Abstract
Aim
To evaluate the efficacy and safety of greater occipital nerve (GON) block combined with triple therapy versus triple therapy alone in the treatment of acute migraine in the emergency department for severe or refractory cases setting.
Methods
We conducted a prospective, randomized, controlled trial without use of placebo control in adult patients with migraine according to International Classification of Headache Disorders, 3rd editon criteria. Patients were randomly assigned (1:1), without blinding method, to receive either a GON block (methylprednisolone 80 mg + lidocaine 20 mg) plus triple intravenous therapy (ketorolac, paracetamol, metoclopramide) or triple therapy alone. The primary outcome was the proportion of patients achieving ≥50% reduction in headache intensity on a visual analog scale (VAS) two hours post-treatment. Secondary outcomes included changes in monthly migraine days, pain-free days, Headache Impact Test-6 (HIT-6) scores and hospital readmissions at 30-day follow-up.
Results
Forty-two patients were enrolled (21 per group). A ≥50% VAS reduction at two hours occurred in 95.2% of patients receiving the GON block versus 47.6% in the control group (p = 0.0003). Median pain reduction was 6.0 vs. 3.0 points, respectively (p < 0.001). At 30 days, the intervention group reported fewer migraine days (median 3.0 vs. 7.0 days; p = 0.0355), more pain-free days (14.0 vs. 5.0 days; p = 0.0379) and lower HIT-6 scores (59.0 vs. 65.0; p = 0.1584). Readmission rates were lower in the intervention group (9.5% vs. 23.8%) but not statistically significant. Adverse events associated with the GON block were mild and transient, including local pain (47.6%) and minor bleeding (14.3%).
Conclusions
GON block as an adjunct to triple therapy is effective and well tolerated for the acute treatment of migraine, providing significant short-term relief and improving outcomes at 30-day follow-up. Our failure to blind the outcome assessors limit the validity of our results, which supports the use of GON block as an adjunct to parenteral therapy for patients with migraine in the emergency department.
Trial Registration
This study was not registered in a public trial registry.
This is a visual representation of the abstract.
Keywords
Introduction
Migraine is a disabling primary headache disorder affecting approximately 14% of the global population and represents the second leading cause of years lived with disability worldwide (1). Current diagnosis is based on clinical findings established in the third edition of the International Classification of Headache Disorders, 3rd editon (ICHD-3), published in 2018 (2,3). Migraine accounts for 19% of all the headache related visits to the emergency department (ED) in Mexico, indicating a substantial burden to the health system, and tend to seek attention due to severe or refractory symptoms (4).
Migraine attack physiology is classically divided into four sequential phases: prodrome, aura, headache and postdrome. The underlying mechanisms are considered to involve cortical spreading depolarization, trigeminovascular complex activation and consecutive pathway sensitization (5–7). In this context, afferent input from cervical and occipital skin and muscles, in conjunction with trigeminal branches, converges onto second-order neurons within the trigeminocervical complex (TCC). This convergence facilitates the sensitization of central TCC and thalamic neurons, clinically manifesting as cephalic allodynia (6). Nonetheless, the pathogenesis of migraine remains only partially understood, with several pathophysiological processes yet to be described (8,9).
Treatment strategies encompass both acute and preventive pharmacologic agents, alongside an array of non-pharmacologic interventions. Despite the availability of these therapeutic options and exhaustive diagnostic criteria, clinical management remains inadequate due to high rates of misdiagnosis and undertreatment (10).
In recent decades, the therapeutic potential of peripheral nerve blocks has gained attention and been used as an alternative treatment for various primary headaches (11,12). Among these, the greater occipital nerve (GON) block is the most extensively employed technique (13). Its efficacy is likely related to the anatomical and functional convergence of sensory cervical and trigeminal fibers. The caudal nucleus of the trigeminal nerve is the largest cranial nerve nucleus and extends from the brainstem to the spinal segment C2. Reduction of nociceptive signaling arriving at TCC neurons, as shown through non-invasive electrical stimulation, allows for cranial desensitization. Therefore, GON blocks can lessen perceived allodynia and migraine attack pain (6,14). Moreover, their analgesic effect tends outlast the anesthetic effect, implying an effect on central pain modulation, which provides some patients with pain-free weeks or even months (13).
