Abstract
Introduction:
Epicrania fugax (EF) is an epicranial headache syndrome characterized by short lasting headache paroxysms which spread in a typical linear/zigzag pattern. The definition of EF has evolved over time, and new phenotypes have been observed in various studies.
Aim:
We present eight cases diagnosed as EF according to the International Classification of Headaches Disorders, 3rd edition criteria and highlight certain novel characteristics which will help in the further categorization of this disorder.
Methods:
We prospectively studied eight cases of EF who presented to our headache clinic from January 2016 to September 2020. Demographic and detailed clinical data were collected and analyzed.
Results:
The mean age of onset of the cohort was 42 ± 10.47 years with a median duration of symptoms being 1 year. There were 5 male and 3 female patients. The mean visual analog scale score was 5.25 ± 1.83. Headache frequency had high intra and interindividual variability. Four patients had a typical spread of pain along the distribution of greater occipital, and supraorbital, nerves. Four patients had an atypical presentation with midline headache, transverse occipital, and bilateral simultaneous hemicranium involvement. Headache paroxysm was followed by a prolonged fixed pain in the same linear distribution in three patients. One patient had ipsilateral redness in the eye. All eight patients required medical intervention with amitriptyline or neuromodulators, five of whom reported a significant reduction in their headache symptoms.
Conclusion:
Our case series highlights a few interesting features which need further exploration with larger studies. Firstly, the atypical distribution of the pain trajectory forces us to dig deeper into the pathogenesis of the disorder. Secondly, our data also suggest a possibility of EF triggered “linear headaches.” EF is a relatively uncommon headache disorder that can be managed easily with neuromodulators but the lack of awareness of the condition often leads to delayed diagnosis.
Introduction
Epicrania fugax (EF) is a rare type of epicranial headache syndrome that was first described by Pareja et al. in 2008 based on their observations in 10 patients who presented with short lasting headache paroxysms which would originate from occipital region and travel forward up to the forehead in a linear or a zigzag trajectory hemicranially without any other characteristic associated features. 1 It was introduced as a distinct headache disorder in the International Classification of Headaches Disorders, 3rd edition (ICHD3) in 2018. 2 Its diagnosis requires stabbing pain across the surface of hemicranium moving with a linear or zigzag trajectory lasting up to 10 s and commencing and terminating in the distribution of different nerves. 2 The most distinguishing feature of EF is the linear trajectory along which the pain spreads and it helps in differentiating EF from its close mimics such as idiopathic stabbing headache, SUNCT, SUNA, and trigeminal neuralgia. Since the original description of posteroanterior linear trajectory, various phenotypic variants of EF have been described following other trajectories and the direction of the propagation of pain. A recent report of patients with facial EF has also been published 3 . 3 The origin of the pain in EF is considered to be peripheral, although central mechanisms might also play some part. We report our experience with eight patients of EF seen over a period of 4 years with some novel features and discuss the potential implications.
Patients and methods
We prospectively studied eight patients with epicranial headaches with EF phenotype attending a tertiary care headache clinic from January 2016 to September 2019. The patients who were recruited before 2018 were diagnosed as EF using the ICHD3 beta appendix criteria, whereas ICHD3 criteria were used for the patients recruited after 2018. Three patients had additional headache features after satisfying EF criteria and are discussed separately.
All the patients and their previous records were reviewed in detail. Both demographic and clinical data were noted. A detailed history of headaches and pain paroxysms were obtained which included age of onset, duration of the headache disorder, headache frequency, site, trajectory, character, and intensity in terms of visual analog scale (VAS), duration of the attack, associated features and aggravating and relieving factors of the pain. Medical history of any chronic illness and any treatment being taken for the same were also reviewed. We carefully excluded the mimics of EF. Referral patterns and physician diagnoses were also captured.
All patients had a normal systemic and neurological examination. Imaging and routine laboratory workup was performed in all patients to exclude secondary headaches. All our eight patients received treatment. They were prescribed amitriptyline or neuromodulator drugs namely gabapentin and oxcarbamazepine as monotherapy or in combination. Five patients had a minimum period of 3 months of follow-up. On the follow-up, the response to therapy in terms of headache frequency and severity were noted.
