Abstract
Cluster headache (CH) is one of the most severe forms of headache, but the number of effective treatments is still limited. Recently, we reported the case of a drug-resistant CH patient responsive to the rotigotine transdermal patch, which is used in the treatment of Parkinson’s disease. This report formed the basis for a case series where other drug-resistant CH patients were treated with rotigotine. Here are the results of this study. Twenty-two CH patients underwent the treatment. Eight were episodic cluster headache (ECH) patients and 14 were chronic cluster headache (CCH) patients. Of the eight ECH patients, four reported that their CH had been stopped by the treatment. Of the 14 CCH patients, 11 were considered responders to the treatment (5 experienced a full resolution of headache, and 6 had a headache reduction of at least 50% in terms of mean monthly number of attacks). Our case series confirms the previous observation that rotigotine could be helpful in the treatment of CH. It may even influence the monoaminergic system that has a key role in the pathogenesis of CH.
Introduction
Cluster headache (CH) is characterized by repeated attacks (0.5–8 per day, lasting 15–180 min) of periorbital unilateral pain, related to a co-activation of trigeminovascular and autonomic system. If attacks are gathered in limited periods (lasting for weeks or months), separated by full remission phases (≥3 month), the CH is classified as episodic (ECH). In absence of remission periods (10–15% of cases), the CH is classified as chronic (CCH). 1
The number of effective treatments in CH is very limited and, 2 while attacks medications (subcutaneous and intranasal sumatriptan, intranasal zolmitriptan, oxygen) are effective in most cases, often the preventive strategies offered by physicians do not fulfil patients’ expectations, thus several patients, looking for relief, often resort to alternative medicines or illicit substances. 3
Our group has previously described the case of a refractory CCH patient who experienced a complete and sustained remission when treated with the rotigotine patch – a non-ergoline D3-like receptor agonist, also with a 5-hydroxytryptamine type 1A (5HT1A) effect, with a transdermal route of administration. 4 In particular, rotigotine is a non-selective agonist of the dopamine D1, D2, D3, and, to a lesser extent, D4 and D5 receptors, with highest affinity for the D3 receptor. 5 Compared to other dopamine agonists, rotigotine is one of the most tolerated treatments used for Parkinson’s disease (PD), both in PD patients 6 and in healthy volunteers. 7
The positive outcome of the said patient and the well-known tolerability of rotigotine prompted us to try this treatment for other patients with intractable CH. We did this in conjunction with other headache clinics, to collect a broader multicentric retrospective case series report about the use of rotigotine in CH.
Case series
We retrospectively collected clinical records of 22 patients referred to our Headache-Clinics between 2014 and 2016 with CH (8 difficult to treat ECH and 14 medically refractory CCH 8 ), all of whom received a prescription of transdermal rotigotine to treat their headache. All were formally diagnosed with CH using the International Classification of Headache Disorders, 3rd edition, beta criteria. 1 All patients’ clinical examination and brain MRI was normal.
Eight of these patients were diagnosed with ECH and treated with the rotigotine patch at the start of their active phase based on the length of previous bouts, the expected duration of the active phase is at least 1 month. Each patient had previously tried at least three different prophylactic drugs and reported them as ineffective or not tolerated. Treatments included verapamil (240–960 mg/day), lithium (up to 900 mg/day) and topiramate (up to 300 mg/day). In addition, various corticosteroids were tried by patients. Further drugs, such as topiramate (up to 300 mg/day), gabapentin (up to 1600 mg/day), amitriptyline (up to 75 mg/day), sodium valproate (up to 1000 mg/day), lamotrigine (up to 400 mg/day), cinnarizine (up to 150 mg/day) and methysergide (up to 16 mg/day), were combined with the above-mentioned treatments. Five patients also underwent the greater occipital nerve (GON) block (steroids and lidocaine). For other clinical characteristics, see Table 1.
