Abstract
Background:
Cluster headache (CH) is one of the most painful conditions in humans and there is limited epidemiological data on this debilitating condition.
Objectives:
To describe the epidemiology of CH in Norway
Methods:
We conducted a nationwide study to investigate the prevalence, incidence, and comorbidity of CH in Norway between January 1 2008 and December 31 2016. Treatment and outcome data from the Norwegian patient registry and the Norwegian prescription database were linked on an individual basis.
Results:
Among 3,892,260 individuals ≥18 years old of age, we identified a total of 1891 patients with CH. The prevalence of CH was 48.6 per 100,000, and the male-to-female ratio was 1.47. The estimated incidence of CH was 3.0 per 100,000/year. Among patients with CH, increased age and sex adjusted odds ratios ([OR], all with p-values <0.0001, were observed for medication-induced headache (OR 50.7, 95% CI 36.7–69.9), migraine (OR 25.2, 95% CI 22.5–28.3), chronic posttraumatic headache (OR 22.2, 95% CI 12.8–38.45), history of cranial trauma (OR 1.9, 95% CI 1.5–2.4), somatoform disorders (OR 4.2, 95% CI 3.0–5.8), suicide attempt (OR 3.9, 95% CI 2.6–5.8), personality disorder (OR 3.6, 95% CI 2.6–4.9), bipolar disorder (OR 3.6, 95% CI 2.8–4.8), peptic ulcer (OR 2.8, 95% CI 2.3–3.3), depression (OR 2.8, 95% CI 2.4–3.1), substance abuse (OR 2.6, 95% CI 2.0–3.3), and cerebrovascular disease (OR 2.4, 95% CI 1.8–3.1). Use of opioid analgesics during the study period was more common among patients with CH compared to others (81% vs. 22%, sex and age adjusted OR 23.4, 95% CI 20.8–26.2, p < 0.0001).
Introduction
Cluster headache (CH) is an excruciating and highly disabling primary headache disorder that is widely considered to be one of the most painful human conditions. 1,2 Further, CH has been associated with reduced quality of life, increased risk of depression, marked functional disability, and suicide. 1,3 –6 There have been several studies assessing CH’s prevalence (Table 1). There is limited high-quality epidemiological data on CH and methodologically rigorous population-based studies are scarce. 29 CH’s comorbidity remains also insufficiently documented 30 (Table 2). This Norwegian nationwide study aims to investigate the prevalence and incidence of CH. Further, we sought to assess comorbidity and use of prescription drugs in patients with CH compared to the general population.
CH: cluster headache. ECH: episodic cluster headache. CCH: chronic cluster headache.
OR: odds ratio.
a Model adjusted by age, sex and group (CH/controls).
b Other GI (gastrointestinal) problems: other than celiac disease, GERD (gastroesophageal reflux disease). Only statistically significant differences found in the different studies presented in this table are included.
c Nocturnal dipping: normal decrease of 10–20% in nocturnal blood pressure.
d IMT: intima-media thickness (measured by carotid ultrasound).
e Charlson’s score: method of predicting mortality by classifying or weighting comorbid conditions. MA+: migraine with aura; MA−: migraine without aura. MCMI-III: Millon Clinical Multiaxial Inventory-III.
Methods
Ethical approval and waiver of the requirement for obtaining patient consent were granted by the Regional Committee for Medical Research (REK 2016/1504). Reporting is consistent with the STROBE statement (S1 Checklist).
Study population
This study was conducted within the 3.8 million inhabitants of Norway ≥18 years of age (2016 census) from January 1, 2008, to December 31, 2016. Two administrative registries were linked on an individual level by a unique 11-digit personal identifier. The registries linked were: (A) the Norwegian Patient Registry (NPR), which contains information about inpatient and outpatient treatment in Norwegian hospitals since 2008 with diagnoses coded according to the 10th revision of the International Classification of Diseases (ICD-10) 51 and (B) The Norwegian Prescription Database (NorPD), which contains information about all prescriptions dispensed in Norway since 2004 including type of drug according to the Anatomical Therapeutic Chemical classification system. 52,53 Diagnoses are registered for medications with reimbursement according to ICD-10 or the International Classification of Primary Care version 2 (ICPC-2). All residents ≥18 years during the study period included in NPR and/or NorPD were eligible for inclusion. Data from NPR and NorPD were de-identified before being provided to the study authors, and the exact dates for prescription dispensing and death were not made available to the authors. NPR receives data monthly and is used for reimbursement purposes, hospital statistics, and research. 54 Sensitivity, specificity, and positive predictive values for neurological diagnoses in NPR are found to be high, indicating that the registry is well suited for this epidemiological study. 51 Nonetheless, the NPR has not been specifically validated for headache diagnoses.