Results
Demography
The demographic and clinical data of the patients are presented in Table 1. There were five males and three female patients. Mean age of presentation was 43.75 ± 10.42 years (range 27–54 years), and the mean age of onset was 42 ± 10.47 years (range 22–54 years). The duration of symptoms ranged from 20 days to 6 years (median 1 year).
Details of the patients.
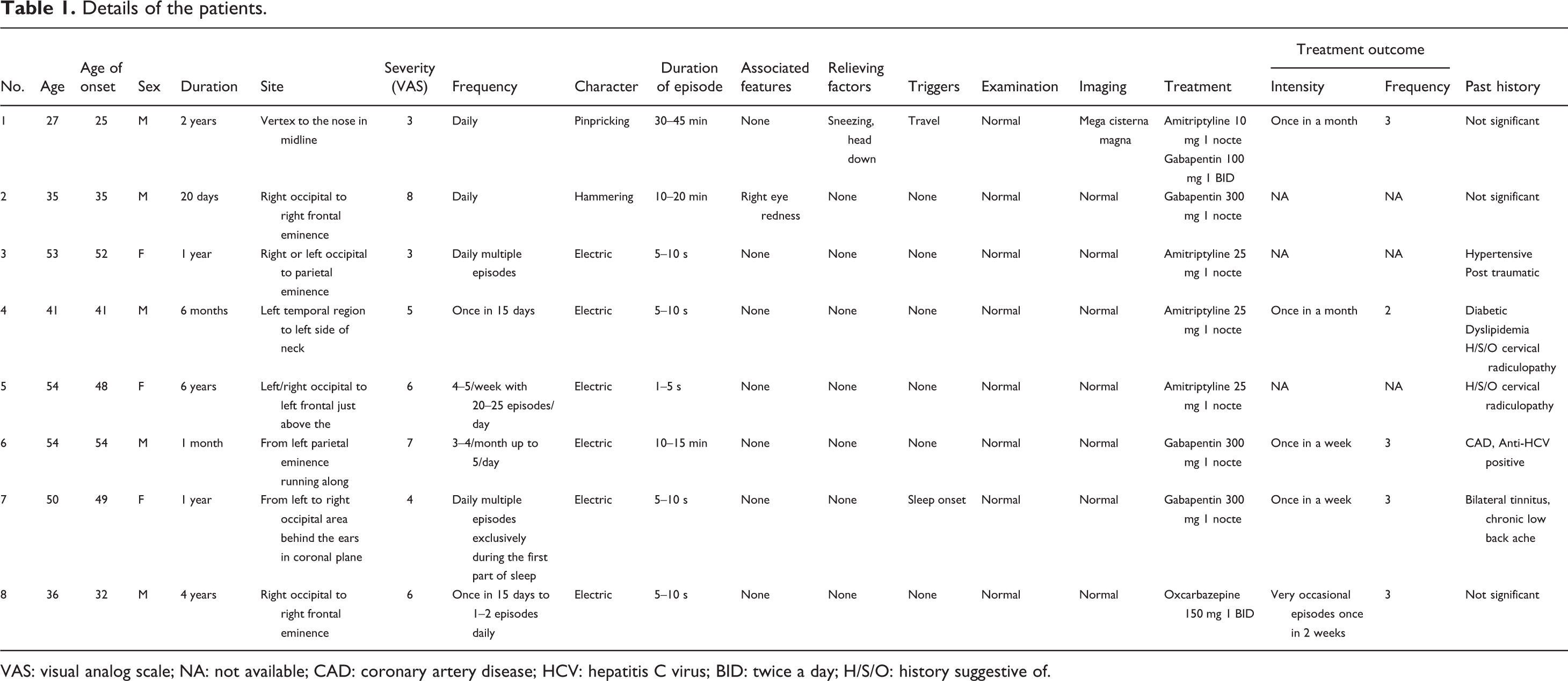
VAS: visual analog scale; NA: not available; CAD: coronary artery disease; HCV: hepatitis C virus; BID: twice a day; H/S/O: history suggestive of.