Sociodemographic and clinical characteristics of eight ECH patients.a
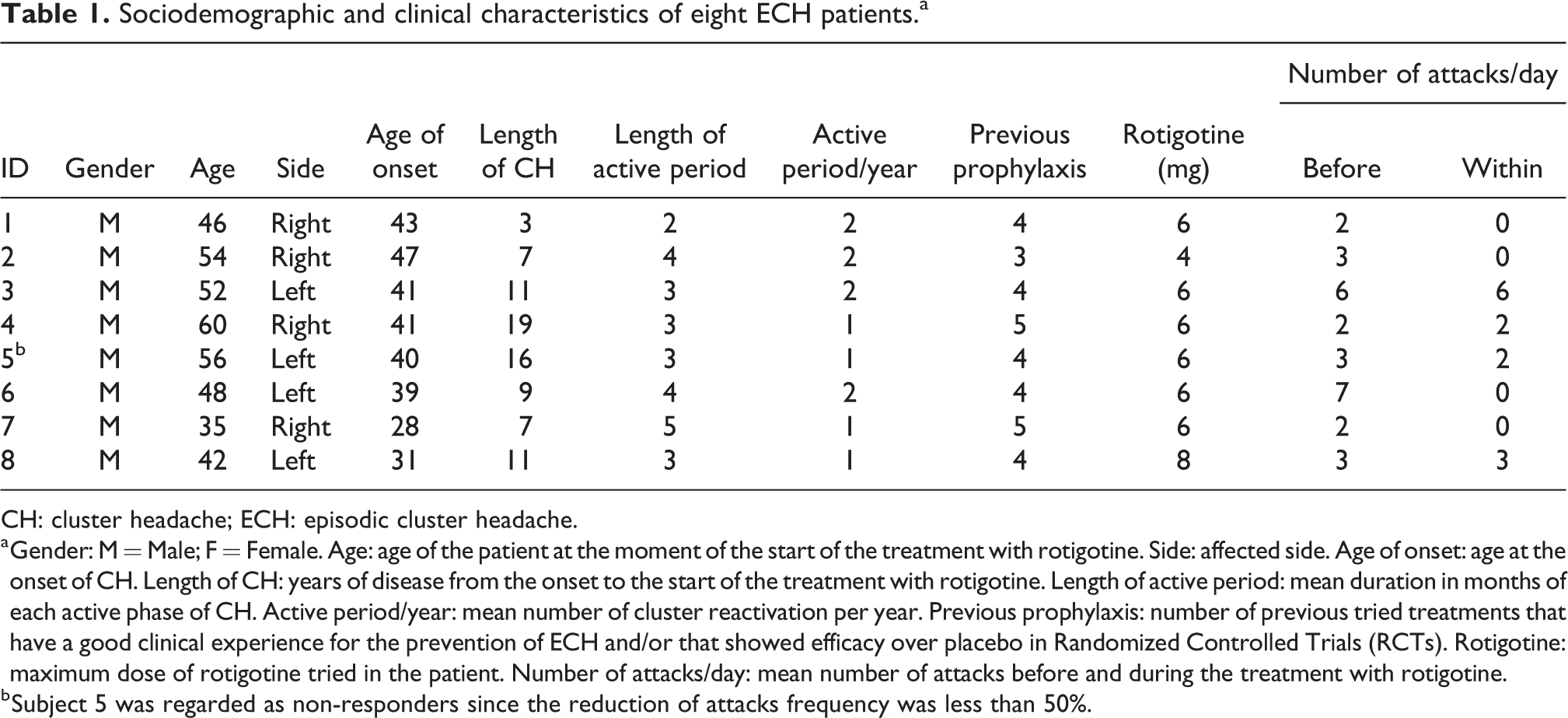
CH: cluster headache; ECH: episodic cluster headache.
