Use of Laboratory Animals (1996, published by National Academy Press, 2101 Constitution Ave. NW, Washington, DC 20055, USA). All pain testing procedures were performed by an observer double‐blinded to the study.
All the experiments were conducted in compliance with the ARRIVE guidelines.
2.2 Drugs and antibodies
Neuronostatin (Leu‐Arg‐Gln‐Phe‐Leu‐Gln‐Lys‐ Ser‐Leu‐Ala‐Ala‐Ala‐Ala‐NH2) used in this study was purchased from Hangzhou Chinese Peptide Biochemical Co. (Jiangsu, China), and was dissolved in artificial cerebrospinal fluid (CSF; 126.6 mM NaCl, 27.4 mM NaHCO3, 2.4 mM KCl, 0.5 mM KH2PO4, 0.89 mM CaCl2, 0.8 mM MgCl2, 0.48 mM Na2HPO4, and 7.1 mM glucose, pH 7.4). The naloxone hydrochloride dehydrates, β‐funaltrexamine hydrochloride (β‐FNA) and bicuculline methiodide, were purchased from Sigma (MO, USA). Morphine hydrochloride was acquired from Shenyang First Pharmaceutical Factory (Liaoning, China). These were dissolved in dimethyl sulfoxide (DMSO) and diluted in artificial CSF. The final concentration of DMSO was less than 0.1%. The following antibodies were purchased from Cell Signaling Technology Co. (MA, USA): PKAc (1:1000; Catalog number 4782); phospho‐PKAc Thr197 (1:1000; Catalog number 4781); and phospho‐Akt Ser473 (1:1000; Catalog number 9271). The GPR107 (1:1000; Catalog number 25076‐1‐AP) and Akt (1:1000; Catalog number 10176‐2‐AP) were obtained from Proteintech Co. (Hebei, China). GAPDH (Catalog number K106389P) was provided by Solarbio Co. (Beijing, China). Appropriate secondary antibodies were obtained from Solarbio Co. (Beijing, China).
2.3 Intrathecal injection
Intrathecal (i.t.) administration was performed manually as previously described [21]. The unanesthetized mice were held by a pelvic girdle after sterilization, and a 27‐gauge needle attached to a 25 μL Hamilton micro‐syringe was inserted between the L5 and L6 lumbar vertebrae. A sudden tail slight flick or “S” curl was used as the indicator for appropriate insertion into the subarachnoid space. Then, 5 μL of either drug or vehicle was administered intrathecally at a speed of 5 μL/10 s according to different experiment time. GPR107 siRNA (100 nmol; 5’ ‐GAUCGGAGUAAGAACGAUGGCUTT‐3’) was injected into the spinal cord (2 μL in total) at the same location. After two days, spinal cord tissues were collected for Western blot analysis. Intrathecal co‐injection was performed as previously described [22], and the antagonists were administered i.t. 10 min prior to injection of NST with different syringes. Morphine was co‐injected i.t. with NST in accordance with the method described previously [18]. Following completion of the behavioral experiments, the placement of cannula was verified by administration and localization of a 1% methylene blue dye, which diffused into the proximal and distal regions of the spinal cords, but was not found in the brain tissue following anatomical dissection.
2.4 Acetic acid‐induced writhing test
Chemical‐induced visceral pain was evaluated using the acetic acid‐induced writhing test as described previously [22]. Drugs or vehicle CSF were administered 5 min prior to the administration of acetic acid. A 1% acetic acid solution (10 mL/kg) was intraperitoneally (i.p.) injected according to the method used in a previous study [23]. The mice were placed in individual cages for observation. The number of writhes or abdominal constrictions, such as elongation of the body and extension of the forelimb, development of tension in the abdominal muscles, and arching of the back, were considered visceral pain reactions. These behaviors were recorded during a 30‐minute period. In order to avoid unnecessary pain to the experimental animals, the durations of experiments were as short as possible, and the mice were immediately anesthetized following the treatment period.
