Abstract
Objective
We tested the hypothesis of a defective functional connectivity between the posterior hypothalamus and diencephalic-mesencephalic regions in chronic cluster headache based on: a) clinical and neuro-endocrinological findings in cluster headache patients; b) neuroimaging findings during cluster headache attacks; c) neuroimaging findings in drug-refractory chronic cluster headache patients improved after successful deep brain stimulation.
Methods
Resting state functional magnetic resonance imaging, associated with a seed-based approach, was employed to investigate the functional connectivity of the posterior hypothalamus in chronic cluster headache patients (n = 17) compared to age and sex-matched healthy subjects (n = 16). Random-effect analyses were performed to study differences between patients and controls in ipsilateral and contralateral-to-the-pain posterior hypothalamus functional connectivity.
Results
Cluster headache patients showed an increased functional connectivity between the ipsilateral posterior hypothalamus and a number of diencephalic-mesencephalic structures
Conclusions
The observed deranged functional connectivity between the posterior ipsilateral hypothalamus and diencephalic-mesencephalic regions in chronic cluster headache patients mainly involves structures that are part of (i.e. ventral tegmental area, substantia nigra) or modulate (dorsal nuclei of raphe, sub-thalamic nucleus) the midbrain dopaminergic systems. The midbrain dopaminergic systems could play a role in cluster headache pathophysiology and in particular in the chronicization process. Future studies are needed to better clarify if this finding is specific to cluster headache or if it represents an unspecific response to chronic pain.
Introduction
Cluster headache (CH) is characterized by extremely severe unilateral head pain and ipsilateral cranial-facial autonomic symptoms (1). Clinical, neuro-endocrinological, and animal findings provided converging evidence of hypothalamic involvement in CH pathophysiology (2,3).
In this framework, the seminal neuroimaging study of May et al. (4), acquired during CH attacks, showed ipsilateral (to the cranial pain) activation in the posterior hypothalamus, then suggested as a cluster generator. The neuroimaging findings (4,5) led to the pioneering successful treatment of refractory chronic CH (CCH) with hypothalamic deep brain stimulation (DBS) (6).
It has been observed that the activated area in the May et al. study (4) lies immediately posteriorly to the posterior boundaries of the hypothalamus, i.e. in the midbrain tegmentum (7,8). A number of sub-cortical areas, that is, diencephalic-mesencephalic structures such as the red nucleus, dorsal nuclei raphe, periaqueductal grey matter (9–11) are activated together with the hypothalamus, suggesting that both hypothalamus and diencephalic-mesencephalic structures are involved in CH attack pathophysiology (8). In this respect, it is noteworthy that the electrode tip in hypothalamic stimulated CH patients (12) is equally located in responders and non-responders, both posterior to the posterior hypothalamus and regions of the diencephalic-mesencephalic junction (13). In agreement with this observation, robust anatomical connections were shown between DBS targeted areas and both the hypothalamus and the reticular nucleus (14).
To test the hypothesis that a dysfunctional connectivity between the posterior hypothalamus and regions of the diencephalic-mesencephalic junction occurs in CH, intrinsic functional connectivity (FC) of the posterior hypothalamus was investigated in CCH patients and compared to a sex and age-matched healthy control group.
Materials and methods
Participants
Demographical and clinical details of chronic cluster headache patients.

VAS: visual analogue scale (from 0–10) to evaluate the headache pain experienced before entering the scanning session.
MRI data acquisitions
All participants were scanned with a 3 T scanner equipped with a 32-channel coil (Achieva TX, Philips Healthcare BV, Best, NL). Volumetric T1-weighted as well as volumetric T2-weighted images and RS-fMRI images were acquired (GE-EPI volumes, TR = 2800 ms, TE = 30 ms, α = 70°, 2.5 mm isotropic voxel size, matrix size 90 × 95, 50 slices with 10% gap acquired in ascending order, volume number = 200). The total RS-fMRI acquisition time was 9.33 min. T2-weighted images were acquired to better explore the anatomy of the diencephalic-mesencephalic junction areas (16) and to identify the anatomical seeds of the hypothalamus, which is better visualized on such images than on T1-weighted images.