Several double blind, randomized controlled clinical trial have established superiority of GON block against placebo (15,16). A meta-analysis evaluating occipital nerve blocks for chronic migraine reported a significant reduction in pain intensity and headache frequency during the first two months of intervention, with no increase in adverse events relative to placebo (17). Similarly, a 2022 systematic review on chronic migraine patients identified a significant reduction in headache days although it did not find a significant effect on acute medication use (14,18). Additionally, similar comparative study without placebo controls demonstrated a reduction in headache days with occipital nerve block against placebo (19,20). However, there is double blind, double dummy, randomized controlled trial that did not establish the superiority of GON block against metoclopramide drip (21).
In this prospective, randomized, controlled trial without use of placebo control, we aimed to determine the efficacy and safety of GON block combined with triple therapy versus triple therapy alone in patients with severe or refractory acute migraine in the ED.
Methods
Standard protocol approvals, registrations and patient consents
The study was approved by the Institutional Research and Ethics Committee of the National Institute of Neurology and Neurosurgery “Manuel Velasco Suarez” (Instituto Nacional de Neurologia y Neurocirugia “Manuel Velasco Suarez”, Mexico City). All procedures were conducted in compliance with current Good Clinical Practice (GCP) guidelines, national regulatory standards and the principles of the Declaration of Helsinki. Written informed consent was obtained from all participants before enrollment. This trial was not registered in a public clinical trial database prior to enrollment.
Study design and participants
This was a prospective, randomized, controlled, single-center clinical trial conducted from March 2024 to June 2025 at the ED for severe or refractory cases of the National Institute of Neurology and Neurosurgery “Manuel Velasco Suarez”. The trial was designed and reported in accordance with the CONSORT 2010 guidelines for randomized controlled trials (22). Eligible participants were adults (≥18 years) presenting with an acute migraine episode, with a prior diagnosis of migraine with or without aura based on the International Classification of Headache Disorders, 3rd edition (ICHD-3). An acute migraine attack was defined as an ongoing episode that met ICHD-3 criteria for migraine with or without migraine, and that was severe enough to prompt the patient to seek emergency care.
Patients were excluded if they were pregnant, due to the associated teratogenic risk, or had a history of occipital craniectomy, active skin infection at the injection site, known allergy to any medication used in the intervention and/or refused to sign informed consent. Patients who did not complete post-intervention assessments were also excluded from analysis.
Randomization was performed using a computer-generated sequence developed by a member of the research team who was not involved in patient enrollment, ensuring a 1:1 allocation ratio between the control and intervention groups. Group assignments were revealed at the time of enrollment using the electronic sequence. Neither participants, providers, nor outcome assessors were blinded to group allocation due to the nature of the intervention.
Study procedures
Upon presentation to the ED for severe or refractory cases, all patients were clinically evaluated by the neurology team participating in the study to confirm the diagnosis of migraine according to ICHD-3 criteria. At this stage, detailed clinical information was collected, including migraine subtype (with or without aura), history of status migrainosus, duration of migraine diagnosis, prior use of prophylactic treatment and demographic characteristics.
All patients received triple acute migraine therapy consisting of intravenous paracetamol (1 g), ketorolac (30 mg) and metoclopramide (10 mg). In addition to triple therapy, patients in the intervention group received a unilateral GON block in all cases. For patients presenting with bilateral headache, the injection was administered on the side with greater pain intensity, based on patient report.
The block was administered using 80 mg of methylprednisolone (2 ml of 40 mg/ml) combined with 20 mg of lidocaine (1 ml of 2%), chosen due to availability and rapid onset of action. The patient was seated with the head slightly flexed. The anatomical target was identified at the medial one-third of the distance between the inion and the mastoid process, approximately 2 cm lateral and 1.5–2 cm below the inion. A 22-gauge needle was used for a subcutaneous injection, performed under aseptic conditions.
Outcome measures
The primary outcome was the proportion of patients achieving a ≥50% reduction in headache intensity two hours after treatment, assessed using a visual analog scale (VAS). A printed version of the same VAS was presented to patients before and two hours after the intervention to ensure consistency in self-reported pain scores.
