Abstract
The Qing-Re-Yi-Liu decoction (QRYLD) is a clinically effective prescription for treatment of breast cancer. Our preliminary studies found that QRYLD treatment interfered with the Warburg effect in breast cancer cells. However, its chemical components and molecular mechanisms have not been clarified. This study analyzed the bioactive components of QRYLD aqueous extracts by HPLC. The effects of QRYLD on the malignant behaviors of MCF-7 cells and potential mechanisms were analyzed by CCK-8, transwell invasion, wound-healing assays, flow cytometry, Western blot assays and transcriptomic analysis as well as in vivo mouse tumor model. QRYLD aqueous extracts contained several bioactive components. Transcriptomic analysis indicated that QRYLD treatment altered the expression of several genes involved in biological processes and signaling pathways, such as manganese superoxide dismutase (MnSOD). Functionally, QRYLD, similar to MnSOD silencing, inhibited the malignant behaviors of MCF-7 cells and enhanced their apoptosis, whereas MnSOD overexpression had the opposite effects. Moreover, QRYLD treatment inhibited the Warburg effect by limiting glucose uptake and lactic acid production, and decreasing the relative expression of glucose transporter-1, hypoxia-inducible factor 1α, c-Myc, hexokinase-2, phosphofructokinase 1, lactate dehydrogenase A, pyruvate kinase isozyme 2, MnSOD, calmodulin-dependent kinase II (CaMKII), and AMP-activated protein kinase (AMPK) in MCF-7 cells. Finally, treatment with QRYLD significantly inhibited the growth of xenografted MCF-7 tumors in mice and reduced the tumor expression of MnSOD, CaMKII, and AMPK. These data suggest that QRYLD may target MnSOD to attenuate the MnSOD/CaMKII/AMPK signaling, inhibiting the Warburg effect and malignant behaviors in breast cancer.
Introduction
Breast cancer is the most common malignant tumor in women globally. Currently, therapeutic strategies for breast cancer include surgical resection and neoadjuvant and postoperative therapies, such as chemotherapy, target therapy, radiotherapy, and immunotherapy. Traditional Chinese medicine (TCM) has been used to treat breast cancer.
In the theory of TCM, tumor development and progression are attributed to the accumulation of heat and toxin, and therapeutic strategies to clear heat and detoxify are important for the control of tumor growth. However, little is known on the therapeutic targets and pharmacological mechanisms underlying the anti-cancer activity of TCMs. Previous studies have shown that Scutellaria baicalensis and its bioactive ingredients have potent activities against the malignancy of breast cancer, 1 and treatment with the ethanolic extract of Andrographis paniculata inhibits the proliferation of human breast cancer MCF-7 cells in a dose-dependent manner. 2 Similarly, treatment with Coptidis Rhizoma limits the growth of 4T1 breast cancer in mice by inducing cell cycle arrest in G1 phase and suppressing DNA synthesis, 3 whereas treatment with Radix Isatidis attenuates the malignant behaviors of SK-BR-1 and MDA-MB-231 cells in a time-dependent manner by inducing cell cycle G2/M arrest and apoptosis. 4 Furthermore, treatment with Pulsatilla saponin inhibits the proliferation of MCF-7 cells in a dose-dependent manner. 5 Although many TCMs have been used in clinical practice to treat breast cancer, their therapeutic efficacy remains limited. Hence, the identification of new and safe TCMs, particularly for those with potent heat-clearing and detoxifying activities, is urgently needed.
The Qing-Re-Yi-Liu decoction (QRYLD) is a clinically effective prescription for the treatment of breast cancer given its heat-clearing and detoxification activities. Our preliminary studies found that treatment with QRYLD inhibited the proliferation of MCF-7 cells, and this effect was associated with inhibition of the Warburg effect (aerobic glycolysis). However, its therapeutic target and the pharmacological mechanisms underlying the antitumor activity of QRYLD against the malignancy of MCF-7 breast tumors and the Warburg effect have not been explored.