2.5 Locomotor activity test
The spontaneous locomotor activity of mice was examined and moderated by a ZZ‐6 Autonomous Movement Instrument for Mice (Chengdu TME Technology Co. Ltd., China) in accordance with the previously described method [8]. This instrument is comprised of separated rooms; the mice were habituated in the apparatus for 10 min prior to i.t. administration, and then immediately placed into their former rooms to measuring their locomotor activities at 5‐min intervals for 30 min. The sum of both horizontal and rearing movements were recorded. After each test session, the apparatus was cleaned to minimize influence of any odors, such as the urine and feces of the previously examined mouse.
2.6 Western blot analysis
10 min after NST or artificial CSF administration, mice were anesthetized with sodium pentobarbital (90 mg/kg, i.p.) and the lumbar segment of spinal cords were quickly removed. The spinal dorsal horns were dissected from this tissue and homogenized in a 10‐fold excess volume of ice cold RIPA protein lysis buffer (Solarbio, Beijing, China), which contained 1 mM PMSF, 0.1% (v/v) 2‐mercaptoethanol, and a 1:100 dilution of protease inhibitor cocktail. The homogenates were left on ice and centrifuged at 14 000g for 15 min at 4 ℃. Supernatants were collected, and protein concentrations were determined using the Bradford method [24]. 30 μg of total protein extracts were boiled for 5 min and separated using a 7.5% to 10% SDS‐PAGE run in SDS buffer [25 mM Tris‐HCl, pH 8.5, 192 mM glycine, 0.1% (w/v) SDS] for 60–100 min at 150 V, depending on the percentage of the gel. The proteins were transferred to nitrocellulose membranes (Whatman, Maidstone, UK) using a wet transfer system in transfer buffer (25 mM Tris‐HCl, pH 8.5, 192 mM glycine, 20% methanol) for 90 min at 100 V. The membranes were incubated with primary antibodies in 0.5% BSA/1× TBST overnight at 4 ℃ after blocking with 10% nonfat milk. The specimens were then immunoblotted with corresponding horseradish peroxidase‐conjugated secondary antibodies (1:5 000) for 1.5 h at room temperature. Blots were visualized using an enhanced chemiluminescence (ECL) reagent, detected using a ChemiDoc MP Imaging System (Bio‐Rad, Hercules, California, USA), and quantified using ImageJ software.
2.7 Experimental design
In order to investigate the effects of NST on acetic acid‐induced visceral pain in mice, NST was administered i.t. at a dose of 1, 3, 6, or 12 nmol/mouse, respectively, 5 min prior to acetic acid injection. Control animals received no NST, but artificial CSF. In order to evaluate whether synergistic analgesic effects between NST and morphine were present at the spinal level, the mice were injected with morphine (2.5, 5, or 10 μg/kg) alone or co‐administered (i.t.) with the minimal analgesic dose of NST (1 nmol/mouse), respectively. Dosages of NST were based on the available literature and this study’s preliminary experimental results [19, 25].
For the purpose of determining pain mechanisms affected by NST, the γ‐aminobutyric acid receptor antagonist bicucullin (1.086 nmol/ mouse), classical opioid receptor antagonist naloxone (10 nmol/mouse), and the μ‐opioid receptor antagonist β‐FNA (10 nmol/mouse) were used. The dosages of these antagonists were chosen in accordance with previous related studies [18, 19, 25].
To determine the role of GPR107 in the antinociceptive effect induced by NST, GPR107 siRNA (100 nmol/mouse) was administered i.t. 2 days prior to NST injection. The dosage of GPR107 siRNA was chosen according to previous results [8]. To further examine pain mechanisms, Western blot was used to examine signaling mediators of the GPR107/PKA/Akt signaling pathway after i.t. administration of NST.
2.8 Statistical analysis
All data are represented as means ± standard error of mean (SEM), and were assessed using SPSS software version 19.0 (SPSS Inc., Chicago, IL, USA). The statistical methods were chosen in accordance with previous related studies [8, 18, 19, 25]. The data obtained from behavioral and Western blot assays were assessed by one‐way or two‐way ANOVA followed by post hoc analysis using Dunnett’s test. The differences between two groups were determined using an unpaired, two‐tailed Student’s t test. P values < 0.05 were considered statistically significant.