Secondary outcomes included the change in Headache Impact Test-6 (HIT-6) score at 30 days, the number of migraine days and pain-free days during the 30-day follow-up, and hospital readmission due to migraine within 30 days after treatment. These outcomes were assessed through a structured telephone follow-up conducted 30 days after the intervention. During the call, the HIT-6 questionnaire was administered, and patients were asked to report the frequency of migraine episodes, pain-free days and any hospital visits or readmissions related to migraine. To ensure successful follow-up, at least three telephone contact numbers were obtained for each participant, including close relatives or household members when available. Patients were informed in advance of the exact date of follow-up and the telephone number from which the call would be made, aiming to reduce the risk of missed or rejected calls.
Safety outcomes included local adverse events at the injection site, such as bleeding, pain, or paresthesias, assessed within the first two hours post-intervention and again at 30-day follow-up. Serious adverse events were defined as any medical occurrence that resulted in death, was life-threatening, required inpatient hospitalization or prolongation of existing hospitalization, resulted in persistent or significant disability/incapacity, or was otherwise considered a medically important condition. All adverse events were recorded systematically during the initial evaluation and follow-up period.
Statistical analysis
The sample size was calculated using Cochran's formula for finite populations. Based on an estimated annual population of 72 patients with acute migraine presenting to the ED for severe or refractory cases, a confidence level of 95% (Z = 1.96), a statistical power of 80%, a standard deviation of 1.25 and a margin of error of 10%, a minimum sample size of 42 patients was determined. This estimate was derived by first calculating the required sample size for an infinite population and then applying the finite population correction.
All statistical analyses were performed using SPSS, version 25 (IBM Corp.). The normality of continuous variables was assessed using the Shapiro–Wilk test. Because most variables did not follow a normal distribution, non-parametric tests were applied consistently throughout the analysis.
Baseline characteristics were summarized as medians and ranges for continuous variables, and as absolute and relative frequencies for categorical variables. Comparisons between groups were made using the Mann–Whitney U test for continuous variables and the chi-squared or Fisher's exact test for categorical variables.
The primary outcome was analyzed in two ways: (1) as the proportion of patients achieving a ≥50% reduction in headache intensity, compared using the chi-squared test, and (2) as the absolute reduction in VAS scores from baseline to two hours, compared using the Mann–Whitney U test.
Secondary outcomes were analyzed using the Mann–Whitney U test for continuous variables and Fisher's exact test for categorical outcomes. Subgroup analyses were conducted to compare responses between patients with and without aura, and between those with or without status migrainosus (prolonged migraine attacks >72 hours). These analyses were exploratory and not pre-specified in the study protocol.
To evaluate the potential influence of migraine prophylactic treatment on outcomes, separate analyses were conducted comparing patients who were already receiving preventive treatment prior to the intervention versus those who were not, as well as between those who were prescribed prophylactic treatment after the intervention and those who were not. These comparisons used the Mann–Whitney U test for continuous outcomes and Fisher's exact test for categorical outcomes.
Safety outcomes were summarized descriptively using frequencies and percentages. No inferential statistical tests were performed on adverse event data due to the small sample size and exploratory nature of the safety analysis.
All statistical tests were two-tailed and p < 0.05 was considered statistically significant.
Results
In total, 78 patients were initially assessed for eligibility, with 36 of them not meeting inclusion criteria and identified as pertaining to other primary headaches. Forty-two patients with acute migraine were enrolled and randomized in a 1:1 ratio to receive either triple therapy alone (n = 21) or triple therapy plus GON block (n = 21). All patients completed the intervention and follow-up assessments. There were no dropouts or missing data (Figure 1).

Flow diagram of the progress through the phases of the study of standard treatment group vs. standard treatment with greater occipital nerve (GON) block group according to CONSORT guidelines.
Baseline characteristics
Baseline demographic and clinical characteristics were comparable between groups (Table 1). The median age was 40.0 years (range 20–53 years) in the control group and 34.0 years (range 27–64 years) in the intervention group (p = 0.3988). The majority of participants were female: 81.0% (17/21) in the control group and 90.5% (19/21) in the intervention group (p = 0.6592).
Baseline characteristics of patients by treatment group.

Migraine without aura was the most frequent subtype: 76.2% in the control group and 66.7% in the intervention group, while migraine with aura occurred in 23.8% and 33.3%, respectively (p = 0.7327). Status migrainosus was present in 23.8% of patients in the control group and 14.3% in the intervention group (p = 0.6944).
