Abstract
Background:
Parenteral non-steroidal anti-inflammatory drugs (NSAIDs) are important alternatives to oral NSAIDs, especially in patients with severe migraine who have emesis or gastroparesis. With increasing research on using parenteral NSAIDs for acute migraine, it is critical to examine the quality of these studies. Our goal was to assess the adherence of these trials to the International Headache Society (IHS) controlled trial guidelines for acute treatment of migraine.
Methods:
We queried PubMed for clinical trials investigating parenteral NSAIDs for acute treatment of migraine in adult patients. We developed a 14-point scoring system based on the essential components of the IHS guidelines. To date, four versions of the IHS’s Guidelines for controlled trials of acute treatment of migraine attacks have been published. Each trial was evaluated with the appropriate edition of the guidelines.
Results:
We identified 216 studies and assessed 27 eligible clinical trials. The mean score was 6.7 ± 2.1 (2–11). Most trials followed the IHS migraine diagnosis criteria (85.2%), but only six (22.2%) selected patients based on the recommended headache frequency. Most trials were randomized (88.8%), but fewer were double-blinded (74.1%) or placebo-controlled (11.1%). Almost every trial clearly explained the pain scale (96.3%), and three-quarters (77.8%) assessed headache-associated symptoms. However, no trial utilized the recommended primary endpoint: pain-freedom at 2-hours.
Conclusions:
Most clinical trials on parenteral NSAIDs for acute migraine did not fully adhere to the IHS recommendations. Future studies should pay special attention to the IHS guideline to improve the quality of clinical trials for the acute treatment of migraine.
Introduction
In 1991, the International Headache Society’s (IHS) Clinical Trials Standing Committee published its first Guidelines for controlled trials of acute migraine attacks to facilitate the use of more rigorous scientific standards in clinical research. 1 Later, the Committee published the second, third, and fourth editions of the Guidelines in 2000, 2012, and 2019, respectively. 2 –4 As the number of trials for acute migraine has substantially increased over the years, the IHS guidelines have been necessary to provide clear standards for investigators. Each edition of the Guidelines has incorporated data and experience from successive clinical trials. And ultimately, the goal of the Guidelines is to improve the quality of controlled clinical trials in migraine.
It is critical that the adherence of clinical trials to the Guidelines be evaluated. This investigation allows for a better understanding of the quality of research for acute trials, and may inform how guidelines should be adapted or changed in the future. Previous study from Deen et al. examined the adherence of trials to the IHS Guidelines for preventive treatment of migraine. 5 Additionally, Alpuente et al. reviewed the adherence of trials to the third edition of the Guidelines for acute treatment of migraine. 6 They identified migraine-specific and end-point-related recommendations that deserve special attention and found that some guideline recommendations were not thoroughly followed. Investigations such as these are valuable in providing a better understanding of study quality and important groundwork for developing future editions of the IHS Guidelines for controlled trials of acute treatment of migraine.
Parental nonsteroidal anti-inflammatory drugs (NSAIDs) are an attractive option for treating acute migraine and deserve serious consideration for their use. Patients who present to the emergency department or other ambulatory settings with severe migraine have limited options for treatment. Parenteral NSAIDs are one first-line treatment that avoids many drawbacks associated with other treatment options. For example, opioids are associated with respiratory depression, urinary retention, and ileus. Antidopaminergic agents may precipitate extrapyramidal side effects. Dihydroergotamines may cause gastrointestinal and vasoconstrictive associated problems. Parenteral delivery of NSAIDs may also be more tolerable in patients that present with nausea and vomiting. Therefore, the objective of this study was to examine the quality of controlled trials of parenteral NSAIDs for acute treatment of migraine.
Methods
Search strategy and study selection
A PubMed search was performed for clinical trial articles published between January 1, 1991, and November 1, 2021, on prospective clinical trials for acute treatment of migraine. We used a search term to identify all articles that fulfilled the following three criteria: used the word migraine or headache in the title/abstract, used the name of any NSAID in the title/abstract, and mentioned intravenous, IV, intramuscular, or IM in the title/abstract (full search term is included in the supplementary material). Inclusion criteria include a) trial on adult migraine patients; b) investigation of acute intravenous or intramuscular treatments; and c) study designed and conducted after 1991 (the publication year of the first IHS guideline for acute clinical trials). Exclusion criteria were: a) trial on pediatric subjects; b) study without NSAID treatment; c) trials that did not have a primary focus on acute treatment of migraine; d) papers without an available English text. Titles and abstracts collected by PubMed search were screened for eligibility. If inclusion or exclusion by title and abstract alone was not adequate, the full text was used for screening. The manuscripts were evaluated and graded independently. Final approval was determined by the senior investigator.