Headache paroxysms and their localization
Two patients had unilateral hemicranial pain paroxysms localized to the right side. Three patients (patients 3, 5, and 7) had bilateral hemicranial paroxysms. Pain in patients 3 and 5 moved classically from posterior to anterior but involved both the hemicrania simultaneously. Patient 7 had horizontal (transverse) occipital paroxysm from the right and left side up to midline. Patient 1 had midline (sagittal) localization of the pain. Pain trajectory was confined to the head in six patients, whereas in two patients pain trajectories extended beyond the head region. In patient 4, pain radiated from the left temporal to the left side of the neck just down to the mid one-third of the sternocleidomastoid. In patient 6, pain originated from the left parietal, radiated along the inner canthus of the left eye up to the lateral surface of the left mandible. The pain paroxysms originated from an occipital region in five patients, parietal region in one patient, in one patient it stemmed from the temporal area, and in another patient it originated from the area just posterior to the vertex in the midline. Figure 1 demonstrates the pain trajectories of these eight patients.

Pain trajectories in eight patients of epicrania fugax. (a) Patient 1: vertex to the nose in midline. (b) Patient 2: right occipital to right frontal eminence. (c)Patient 3: right or left occipital to parietal eminence. (d) Patient 4: left temporal region to left side of the neck. (e) Patient 5: left/ right occipital to left frontal just above the eyebrow. (f) Patient 6: left parietal eminence running along inner canthus down to mandibular angle. (g) Patient 7: left to right occipital area behind the ears in the coronal plane. (h) Patient 8: right occipital to the right frontal.
Headache features
The mean VAS score of headache attacks was 5.25 ± 1.83 (range 3–8). Headache character was defined as electric current like in six patients, pinpricking and hammering type in one each. Five patients had headache duration of a few seconds up to a maximum of 10 s. In three patients (patients 1, 4, and 6), the pain would begin like a paroxysm that would last for 5–10 s but later would become fixed in the linear trajectory. In two patients (patients 4 and 6), headache attacks lasted in the linear distribution of 5–10 mm for up to 20 min and in one patient (patient 1) for 30–45 min. The character of pain during the fixed period was described as “sore” by one patient, throbbing by one patient, and stretching by one patient.
Associated feature of redness of ipsilateral eye was noted by one patient after the subsidence of the attack. None of the other patients reported any associated features. There was no sensory loss. Two patients had mild tenderness over the greater occipital nerve.
High intraindividual and interindividual variability was noticed in the frequency of the headaches. Patients 1 and 2 had a frequency of one episode daily, whereas patient 3 suffered multiple episodes per day. Patient 7 has daily episodes linked to sleep onset. On the contrary, patients 5 and 8 had very infrequent episodes of once in 15 days. Patient 5 had 4–5 episodes/week to a maximum of 20–25 episodes/day. Patient 6 reported an attack frequency of four per month to five attacks per day. Systemic and neurological examination in all patients was unremarkable. Local tenderness at the site of origin of pain was not found in any of the patients.
All except one patient reported spontaneous pain. Traveling was identified as a trigger by one patient. One patient had headaches (patient 7) triggered by the onset of sleep who also had bilateral tinnitus. In none of the patients could the pain be triggered by applying pressure over the emerging point of the nerves. Patient 1 also gave a history of chronic tension-type headache. All patients denied any significant anxiety or depression. None of them had any psychiatric disorders in the past.
Investigations
Magnetic resonance imaging (MRI) of the brain and cervical spine (3 tesla) was normal in all patients. Since patient 7 had headaches often triggered by sleep, polysomnography (PSG) was done rule out obstructive sleep apnea. PSG was normal.
All but one patient were referred to us from other secondary/tertiary care hospitals in the city. One patient with symptoms of 20 days had come directly to our center. Four patients were just given a diagnosis of “headache” without any subclassification. One patient was labeled as tension-type headache and two patients as psychogenic headache. The patients were worried about their headache as their physicians could not explain the cause of their headaches and could not allay their fears about an underlying sinister cause.