Prvious studies have shown that the MnSOD/CaMKII/AMPK signaling is crucial for the Warburg effect, which is preferred by tumor cells. 6 The MnSOD is a manganese-containing homotetramer in the mitochondrial matrix. 7 MnSOD expression is up-regulated in breast cacner, and is associated with its aggrasiveness and worse prognosis. 8 MnSOD overexpression promotes metastasis and drug resistance in breast cancer, but it also inhibits the proliferation of breast cancer cells by reducing ROS production and attenuating the PI3K/AKT and MAPK/ERK signaling. 9 Furthermore, MnSOD can also activate CaMKII and AMPK to enhance glycolysis by enhancing its key enxyme expression in breast cancer cells.10,11 Thus, MnSOD may act as a tumor suppressor or oncogenic factor. However, it is unclear whether QRYLD treatment could modulate the MnSOD/CaMKII/AMPK signlaing and subsequent the Warburg effect in breast cancer cells.
Materials and Methods
QRYLD
QRYLD is composed of Taraxacum officinale L., Iphigenia indica L., Forsythia suspensa L., Cortex Moutan, Hedyotis diffusa L., Lonicera japonica L., Prunella vulgaris L., and Lycii Cortex L. Further details about these components are presented in Table 1 (the plant names were verified at www.worldfloraonline.org). These medicinal herbs were obtained from the TCM Pharmacy of the First Affiliated Hospital of Xi’an Jiaotong University (Xi’an, Shaanxi, China) and identified by experts in the Pharmacy Department. QRYLD was soaked in 10 volumes of water and heated at 100 °C for 1 h. The decoction was collected, and the herbs were heated at 100 °C with another 10 volumes of water for 40 min for further decoction collection. The collected decoctions were mixed, filtered through a filter, concentrated, and dried using a spray dryer (Lemal, Changzhou, China). The dried QRYLD powder was dissolved in water, and the chemical components in the QRYLD aqueous extracts were identified by HPLC using an Agilent 1260 Infinity II HPLC system (Agilent Technologies, Santa Clara, CA, USA).
The Composition of QRYLD.

The plant names have been checked at http://www.theplantlist.org and www.worldfloraonline.org.
Preparation of QRYLD-Supplemented Serum
BALB/c mice at 3–5 weeks of age were obtained from the Laboratory Animal Center, Xi’an Jiaotong University Health Science Center. The mice were randomized and treated with the control physiological saline or 0.3 ml of QRYLD in physiological saline at a low (LD, 5.6875 g·kg−1·day−1), medium (MD, 11.375 g·kg−1·day−1) or high dose (HD, 22.75 g·kg−1·day−1) by gavage daily for 7 consecutive days. The daily medium dose of QRYLD for mice was calculated according to that individual adults with an average of body surface area of 1.7 m2 take about 400 ml of QRYLD per day and then converted into mouse dose, based on the ratio of body surface area of humans to mice. Two hours after the last gavage, their blood samples were collected and prepared for serum samples. After being heated at 56 °C for 30 min, the serum samples were filtered through a 0.45-µm filter and used as the QRYLD-supplemented sera for cell culture. RPMI-1640 medium (Solarbio, Beijing, China) containing 10% QRYLD-supplemented serum was prepared.
Cell Culture
Human breast cancer MCF-7 cells were obtained from Shanghai Institute of Cell Biology (Shanghai, China) and cultured in RPMI-1640 medium containing 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin at 37°C in a 5% CO2 incubator.