Data extraction and analysis
Based on each IHS guideline for acute trials, we developed a 14-point scoring system of the most critical recommendations (Table 1). Similar scoring methods have been used in the past to examine trial adherence to specific IHS guidelines. 5 –7 Although many of the recommendations outlined in the original IHS guideline have remained unchanged (double-blind, placebo-controlled, randomized, etc.), some recommendations show discrepancies between editions. For these recommendations, we captured each article under a common category and then graded each article based on its relevant IHS guideline. For example, the first three editions specified that subjects with acute migraine should have 1–6 attacks per month, while the fourth edition specifies 2–8 attacks per month. We grouped these recommendations under the common heading of appropriate attacks per month, but the grading was specific to the appropriate guideline. Studies were graded based on our best estimation of which edition of the Guidelines was most current at the time of trial design. If the study’s start date was available, we grouped trials based on the study start date with a 2-year leeway. For example, if a trial had a start date of December 2002, we would consider it under the second edition of the Guidelines (published November 2000). If the study’s start date was unknown, we utilized publication date with a 4-year leeway. For example, a trial with a publication date of April 2021 was considered under the third edition of the Guidelines (published January 2012). Overall, they were graded into four groups: pre-first (labeled 0th), 1st, 2nd, and 3rd edition. No trial was grouped under the 4th edition of the guideline.
Grading system for evaluating trials for acute treatment of migraine.

Minimum score = 0, Maximum score = 14.
Relevant data was then collected from articles identified in our search. The full text of each article was analyzed for adherence to our scoring system. Satisfactory adherence to each guideline was associated with 1 point, while non-adherence was denoted as 0 points. Therefore, the minimum score for each article was 0, and the maximum score was 14. All trials were evaluated independently by two investigators. Disagreement or incomplete agreement was resolved by discussion and finalized by the senior investigator. Data were presented in percentage or mean ± standard deviation. To examine the adherence difference (individual score to each recommendation, total score) between each guideline edition, Fisher’s one-way ANOVA was performed, followed by Tukey post hoc pairwise comparison. The statistical analysis was done using the jamovi project (2021) from https://www.jamovi.org.
Key Similarities and differences between the four editions of IHS guidelines
Patient Selection. Across all editions, the IHS guidelines consistently recommend fulfillment of ICHD criteria for acute migraine as the standard for study inclusion. The recommended frequency of attacks for study inclusion varies slightly with each edition. The first edition specifies 1–6 attacks per month with at least 24 hours of headache freedom between each attack. The second and third editions stipulate 1–6 attacks per month with a maximum of 6 headache days per month. And the fourth edition indicates 2–8 attacks per month with a maximum of 15 headache days per month. Lastly, the Guidelines consistently recommend a clear explanation for what medications subjects are permitted to take alongside the study drug.
Trial design. Each edition of the Guidelines recommends performing randomized, double-blinded, placebo-controlled trials when studying the acute treatment of migraine. The Guidelines consistently recommend that a sample size calculation be performed. The two most recent editions of the IHS Guidelines place an emphasis on utilizing intention-to-treat (ITT) analysis. We chose not to utilize this metric for our overall grading as the first and second edition of the Guidelines did not include ITT analysis as a recommendation.
Evaluation of results. Each version of the Guidelines clarifies that pain-freedom at 2 hours post-treatment should be used as the primary endpoint. The fourth edition of the Guidelines also states that the absence of the most bothersome migraine-associated symptoms may be used as a co-primary endpoint. Using a 4-point scale for measuring headache intensity, reporting secondary headache symptoms such as nausea, recording a global evaluation of the study drug from subjects, and reporting adverse events are also consistently recommended. For the purposes of this paper, we accepted any clear explanation of the scale used to measure headache intensity (e.g., 4-point verbal, 100 mm VAS, 0–10 numeric) as adequate fulfillment of the IHS guideline. Recommended secondary outcomes vary significantly between editions of the Guidelines. Although the method for data collection has changed over the time that the Guidelines have been published, each edition recommends using a headache report form that is clearly explained in the paper’s text. Lastly, the third and fourth editions of the IHS Guidelines recommend blinding assessment. This category was not included in the scoring system.