Outcome measures
The primary outcome measures were prevalence and incidence of CH according to ICD-10. The diagnoses screened in this study are presented in Supplementary Table 1. The ICD-10 code for CH (G44.0) is the same used for chronic paroxysmal hemicrania. A diagnostic criterion for chronic paroxysmal hemicrania is the prevention of attacks by indomethacin, and patients who had dispensed the arbitrary amount of >200 tablets were not considered to have CH. ICD-10 does not provide different codes for episodic and chronic CH.
In order to estimate incidence of CH we divided our database in two periods: period 1 (2008–2011) and period 2 (2012–2016). Patients who had not received the diagnosis CH in period 1 but who received the diagnosis in period 2 were used to calculate an incidence rate in the second period. Secondary endpoints were assessment for comorbidities and use of prescription drugs (Supplementary Table 2).
The Norwegian health care system
Norway has a public health care system with evenly distributed resources. Most patients with CH are treated by neurologists working in the public health care system. Those who are treated in the private sector are also registered in the NPR. Health authorities cover all inpatient treatment for patients with CH, and costs concerning established treatment options are not a concern for the individual patients. Except for a maximum annual deductible of 2185 Norwegian Kroner (210 USD, 2020), prescription drugs are provided to patients without further costs. This ensures access to medicines regardless of financial situation.
Statistics
Analyses were performed with R 3.1 (R-Foundation for Statistical Computing), SPSS 25.0 (IBM Corp., Chicago), and MySQL (Oracle). Due to multiple comparisons, statistical significance level was defined as p ≤ 0.001 when investigating comorbidity and drug use in CH patients and people without CH in the NPR. For other statistical tests the statistical significance level was defined as p ≤ 0.05. Exposure to a drug was defined as having occurred when the drug was prescribed. Patients were followed until death, emigration, or end of study period.
Results
The average population in Norway ≥18 years of age during the study period was 3,892,260. The number of patients who received the diagnoses G44.0 was 2734. We identified a total of 1891 patients who fulfilled our criteria for having CH, providing a prevalence of CH of 48.6 per 100,000 (between January 1, 2008 and December 31, 2016). We identified 584 patients who received the diagnosis in the period between 2012 and 2016. The estimated incidence of CH was 3.0 per 100,000 per year.
Among 1891 patients with CH 1126 were men (59.5%), providing a male-to-female ratio of 1.47. The mean age of the whole population was 55.8 (±18.9) years. The median age of the patients with CH was 42 years (interquartile ratio: 20). Figure 1 shows the age distribution of patients with CH.

Age distribution of patients with CH in our nationwide sample.
Forty-two patients with CH (2.2%) died between 2008 and 2016. The expected number of deaths in the CH group according to the mortality in the Norwegian population in 2016 adjusted for age and gender would have been 30.3 deaths in that period of time (HR: 1.4, CI 1.0–1.9, p = 0.044).
The main comorbid headache disorders in patients with CH were drug-induced headache, trigeminal neuralgia (TN), tension-type headache (TTH), migraine, atypical facial pain and chronic posttraumatic headache (Table 3 and Figure 2). Other common medical comorbidities were peptic ulcer, cerebrovascular disease, hemorrhagic complications, hypertension, thromboembolism, and chronic renal failure. The main psychiatric comorbidities were somatoform disorders, suicide attempt, personality disorders, bipolar disorders, depression and substance abuse. After adjusting by age and gender, there was no association with increased risk for intracranial hemorrhage, cognitive symptoms, chronic heart failure, atrial fibrillation, liver disease or diabetes mellitus.