Transfection and Transduction
293 T cells were transfected with plasmids for lentivirus packaging and MnSOD expression or control GFP expression using Lipofectamine 3000 to generate the lentivirus virions carrying cDNA for the expression of MnSOD (Lent-MnSOD) and control GFP (Lent-GFP), respectively. Similarly, HEK293 cells were transfected with adenovirus packaging plasmids and plasmids for the expression of MnSOD-specific shRNA or control shRNA to generate Ad-sh-MnSOD and control Ad-sh virions, respectively. These lentivirus and adenovirus virions were produced by Jinsirui Biotechnology (Nanjing, China). MCF-7 cells were transduced with lent-MnSOD virions at a multiplicity of infection (MOI) of 10. The transduced cells were treated with 4 µg/ml puromycin to induce stable MnSOD expressing Lent-MnSOD MCF-7 cells. Furthermore, MCF-7 cells were infected with Ad-sh-MnSOD or control Ad-sh at a MOI of 10 and treated with 2 µg/ml puromycin to generate stable MnSOD silencing Ad-sh-MnSOD MCF-7 cells. The efficiency of MnSOD overexpression or silencing was examined by Western blot.
CCK-8 Assay
Wild-type MCF-7 cells (1 × 104 cells/well) were cultured in 96-well plates and treated in triplicate with 10% control mouse serum or 10% LD, MD, or HD QRYLD-supplemented serum, together with Lent-MnSOD and Ad-sh-MnSOD MCF-7 cells in RPMI 1640 medium supplemented with 10% fetal bovine serum (FBS) for 24, 48 and 72 h. During the last 4 h culture, individual wells of cells were added with 10 μL of CCK-8 solution (Beyotime, Shanghai, China), and the viability of each group of cells was measured for the absorbance at 450 nm using a microplate reader.
Transcriptomic Analysis
MCF-7 cells were treated in triplicate with control mouse serum as the control or MD QRYLD-supplemented serum for 48 h. Their total RNA was extracted using TRIzol (Thermo Fisher Scientific, Waltham, MA, USA), and their mRNAs were sorted using an Oligotex mRNA sorting kit (Qiagen, Hilden, Germany) per the manufacturer's instructions. After qualification and quantification of mRNA using the Illumina HiSeq™ 2000 system, the mRNA samples from each group were reverse-transcribed into cDNA to generate cDNA libraries using a SMART-Seq V4 kit (Takara, Kyoto, Japan). The cDNA libraries were sequenced in the NovaSeq 6000 system (Illumina, San Diego, CA, USA). The differentially expressed genes (DEGs) were identified by a P-value of <.05 or a fold change of >|2.0|. The biological nature of DEGs was analyzed using DAVID Bioinformatics Resources 6.7, and their biological processes or signaling pathways were analyzed by Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) analyses. The transcriptomic analysis was conducted by MED-X institute, the First Affiliated Hospital of Medical School of Xi’an Jiaotong University.
Transwell Invasion Assay
MCF-7 cells (4 × 104 cells/well) were treated with control mouse serum or MD QRYLD-supplemented serum for 48 h. The cells, together with the same number of Ad-sh-MnSOD MCF-7 or Lent-MnSOD MCF-7 cells, were cultured into the upper chamber, which had been coated with Matrigel (Thermo Fisher Scientific), and the bottom chamber was filled with 10% FBS-containing medium. After 24-h culture, the cells on the upper chamber surface membrane were removed, and the invaded cells on the upper chamber bottom surface were fixed in 4.0% paraformaldehyde and stained with 0.1% crystal violet solution for 10 min, followed by photoimaging under a Leica DMil inverted microscope (Leica, Wetzlar, Germany). The number of invaded cells was counted in a blinded manner.
Wound-Healing Assay
MCF-7 cells were treated with control mouse serum or MD QRYLD-supplemented serum for 48 h. The cells, together with the same number of Ad-sh-MnSOD MCF-7 or Lent-MnSOD MCF-7 cells (1 × 106 cells/well), were cultured in six-well plates. When the cells reached 95% confluency, the monolayer cells were wounded using a 10-μL sterile pipette tip. The wounded areas were photographed immediately after scratch and after 48 h of culture. The wound-healing ability of each group of cells was analyzed using ImageJ software (US National Institutes of Health, Bethesda, MD, USA).