3 Results
3.1 NST produced antinociception in the acetic acid‐induced visceral pain model and enhanced analgesia caused by morphine
I.t. administration of NST (1–12 nmol/mouse) decreased the number of writhes in a dosedependent manner in the acetic acid‐induced writhing test of visceral pain [Fig. 1(A)]. The total number of contractions was statistically reduced at doses of 3, 6, and 12 nmol NST/mouse when compared with control group (P < 0.05, P < 0.01, P < 0.05, respectively). The most effective dose was 6 nmol/mouse. In order to determine whether this analgesic effect was dependent on the opioid receptor or GABA system, naloxone, β‐FNA, or bicuculline was co‐injected i.t. with NST. Naloxone and β‐FNA both significantly antagonized the reduction in the total number of writhes caused by NST (P < 0.01 and P < 0.05, respectively) [Fig. 1(B) and (C)]. However, naloxone or β‐FNA alone did not affect the number of writhes. These results demonstrated that the μ‐opioid receptor was involved in the antinociceptive effect of NST in the visceral pain model. However, i.t. injection of bicuculline alone, or in combination with NST, did not change the total number of writhes, suggesting that the GABAA system was not involved in this process [Fig. 1(D)]. Furthermore, GPR107 siRNA significantly eliminated the reduction in the number of writhes when compared with NST treatment [Fig. 1(E), P < 0.01]. In order to exclude the effect of motor ability on pain‐related behaviors, the locomotor activity was determined. The 30‐min spontaneous locomotor activity counts at 5‐min intervals were not found to be statistically different among all treatment groups [Fig. 1(F)], indicating that the abdominal constrictions were not caused by locomotor inhibition.
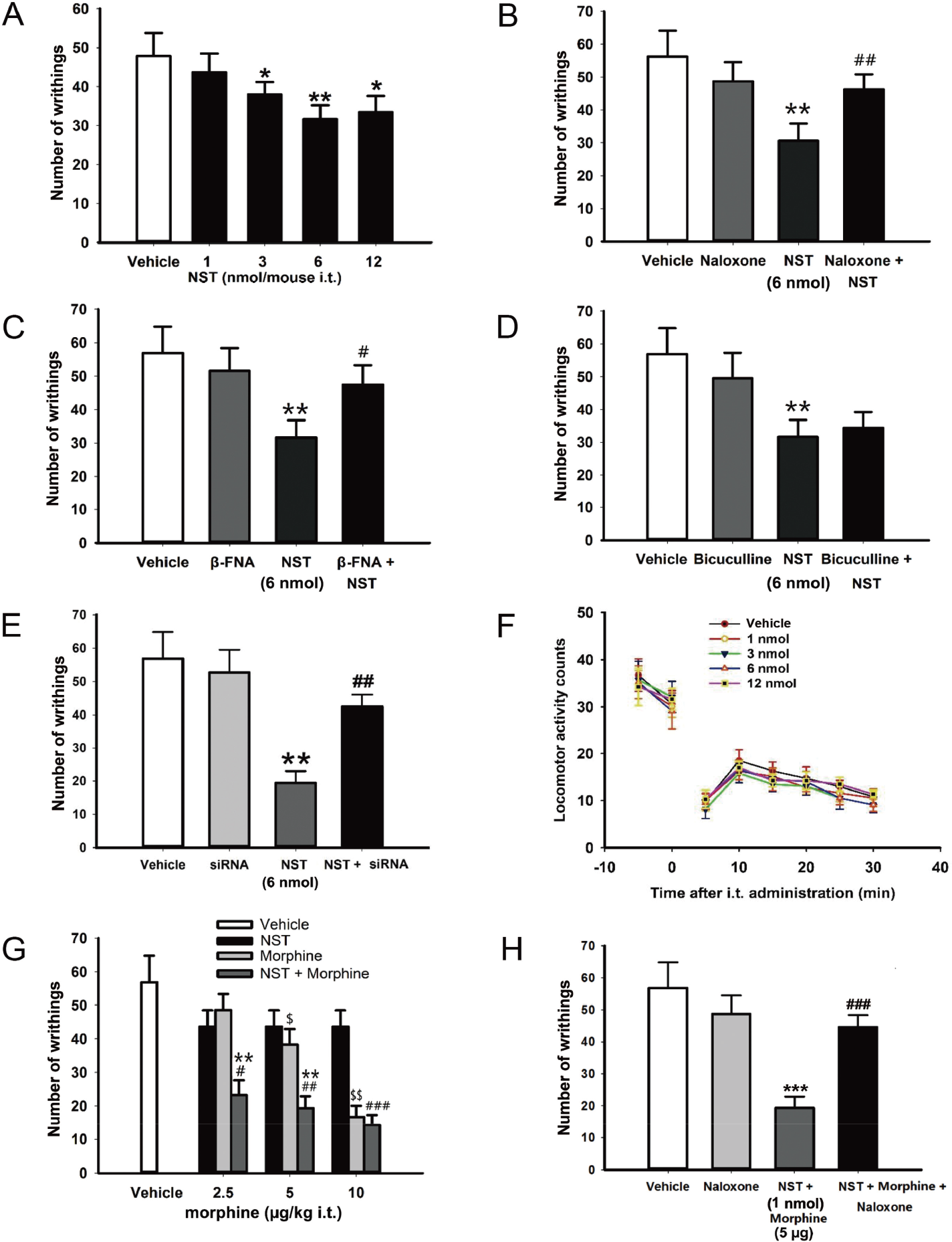