a Gender: M = Male; F = Female. Age: age of the patient at the moment of the start of the treatment with rotigotine. Side: affected side. Age of onset: age at the onset of CH. Length of CH: years of disease from the onset to the start of the treatment with rotigotine. Length of active period: mean duration in months of each active phase of CH. Active period/year: mean number of cluster reactivation per year. Previous prophylaxis: number of previous tried treatments that have a good clinical experience for the prevention of ECH and/or that showed efficacy over placebo in Randomized Controlled Trials (RCTs). Rotigotine: maximum dose of rotigotine tried in the patient. Number of attacks/day: mean number of attacks before and during the treatment with rotigotine.
b Subject 5 was regarded as non-responders since the reduction of attacks frequency was less than 50%.
Fourteen were diagnosed as CCH sufferers. Each patient had previously tried at least four different prophylactic drugs and reported them as ineffective or not tolerated. Treatments included verapamil (up to 1080 mg/day), lithium (up to 900 mg/day) and topiramate (up to 500 mg/day). Also oral, i.m and i.v. corticosteroids were tried by some patients. Further drugs, such as gabapentin (up to 2400 mg/day), amitriptyline (up to 150 mg/day), sodium valproate (up to 1200 mg/day), lamotrigine (up to 400 mg/day), levetiracetam (up to 2000 mg/day), carbamazepine (up to 1200 mg/day), flunarizine (up to 10 mg/day), were combined with the above-mentioned treatments. Eleven patients also underwent the GON block (steroids and lidocaine), four had the sphenopalatine ganglion block, two had occipital nerve stimulation and one had hypothalamic deep brain stimulation. For other clinical characteristics, see Table 2.
Sociodemographic and clinical characteristics of 14 CCH patients.a

CH: cluster headache; CCH: chronic cluster headache.
a Gender: M = Male; F = Female. Age: age of the patient at the moment of the start of the treatment with rotigotine. Side: affected side. Age of onset: age at the onset of CH. Length of CH: years of disease from the onset to the start of the treatment with rotigotine. Chronic from: number of years from the chronification of CH. Previous prophylaxis: number of previous tried treatments that have a good clinical experience for the prevention of CCH and/or that showed efficacy over placebo in RCTs. Rotigotine: maximum dose of rotigotine tried in the patient. Number of attacks/day: Mean number of attacks before and during the treatment with rotigotine.
According to our previous report, all patients started rotigotine at the dose of 2 mg/24 h, which was increased weekly by 2 mg up to a maximum dose of 8 mg/24 h.
Results are reported in Tables 1 and 2 and in Figure 1. In brief, of the eight ECH patients, four found that the treatment stopped their cluster. All the responders experienced improvement early on at 4 mg (in three cases an abrupt stop of cluster). It was decided that after 1 week at 4 mg, the dose should not be increased in the three subjects who responded fully. Two patients had cluster reactivation, and their dosage was increased to 6 mg with a definitive response. In all the responders, the treatment continued for a period lasting 1 month longer than the expected duration of the active phase, then they slowly tapered the dose by 2 mg/week until the suspension of the rotigotine. Among the non-responders, two patients were also being treated with verapamil (360 and 480 mg); in another case, rotigotine was increased up to 8 mg/24 h, without benefits, and slowly the treatment was suspended.

Mean number of attacks per day before and within rotigotine treatment in episodic (a) and chronic (b) cluster headache patients.
Of the 14 CCH patients, 11 were considered responders to the treatment (5 experienced a full resolution of headache, and 6 had a headache reduction of at least 50% in terms of number of attacks). Six of the responders experienced an early improvement at 4 mg, but later worsened. The dosage was increased, with a stable response at 6 mg. Among the six patients with a partial response to rotigotine (attack frequency reduction >50%, but did not disappear), four increased the dose up to 8 mg/24 h. In addition, one patient of the group that experienced a full resolution of CH attacks did so only at the dose of 8 mg/24 h. Two patients suddenly discontinued treatment and switched to pramipexole slow-release 2.01 mg, due to the late dermatological reactions; one of them remained CH free after the switch. All the non-responders reached the dose of 8 mg/24 h before the tapering off and interruption of the treatment.