RS-fMRI data processing
All the analyses were conducted using Brain Voyager QX (http://www.brainvoyager.com). For each participant, the RS-fMRI data pre-processing included spatial realignment, slice-timing correction, spatial smoothing (full width at half maximum – FWHM = 5 mm) and normalization to the Talairach space. To perform this last step, we co-registered the RS-fMRI images to the T2-weighted images: After the automatic step, co-registration accuracy was visually inspected and manually corrected. The T2-weighted images were then normalized to the Talairach space and the obtained normalization parameters were applied to normalize the co-registered RS-fMRI images. In agreement with other authors’ works (17–19), we flipped along the midsagittal plane the RS-fMRI images of CCH patients with left-sided attacks, so that all patients presented the ipsilateral-to-the-pain hypothalamus on the same side (right). Coherently, in the CCH group we will refer to ipsilateral brain structures to the cranial pain during the attack as ‘ipsilateral’ (e.g. ipsilateral hypothalamus), and to contralateral brain structures to the cranial pain as ‘contralateral’ (e.g. contralateral hypothalamus). Similarly, in the CTRL group, we will refer to the right brain structures as ipsilateral and to the left brain structures as contralateral. Further supporting the RS-fMRI images’ flipping, we tested whether the CTRL group presented differences between ipsilateral and contralateral hypothalamus FC.
To test resting-state FC abnormalities of the ipsilateral and contralateral posterior hypothalamus, we selected these regions as seeds and, by means of voxel-wise multiple–regression, we produced correlation t-maps between such areas and the rest of the brain.
To this purpose, for each single participant, an expert neuro-radiologist (L.C.) manually drew the seeds of the posterior ipsilateral and contralateral hypothalami (Figure 1) on the anatomical MRI images in the normalized Talairach space, blinded to the condition of the individual (CCH or CTRL). Hypothalamic seeds were defined based on the anatomical boundaries of the posterior hypothalamus as described in Baroncini et al. (20): The inferior extent was limited by the mammillary bodies, the medial extent by the III ventricle, the lateral extent by the mammillo-thalamic tract. Since caudally the posterior hypothalamic nuclei are in continuity with the periaqueductal grey, the VOIs have been drawn on coronal slices encompassing 3 mm in the y direction, approximately 7 to 10 mm posterior to the anterior commissure. This allowed an unbiased approach, only based on the anatomy of each single individual, to the investigation of the posterior hypothalamus FC. To verify the goodness of the segmentation, another researcher (S.F.) was trained to carry out the segmentation, performed, in this case, on all the CCH individuals for both the left and right hypothalamus. Then, inter-rater agreement was assessed using the intra-class correlation coefficient (ICC) of the coordinates of the center of gravity of each identified seed (total number of seeds = 34). The ICC (0.83) showed a good inter-rater agreement.