Effect of i.t. injection of neuronostatin (NST) on nociception in the acetic acid‐induced writhing test. (A) Numbers of writhes in the acetic acid‐induced writhing test are shown in response to different doses of NST (1, 3, 6, and 12 nmol/mouse). All data are presented as means ± SEM of n = 12 mice per group. * P < 0.05 and ** P < 0.01 when compared with the vehicle group. Number of writhes in the acetic acid‐induced writing test following i.t. co‐administration of (B) opioid receptor antagonist naloxone (10 nmol/mouse), (C) μ‐opioid receptor antagonist β‐FNA (10 nmol/mouse), (D) γ‐aminobutyric acid receptor antagonist bicuculline (1.086 nmol/mouse), and (E) GPR107 siRNA (100 nmol) with NST (6 nmol/mouse), respectively (n = 11 mice per group, ** P < 0.01 when compared with the vehicle group, and ## P < 0.01 when compared with NST group). (F) Locomotor activity was recorded following NST treatment, in which complete time‐courses at 5‐min intervals for 30 min were calculated (n = 10 mice per group). (G) Antinociceptive response to morphine following co‐injection (i.t.) of NST (1 nmol/mouse) and morphine (2.5 and 5 μg/kg) in the acetic acid‐induced writhing test (n = 12 mice per group, $ P < 0.05 and $$ P < 0.01 when compared with the vehicle group; ** P < 0.01 indicates difference between NST + morphine versus morphine alone; # P < 0.05 ## P < 0.01, and ### P < 0.001 indicate statistically significant differences between the NST + morphine versus NST). (H) Effect of i.t. administration of naloxone (10 nmol/mouse) on the synergistic analgesic response of i.t. co‐injection of NST (1 nmol/mouse) and morphine (5 μg/kg) in the writhing test (*** P < 0.001 when compared with the vehicle group; ### P < 0.001 indicates statistically significant difference between NST + morphine versus NST + morphine + naloxone, n = 14 mice per group). Data were assessed using one‐way or two‐way ANOVA followed by post hoc analysis using Dunnett’s test. Difference between two groups was determined by the unpaired, two‐tailed Student’s t test. NST, neuronostatin; β‐FNA, β‐funaltrexamine hydrochloride; Vehicle refers to artificial CSF; Nal, naloxone; BIC, bicuculline.