We started treatment in all patients with gabapentin, amitriptyline, or oxcarbazepine. Patients 3, 4, and 5 were treated with amitriptyline 25 mg once at night and patients 2, 6, and 7 had significant improvement with gabapentin 300 mg once at night. Only one patient (patient 1) required combination therapy including amitriptyline 10 mg once and gabapentin 300 mg twice daily. Patient 8 was treated with 150 mg of oxcarbazepine twice a day and responded well. In relation to treatment outcome, three patients (patients 2, 3, and 5) did not follow-up, but the remaining five patients reported significant benefit in terms of a reduction in frequency and intensity of the headache episodes.
Discussion
EF was described as a distinct headache syndrome by Pareja et al. in 2008. 1 Epicrania is an umbrella term also introduced by Pareja et al. in 2003 to describe the headache disorders which probably originated from the superficial or extracranial structures. 4 It included disorders such as nummular headache, occipital, and supraorbital neuralgias. The feature of EF which sets it apart from other disorders is the fixed area of origin of pain and its stereotypical spread along a linear or zigzag trajectory and termination in a different nerve territory. Table 2 describes the salient features of the various mimics of EF which might help in distinguishing them from the EF. Other features that might be seen with EF are tenderness over the peripheral nerves or the peripheral tissue where the pain apparently stems from and sensory dysfunction over the symptomatic area. 3 In our series, we found mild tenderness in the greater occipital nerve in two patients on palpation. No patient had any sensory deficits.
Differentiating features of epicrania fugax and its mimics.

SUNCT: short-lasting unilateral neuralgiform headache with conjunctival injection and tearing; SUNA: short-lasting unilateral neuralgiform headache attacks with cranial autonomic.
Female preponderance in EF has been reported in the limited case series. 5 –7 In our case series, we found a male predominance (M:F—5:3). The mean age of presentation was 43.75 ± 10.42, and the mean age of onset was 42 ± 10.47 which was similar to that found in other studies. 5 –7
All of our patients had forward radiation of the pain, but several variants have been reported including backward, multidirectional, coronal, and parallel radiation. 6 –9 The spread of pain from one fixed point to another was common to all these variants. Except for the topography all other characteristics of the headache were similar. Before labeling EF as a primary headache disorder, secondary causes need to be ruled out. It is imperative to do a detailed examination and an MRI of every patient suspected of EF to rule out a secondary cause. 10 –12 All our patients had normal MRI brain and cervical spine.
In 62.5% of our patients, the pain began from the occipital region, similar to the earliest described patients of EF along the peripheral nerve distribution of the greater occipital nerve. One patient (patient 1) had a midline headache originating from the vertex radiating to just above the nasion. Only one similar case 13 had been reported so far to the best of our knowledge. The origin of pain, in this case, can be postulated to be due to terminal twigs of greater occipital nerve involvement (C2), which innervates the skin of medial occipital bone till the vertex. Pain beyond the vertex however cannot be explained by occipital nerves. Pareja et al. have postulated that the nerve impulses from the terminal branches of greater occipital nerve can spread to the supraorbital nerve by cross talk between the fibers of both the nerves. 1 They also argued that the pain fibers that cross the bones of the skull and the pain fibers that run longitudinally within the endosteum as seen in mice can also result in transdiploic transmission. 1
Patient 7 had transverse occipital involvement up to midline involving both the hemicrania. The pain would start behind each ear (lesser occipital nerves) simultaneously and travel down to midline near the occipital protuberance (greater occipital nerve). Such a pattern has not been described previously to the best of our knowledge. A case of multidirectional EF has also been reported in whom the transverse pain trajectory would cross midline bidirectionally along with other directional trajectories. 8
Patient 6 had extracephalic irradiation of pain from left temporal region to the neck. Similar extracephalic irradiation of pain has been described by Gutiérrez-Sánchez et al. 14 in a series of five patients where the pain radiated from cranium to neck, shoulder, and limbs. The authors argued that the long trajectory of pain radiation cannot be explained solely on peripheral mechanisms and suggested the possible role central pain generators resulting from hyperexcitability and/or ephaptic transmission involving the pain pathways within the thalamus, brainstem, and/or the spinal cord. Further the report of secondary EF involving brain structures also suggest a role of central pain mechanisms for the causation of pain in EF. 10 –12 It has been shown by anatomical studies the convergence of sensory input from upper cervical spinal cord neurons and trigeminal fibers occurs at trigeminal nucleus caudalis. 15 Experimentally, it has also been shown that the stimulation of the greater occipital nerve induces increased central excitability of dural afferent input. 16 Therefore, it is possible that central mechanisms might also play a role in the genesis of pain in EF.