Individual ipsilateral and contralateral posterior hypothalamic regions of interest (ROIs) superimposed on T2-weighted image of a single participant. I: ipsilateral; C: contralateral; A: anterior; P: posterior; SAG: sagittal; COR: coronal; AX: axial.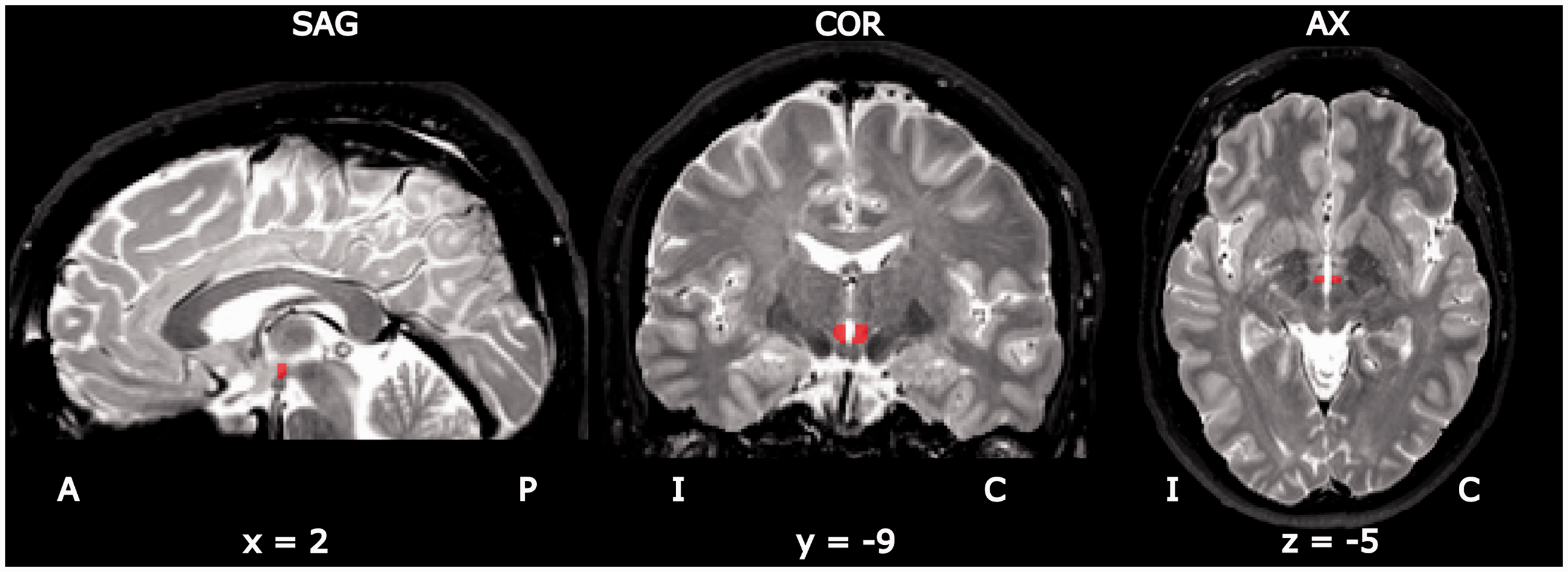
Moreover, in each single patient, we drew masks for the third ventricle and of a selected four spherical regions in the supratentorial white matter (5 mm spherical masks in genu and splenium of corpus callosum and 3 mm spherical masks in the bilateral cortico-spinal tract). For each of the seed and mask regions, we extracted and computed the average signal time-courses. As a first step, we produced, in the framework of the general linear model (GLM), residuals t-maps. To reduce the impact of non-neural noise sources on FC, nuisance signals, the average signal time-courses of the third ventricle and of the supratentorial white matter regions (20) were regressed out from the single voxel time series. To control for possible artifacts in FC estimation due to micro-motion during RS-fMRI acquisition, we computed, for each participant, the mean framewise displacement (FD). Mean FD was obtained in agreement with the work of Power et al. (21). Each single subject presented a mean FD inferior to 0.5 mm (22) and no significant differences were found between the CCH patients and healthy participants’ groups (two-tailed t-test for independent sample: p = 0.36). We did not regress out the average global brain signal, which is known to bring into play spurious negative correlations between brain regions (23). On such residuals t-maps, we performed single-subject analyses, using as predictors the average signal time-courses of each hypothalamus (ipsilateral and contralateral). Predictors were z-transformed and connectivity t-maps were produced. These single-subject connectivity t-maps express the correlation between the seed average signal time-series and the signal time-series of each voxel of the brain.
To further support, from a neurophysiological point of view, the flipping of RS-fMRI images, we produced flipped residual t-maps for every CTRL subject and we proceeded, as described above, to perform single subject analyses. Next, we computed the group analyses using a GLM random-effect model to assess whether significant differences in FC existed between the ipsilateral and contralateral hypothalamus in the CTRL group. Both non-flipped and flipped single-subject connectivity t-maps of the CTRL group were entered in a GLM random-effect analysis with one within-subjects factor (two levels: Ipsilateral and contralateral hypothalamus) and one between-subjects factor (two levels: CTRL non-flipped and CTRL flipped). The relevant t-map was produced for the comparison between the ipsilateral and contralateral hypothalamus. Because we were interested in finding evidence of differences between the hypothalami, we used a permissive threshold (p < 0.05 FDR-corrected; cluster threshold: 10 voxels). Importantly, this analysis showed that in the CTRL group, the ipsilateral and contralateral hypothalami did not differ in their FC with the diencephalic-mesencephalic regions, further justifying the RS-fMRI flipping procedure (17–19).