Results
Identification of relevant trials
Our PubMed search produced 216 records. Four articles were unretrievable, leaving us with 211 titles and abstracts to screen for relevance. Of these 211, we eliminated 185 articles that either did not satisfy inclusion criteria or met one or more exclusion criteria. Twenty-seven studies remained for further evaluation and grading (Figure 1).

Data collection flow diagram.
Evaluation of trials
From the 27 trials, 8 –34 11 studies investigated the use of intramuscular (IM) medication for acute migraine, 8 –16,22,29 while 16 investigated intravenous (IV) use. 17 –21,23 –28,30 –34 Fourteen articles were for ketorolac, 5 for dexketoprofen, 3 for acetylsalicylic acid, 2 for diclofenac, 2 for ibuprofen, and 1 for lysine cloxinate (Table 2).
Total scores of trial adherence to IHS guideline for acute treatment of migraine.

Articles are listed by publication date. IV: intravenous; IM: intramuscular, ICHD: follows IHS criteria for diagnosis of migraine; Migr freq: subjects experienced the appropriate number of headache days per month for inclusion into the trial; Med use: clearly described which concomitant medications were permissible for inclusion into the trial; DB: double-blinded, PC: placebo-controlled; RDZ: subjects randomized at entry; SS: sample size calculation performed; Pain free: primary endpoint is pain freedom at 2 hours; Scale: trial used a clearly defined scale to record headache intensity; 2o symp: secondary symptoms such as photophobia, phonophobia, nausea, etc. were recorded; Global eval: subjects global evaluation of the treatment they received was recorded; AE: adverse events report; a priori: outcomes specified a priori; NCT: trial was registered before start of enrollment.
Three (3/27, 11.1%) trials were randomized, double-blind, and placebo-controlled. Most trials were performed in the emergency department (21/27, 77.8%), while other trials occurred either in the clinic (5/27, 18.5%) or by self-administration at home (1/27, 3.7%). Most trials utilized IHS criteria for the diagnosis of migraine (23/27, 85.2%), were randomized (24/27, 88.9%), utilized a proper scale for measuring headache intensity (26/27, 96.3%), and reported adverse events (22/27, 81.5%). Fewer trials mentioned the frequency of migraine attacks for inclusion (6/27, 22.2%), were placebo-controlled (3/27, 11.1%), incorporated a global evaluation of treatment from the subjects (3/27, 11.1%), or specified outcomes a priori (2/27, 7.4%). For trials that did not utilize placebo, the most common active controls were metoclopramide (6/27, 22.2%), meperidine (4/27, 14.8%), and acetaminophen (3/27, 11.1%). No trials utilized the recommended primary outcome of pain-freedom at 2 hours post-intervention (Table 2). Additionally, none of the trials reported the absence of most bothersome headache-associated symptoms as an outcome measure, which is considered a potential co-primary endpoint in the fourth edition.
The mean score of all trials was 6.70 ± 2.07. Additionally, the mean score for IM trials was 5.91 ± 2.26, and the mean score for IV trials was 7.25 ± 1.88. We examined the differences in trial scores based on their appropriate edition of the IHS guidelines. Eight were grouped under pre-guideline; they had a mean score of 4.88 ± 1.46. Studies under the first edition of the Guidelines had a mean score of 6.20 ± 1.79, second edition of 8.17 ± 2.32, and third edition of 7.75 ± 1.28. There was a statistically significant difference between groups as determined by Fisher’s one-way ANOVA (F(3,23) = 5.76, p = 0.004). Tukey post hoc test revealed significant differences between pre-guideline vs. second edition (p = 0.008) and vs. third edition trials (p = 0.012) (Figure 2). We further examined which recommendations had significant differences in adherence across these four groups. Table 3 showed the between-group difference significant for IHS diagnostic criteria (F(3,23) = 5.40, p = 0.006), randomization (F(3,23) = 3.24, p = 0.041), and trial registered before enrollment (F(3,23) = 5.12, p = 0.007). For IHS diagnosis criteria, within-group differences were found between pre-guideline vs. 1st edition (p = 0.032), vs. 2nd edition (p = 0.022), and vs. 3rd edition (p = 0.012). No within group difference was found for randomization. For trial registration before enrollment, within-group differences were found between pre-guideline vs. 3rd edition (p = 0.017) and 1st edition vs. 3rd edition (p = 0.041).