Median duration since diagnosis was 3.0 years (range 1–39 years) in the control group and 6.0 years (range 1–23 years) in the intervention group (p = 0.897). Prior use of prophylactic migraine treatment was reported in 14.3% of patients in each group (3/21, p = 1.0). Among patients with previous prophylaxis, the most commonly used agents were propranolol (2/6; 33.3%), topiramate (2/6; 33.3%) and amitriptyline (2/6; 33.3%).
Primary outcome
At two hours post-treatment, a ≥50% reduction in pain intensity on the VAS was achieved by 20/21 patients (95.2%) in the intervention group and 10/21 (47.6%) in the control group with an absolute difference of 47.6% (95% confidence interval (CI) = 24.4–70.8; p = 0.0003) and the relative risk was 2.00 (95% CI = 1.27–3.14). The median VAS reduction was 6.0 points (range 4–9) in the intervention group versus 3.0 points (range 1–5) in the control group (p < 0.001) (Figure 2 and Table 2).
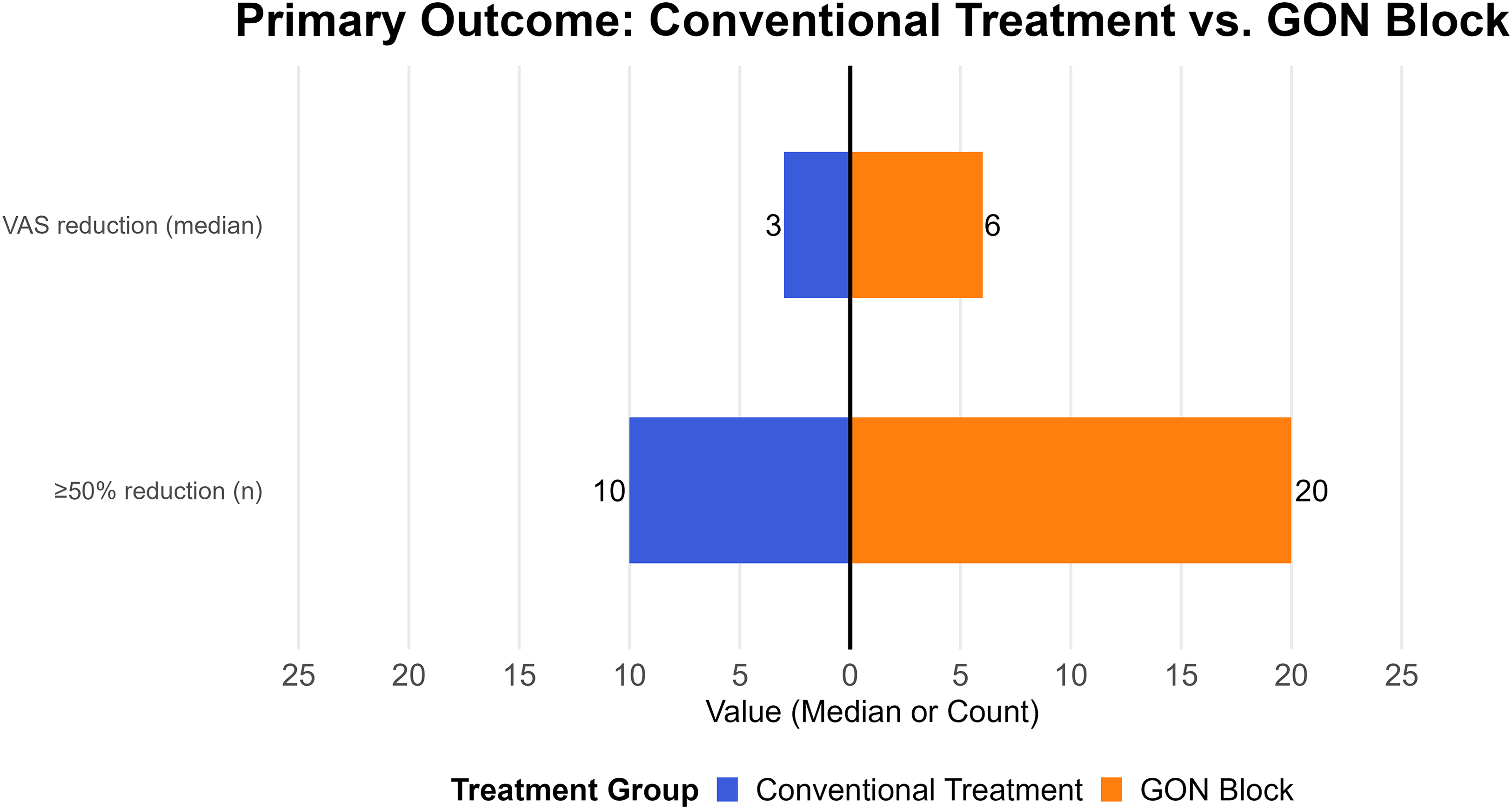
Primary outcome measures comparing conventional treatment vs. greater occipital nerve (GON) block. Bar plot showing the median reduction in visual analog scale (VAS) scores and the number of patients achieving ≥50% pain reduction two hours after intervention, comparing the conventional treatment group (blue) and the GON block group (orange).