Apoptosis Detection by Flow Cytometry
Ad-sh-MnSOD MCF-7, Lent-MnSOD MCF-7, and wild-type MCF-7 cells were treated in triplicate with control mouse serum or MD QRYLD-supplemented serum for 48 h, harvested, and stained with Annexin V–fluorescein isothiocyanate (Solarbio) and propidium iodide (Solarbio) for 10 min in the dark. The percentage of apoptotic cells was analyzed by flow cytometry, and the data were analyzed using FlowJo software (Becton, Dickenson, Franklin Lakes, NJ, USA).
Western Blot
Ad-sh-MnSOD MCF-7, Lent-MnSOD MCF-7, and MCF-7 cells were treated with control serum or MD QRYLD-supplemented serum for 48 h, harvested, and lysed in lysis buffer, followed by centrifugation. After measuring protein concentrations using a BCA kit, the cell lysates (30 µg/lane) were resolved by sodium dodecyl-sulfate polyacrylamide gel electrophoresis (SDS-PAGE) on 10% gels and transferred onto polyvinylidene difluoride membranes. After being blocked with 5% skimmed dry milk powder in TBST for 2 h, the membranes were probed with primary antibodies against MnSOD, CaMKII, AMPK, HIF-1α, Glut-1, c-Myc, hexokinase 2 (HK-2), phosphofructokinase 1 (PFK-1), lactate dehydrogenase A (LDH-A), pyruvate kinase isozyme 2 (PKM-2), and β-actin (1:1000, all from Abiowell Biotechnology, Changsha, China) at 4°C overnight, The bound antibody was reacted with horseradish peroxidase-conjugated secondary antibodies (Abiowell Biotechnology), followed by visualizing with enhanced chemiluminescence reagents. The data were analyzed by densitometric scanning using ImageJ.
Glucose Uptake and Lactate Levels
MCF-7 cells were treated with control mouse serum or MD QRYLD-supplemented serum for 48 h. The impact of QRYLD treatment on glucose uptake in those cells, together with the same number of Ad-sh-MnSOD MCF-7 or Lent-MnSOD MCF-7 cells was tested using a 2-deoxyglucose (2-DG)–based colorimetric glucose uptake assay kit (Abcam, Cambridge, UK) per the manufacturer's protocol. Briefly, the cells were glucose-starved for 40 min, stimulated with insulin for 20 min, and exposed to 1 mM 2-DG for 20 min, followed by washing. The cells were lysed, and heated at 80 °C for 40 min. After neutralization, the samples were incubated with the reaction mixture at 37 °C for 30 min, neutralized, and incubated with reaction B (Lactate oxidase and Horseradish peroxidase). The levels of 2-DG6P–related NADPH in each group of samples were measured for the absorbance at 412 nm in a microplate reader. The concentrations of accumulated glucose were calculated according to the standard curve established using standard 2-DG6P provided by the manufacturer. In addition, the levels of lactic acid in the supernatants of cultured cells were measured in a fully automated biochemical analyzer (Solarbio).
Animal Studies
Female BALB/c nude mice were obtained from the Laboratory Animal Center, Xi’an Jiaotong University Health Science Center and housed in a specific pathogen-free facility in our university. The mice were randomized, and implanted with 3 × 106 MCF-7, Ad-sh-MnSOD MCF-7, or Lent-MnSOD MCF-7 cells in 100 µl of saline into their breast fat pads. Some mice were intravenously administered 11.375 g kg−1 day−1 QRYLD in 0.3 ml of saline daily beginning on day 5 post-inoculation for 10 consecutive days. Other groups of mice received the same volume of saline (n = 6 per group). The volumes of implanted tumors and body weights of mice were measured every other day for 4 weeks. At the end of the experiment, the mice were euthanized, and their tumor tissues were dissected, imaged, and weighed. The tumor tissues were fixed in 10% formalin and embedded in paraffin. The tumor tissue sections (4 µm) were subjected to immunohistochemistry using primary antibodies against MnSOD (1:100), CaMKII (1:200), and AMPK (1:2000, all from Abiowell Biotechnology); counterstained with hematoxylin; and analyzed using ImageJ software. All animal experiments were performed strictly following applicable national and institutional guidelines and approved by the Ethics Committee of the First Affiliated Hospital of Xi’an Jiaotong University (No. 2017 lunshenkezi No. 44, dated March 2, 2017).
