Abstract
Objective:
We report a case series of patients diagnosed with the syndrome of transient headache and neurological deficits with cerebrospinal fluid lymphocytosis (HaNDL) responding to high dose Valproic acid.
Background:
HaNDL syndrome is an infrequent entity, presenting with neurological impairment episodes and severe headache. A treatment to prevent the attacks has not been proposed yet.
Results:
We describe 6 patients with a definite diagnosis of HaNDL, responding to Valproic acid 1000–1500 mg/day.
Conclusion:
Although HaNDL’s self-limiting nature, episodes may cause important disability and can last up to 3 months. Valproic acid may be a good choice to prevent attacks due to its effect on cortical depression and fast titration.
The syndrome of transient headache and neurological deficits with cerebrospinal fluid lymphocytosis (HaNDL) is defined as migraine-like headache episodes accompanied or shortly preceded by hemiparaesthesia, hemiparesis, and/or dysphasia, lasting at least 4 hours and associated with cerebrospinal fluid (CSF) lymphocytic pleocytosis (>15 white cells per µl). 1 Although the disorder resolves spontaneously within 3 months, the nature of this syndrome which presents with recurring episodes of neurological impairment up to 12 times, may cause significant disability in patients. 1,2 However, there are no proven treatment options to abort the episodes or prevent recurrence; treatments have only been described in case series/reports, especially when there are signs of accompanying intracranial hypertension. 3
In this case series, we report 6 patients presenting with HaNDL syndrome and responding to Valproic acid. This is the first case series reporting improvement of HaNDL syndrome episodes by using Valproic acid to the best of our knowledge.
The clinical, CSF, electroencephalographic and radiological findings, duration and dosage of valproic acid treatments are summarized in Table 1. Patients’ neurological and fundoscopic examination, magnetic resonance venography and angiography were normal.
Synopsis of clinical data in HaNDL patients.
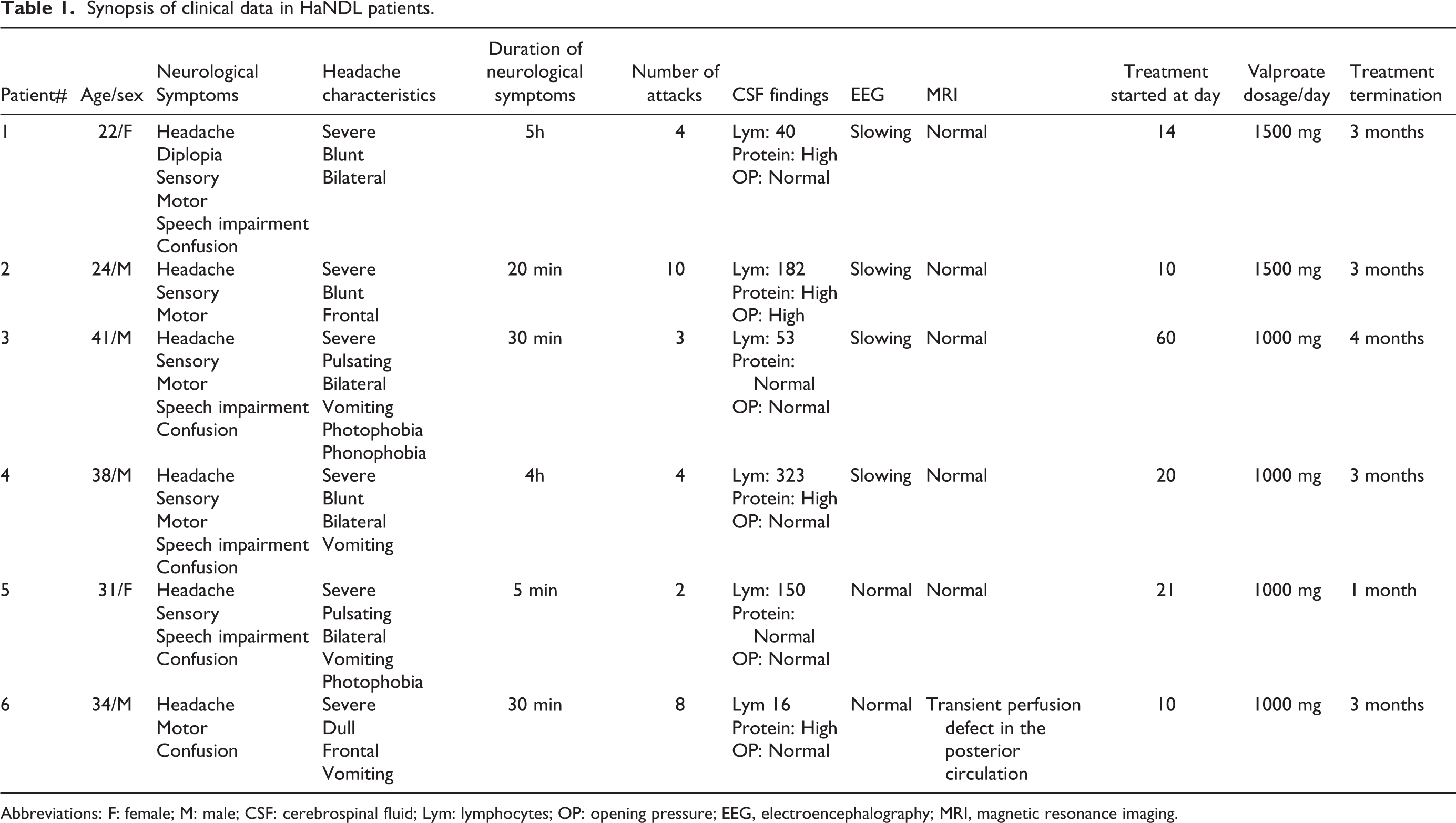
Abbreviations: F: female; M: male; CSF: cerebrospinal fluid; Lym: lymphocytes; OP: opening pressure; EEG, electroencephalography; MRI, magnetic resonance imaging.