Finally, we compared the FC between the CCH and the CTRL groups. To this end, single-subject connectivity t-maps of CCH and CTRL participants were entered into a second GLM random-effect analysis: We performed a mixed-design ANOVA, with one within-subjects factor (two levels: Ipsilateral and contralateral hypothalamus) and one between-subjects factor (two levels: CCH and CTRL group). Relevant t-maps (p < 0.005 FDR-corrected; cluster threshold: 10 voxels) were produced for the comparisons of interest: We tested for the main effects of ipsilateral and contralateral hypothalami FC separately in the CCH and CTRL groups and for differences between groups in the FC of the ipsilateral hypothalamus (CCH ipsilateral hypothalamus > CTRL ipsilateral hypothalamus; CTRL ipsilateral hypothalamus > CCH ipsilateral hypothalamus), and of the contralateral hypothalamus (CCH contralateral hypothalamus vs. CTRL contralateral hypothalamus; CTRL contralateral hypothalamus vs. CCH contralateral hypothalamus).
Results
Between groups comparisons
Differences in functional connectivity between groups in ipsilateral and contralateral hypothalamus computed at p < 0.005 FDR-corrected.

CCH: chronic cluster headache; CTRL: control; FDR: false discovery rate; VTA: ventral tegmental area.

T-maps showing the main effects of intrinsic functional connectivity (FC) of ipsilateral hypothalamus (a) in chronic cluster headache patients and (b) in the control participants; (c) t-maps showing the differences in intrinsic functional connectivity of ipsilateral hypothalamus between (a) the chronic cluster headache patients and (b) in the control participants. CCH: chronic cluster headache; CTRL: control; I: ipsilateral; C: contralateral; A: anterior; P: posterior; SAG: sagittal; COR: coronal; AX: axial; FDR: false discovery rate.
Remarkably, the FC of the contralateral hypothalamus did not differ between the two groups (CCH contralateral hypothalamus > CTRL contralateral hypothalamus and CTRL contralateral hypothalamus > CCH contralateral hypothalamus).
CH patients
Regions showing increased functional connectivity with ipsilateral or contralateral hypothalamus in chronic cluster headache (CCH) and control (CTRL) participants computed at p < 0.005 FDR-corrected.

FDR: false discovery rate.
Controls
Significant FC of the ipsilateral hypothalamus with the ipsilateral substantia nigra, thalamus, and cerebellum (p < 0.005 FDR-corrected; cluster threshold: 10 voxels) was found in healthy controls. The contralateral hypothalamus showed significant FC with the contralateral substantia nigra, thalamus, nucleus caudatus, and subthalamic nucleus (Table 3, Figure 2).
Discussion
Our RS-fMRI study shows a deranged FC between the posterior ipsilateral-to-the-pain hypothalamus and regions of the diencephalic-mesencephalic junction in CCH patients. These abnormalities have been detected outside CH attacks (Table 2). Remarkably, no difference between patients and controls was observed in the contralateral hypothalamic FC (Table 3). This observation lends further support to the importance of the ipsilateral-to-the-pain hypothalamus in CH pathophysiology (4,18).
In particular, the present study revealed increased FC between the ipsilateral-to-the-pain hypothalamus and the VTA, bilateral substantia nigra, sub-thalamic nucleus, dorsal nuclei of raphe, and red nucleus in CCH patients.
VTA, bilateral substantia nigra, and sub-thalamic nucleus belong to the midbrain dopamine (DA) pathways (24,25), suggesting an involvement of the DA networks in CCH (2). An involvement of DA systems in CH is supported by the decreased growth hormone response to apomorphine (26) and increased plasma levels of DA and other related neurotransmitters in CH patients (27). Efficacy of VTA DBS in the treatment of CCH (28), as well as of other intractable trigeminal autonomic cephalalgias (29), further points to the involvement of the midbrain DA systems in CH pathophysiology.