Box plot of trial scores based on appropriate edition of the IHS guidelines for acute trials. There was a statistically significant difference between groups as determined by Fisher’s one-way ANOVA (F(3,23) = 5.76, p = 0.004). A Tukey post hoc test revealed significant differences between pre-guideline (0th edition) vs. 2nd edition trials (p = 0.008) and vs. 3rd edition trials (p = 0.012).
Individual scores by each guideline edition.
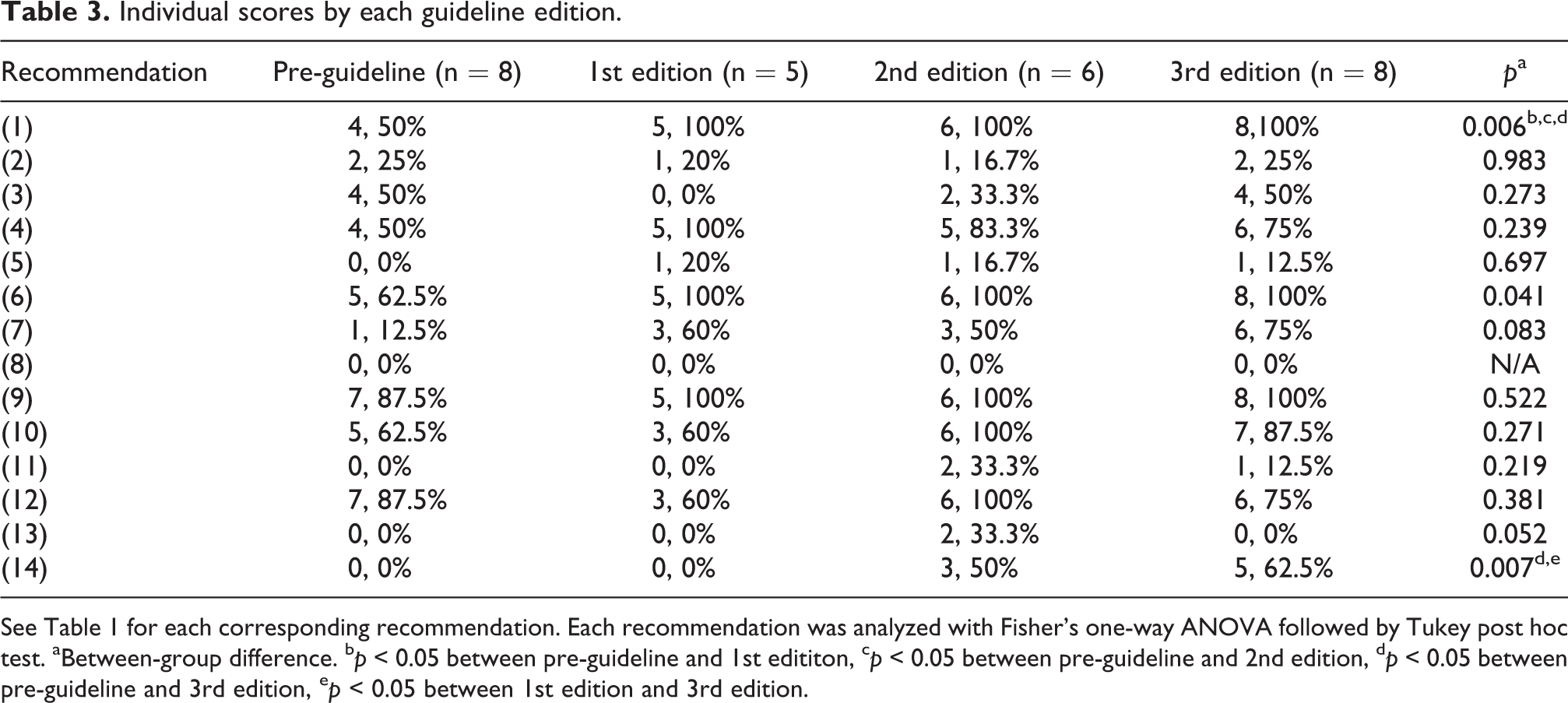
See Table 1 for each corresponding recommendation. Each recommendation was analyzed with Fisher’s one-way ANOVA followed by Tukey post hoc test. aBetween-group difference. b p < 0.05 between pre-guideline and 1st edititon, c p < 0.05 between pre-guideline and 2nd edition, d p < 0.05 between pre-guideline and 3rd edition, e p < 0.05 between 1st edition and 3rd edition.