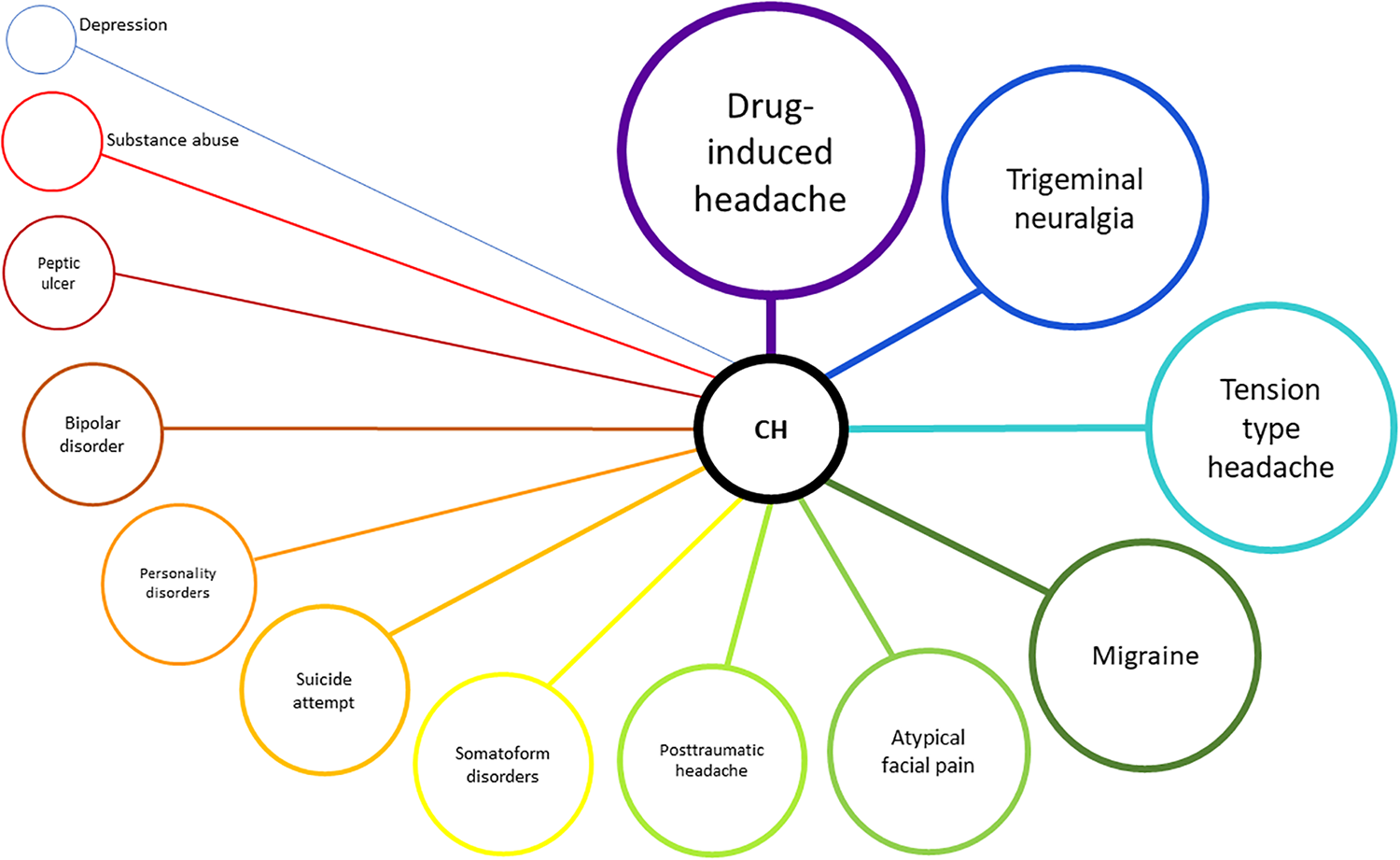
Visualization of the most common comorbid diagnosis found in this nationwide study (see Table 3 for further information). CH: cluster headache.
Unadjusted and adjusted odds ratios for age and sex for different comorbidities in our nationwide sample.