All responders CCH patients do not yet interrupt the treatment. At the moment of manuscript writing, we have had a follow-up duration ranging from 6 months to 3 years.
Other than the above-mentioned late dermatological reactions, patients reported no other side effects or complications, when interrogated about them.
Discussion
The result of the case series is consistent with our early observations that the transdermal patch of rotigotine could be effective as a preventive treatment in refractory CH. 4 Furthermore, we observed that rotigotine could be increased up to 8 mg if necessary. Finally, the treatment seems to be well tolerated by CH patients, since only dermatological complications occurred later in the treatment.
Although CH is a relatively rare condition, its refractory form is still an open challenge in the clinical management of headaches that could induce patients to require invasive procedures 2 or to abandon the medical advice and self-refer to illicit substances looking for relief, in particular lysergic acid diethylamide and psilocybin. 3 The latter was the case of the patient we described in our first report. 4 In answer to his request to try an ergolinic drug, we decided to try an off-label treatment with rotigotine, a non-ergoline agonist of dopamine receptors also characterized by a 5HT1A agonism, 9 this characteristic is shared with hallucinogens and could contribute to achieve an analgesic effect. 10 The choice of this class of drug was due to a previous report about pramipexole (another non-ergolinic dopamine agonist). 11 That paper inspired us to try dopaminergic treatment in CH patients and prompted us to use pramipexole in the two patients who developed late dermatological reactions by the prolonged use of the patches. In one case, the ‘overnight-switch’ to 2.01 mg of pramipexole slow-release gave the persistence of CH clinical response.
Rotigotine is one of the most tolerated dopamine agonists, 6 possibly because of the transdermal route of administration, accounting for a more physiological, continuous dopaminergic stimulation. Moreover, rotigotine is generally well tolerated even if the duration of treatment lasts up to 6 years, without serious adverse side-effects (the most common of which are somnolence, falls, peripheral oedema, nausea and reactions at the application site). 12,13
So far, a possible relationship between CH and the dopaminergic system is under scrutiny, although CH is associated with impaired dopaminergic stimulation, 14 whereas dopamine inhibits the nociception in the trigeminovascular transmission in animals. 15 Moreover, it is known that the dopaminergic system is strictly involved with pain modulation 16 and its chronification. 17 On the other hand, rotigotine has proven to improve PD-associated pain. 18 Finally, in our previous report, we observed that treatment with rotigotine reverted the deficit of habituation in the nociceptive blink reflex of the affected side, 4 a typical neurophysiological marker of CH patients, 19,20 suggesting a brainstem modulation of rotigotine on pathophysiological mechanism of disease.
The main shortcoming of this report is the retrospective observational nature of our survey, nonetheless, the case series and open label trials are in line with other recent papers in the field of refractory CH. 21 –23 Another important limit of our report is the little sample of observed subjects that makes impossible to generalize our results. In particular, ECH patients were only eight and it could induce to regard them as less responsive than CCH; it is possible that a more ample cohort of subjects would not confirm this impression. On the other hand, we cannot exclude that the bout of some ECH patients has spontaneously stopped. Although non-conclusive, our preliminary results about dopaminergic stimulation in CH seem to be promising and we predict further studies.
Footnotes
Author note
Gianluca Coppola is also affiliated to Department of Medico-Surgical Sciences and Biotechnologies, ‘Sapienza’ University of Rome Polo Pontino, Latina, Italy.
Acknowledgements
The authors would like to thank Deirdre Ní Mhórdha for editorial assistance. The contribution of the G.B. Bietti Foundation in this article was supported by the Italian Ministry of Health and Fondazione Roma.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Fondazione Roma and the Italian Ministry of Health.