DA VTA neurons are innervated by dense direct projections from the hypothalamic orexinergic cells (30,31), thought to play a major role in the pathophysiology of CH attacks (32). DA VTA neurons inhibit hypothalamic orexin cells (33,34), while the latter activate the former (35), possibly in a feedback system. A dysfunction of such a system could favor the chronicization of the disease. In this respect, it has been shown that long term DBS of the posterior hypothalamus-VTA can revert the chronic course of CH (2) to episodic.
Impairments of the DA system could favor both the chronicization of pain and the addictive behavior observed in CH patients (36). In this respect, it is noteworthy that in episodic CH patients there is no evidence of dysfunctional connectivity between the hypothalamus and diencephalic-mesencephalic regions (17,37–39).
A deranged FC between the posterior hypothalamus and other DA structures such as the subthalamic nuclei and substantia nigra seems to confirm a dysfunction of the midbrain DA system in our CCH patients.
Hypothalamic orexinergic neurons also project to the serotonergic dorsal nuclei of raphe (40–42), involved in analgesia. Deranged FC between the posterior hypothalamus and dorsal nuclei raphe in CCH patients could reflect a dysfunction of the pain control mechanism. Effective long-term DBS of the posterior hypothalamus increases the pain threshold in stimulated CCH patients (43), which could be mediated by a restored FC between the posterior hypothalamus and the dorsal nuclei of raphe.
FC between the red nucleus and the hypothalamus has been reported in humans (44). In animals, chemical or electrical stimulation of the red nucleus has an analgesic effect (45); and it was shown that red nucleus activation occurs during CH attacks (11). Taken together, these observations may suggest a role of the red nucleus in pain control in CH patients. The altered FC between the posterior hypothalamus and red nucleus in our patients might again reflect a derangement of the pain control system in CCH. It is worth noting that the observed deranged FC might be linked or not to possible structural alterations of the hypothalamus (46), which was not investigated in this study. Notably, we used the visual inspection and the manual segmentation to identify the posterior hypothalamus for two main reasons: a) the most part of the previous CH studies using RS-fMRI (37–39) to investigate the FC of the hypothalamus used spherical or cubical volumes located in the standard coordinates that emerged in the study of May et al. (9); however, reconsideration of these coordinates led to the hypothesis that they are relative to the midbrain areas (8,47); b) the MRI atlas of the human hypothalamus by Baroncini et al. (20) showed that there is a significant variability between individuals in the hypothalamus even in the normalized brain.
Despite all the above relevant findings, our results need to be taken with caution for two reasons: a) all the CCH patients investigated in this study were under prophylactic medications during the RS-fMRI acquisitions; therefore, we cannot exclude that the observed hypothalamic dysfunctional connectivity might be related to the taken medications; b) the inherent difficulties in investigating the brainstem: It does not reliably align to the standard space and it presents a reduced BOLD signal-to-noise ratio compared to cortical areas. However, it should be noted that we have investigated not the whole brainstem but only the midbrain, on which successful fMRI studies were conducted (48).
Conclusion
The present study provides evidence of a deranged FC between the posterior hypothalamus and diencephalic-mesencephalic regions in CCH. This defective FC could reflect an imbalance between the hypothalamus-hypocretin/orexin system and the midbrain DA pathways. Such an imbalance might be involved in both pain chronicization of CCH and in the addictive behavior observed in CH patients (36). Future studies should clarify whether this defective connectivity is a specific pattern of CCH or a non-specific response to chronic pain.
Key findings
We provided evidence of defective functional connectivity between the posterior hypothalamus and regions of the diencephalic-mesencephalic junction in chronic cluster headache. The observed deranged functional connectivity mainly involves structures that are part of (i.e. ventral tegmental area, substantia nigra) or modulate (dorsal nuclei of raphe, sub-thalamic nucleus) the midbrain dopaminergic systems.
Footnotes
Acknowledgement
We are particulary grateful to Prof. Gennaro Bussone for his inspiring and continuous support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been performed with the economical support of F.I.CEF. Onlus–Fondazione Italiana Cefalee Onlus.