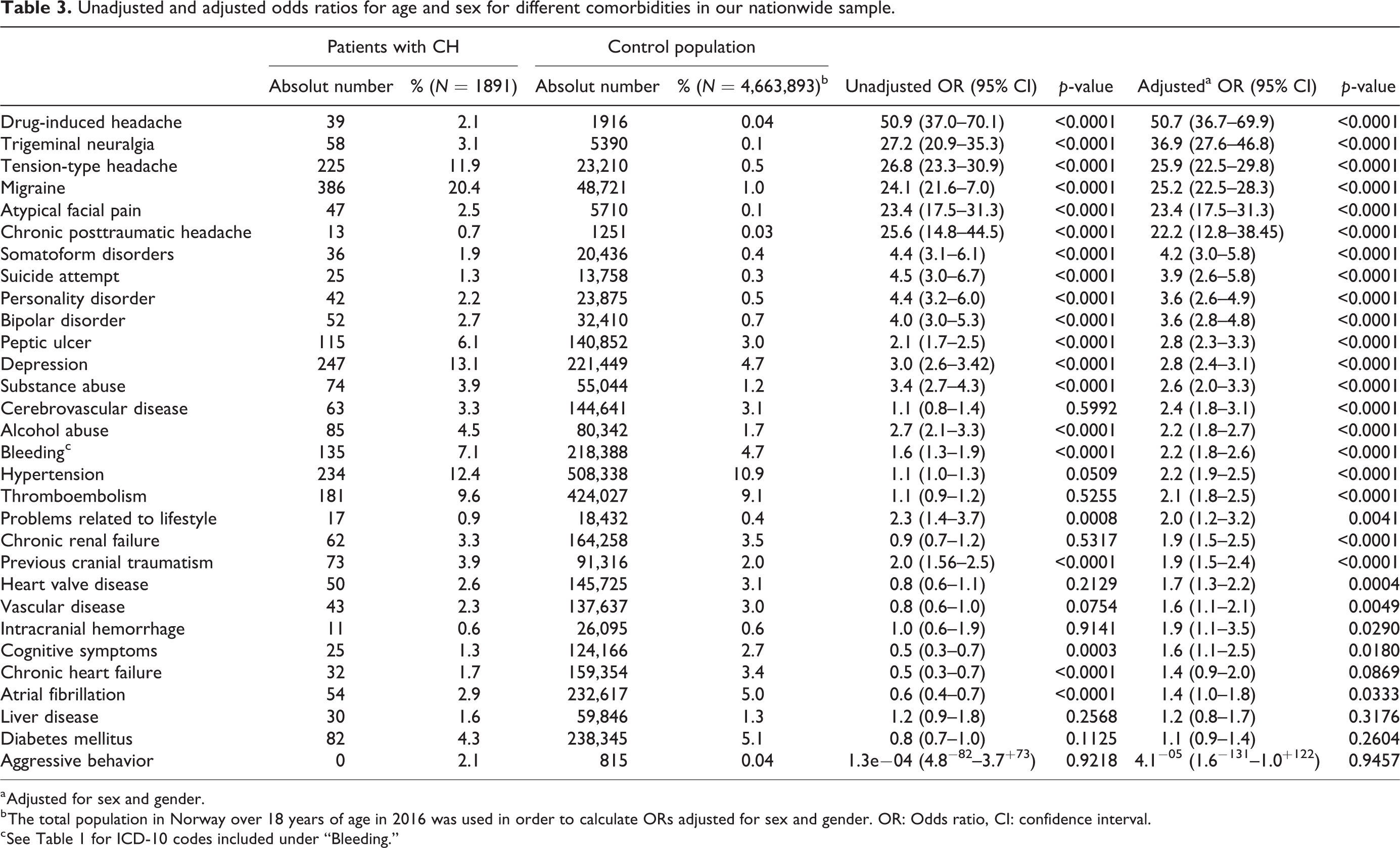
a Adjusted for sex and gender.
b The total population in Norway over 18 years of age in 2016 was used in order to calculate ORs adjusted for sex and gender. OR: Odds ratio, CI: confidence interval.
c See Table 1 for ICD-10 codes included under “Bleeding.”