In our case series, we had three patients who had an atypical EF phenotype in which the paroxysms would be followed by fixed pain in the same linear distribution lasting for a much longer duration. Wang et al. in 2014 described a new headache syndrome known as “linear headache” which was postulated to be a variant of migraine headache and the authors postulated a central mechanism for their causation. 17 These are characterized by paroxysmal or chronic fixed head pain restricted in a linear trajectory linking the occipital or occipitocervical region with the ipsilateral nasion or forehead region without any evidence of short duration marching pain of EF. These headaches typically were of long duration lasting from hours to days although 3 of 12 patients had duration of headaches lasting minutes as in our group of 3 patients. Contrarily, none of our patients had any migrainous features. It is of interest to note that 3 of 12 patients of Wang et al. also did not have any migrainous features. However, the duration of headaches in our case was long but never lasted more than 60 min and also had the characteristic march/paroxysm seen in EF at the beginning of the attack before they became fixed. This raised the possibility of an interictal persistent line-shaped head pain in EF as reported by Pareja and Bandrés in 2015. 18 Similar to our patient number 6, they reported a case of a 39-year-old female who had EF, followed by liner pain in the same trajectory involving right hemicranium up to inner canthus of the ipsilateral eye. However, this interictal linear pain was persistent contrary to our patients, where the linear pain was recurrent following recurrent attacks of EF. This linear pain should also not be confused with intermittent or continuous tenderness and/or pain at the stemming area that has been described between the EF paroxysms in more than half of the patients. While the majority of such pains may have ill-defined borders, in one-fourth, the pain may be well-circumscribed and fulfill the diagnostic criteria of nummular headache. 5,6,19
Hence, by excluding the above possibilities we propose that EF headache in three of our patients might have triggered a linear headache. EF can trigger attacks of headaches with clear central mechanisms such as cluster headache or migraine as well. 20 Linear headaches have also been postulated to have central mechanisms of causation. 21 Table 3 represented the comparison of the clinical picture of our patients with other case series of EF. 1,5,6,21
Comparison of clinical features of patients from various studies.
