Abstract
Background:
Indomethacin is a non-steroidal anti-inflammatory used to diagnose and treat hemicrania continua and paroxysmal hemicrania. Treatment can be complicated by gastrointestinal adverse effects; less commonly reported are idiosyncratic neuropsychiatric adverse effects with indomethacin.
Methods:
A 50-year-old male with lateralized brief attacks of headache associated with cranial autonomic symptoms was administered a single 200 mg dose of intramuscular indomethacin. Within an hour, he developed acute psychosis, with paranoid delusions and verbal and physical aggression lasting 5 h, followed by recovery to baseline. We used search terms “indomethacin psychosis,” “indomethacin psychiatric,” “indomethacin side effects,” “non-steroidal anti-inflammatory psychosis,” and “non-steroidal anti-inflammatory psychiatric” within PubMed to identify previous reports and literature in this area.
Results:
Neuropsychiatric adverse effects of indomethacin have been reported since 1965 in a dose-dependent manner, usually with oral courses. They may be more common in the elderly, postpartum women and postoperative patients.
Conclusion:
Neuropsychiatric adverse effects should be considered in headache medicine, particularly in at-risk groups when indomethacin is administered. Patients, particularly those at highest risk, should be counseled about the risk of neuropsychiatric side effects on indomethacin which may be dose-dependent and are generally reversible on stopping the drug.
Introduction
Indomethacin is a non-steroidal anti-inflammatory drug (NSAID) with anti-pyretic, anti-inflammatory, and analgesic properties. 1 The drug is used in headache medicine in the diagnosis and treatment of paroxysmal hemicrania (PH) and hemicrania continua (HC), the so-called “indomethacin-sensitive headache disorders.”
The mechanism of action of indomethacin in PH and HC is not well understood. NSAIDs penetrate the blood-brain barrier, as they are highly lipophilic, and indomethacin is present in the cerebrospinal fluid after just a single intramuscular dose. 2 The absolute response of PH and HC to indomethacin, and less so to other NSAIDs, 3 has been of neurobiological and therapeutic interest in headache research. Unfortunately, adverse gastrointestinal effects often result in prolonged indomethacin treatment being poorly tolerated. Other side effects of indomethacin, in particular central nervous system (CNS) effects, are recognized but less well reported. These include fatigue (1–3%), dizziness (3–9%), mood change (1–3%), and even less commonly in less than 1%, neuropsychiatric disturbance, including hallucinations, nightmares, and confusion. 2
Use of the inpatient intramuscular placebo-controlled double blind indomethacin test (the INDOtest) using 100–200 mg has been used by the authors to ascertain patients’ response to indomethacin and therefore accurate diagnosis. 4,5 Patients are typically admitted for 48 h, with hourly headache diaries starting a week prior to admission. Intramuscular indomethacin is administered as two separate intramuscular injections on one day, and two placebo injections are administered at the same time on the other day, in a double-blinded, randomized crossover fashion (both the patient and diary interpreting clinican are blinded to what is being administered on each day). Hourly headache diaries are reviewed before and after injections by a blinded clinician, in order to make an unbiased headache diagnosis.
We report here a case of a male who developed an acute neuropsychiatric adverse event in response to a single dose of intramuscular indomethacin during an inpatient INDOtest.
Case
We reviewed a 50-year male in clinic, who had presented to an autonomic neurologist with an 8-year history of recurrent multiple daily episodes of neurocardiogenic syncope, some associated with myoclonic jerks. He had been seen by a cardiologist and had 30 min of asystole 9 min after a glyceryl trinitrate challenge during a tilt-table test. When examined by a neurologist on recovery, there was left-sided headache, left ptosis, and facial asymmetry. He was admitted for further neurological assessment.
On further history, just before the majority of the loss of consciousness episodes, his wife witnessed left facial itching, facial droop, and drooling of saliva. The patient complained of feeling very hot and sweaty before or after the collapses and he was sleepy, disorientated, and cognitively slow for several hours after. He also gave a history of headaches preceding or following some of these episodes, starting 2 years into the presentation.