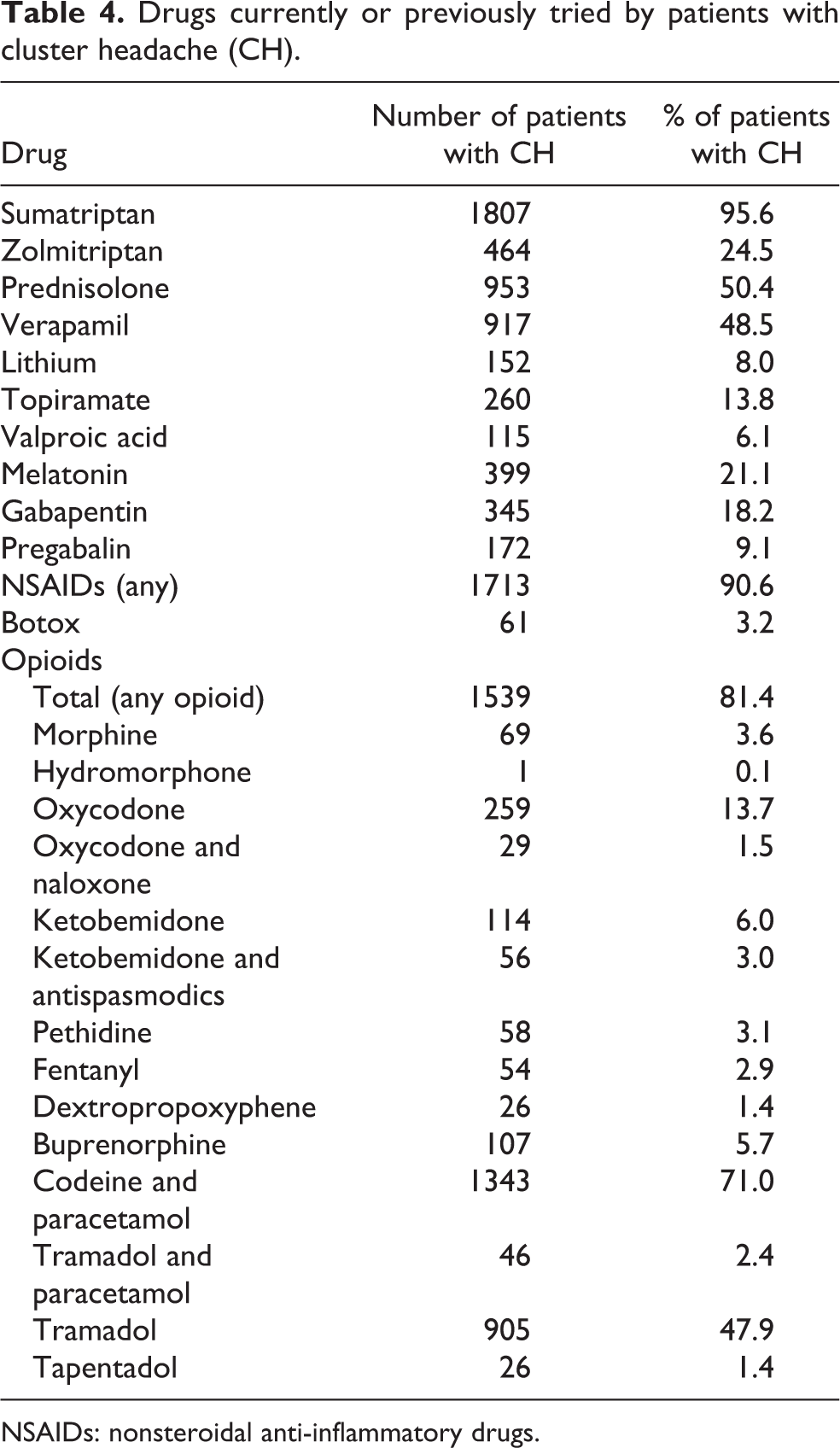
Most of the CH patients had tried sumatriptan and prednisolone was dispensed in half of them (Table 4). Eighty-one percent (1539) of the CH patients were dispensed opioids during the study period versus 22% (854,470) of the rest of the population, unadjusted OR: 19.3 (CI: 17.2–21.7, p < 0.0001); adjusted OR by sex and age: 23.4 (95% CI 20.8–26.2 p < 0.0001). Seven CH patients (0.37%) had an occipital stimulator implanted between 2008 and 2016.
Drugs currently or previously tried by patients with cluster headache (CH).
NSAIDs: nonsteroidal anti-inflammatory drugs.
Discussion
In this nationwide study of CH, we report a prevalence of 48.6 per 100,000 and annual incidence of 3.0 per 100,000. We further show that CH is associated with a higher risk of potentially severe medical, and psychiatric comorbidities and higher use of opioid analgesics. High-quality population-based studies on prevalence, incidence, and comorbidity are scarce. 29,30 A population-based study based on direct interviews in a small, but well-defined geographical catchment area in Norway, found that 0.38% (7/1838) fulfilled the criteria of CH and that among these only two had consulted a physician. 7 In another study in Sweden, only four out of nine patients with CH had sought professional help 8 even though they had experienced CH for 6 years on average. This raises the possibility that our study underestimates the real prevalence. Prevalence in studies conducted in Africa range between 30 and 1298.7 per 100,000. 9,10 The lifetime prevalence of CH has been estimated around 0.2–0.3%. 55 Studies that report 1-year prevalence of CH have been criticized for possibly underestimating the real prevalence as some patients might not have bouts during the observation period. Our prevalence of 48.6 per 100,000 is lower than the median prevalence of 121.5 per 100,000 observed in other studies (Table 1). This might be due to several factors including selection bias, non-consulting, misdiagnosis, or lack of specialist visit between 2008 and 2016.