Combined summary of primary outcome (pain reduction).

CI = confidence interval; VAS = visual analog scale.
Secondary outcomes
At 30-day follow-up, the intervention group had significantly fewer migraine days per month (median 3.0 days, range 0–11 days) compared to the control group (median 7.0 days, range 2–15 days; p = 0.0355). Pain-free days were more frequent in the intervention group (median 14.0 days, range 4–26 days) versus the control group (median 5.0 days, range 0–20 days; p = 0.0379). The median HIT-6 score was 59.0 (range 48–72) in the intervention group and 65.0 (range 56–76) in the control group (p = 0.1584). Hospital readmission for migraine occurred in 2/21 patients (9.5%) in the intervention group and 5/21 (23.8%) in the control group with an absolute difference of 14.3% (95% CI = −7.8 to 36.4; p = 0.4076) corresponding to a relative risk of 0.40 (95% CI = 0.09–1.84; p = 0.4076) (Figure 3 and Table 3).
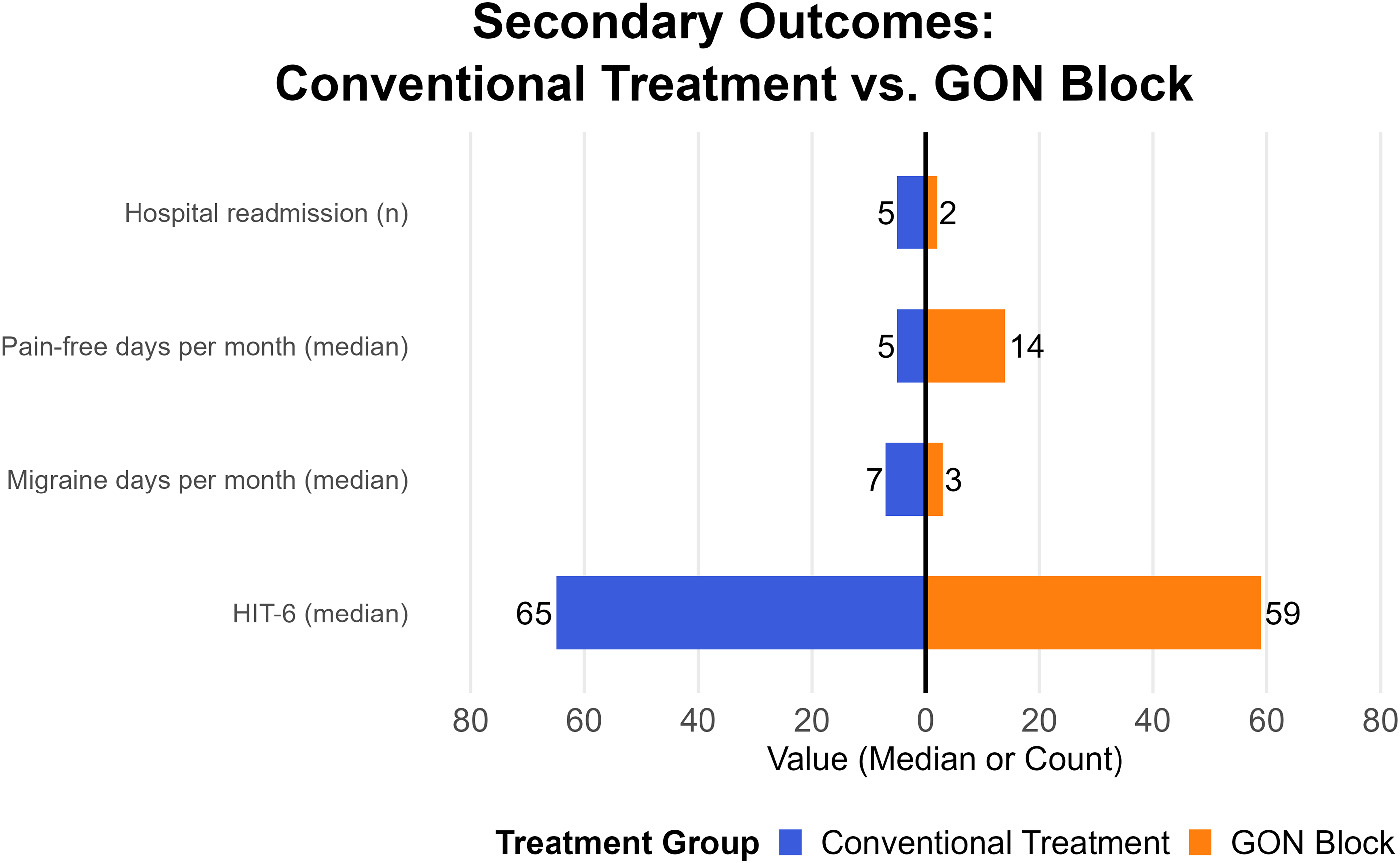
Secondary outcomes at 30-day follow-up comparing conventional treatment vs. greater occipital nerve (GON) block. Bar plot showing secondary outcomes at 30-day follow-up, including median Headache Impact Test-6 (HIT-6) scores, monthly migraine days, monthly pain-free days and number of hospital readmissions. Comparisons are made between the conventional treatment group (blue) and the GON block group (orange).
30-day follow-up: comparison between groups.

HIT-6 = Headache Impact Test-6.
Subgroup analysis