Statistical Analysis
Data are presented as the mean ± SEM. The difference among three or more groups was analyzed by one-way ANOVA and post hoc Tukey's test. The comparison between groups was performed using Student's t-test. All statistical analyses were conducted using GraphPad Prism 8 software (GraphPad Software, Boston, MA, USA). Statistical significance was defined when a P-value of < .05.
Results
QRYLD Aqueous Extracts Contain Several Bioactive Components
To understand the biological functions of QRYLD, we prepared the aqueous extracts of QRYLD using 3300 g QRYLD and obtained 410 g QRYLD dry powder, leading to a powder recovery rate of approximately 12.4%. HPLC analyses revealed six main chemical compounds at high concentrations in QRYLD (Figure 1), namely chlorogenic acid (133.21 ± 19.05 mg/g), caffeic acid (151.22 ± 11.38 mg/g), quercetin (125 ± 2.96 mg/g), rutin (145.11 ± 16.29 mg/g), ferulic acid (179.53 ± 23.58 mg/g), and luteolin (102.39 ± 8.23 mg/g). Hence, the QRYLD aqueous extracts contained several chemical compounds with high bioactivity, and their interactions might contribute to the functions of QRYLD.

HPLC Analysis of the Chemical Components in QRYLD Aqueous Extracts. (A) Drug Standards. (B) QRYLD Aqueous Extracts. 1, Chlorogenic Acid; 2, Caffeic Acid; 3, Quercetin; 4, Rutin; 5, Ferulic Acid; 6, Luteolin.
QRYLD Targets Several Biological Processes and Signaling Pathways in Breast Cancer Cells
Next, we explored whether QRYLD treatment could modulate the transcriptome in MCF-7 cells. Following treatment of MCF-7 cells with control mouse serum or QRYLD-supplemented serum for 48 h, the transcriptomes of control and QRYLD-treated MCF-7 cells were analyzed by RNA sequencing. Compared with the control serum-treated MCF-7 cells, treatment with QRYLD-supplemented serum caused 2918 DEGs, including 1693 upregulated and 1225 downregulated genes, in the QRYLD-treated cells. Notably, 293 of these DEGs, including 133 downregulated and 160 upregulated DEGs, were oncogenes and tumor suppressor genes (Figure 2A). Bioinformatic analyses predicted that these 293 DEGs were mainly involved in metabolism-related signaling pathways or biological processes. GO and KEGG analyses unveiled that the top 50 DEGs were directly related to the Warburg effect (Figure 2B). The connections among these targets and signal pathways predicted that these 293 DEGs were mainly involved in response to oxidative stress, cancer-related signaling pathways, glycogen synthesis and metabolism, the pentose phosphate pathway, the TCA cycle, glucose metabolism regulation, gluconeogenesis, glycolysis, and the MnSOD/MAPK signaling (Figure 2C). Possibly, QRYLD may target MnSOD in breast cancer.

Bioinformatic Analysis of the Transcriptomes of QRYLD-Treated MCF-7 Cells. (A) Heatmap Analysis of DEGs. 1–3, the Control MCF-7 Cells; 4–6, the QRYLD-Treated MCF-7 Cells. (B) PPI Analysis among the Targets of QRYLD. (C) Enrichment Analysis of DEGs.