The results of a previous related study revealed the synergistic analgesic effects of NST and morphine at the supraspinal level in the tail immersion test [18]. NST (1 nmol/mouse i.t.) markedly enhanced the antinociceptive effect of morphine (2.5 or 5 μg/kg) in the acetic acidinduced visceral pain model in conscious mice compared to morphine alone (P < 0.01) [Fig. 1(G)]. However, it was observed that NST (1 nmol/mouse) did not improve the antinociceptive effect induced by the highest dose of morphine (10 μg/kg). Moreover, the analgesic effects of i.t. co‐injection of NST (1 nmol/mouse) and morphine (5 μg/kg) was inhibited by naloxone, but naloxone (10 nmol/mouse) alone did not influence the analgesic response [Fig. 1(H)].
3.2 NST increased GPR107 protein expression and PKA phosphorylation in the spinal dorsal horn
In order to identify the pain mechanisms induced by i.t. administration of NST in mice, GPR107 expression and PKA phosphorylation in the spinal cord were analyzed. GPR107 protein expression levels were significantly increased following i.t. injection of NST in a dosedependent manner when compared with the vehicle group (3 nmol NST, P < 0.05; 6 and 12 nmol NST, P < 0.01; Fig. 2). PKA was consistently phosphorylated at the Thr197 residue, which increased in the spinal dorsal horn after i.t. administration of NST in mice when compared with vehicle‐treated group (6 nmol NST, P < 0.01; and 12 nmol NST, P < 0.05). However, there was no difference in the phosphorylation of Akt at Ser473 between the NST and vehicle‐treated groups (P > 0.05). I.t. injection of GPR107 siRNA significantly prevented PKA phosphorylation and GPR107 expression induced by NST. These results indicate that the analgesic response induced by i.t. administration of NST is mediated via the GPR107/PKA signaling pathway in the spinal dorsal horn.
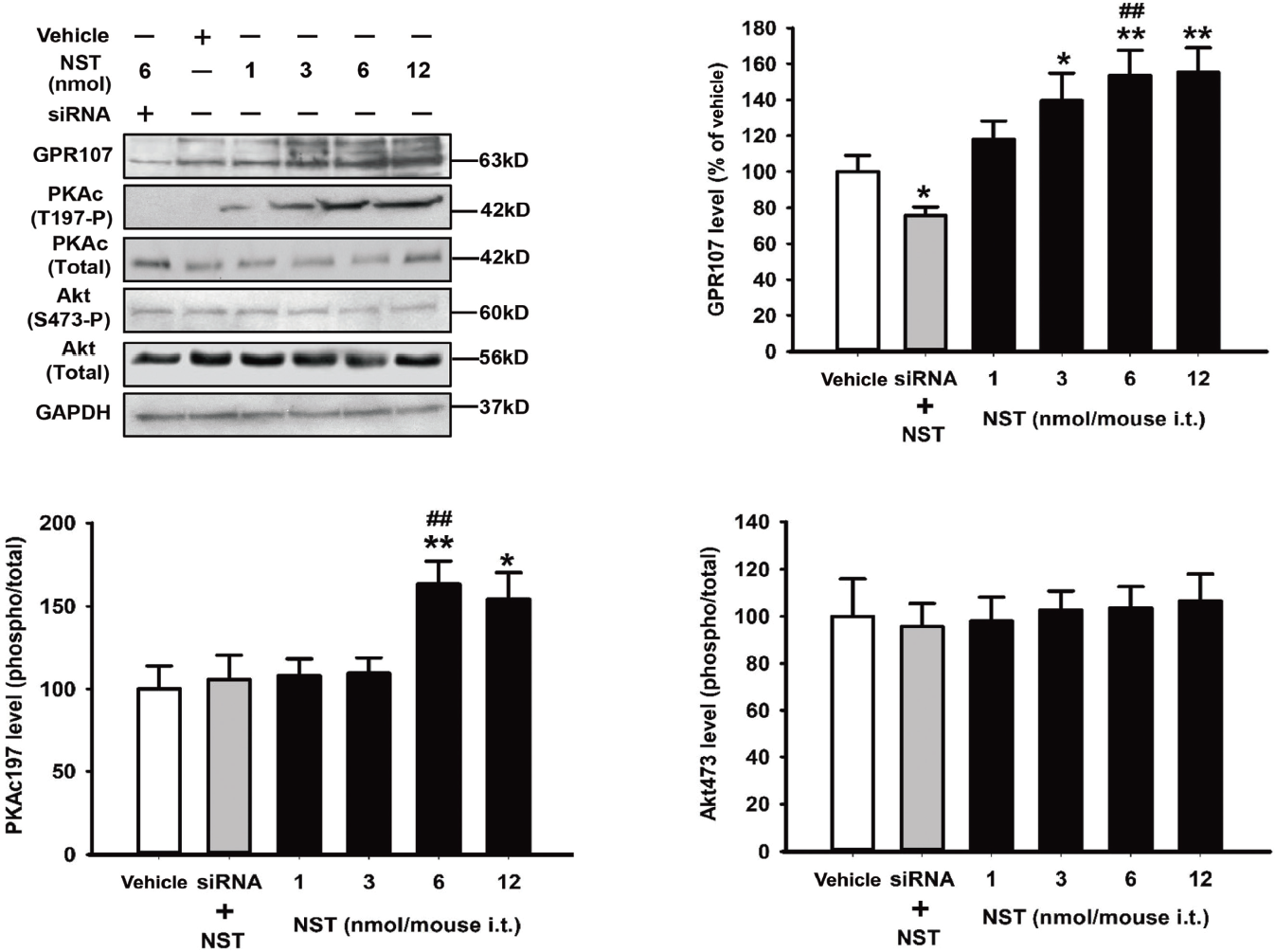
Effect of i.t. administration of neuronostatin (NST) on the GPR107/PKA/Akt signaling pathway in the spinal dorsal horn. NST was administered i.t. After 10 min, L4–L5 spinal dorsal horn samples were collected and immunoblotting analysis was performed. Band densitometry quantification of GPR107 was normalized to GAPDH levels; band densitometry quantification of the ratio between phosphorylated (phosphorylation of PKA at Thr197 and Akt at Ser473 site) and total protein levels (PKA and Akt) are presented at the right, or expressed as a percentage of the vehicle (artificial CSF) in lower panels. The data are represented by means ± SEM (n = 6 mice/group); * P < 0.05 and ** P < 0.01 compared to the control using one‐way ANOVA followed by post hoc analysis using Dunnett’s test.
4 Discussion
The present study examined the effects of i.t. administration of NST on visceral nociception in mice. The results show that NST has an antinociceptive effect using the acetic acidinduced writhing test, and that the endogenous opioid system and GPR107/PKA signaling pathway are involved in this antinociception.
Visceral pain is closely related with visceral hypersensitivity, due to the sensitization of afferent nerve pathways. Intestinal afferents convey sensory information of visceral pain to the CNS, and the activities of spinal neurons play an important role in the process [17]. Acetic acid‐induced visceral pain is a suitable model for visceral hypersensitivity, as it induces transient low‐level inflammation [15, 16]. NST had an antinociceptive effect in the acetic acid‐induced writhing test at the L4 and L5 dorsal root ganglia, where NST is widely expressed (data not shown), indicating that exogenous NST contributes to pain sensing at the spinal level. However, the visceral region is also innervated by neurons located in the L6 dorsal root ganglia; NST may regulate visceral pain sensations located in the L6 dorsal root ganglia which project to the L4 and L5 spinal cord segments [26]. It was observed that the most effective analgesic response in the acetic acid‐induced visceral pain model was evoked by the dose of 6 nmol/mouse NST, and there were no further increases in analgesia caused by either higher or lower doses of NST. A bell‐shaped dose response curve was observed for NST, which was similar to those reported for other bioactive peptides and for the mean arterial pressure increase caused by NST [4, 8, 18, 19, 25]. These phenomena suggest that NST has different physiological activity in different doses, and activity may also depend on the number of receptors present in a specific tissue. However, this hypothesis requires further confirmation.