In patients with migraine with aura (n = 12), median VAS decreased from 9.0 (range 7–10) to 4.5 (range 2–7). In those without aura (n = 30), VAS decreased from 9.0 (range 7–10) to 3.0 (range 1–7) (p = 0.0633 with respect to comparison at two hours).
Among patients with status migrainosus (n = 8), median VAS decreased from 8.5 (range 7–10) to 4.5 (range 2–6), and, in those without status migrainosus (n = 34), from 9.0 (range 7–10) to 3.5 (range 1–7) (p = 0.5069). No statistically significant differences were found between subgroups (Table 4).
Subgroup analysis: initial VAS and VAS at two hours

VAS = visual analog scale.
Prophylactic treatment analysis
After the intervention, 33 patients (78.6%) were prescribed migraine prophylactic treatment: 17 in the intervention group and 16 in the control group. The most frequently prescribed agents were amitriptyline (n = 22; 66.7%), topiramate (n = 5; 15.2%), propranolol (n = 5; 15.2%) and valproate (n = 1, 3.0%).
No statistically significant differences were observed in 30-day outcomes based on prior or newly prescribed prophylaxis. Among patients who were already on prophylactic treatment at baseline (n = 6), median HIT-6 = 63.0, migraine days = 5.0, pain-free days = 7.0 and hospital readmission occurred in 18.8%. In patients not on prior prophylaxis (n = 36), median HIT-6 = 64.0, migraine days = 5.0, pain-free days = 7.0 and hospital readmission occurred in 15.4%, respectively (Table 5).
30-day follow-up comparison according to prophylactic treatment use before intervention.