Data were retrospectively analyzed of patients followed up by two headache centers with a definite diagnosis of HaNDL, and for who we have used oral Valproic acid as a treatment were included. The numeric pain rating scale was (NPRS) used to record the patient’s head pain intensity. The indication of no pain was 0; the worst pain was 10 on a self-reported eleven-point scale . The most common side effects of treatment were nausea, asthenia, somnolence and therefore three of our patients (Patient 3, 4 and 5) needed a dosage reduction to 1000 mg/day.
All patients gave written informed consent to be published.
Patient 1
A 22-year-old female patient presented with numbness episodes in the right facial half, right arm, and leg that started 2 weeks ago, recurring twice, and lasted 4–5 hours. This complaint was accompanied by loss of strength in the right arm, dysarthria, and confusion. However, the headache did not occur with these symptoms; it appeared twice on different days, bilaterally, the intensity of numeric pain rating scale (NPRS) 8–9, blunt, and regressed in about 1 hour. Confusion and diplopia accompanied the headache. Flunarizine recommended by another center was stopped, and 1500 mg of Valproic acid prevented further episodes of confusion, double vision and headache after the first dose.
Patient 2
A 24-year-old male patient experienced a headache that awakened him in the morning and caused him to admit to the emergency room 10 days ago. Headache intensity was NPRS 9, frontal, blunt, followed by vomiting. The headache became recurrent in the days after that, self-limiting in around 20 minutes, accompanied by marching numbness in the left face, left arm and leg, and paresis in the left half of the face. With valproic acid (1500 mg dose), the disabling headache decreased to NPRS 6 intensity 3 days later, and vomiting did not recur. Also, the frequency of paresthesia attacks during the day decreased and resolved entirely within 2 weeks.
Patient 3
A 41-year-old man presented with numbness episodes in the left half of the face, arm, and leg. Additionally, he showed loss of strength in the left leg, impaired speech, and confusion, repeating 3 times in a 2-month period, lasting approximately 30 minutes. Bilateral pulsating headache intensity of NPRS 9, followed by vomiting, photophobia, and phonophobia accompanied and remained for about 6 hours. He exhibited an irritable personality trait between the attacks, had noticeable forgetfulness. He received antiviral, antibiotic treatment, and lamotrigine in two other centers. The patient’s neurological episodes did not reappear after the first dose of 1500 mg Valproic acid, but due to side effects, the amount was reduced to 1000 mg. His wife observed a pronounced improvement in his aggression and restlessness within the first week of treatment.