It has been well documented that the opioid system modulates nociception at multiple sites in the CNS, including the spinal cord, midbrain, and thalamus [27 –29]. Peripheral tissue trauma elicits an increase in production and release of the two opioid peptide families, dynorphin and enkephalin, in the dorsal horn of the spinal cord [22, 30]. Previous studies have also indicated that different actions induced by NST are mediated by opioid receptors [18, 19, 25]. Coincidentally, it was observed that naloxone significantly attenuated the antinociception effect in the acetic acid‐induced writhing test, and the μ‐opioid receptor antagonist β‐FNA was also able to block the reduction in the amount of writhing induced by NST, suggesting that the endogenous opioid system is involved in modulation of the visceral pain response at the spinal level. Therefore, the interaction between NST and endogenous opioid system is a critical event in the modulation of analgesia. We found that naloxone could effectively abolish the enhanced analgesic response of morphine, suggesting that opioid receptors were involved in improved analgesia induced by NST. These results indicated that NST dampened visceral pain effects on nociception via the μ‐opioid receptor at the spinal level.
Morphine is a potent analgesic, used in the long‐term treatment of acute and chronic pain, that acts via the opioid receptor in the spinal cord. However, morphine can be highly addictive, and has displayed negative side effects when administered repeatedly or used at high doses [31]. Previous studies have revealed that NST enhances the antinociceptive effect of morphine at the supraspinal level [18]. The present study has indicated that i.t. administration of NST could improve the analgesia caused by a lower dose of morphine (2.5 or 5 μg/kg). Therefore, a cascade amplification mechanism may potentially exist to modulate pain following co‐administration of NST and morphine in the CNS. On the other hand, i.t. co‐injection of a high‐dose of morphine (10 μg/kg) and NST did not improve the antinociceptive effect of morphine alone. These findings were confirmed by the saturation phenomena of classical opioid receptors, since there were insufficient opioid receptors for the highest dose of morphine and NST [22].
I.t. administration of NST led to increases in GPR107 protein expression and phosphorylation of PKA in the spinal cord. GPR107 is considered to be a candidate receptor for NST [8, 32], indicating that GPR107 may be implicated in the regulation of pain by NST downstream of the PKA signaling pathway. Furthermore, this study also revealed that i.t. injection of GPR107 siRNA not only prevented PKA phosphorylation, but markedly decreased the analgesic effect induced by NST. These results indicated that i.t. administration of NST led to antinociceptive effect in the visceral nociception through the GPR107/PKA signaling pathway in addition to the opioid receptor. NST might also increase the affinity of morphine for the opioid receptors through the GPR107/PKA signaling pathway in the visceral pain response. This hypothesis requires further confirmation.
5 Conclusions
In conclusion, this study demonstrates that i.t. administration of NST induces antinociception in the acetic acid‐induced writhing test. This effect is mediated by the μ‐opioid receptor and the GPR107/PKA signaling pathway. This study also shows that NST significantly enhances the antinociceptive effect of morphine in the writhing test, a response that is also mediated by the opioid system. This indicates that i.t. injection of NST could potentially be used as a new approach in mediating visceral pain.
Footnotes
Conflict of interests
The authors state that there is no conflict of interest in this study.
Acknowledgments
This research study was funded by the Natural Science Foundation of Gansu Province (Grant No. 18JR3RA101), Youth Science and Technology Talents Lifting Project Foundation of Gansu Province, and the Doctoral Launching Foundation of Northwest Normal University.
Author contributions
The conception and design of this research project were completed by S Yang. The experiments were performed by TJ Shao, P Yu and S Yang. The analyses and interpretations of the data were conducted by TJ Shao, S Yang, and P Yu. S Yang completed the drafting of the manuscript.