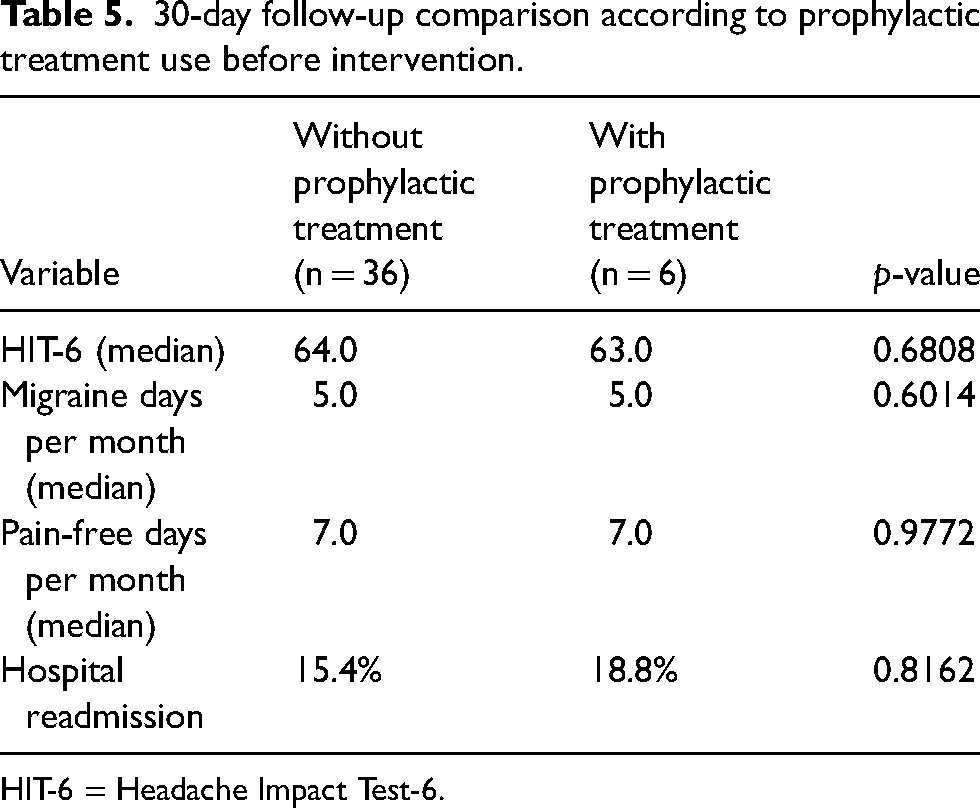
HIT-6 = Headache Impact Test-6.
In those prescribed prophylaxis after intervention (n = 33), median HIT-6 = 62.0, migraine days = 6.0, pain-free days = 7.0 and hospital readmission occurred in 15.2%. In those not prescribed prophylaxis (n = 9), median HIT-6 = 65.0, migraine days = 3.0, pain-free days = 7.0 and hospital readmission occurred in 22.2% (p > 0.05 for all comparisons) (Table 6).
30-day follow-up comparison according to prescription of prophylactic treatment after intervention.

HIT-6 = Headache Impact Test-6.
Safety outcomes
Within the first two hours post-injection, 10 patients (47.6%) reported local pain, three (14.3%) had minor bleeding at the puncture site and one (4.8%) reported transient paresthesia.
At 30-day follow-up, 10 patients (47.6%) continued to experience intermittent pain at the injection site and three (14.3%) had mild paresthesias. All adverse events were self-limited, non-serious and resolved without the need for additional intervention. No serious adverse events were reported during the study (Table 7).
Safety outcomes of the greater occipital nerve block.

Discussion
This prospective, randomized, controlled trial without use of placebo control study demonstrates that the addition of a GON block to triple intravenous therapy for acute migraine in the emergency department results in a statistically significant and clinically meaningful improvement in short-term pain relief. At two hours post-treatment, nearly all patients receiving the intervention (95.2%) achieved a ≥50% reduction in headache intensity, more than doubling the response rate observed in those receiving triple therapy alone (47.6%). Additionally, the magnitude of pain reduction was greater in the intervention group, with a median VAS decrease of 6.0 points compared to 3.0 in the control group. The lack of pain reduction in triple therapy may be due to the selection of severe and refractory migraine acute episodes. These findings support the clinical value of GON block as a rapid and effective adjunctive therapy for acute migraine management.
The benefits of the intervention extended beyond the immediate relief phase. At 30-day follow-up, patients in the GON block group experienced significantly fewer migraine days and more pain-free days compared to controls. While HIT-6 scores were lower in the intervention group, the difference did not reach statistical significance, likely due to the limited sample size. Importantly, these effects occurred regardless of whether patients had received or initiated prophylactic treatment, suggesting that the observed benefit was attributable to the acute intervention itself rather than a confounding preventive regimen, yet it would need further characterization between episodic and chronic migraine.