For unscored metrics (Table 4), we found that few trials explicitly mentioned utilizing ITT for primary analysis (3/27, 11.1%) or clearly defined the report form or diary used to record headache severity (4/27, 14.8%). Additionally, no trials reported completing a blinding assessment (0/27, 0%). Most trials (24/27, 88.9%) evaluated secondary outcomes such as average time to headache improvement, rescue medication use, and changes in accompanying symptoms (e.g., nausea, vomiting, photophobia, or phonophobia).
Main endpoints and other characteristics of included trials for discussion.

ITT: study specified the use of intention-to-treat for primary analysis. Report Form: trial clarified the use of a report form or diary for recording subjects’ headache intensity. Blinding Assess: trial explicitly stated that they performed a blinding assessment. A “-” indicates that the study did not meet and a “x” indicates that the study did meet the criteria.
Discussion
Examining adherence to the IHS Guidelines for controlled trials of acute treatment of migraine attacks allows us to better understand the quality of published research. Our evaluation on parenteral NSAID trials found that not all recommendations were followed. Criteria such as utilizing ICHD diagnostic criteria for study inclusion, specifying a clear scale to measure headache severity, and reporting adverse events were almost entirely followed by the trials we examined. Other recommendations, such as applying the recommended primary endpoint of pain-freedom at 2 hours and employing a placebo control, were not as well incorporated into these acute trials.
Regarding the selection of patients, only a minority of trials reported a clear definition of headache frequency or which concomitant medications were permissible for study inclusion. Most trials stipulated ICHD migraine criteria for trial inclusion, but few clarified the specific attack frequency, which was recommended 2–8 per month (<15 monthly headache days) in the latest guideline. It is important to differentiate episodic from chronic migraine as the treatment response of each group may vary significantly. 35 Additionally, about a third of trials mentioned the use of concomitant medications. Although this recommendation varies between the four editions of the IHS guidelines, we found that few trials provided a clear explanation of what concomitant medications were allowed or if a washout period was used before randomization. It is essential to clearly define this aspect of trial inclusion as simultaneous use of other medications for migraine or carryover effects from recently discontinued medications could affect study outcomes.
For the trial design, around three-quarters of the trials we graded were double-blinded, and close to all trials were randomized. Most trials that did not follow these guidelines were published when the first edition was released; they were most likely designed before the Guidelines were released. This may reflect the initial need for the IHS Guidelines to standardize trials and provide clear recommendations for performing evidence-based investigations of therapies for the acute treatment of migraine. It is an encouraging sign that trials have become more adherent with each new edition of the Guidelines (Figure 2). More specifically, the recommendations to utilize ICHD criteria, randomization, and trial registration make up most of this increase in adherence (Table 3). Although this is an underpowered study, the increase in adherence score may reflect an enhanced awareness and improved understanding of the clinical trial operation. Less adhered to recommendations in this section of the Guidelines were performing sample size calculations and utilizing a placebo control. The placebo effect is a well-characterized effect seen in many clinical trials. In acute trials for migraine, the placebo effect varies significantly. A meta-analysis of the placebo response in migraine found that across 196 trials of acute migraine, 28.6% of patients in the placebo group experienced pain-relief at 2 hours, and 8.8% achieved pain-freedom. 36 This effect may be even more pronounced for parenteral trials. 37,38 This lack of a placebo control in many trials is especially concerning when those same trials also lack appropriate sample size calculations. Trials with active controls often lack assay sensitivity and therefore require larger sample sizes to provide more statistical confidence or rely on previous research to show that the active control itself is superior to the placebo. 39 However, it is worth noting that significant ethical concerns have been raised about the use of placebo controls in the treatment of migraine, with some researchers even suggesting that the IHS Guidelines themselves may violate the Helsinki declaration of ethics by recommending placebo controls. 40
For evaluation of results, no trial utilized the recommended primary endpoint of pain-freedom at 2 hours after treatment administration. Almost every trial utilized pain-relief as their primary endpoint, either at 1 hour, 2 hours, etc. The fourth edition of the IHS Guidelines provides a compelling explanation for the utility of pain-freedom as the primary efficacy measure. Compared to pain-relief, pain-freedom provides a much more rigorous method for treatment investigation. Pain-relief counts partial response as success and may make it more difficult to compare different migraine therapies. 41 Additionally, others have argued that pain-relief may not be clinically relevant for the treatment of migraine. 42 Lastly, and possibly most importantly, pain-freedom is an appropriate primary endpoint as it matches patient’s expectations for migraine treatment. Previous research has shown that only 60–70% of patients that achieve pain-relief were satisfied with treatment. 43 This is in contrast to patients who achieve pain-freedom, who report over 90% satisfaction. 44 It is interesting that trials forego the use of pain-freedom and instead opt for pain-relief. One possible explanation for this is that pain-relief as primary endpoint may make it easier for researchers to achieve a statistically significant or positive result. As discussed previously in this paper, pain-relief is associated with a much higher placebo-response than pain-freedom. It is well documented that trials with negative or non-significant results are less likely to be published or accumulate as many references as compared to trials with positive or significant results. Researchers may therefore be drawn to utilize pain-relief as their primary endpoint because it could make achieving a more favorable result more attainable. With the remaining criteria under the category of evaluation of results, we found that almost all trials reported a clear scale for measuring headache intensity and most appropriately reported adverse events and the prevalence of headache-associated symptoms (photophobia, nausea, etc.). However, fewer trials recorded the subject’s global evaluation of study treatment, specified outcomes a priori, or registered before enrollment.