Few studies have reported incidence of CH (Table 1). Incidence rates vary between 2.1 and 9.8 per 100,000 per year. 11 –13 Whereas the sample size of previous studies ranges between 104 and 36,145, 14 this study included the entire adult Norwegian population of more than 3.8 million individuals. The incidence of 3.0 per 100,000 per year is within the range of previous studies.
While early reports on CH reported up to a 14-fold male predominance, 15 –17 later studies have detected more women with CH. 18,19 The decrease in the male-to-female ratio might be due to increased awareness of the possibility of other primary headache diagnoses in women other than migraine. Our observed male-to-female ratio of 1.47 seems consistent with more recent epidemiological studies, 18 –20 and it is possible that methodologically more rigorous studies in larger populations have simply clarified a more accurate male to female ratio that in the past was overestimated.
Comorbid headache disorders
As seen in other reports, 56,57 our study suggested that medication-induced headache appears to be a major problem in CH patients. Unfortunately, there is no ICD-10 code that perfectly overlaps with the ICHD-3 diagnosis “Medication-overuse headache.” The ICD-10 code that is more analogous is G44.4 (“Drug-induced headache”). Distinguishing atypical facial pain, TN and CH often represents a real challenge in daily clinical practice. 58 –63 Fifty-eight patients with CH (3.1%) in our study also received a diagnosis of TN. This raises the possibility of diagnostic uncertainty. The same problem applies to the 47 patients (2.5%) with CH who also received the diagnosis atypical facial pain. In one study, 16% of the patients with CH initially received the diagnosis of TN before it was revised to CH. 31
Migraine was a common comorbidity in our sample (20.4% vs. 1.0% of people without CH in the NPR). TTH was also more common in CH patients (11.9% vs. 0.5% of people without CH in the NPR). Population-based studies have shown a 1-year prevalence of 43.1% for TTH and of 18.1% for migraine. 21 Other authors have found that 15.6% of patients with CH had comorbid migraine. 32 Most patients with TTH or migraine are not seen by neurologists in our country and that these are less likely to be registered as ICD-10 diagnosis by specialist health care services. This might be the reason why the percentage of people without CH who were diagnosed with migraine was lower than in population-based studies.
As reported by others, 33 we found that patients with CH were more likely to be diagnosed with traumatic head injuries and chronic posttraumatic headache. Due to the observational design of our study we cannot make any causal inferences.
Psychiatric comorbidities
We identified several severe psychiatric comorbidities to be more frequent in CH patients. Somatoform disorders were reported in 15% of patients in an Italian case series 34 ; a figure that is similar to population-based prevalence rates. However, only 2% of patients with CH in our study were diagnosed with somatoform disorders, raising suspicion that clinicians in Norway underreport somatoform disorders in the presence of other severe conditions such as CH. Most of the literature has focused on suicidal ideation rather than on suicide attempts in CH 30,35 –37 (Table 2). Twenty-five (1.3%) of the patients with CH in our study had made a suicide attempt that was serious enough to receive specialized care (vs. 0.3% of the population without CH). We might not have detected cases that did not seek help or where a suicidal attempt was not documented. Between 2008 and 2016 there were a total of 5069 deaths by suicide in Norway. 64 Patients with CH in this study had a higher risk of attempting suicide, putting these patients in a high-risk group. Our dataset did not have information about cause of death and thus we cannot know how many patients with CH did commit suicide during the study period.
Several studies report increased depression scores in CH patients 3,30,38 –41 and depression was a common diagnosis among patients with CH in our sample (%) but lower than most other studies (Table 2). The actual prevalence of depression in our sample might be higher if patients with CH were screened directly.