QRYLD Treatment Limits the Malignant Behaviors of MCF-7 Cells
To understand the importance of MnSOD in the malignant behaviors of breast cancer cells, we generated stable MnSOD-silenced Ad-sh-MnSOD cells and MnSOD-overexpressing Lent-MnSOD MCF-7 cells. Subsequently, wild-type MCF-7 cells were treated with control serum or LD, MD, or HD QRYLD-supplemented serum, and their proliferation was tested by the CCK-8 assay. Compared with the proliferation in the control group, the proliferation of Ad-sh-MnSOD MCF-7 cells was significantly reduced (P < .01), whereas that of Lent-MnSOD MCF-7 cells was significantly enhanced (P < .01). In addition, the proliferation of QRYLD-treated MCF-7 cells (LD, MD, or HD) was significantly reduced (P < .05, Figure 3A). Notably, treatment with MD QRYLD-medicated serum for 48 h inhibited cell proliferation by 50%. Accordingly, we used MD QRYLD-supplemented serum for the subsequent in vitro experiments. Transwell invasion assays indicated that the number of invaded cells was significantly lower in the QRYLD group than in the control and Lent-MnSOD groups, whereas it was slightly greater than that in in the Ad-sh-MnSOD group (Figure 3B). A similar pattern of wound healing was observed among the different groups of cells (Figure 3C).

QRYLD Treatment, Similar to MnSOD Silencing, Supresses the Malignancy of MCF-7 Cells in vitro. (A) the Effect of QRYLD on Cell Viability. MCF-7 Cells Were Treated with Control serum or the Indicated Concentrations of QRYLD-Supplemented serum and Tested Together with Ad-sh-MnSOD and Lent-MnSOD MCF-7 Cells in Triplicate to Assess Their Viability by the CCK-8 Assay at the Indicated Time Points. (B) the Effects of QRYLD on Cell Invasion. (C) the Effects of QRYLD on Wound Healing in MCF-7 Cells. (D) Flow Cytometry Analysis of the Effects of QRYLD on Cell Apoptosis. Scale bar, 50 μm. Data are Presented as the Mean ± SEM of Each Group from Three Separate Experiments, *P < .05, **P < .01 Versus the Control.
Flow cytometry revealed that compared with the control MCF-7 cells, QRYLD treatment significantly increased the percentages of apoptotic MCF-7 cells (P < .05) and MnSOD silencing also further increased the eprcentages of apoptotic MCF-7 cells (P < .01, Figure 3D). In contrast, MnSOD overexpression significantly reduced the percentages of spontaneously apoptotic MCF-7 cells (P < .05). Collectively, the reduced cell proliferation, invasion and wound healing, together with enhanced apoptosis, indicated that QRYLD treatment, similar to MnSOD silencing, limited the malignant behaviors of MCF-7 cells in vitro.
QRYLD Treatment Inhibits Glucose Uptake and Lactic Acid Production and Reduces the Warburg Effect in MCF-7 Cells by Attenuating the MnSOD/CaMKII/AMPK Signaling
To further understand the pharmacological action of QRYLD, we tested its effects on glucose uptake and lactic acid production in MCF-7 cells. Following treatment with MD QRYLD-supplemented serum for 48 h, the levels of glucose uptake in the different groups of MCF-7 cells were measured by 2-DG–based colorimetric assays. Compared with the control MCF-7 cells, the levels of glucose uptake and lactic acid production were significantly lower in the QRYLD-treated MCF-7 and Ad-sh-MnSOD MCF-7 cells while they were significantly higher in the Lent-MnSOD MCF-7 cells (P < .05 or P < .01, Figure 4A). Apparently, treatment with QRYLD, like MnSOD silencing, decreased glucose uptake and lactic acid production to suppress the Warburg effect in MCF-7 cells.