Patient 4
A 38-year-old male patient presented with a headache intensity of NPRS 9–10, which started 20 days ago and appeared daily. The headache was bilateral and blunt, and only vomiting was accompanied. During this period, 4 episodes of dysarthria, alternating weakness and numbness in both arms, and confusional findings regressing within 3–4 hours occurred. Despite a high cerebrospinal fluid pressure, the patient’s neurological and fundoscopic examination was normal. He described a marked relief of severe daily headaches within the first week with 1000 mg Valproic acid and did not experience any neurological episode after the first dose.
Patient 5
A 31-year-old breastfeeding female patient presented with a hemicranial headache intensity of NPRS 9, which started 3 weeks ago and appeared almost every day. She had a medical history of migraine without aura and described her pain as bilateral, throbbing, accompanied by photophobia, nausea, and vomiting. Two episodes of numbness in both arms and confusion, lasting 5 minutes, were accompanied twice by the headache. Verapamil, which was given in another center, did not relieve her complaints. Her headache, which she described as worse than her migraine and non-responsive to painkillers, has totally disappeared 4–5 hours after the first administration of Valproic acid 1000 mg/day. She did not experience any neurological symptoms again.
Patient 6
A 34-year-old male patient presented with a dull frontal headache intensity of NPRS 7–8, occurring every day for 10 days. The headache was exacerbated by transient episodes of left hemiparesis, which developed 8 times, the longest lasting 30 minutes, and was accompanied by confusion, nausea, and vomiting. One of the episodes developed while driving his car, and he had to stop suddenly. Flunarizine, started by another center, was stopped, and Valproic acid 1500 mg relieved his headache and ictal symptoms within 2 days.
Headaches in HaNDL are described as moderate-to-severe, bilateral or unilateral, pulsating and can co-occur, precede or follow these neurological symptoms and rarely headaches can be absent. 2 Our patients, similarly, described their head pain as bilateral, dull or pulsating, severe, disabling, and sometimes lasting for several days, accompanied by vomiting and photophobia. Patient1 experienced some neurological episodes without any headache.
Our patients were admitted at least one time to an emergency unit, sought medical attendance 3 to 5 times, and experienced 2 to 10 episodes. Their condition was defined as viral meningoencephalitis, migraine with aura, sinusitis, and hemiplegic migraine before their definite diagnosis. Usually, auras of migraines precede the headache, are predominantly visual, and last for 1 hour, with normal CSF. Sporadic hemiplegic migraine is a recurring disorder, and the CSF findings don’t show any abnormality. Viral meningoencephalitis commonly presents with clinical findings such as fever and signs of meningeal irritation, and typical CSF findings.
Although the disorder resolves spontaneously within 3 months, the nature of this syndrome which presents with recurring episodes of neurological impairment up to 12 times, may cause significant disability in patients. 2 Some of our patients who had frequent and severe attacks expressed that they have felt very helpless and anxious at that time. Their condition and repeated medical visits had an impact on productivity at school or work. Most likely, the recurrent occurrence of episodes, disabling neurological manifestations may have raised some patient’s expectations to get their condition under control as soon as possible.
Methylprednisolone acetazolamide has been reported as a treatment, especially when an association of HaNDL and intracranial hypertension was detected. 3 –6 Prednisolone, valproic acid, and verapamil were used together in one case, but the effectiveness of these drugs was not mentioned in detail. 7 We have prescribed oral Valproic acid 1000–1500 mg for an average of 1–4 months. In our case series, patients reported an immediate improvement in headache and neurological attacks after administration of Valproic acid. Time elapsed since the beginning of the illness taking Valproic acid was 10–60 days. The fast treatment response that exceeded our expectations might be due to Valproic acid’s effect on cortical spreading depression and the ability to titrate it rapidly to achieve therapeutic levels. 8 This feature led to the use of valproate in the treatment of migraine and acute migraine episodes. It is thought that cortical spreading depression develops in HaNDL syndrome’s pathophysiology, similar to migraine with aura. 9
Clinical implications
HaNDL episodes may cause significant disability in patients
Although HaNDL syndrome is a self-limiting disease, there may be a need to abort episodes
Valproic acid may be a good option to prevent attacks by suppressing cortical spreading depression
Footnotes
Authors’ note
We confirm that we have read the Journal’s position on issues involved in ethical publication and affirm that this report was written with these guidelines.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