The headaches were characterized by strictly left-sided retro-orbital and peri-auricular stabbing pain, typically lasting between 10 and 60 s. He could have some interictal discomfort on the left lasting an hour after an attack. Attacks were occurring seven to eight times a day on most days (25 days a month) and were associated with lateralized left-sided photophobia and cranial allodynia. He reported a maximum attack duration of 1 h (average a minute or less) and a maximum frequency of 20 attacks in 24 h. There was left eye lacrimation, ptosis, eyelid edema, and left aural discomfort. The attacks were not triggerable. There was a positive family history of migraine. There was no personal or family psychiatric history. He had a history of back pain, obstructive sleep apnea, and renal calculi. Neurological and general examinations were normal; he weighed 109.8 kg (BMI 28).
He had never tried any other headache preventives in the past, nor triptans. Prior to presenting to us, all treatments used had been targeted against the syncopal component to his presentation.
An inpatient INDOtest was conducted during his neurological admission. The intramuscular indomethacin dose is divided into two separate injections, which are administered at the same time on one day. Concurrently, an equivalent volume of placebo is administered as two different injections on the other day. On day 1, he was injected with normal saline, with no side effects and only one headache attack following the injections for 24 h. On day 2, 200 mg IM indomethacin was administered into the gluteal muscles, with 100 mg on each side. One hour after the injections, he complained of chest pain and dizziness, and promptly lost consciousness for seconds. He was very confused and agitated on recovery. He denied headache but had a visible left partial ptosis, receptive aphasia, and dysarthria on examination. He had several further episodes brief of loss of consciousness with associated brief jerking, and progressive confusion and disorientation after each episode. After one collapse 5 min into this presentation, he became very agitated, paranoid, and aggressive, trying to break out of the ward to escape. He was isolated to a side room to protect himself and others, and he had further episodes of loss of consciousness (in total 15–20) whilst continuing to display these neuropsychiatric symptoms. This entire episode lasted 5 h, and he then began to become more orientated until he recovered to baseline.
The blood and ECG work up was normal, other than the initial ECG showing sinus arrhythmia at a rate of 108 beats per minute which normalized to normal sinus rhythm thereafter. His blood pituitary profile and MRI brain scan with pituitary views were unremarkable, apart from a likely vascular area of hyperintensity in the left lentiform nucleus. This likely idiosyncratic drug reaction was reported via the Medicines and Healthcare Products Regulatory Agency (MHRA) via the UK Yellow Card Scheme.
Following his discharge from hospital, he was mostly headache free for 1 year, but he continued to experience frequent and daily episodes of loss of consciousness. He subsequently had complete resolution of both syncopal and headache symptoms on 12 mg candesartan for headache prevention without side effects. Symptoms then returned after a few months, with no response to up titration of the candesartan to 24 mg. Attacks failed to respond to greater occipital nerve blockade. Ultimately, he gained sufficient headache control with flunarizine 5 mg. He continued to have infrequent episodes of presyncope without loss of consciousness.
We felt that this patient is likely to have experienced cranial autonomic symptoms preceding his headache attacks (manifesting as sweating, facial swelling appearing as facial weakness and sialorrhea), which we have previously reported as occurring in the premonitory phase, 6 and possible systemic autonomic or brainstem dysfunction thereafter, leading to collapse. The postdrome and interictal headache are typical of the migrainous component of his disorder.
Given the sustained IM indomethacin response, we initially concluded that the patient likely had CPH, but headache recurrence thereafter responded to candesartan and flunarizine, suggesting underlying significant migraine. This prompted us to consider chronic unilateral migraine with primary stabbing headache with an NSAID response as the most likely diagnosis. 1 It remains difficult to ascertain the definite diagnosis in this case given the change in headache frequency initially following a single dose of IM indomethacin, and following migraine prevention. Unfortunately, he cannot be re-exposed to indomethacin safely for further evaluation.
Literature
Psychotic side effects to indomethacin have been reported through case reports in the literature since 1965. 7 These are summarized in Table 1.
A summary of existing reports of neuropsychiatric side effects of indomethacin.