In this paper, we also chose to examine some characteristics of the trials that were not utilized for scoring purposes. Despite the Guidelines recommendation, few trials specified that ITT analysis was utilized. Many of the trials had a small number of subjects, so typically, most subjects who were randomized also followed protocol, were not lost to follow up, and were included in the final analysis. In these situations, choosing between ITT and per-protocol analysis may not be needed. Regardless, future studies should clarify the intention to use ITT from the study outset. Another IHS recommendation included for discussion is the use of a clear report form for subjects to record headache intensity and other outcome measures. Each edition of the IHS Guidelines recommends utilizing a simple report form or, in more recent editions, an electronic diary. We assume that most trials utilized such report forms for data collection but found that few clearly stated the use of these forms in the text of the papers. Lastly, we found that no trial performed a blinding assessment.
Despite the lack of guideline adherence in these studies, parenteral NSAIDs are still often used clinically with visible benefit. And with increased Guidelines adherence and better research quality, their use as important migraine therapy may increase. Improvement in the adherence of trials to these recommendations will require increased awareness to the IHS Guidelines. As researchers and clinicians become more familiar with the benefits of utilizing the Guidelines, we hope to see an increase in their adoption in clinical trials. Clinical decision-making is often evidence-driven. Additionally, the FDA increasingly requires more rigorous data for drug approval. Therefore, we emphasize the importance of utilizing rigorous research standards, like that of the IHS Guidelines, so safe and effective medications such as parenteral NSAIDs may be utilized for acute treatment of migraine.
Our research and analysis in this review have limitations. First, we developed a simplified version of the IHS Guidelines for scoring purposes. Although we incorporated what we considered the most important recommendations, we recognize that many other important recommendations were left out of our final grading system. This is especially true as we attempted to synthesize every edition of the IHS guidelines into one grading system, which discounts the nuance between editions. Additionally, we were limited to scoring trials based solely on the published paper and its supplementary material. Often, even though the trial may have followed IHS guidelines in practice, we were limited to published results and study descriptions. Lastly, we were limited in our ability to group trials based on what edition of the Guidelines was available to trial researchers at the time of study design. Many trials did not include information about the study start date. Although we attempted to develop a grouping system that most accurately captured the appropriate Guideline, some trials may have been erroneously grouped under the wrong edition.
Conclusion
We examined the adherence of parenteral NSAIDs trials for acute treatment of migraine based on the essential recommendations of the IHS Guidelines for controlled trials of acute migraine attacks. Although many trials followed recommendations for patient selection or trial design, many did not implement critical recommendations such as pain-freedom as the primary endpoint or placebo as the control. Future studies of parenteral NSAIDs for migraine should pay special attention to IHS guidelines to improve the validity, quality, and evidence-based acute treatment of migraine.
Article Highlights
Most parenteral NSAID trials do not fully adhere to the IHS guidelines for controlled trials of acute treatment of migraine.
Adherence is low on the use of placebo control and pain-freedom at 2 hours as the primary outcome.
Future studies should follow the guidelines to improve the overall quality of research for the acute treatment of migraine.
Supplemental material
Supplemental Material, sj-pdf-1-rep-10.1177_25158163221114465 - Parenteral NSAIDs for acute treatment of migraine: Adherence to the IHS guidelines for controlled trials
Supplemental Material, sj-pdf-1-rep-10.1177_25158163221114465 for Parenteral NSAIDs for acute treatment of migraine: Adherence to the IHS guidelines for controlled trials by John G Curran, John Waters and Hsiangkuo Yuan in Cephalalgia Reports
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