In this sample, 2.2% of patients with CH received a personality disorder diagnosis (vs. 0.5% of the population without CH). Other authors have reported a much higher prevalence of personality disorder 34,42 when studying CH patients directly with neuropsychological tests (Table 2). Further, we found that patients with CH were twice as likely to receive diagnoses of problems related to lifestyle (ICD-10 code Z72) and life-management difficulty (ICD-10 Z73) compared to the population without CH. ICD-10 codes Z72 and Z73 (Supplementary Table 1) include problems such as lack of physical exercise, inappropriate diet and eating habits, problems related to socioeconomic and psychosocial circumstances, and so on. Substance abuse was also increased in patients with CH. Heavy alcohol consumption has been reported in up to 10% of patients with CH. 33 Diagnoses of alcohol abuse registered by specialized health care services was higher in patients with CH (4.5% vs. 1.7% in people without CH) with numbers in line with another Scandinavian study. 38 Cigarette smoking has also been documented to be increased among patients with CH. 39,43 Unfortunately, we could not collect good quality data on this topic since the coding praxis among specialist health care services in Norway does not constantly register whether a patient smokes.
Comorbid medical diseases
Peptic ulcer has been described to have a significantly higher prevalence in men with CH compared to those without CH 44 and this was also true in our study. This might be related to the common use of nonsteroidal anti-inflammatory drugs (NSAIDs, which had been dispensed for 90.6% of our patients) and prednisolone (dispensed for 50.4% of the patients). It is noteworthy that half of the patients with CH had never tried prednisolone. Oral corticosteroids are often used for transitional prophylaxis but its level of evidence is low. 65 CH patients also had an increased risk for chronic renal failure which might be related to the common use of NSAIDs. Patients with CH had a two-fold increased risk of hemorrhagic complications (e.g. acute bleeding due to gastric ulcer or a duodenal ulcer, hematemesis, melaena, and hematuria; Supplementary Table 1) compared to people without CH in the NPR. We did not find an association between intracranial hemorrhage and CH after adjusting for age and sex.
Many CH patients in this study had not tried all available therapeutic options 65 –67 (Table 4). A detailed medication history, focusing on the drugs that have been tried before, the maximal dose and duration of treatment, as well as partial efficacy and side effect profile, may improve and expand treatment options. Most of the patients with CH had tried at least one opioid. There is no good evidence to recommend the use of opioids in the acute or chronic treatment of CH 65 and clinicians should be especially careful when prescribing opioids because of the risk of habituation, dependence, substance use disorders, comorbid psychiatric disorders, and suicide risk in this population. 68
In a series of 400 patients with CH compared to 200 controls, CH patients had a higher risk for cerebral infarction. 38 In the previous material, only patients between 18 and 65 years of age were included while we included all patients ≥18. In our patients with CH, risk of thromboembolism was doubled compared to the rest of the population (Table 3). The use of intravenous dihydroergotamine has been linked to increased rate of venous thrombosis both in patients with migraine and CH. 69 Unfortunately, we could not document how many patients with CH, migraine or the rest of the population were treated with intravenous dihydroergotamine during the study period as this is not registered in NorPD. Several cardiac conditions have previously been reported to be more prevalent in patients with CH. 38 In our data, patients with CH had an increased risk of developing vascular disease (i.e. coronary artery disease, atherosclerosis, aneurysm or dissection of big arteries and vascular dementia; Supplementary Table 1) or heart valve disease, however this is not a consistent finding in the literature. 44 The prevalence of chronic heart failure or atrial fibrillation were not increased in CH patients in our study after adjusting for age and sex. Vascular risk factors and lifestyle-related factors (alcohol and tobacco abuse) in patients with CH appear to be common, and a clinical evaluation to assess the risk for stroke, heart disease or thromboembolism should be an essential part of the follow-up in most CH patients.
Strengths and limitations
The major strength of this study is the large nationwide sample size collecting real world data in a relatively rare, but important condition. To our knowledge, the sample size exceeds any previous study.
The main limitation of this study is the observational design. Since data are based on diagnostic codes set in clinical practice, the results may be affected by the quality of coding practice.
The ICD-10 code for CH is the same used for chronic paroxysmal hemicrania. There is a possibility that the real prevalence of CH has been underestimated if some of the patients who dispensed more than 200 tablets of indomethacin actually had CH. ICD-10 does not differentiate between episodic and chronic CH. Future studies using ICD-11 will be able to differentiate between these two forms of CH.