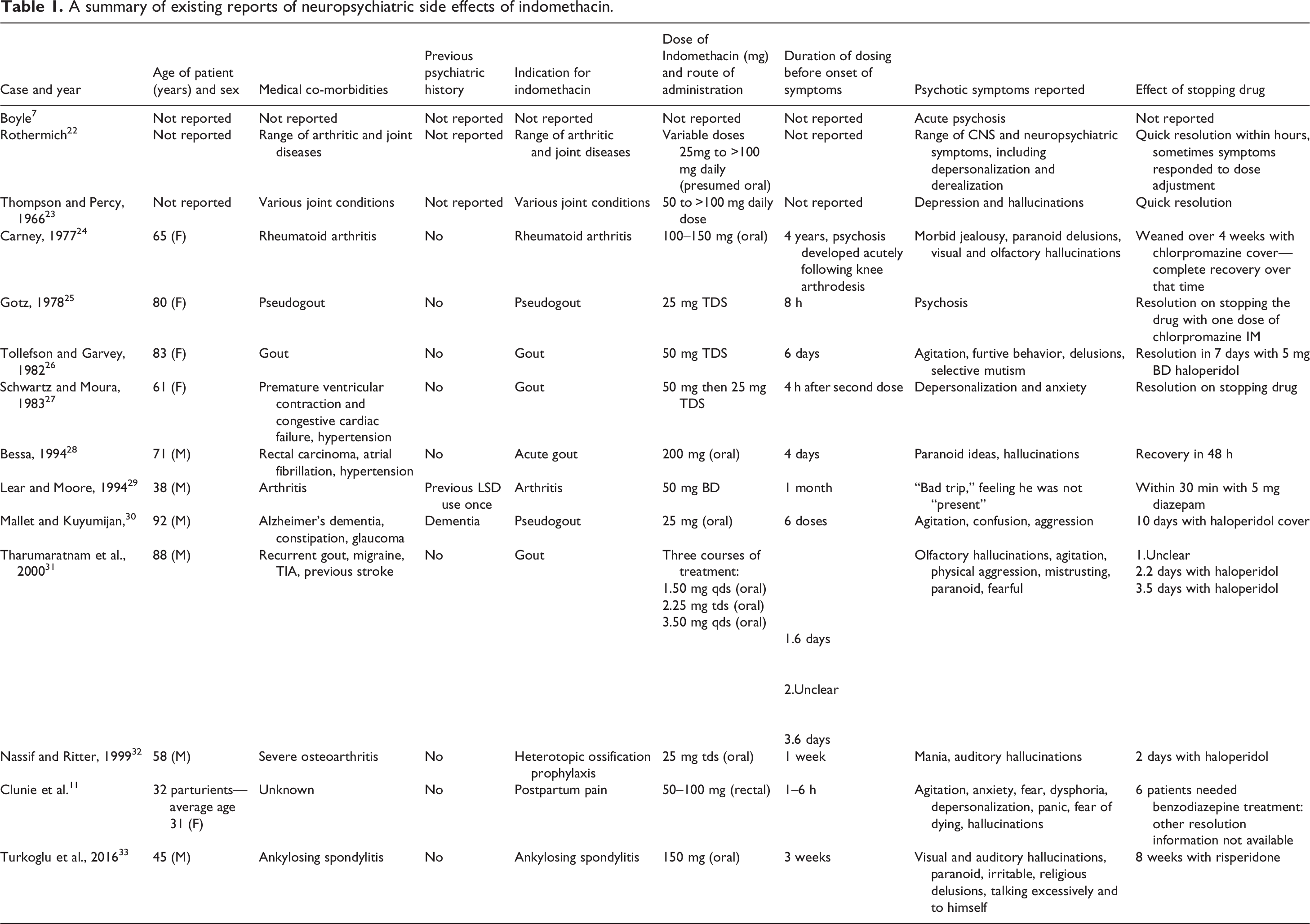
There has been suggestion of an increased risk to these side effects in the elderly and in those with pre-existing psychiatric disease, 8 –10 although they can occur without these factors being present. There has also been a series looking at psychotic side effects of indomethacin in post-partum women. 11 Other NSAID-induced neuropsychiatric adverse events studies suggest that these are no more common in the elderly, with incidence peaking in middle age and falling after the age of 70 years. 12 In a case series, psychiatric side effects of indomethacin were reported to occur most commonly within an hour of the dose, and last on average less than 6 h. 13 Indomethacin and selective cyclo-oxygenase 2 (COX-2) inhibitors seem to share these neuropsychiatric adverse events more so than the other NSAIDs, 13,14 but the occurrence of them with these different drugs suggests a possible shared mechanism via prostaglandins.