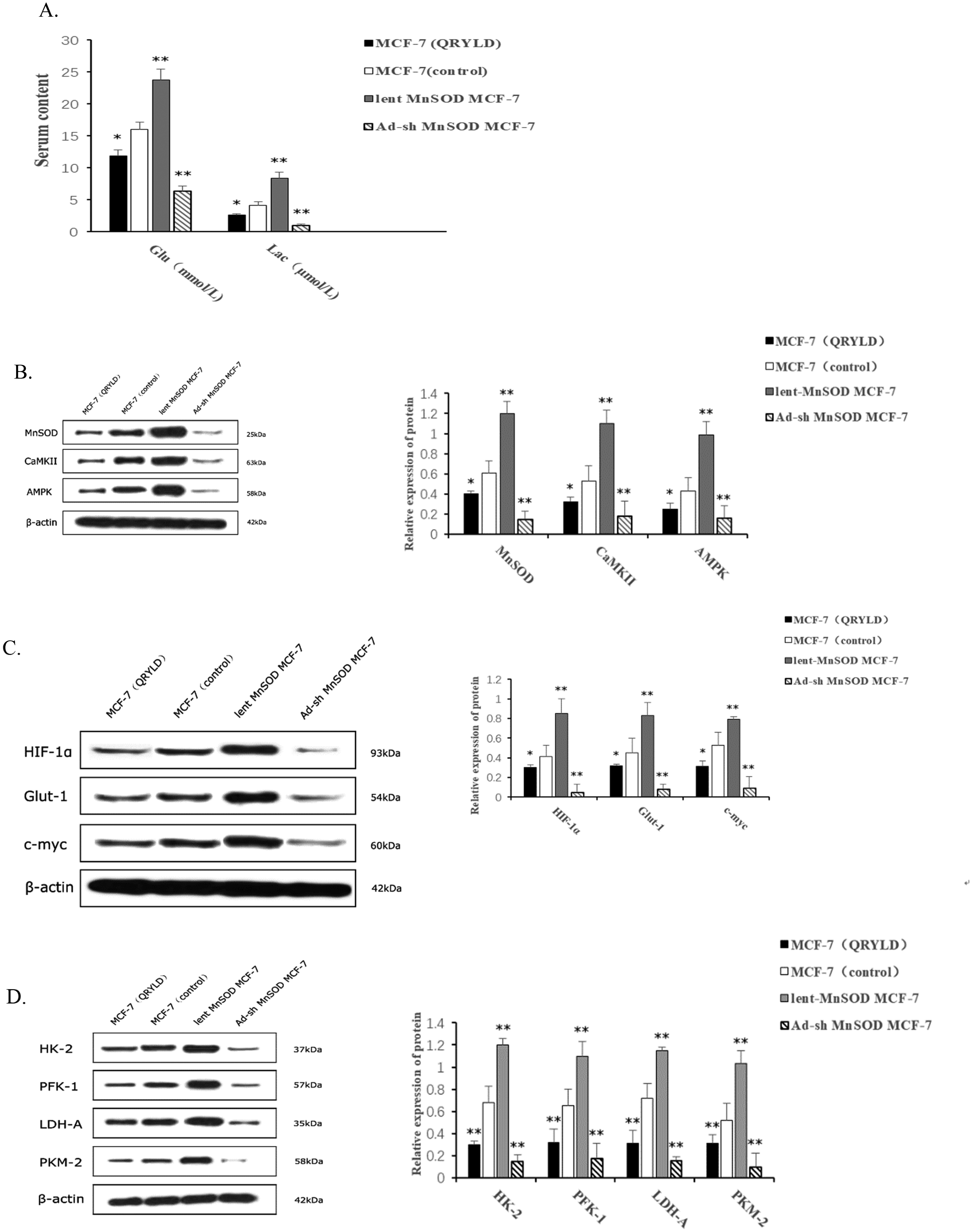
QRYLD Treatment, Similar to MnSOD Silencing, Modulates the Warburg Effect in MCF-7 Cells. (A) the Effect of QRYLD on Glucose Uptake and Lactic Acid Production. (B) the Expression of key Regulatory Factors in the Warburg Effect. (C) the Expression of Metabolic Enzymes Involved in the Warburg Effect. (D) the Expression of MnSOD/CaMKII/AMPK Pathway Proteins in the Warburg Effect. Data are Presented as the Mean ± SEM of Each Group of Cells from Three Separate Experiments, *P < .05, **P < .01 Versus the Control.