Previous studies have explored GON block primarily in the context of chronic migraine, showing favorable reductions in frequency and intensity of attacks. However, there is limited randomized evidence for its efficacy in the acute setting. The present findings are consistent with prior reports that GON block provides rapid and sustained analgesic benefit, likely mediated through modulation of the trigeminocervical complex. Notably, the block was well tolerated, with no serious adverse events reported. Minor local effects such as transient pain or paresthesia at the injection site were frequent but self-limited and required no additional intervention and may be associated with the 22-gauge needle chosen due to its availability.
Only a few studies have evaluated the efficacy of intensity reduction in acute migraine episodes. A clinical trial identified that GON blocks with bupivacaine alone were as effective as intravenous dexketoprofen and metoclopramide, and superior to placebo in acute migraine episodes (16,23). Another double blind, placebo-controlled trial determines that GON block, supraorbital block or combination of both were superior to placebo in acute migraine (15). Alternatively, a double dummy, double blind controlled clinical trial established that metoclopramide drip and sham GON block was equivalent to GON block, with bupivacaine, plus sham drip (21). A recent meta-analysis evaluated intensity reduction of ropivacaine and bupivacaine in acute migraine attacks throughout four clinical trials. A significant reduction of intensity score was noted at 30 minutes and the 45–60-minute timeline, while no statistically significant reduction was reported at 0–15 minutes. Similarly, no prolonged headache relief was shown with GON block (defined as pain-free 2–48 hours. without medication) and no significant reduction in the need for additional acute medication was noted. On the other hand, very few major side effects were observed, acknowledging GON as a safe procedure. Common adverse effects reported were neck pain, dizziness, pain at puncture sites and vertigo. Likewise, nerve blocking was identified as a useful intervention for pain relief and allodynia within 5–20 minutes of an acute migraine episode (24).
The strengths of this study include its prospective randomized design, complete follow-up, and real-world applicability in an emergency care setting. The use of both objective (VAS) and functional (HIT-6) outcome measures strengthens the internal validity of the findings. In addition, the assessment of safety outcomes, both immediately and at 30 days, provides reassurance regarding the tolerability of the procedure.
Several limitations should be noted. This was a single-center study with a modest sample size and no public trial registry. The lack of blinding and differentiation between migraine subtypes may have influenced patient-reported outcomes, though the observed effect size supports a real benefit. Follow-up was limited to 30 days, without data on medication overuse, time from symptom onset, or use of abortive treatments. Outcomes were collected by phone without a headache diary, which limited precision but was appropriate for the emergency care context.
Despite these limitations, this study provides compelling evidence that GON block, when added to triple therapy, enhances pain relief and improves short-term clinical outcomes in patients with acute migraine. Given its low cost, feasibility and favorable safety profile, GON block represents a promising adjunct in the acute management of migraine, particularly in emergency settings where rapid symptom control is a priority. Further multicenter studies with larger sample sizes and longer follow-up are warranted to confirm and expand upon these findings.
Key findings
Adjunctive greater occipital nerve block plus conventional therapy significantly improved acute migraine pain in emergency care. A ≥50% pain reduction at two hours was achieved in 95.2% of the intervention group vs. 47.6% of the control group. The intervention group had fewer migraine days and more pain-free days at 30-day follow-up. No serious adverse events were reported; the combination therapy was safe and well tolerated.
Footnotes
Author contributions
Cesar D. Tamayo had a major role in the acquisition, analysis or interpretation of data, and revised the manuscript critically for important intellectual content. Elliot G. Gama had a major role in the acquisition, analysis or interpretation of data, and revised the manuscript critically for important intellectual content. Felipe A. Paredes contributed to data collection and data entry, and supported the interpretation of study findings. Javier A. Galnares, was responsible for investigations, as well as writing – reviewing and editing, and supervision.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical statement
The study was approved by the Institutional Research and Ethics Committee of the National Institute of Neurology and Neurosurgery “Manuel Velasco Suarez” (Instituto Nacional de Neurologia y Neurocirugia “Manuel Velasco Suarez”, Mexico City). All procedures were conducted in compliance with current Good Clinical Practice (GCP) guidelines, national regulatory standards, and the principles of the Declaration of Helsinki. Written informed consent was obtained from all participants before enrollment.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