Participants who had received the diagnosis G44.0 (CH) only once were also considered cases. Since there was no longitudinal assessment, some of these diagnoses might have been corrected outside the study period and changed to other ICD-10 diagnoses. This is also true for comorbidities detected in our study and some of them might have been misdiagnoses.
The calculation of incidence was limited by the fact that some patients in the 2008–2011 period may have received the diagnosis before 2008, and falsely been registered as “new patients” in this dataset.
In this study, data from January 1 2008 to December 31 2016 was included. The Norwegian patient registry uses ICD-10 codes which do not have a complete overlap with ICHD criteria. ICHD-II criteria were used in the specialist health care system between 2008 and 2013. ICHD-III Beta criteria were used between 2013 and 2016.
Validation of diagnoses and drug exposure in a representative subgroup of patients, would have strengthened our results. Still, previous validation studies have shown that NPR is adequately complete and correct to provide data for neurological epidemiological studies. 51
Conclusion
In this nationwide epidemiological study of cluster headache, we report a prevalence of 48.6 per 100,000 and annual incidence of 3.0 per 100,000. We further show that patients with CH have a higher risk of potentially severe medical and psychiatric comorbidities and higher use of opioid analgesics. This places this patient population at substantial risk of serious adverse health outcomes, beyond the disability caused by the headache disorder. This study emphasizes the need to systematically and comprehensively evaluate these patients from a general medical and psychiatric perspective in addition to a neurological evaluation. There is a risk that some or many of these diseases may be overlooked or not carefully searched for because of the very severe and highly disabling nature of the headache disorder itself.
Conclusion and relevance
CH was associated with higher risk of medical and psychiatric comorbidities, suicide attempts, and opioid use. In addition to deliberate and evidence-based management of CH, screening and appropriate management of comorbid diseases may improve overall outcome in patients with CH. Limitations include those inherent to observational studies including the inability to make causal inferences, assumptions regarding drug exposure, and the possibility of residual confounding.
Clinical implications
– In this Norwegian nationwide study, the prevalence of CH was 48.6 per 100,000 and the incidence was 3.0 per 100,000/year.
– The most common medical comorbidity was migraine requiring hospital specialist care (20.4% vs. 1.0% of people without CH).
– The most common non-headache medical comorbidity was hypertension (12.4% vs. 10.9% of people without CH). The most common psychiatric comorbidity was depression (13.1% vs. 4.7% of people without CH), and 1.3% of the patients had attempted suicide (vs. 0.3% of people without CH). A history of substance abuse was more common (3.9% vs. 1.2% of people without CH).
– The drug most commonly used was sumatriptan (95.6% of the patients). Most of the patients had been prescribed opioids at some point (81.4%).
– Cluster headache is a devastating headache disorder with potentially severe medical and psychiatric comorbidities.
Supplemental material
Supplemental Material, sj-docx-1-rep-10.1177_25158163221075569 - Epidemiology of diagnosed cluster headache in Norway
Supplemental Material, sj-docx-1-rep-10.1177_25158163221075569 for Epidemiology of diagnosed cluster headache in Norway by Joan Crespi, Sasha Gulati, Øyvind Salvesen, Daniel Fossum Bratbak, David W Dodick, Manjit Singh Matharu and Erling Tronvik in Cephalalgia Reports
Supplemental material
Supplemental Material, sj-docx-2-rep-10.1177_25158163221075569 - Epidemiology of diagnosed cluster headache in Norway
Supplemental Material, sj-docx-2-rep-10.1177_25158163221075569 for Epidemiology of diagnosed cluster headache in Norway by Joan Crespi, Sasha Gulati, Øyvind Salvesen, Daniel Fossum Bratbak, David W Dodick, Manjit Singh Matharu and Erling Tronvik in Cephalalgia Reports
Footnotes
Acknowledgments
The authors would like to thank the Norwegian patient registry and the Norwegian prescription database.
Author contributions
All authors read and approved the final manuscript. JC is the guarantor and was involved in planning the study, collection of the data, statistics, and writing of the manuscript. SG and ET gave the original concept of the study and were involved in writing of the manuscript. ØS and JC performed the statistical data analyses. MM and DD revised the final manuscript and were involved in the scientific organization of the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is funded by a grant from the Liaison Committee between the Central Norway Regional Health Authority and the Norwegian University of Science and Technology; grant number 46056923. The funders had no role in the study design, data collection or analysis, decision to publish, or preparation of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