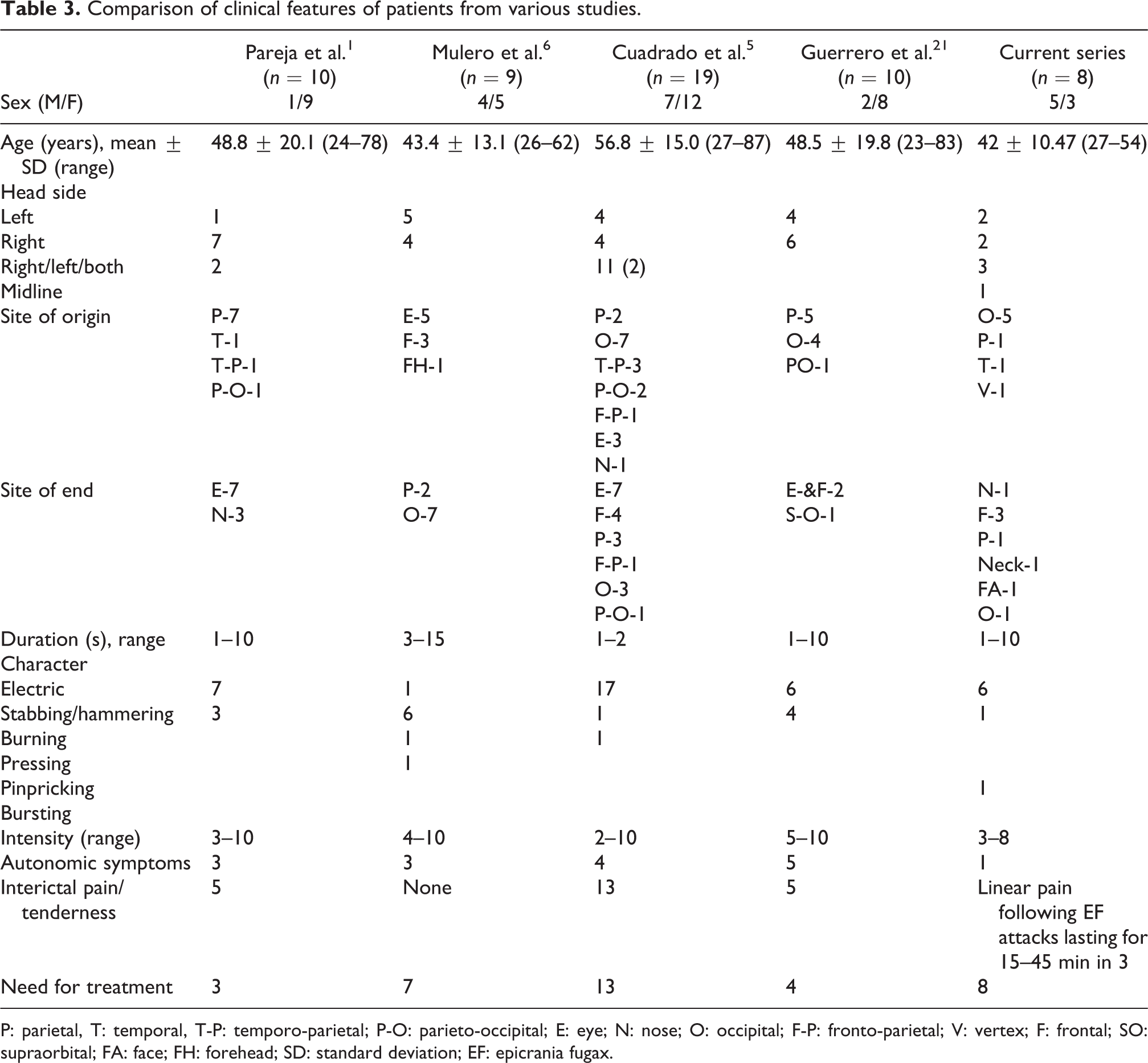
P: parietal, T: temporal, T-P: temporo-parietal; P-O: parieto-occipital; E: eye; N: nose; O: occipital; F-P: fronto-parietal; V: vertex; F: frontal; SO: supraorbital; FA: face; FH: forehead; SD: standard deviation; EF: epicrania fugax.
In conclusion, in our case series besides the classical features of EF, we found two patients with novel features namely mid-sagittal and bilateral occipital transverse trajectory and three patients with the long duration linear headaches with EF phenotype at the onset. The latter group of patients raises the possibility of EF triggered “linear headaches.” This needs to be further explored with larger studies to help expand the phenotypic variations in EF. Our report also shows that there is a general lack of awareness about these uncommon headache disorders leading to not only delayed diagnosis but also significant anxiety and apprehension in the minds of the patients.
Clinical implications
Eight new cases of EF are presented. Midline sagittal trajectory and the bilateral occipital transverse trajectory of pain were the novel variants noted among the patients.
Three patients had unique linear headaches involving the same trajectory just following the EF lasting for 15–45 min. We postulate that EF might have triggered liner headaches in these patients.
It is possible that central mechanisms might also play a role in the genesis of pain in EF as shown by extracephalic radiation of pain in one of our patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