Discussion
The acute paranoid psychosis that the patient developed in this case following a single parenteral indomethacin dose for headache diagnosis, has not, to our knowledge, been previously reported in headache literature.
Most cases previously reported have been in patients on courses of indomethacin. The literature leans toward a propensity for the elderly, postpartum and postoperative populations to develop this adverse effect, and these factors may be seen as independent risk factors. There has been suggestion that the use of indomethacin should be entirely avoided in the elderly. 15
The reasons for the drug to have such effects are unclear, although they are clearly reversible on cessation (none of the cases reported seem to have longstanding symptoms), after which with rapid plasma clearance, the effects do seem to cease. None of the selective COX-2 inhibitors or indomethacin have been studied systematically in homogeneous patient groups to elucidate why they may be more prone to producing this effect compared to other NSAIDs. Interestingly, in many cases, a rechallenge with the same or different NSAID leads to a similar outcome, at least in those with a history of psychiatric disease. 9 NSAIDs inhibit prostaglandin synthesis via COX-2, which is widely located in the CNS, in areas like limbic cortex, hippocampus, and amygdala, 16 and with a predilection for dendrites and dendritic spines, where prostaglandins may play a role in synaptic signaling and CNS neurotransmission. 16 The localization of COX-2 within the brain, and the potential role of prostaglandins in modulating CNS function provide possible mechanisms by which indomethacin could potentiate neuropsychiatric symptoms in some patients, likely via dopaminergic neurotransmission, which has also been implicated in schizophrenia-associated psychosis. 17 It has been suggested that those with schizophrenia may lack the protective effect of prostaglandin-mediated inhibition of dopaminergic neurotransmission. 18 In the same way, NSAIDs could increase dopaminergic neurotransmission by inhibiting prostaglandin synthesis and therefore cause psychotic symptoms. This sensitivity to dopamine may be further modulated by factors such as hormones, advancing age and pre-existing psychiatric disease.
Fatty acid precursors of prostaglandin may also be involved via serotoninergic and cholinergic neurotransmissionin modulating brain function and in mediating neuropsychiatric effects. Fatty acid metabolism has been implicated in other psychiatric disorders like schizophrenia, depression, and attention deficit hyperactivity disorder. 19 The CNS penetration of indomethacin may lead to it acting on serotoninergic targets because of molecular similarity, and a shared indole-ring structure, 20 see Figure 1. Indomethacin may also have contributory effects on cerebral blood flow. 21 The severity of the side effect may be dose-dependent, and there has been suggestion that a single dose of 300 mg could cause some neuropsychiatric symptoms in all patients with feelings of “unreality” or dissociation, and in some personality change and paranoia. 22 In addition, other studies have reported these effects mostly in patients taking more than 100 mg/day (see Table 1).

Molecular similarity between serotonin and indomethacin. Both compounds share an indole-ring structure.
We therefore suggest that this side effect is an important one to ask for and be aware of, as well to counsel patients about, in all populations of patients treated with indomethacin for headache disorders. It may be under-recognized in headache medicine and may complicate treatment, particularly in some at risk groups.
Clinical implications
Patients, particularly those at highest risk, should be counseled about the risk of neuropsychiatric side effects on indomethacin. These include the elderly and those who are postpartum or postoperative.
These effects may be dose-dependent and are generally reversible on stopping the drug.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and patient consent
No specific ethical approval was sought for publication of this case. The patient provided written signed consent for publication.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