It is well known that several regulators are crucial for the Warburg effect in cancers. To explore the molecular mechanism by which QRYLD treatment suppressed the Warburg effect in MCF-7 cells, we tested the relative levels of HIF-1α, Glut-1, and C-Myc expression in the different groups of cells by Western blot. Indead, the relative levels of HIF-1α, Glut-1, and C-Myc were significantly lower in the QRYLD-treated MCF-7 cells than in the control and Lent-MnSOD MCF-7 cells but higher than that in the Ad-sh-MnSOD MCF-7 cells (Figure 4B). Similar results were observed in the relative levels of HK-2, PFK-1, LDH-A, and PKM-2 expression in the different groups of MCF-7 cells (Figure 4C). These data indicated that QRYLD, similar to MnSOD silencing, inhibited the expression of these regulators and metabolic enzymes, leading to its inhibition on the Warburg effect in MCF-7 cells.
Given the importance of MnSOD in the pharmacological functions of QRYLD, we tested whether QRYLD treatment could modulate the MnSOD/CaMKII/AMPK signaling in MCF-7 cells by Western blot. As expected, the relative levels of MnSOD expression were significantly lower in the QRYLD-treated MCF-7 cells than that in the control and Lent-MnSOD MCF-7 cells, but significantly higher than that in the Ad-sh-MnSOD MCF-7 cells (Figure 4D). These data demonstrated the efficiency of MnSOD overexpression and silencing in MCF-7 cells and suggest that QRYLD may target MnSOD expression in MCF-7 cells. A similar pattern of CaMKII and AMPK expression was observed in the different groups of MCF-7 cells. Collectively, these data provided novel evidence to demonstrate that QRYLD, similar to MnSOD silencing, inhibited glucose uptake and lactic acid production, reducing the Warburg effect in MCF-7 cells by suppressing the expression of several regulators and metabolic enzymes and attenuating the MnSOD/CaMKII/AMPK signaling.
QRYLD Treatment Inhibits the Growth of Breast Cancers in Mice
Finally, we tested the therapeutic effect of QRYLD on the growth of xenografted breast cancers in mice. After establishment of wild-type, Ad-sh-MnSOD, or Lent-MnSOD MCF-7 xenograft tumors, the wild-type tumor-bearing mice were randomized and treated intravenously with vehicle saline or MD QRYLD daily from day 5 to day 15. The dynamic growth of implanted tumors in the different groups of mice was monitored. Compared with the control group, QRYLD treatment, similar to MnSOD silencing, significantly decreased tumor volumes and weights in mice, whereas induction of MnSOD overexpression significantly increased tumor volumes and weights in mice (Figure 5A and C). Conversely, there was no significant difference in body weights among the different groups of mice (Figure 5B). Immunohistochemistry illustrated that compared with the control tumors, the tumors from QRYLD-treated mice, similar to that from the Ad-sh-MnSOD mice, exhibited a reduced level of MnSOD, CaMKII, and AMPK expression, whereas their expression was significantly up-regulated in the Lent-MnSOD tumors (Figure 5D). Together, these data indicated that MnSOD overexpression significantly enhanced the MnSOD/CaMKII/AMPK signaling to promote the growth of implanted MCF-7 tumors in mice and QRYLD treatment, similar to MnSOD silencing significantly inhibited the growth of implanted breast tumors in mice by attenuating the MnSOD/CaMKII/AMPK signaling.

QRYLD Treatment Suppresses the Tumor Growth in Mice. (A) Tumor Volumes Were Measured in Mice Every Other day for 4 Consecutive Weeks. (B) the Body Weights of Mice Were Measured Every Other day for 4 Consecutive Weeks. (C) Images of all Dissected Tumors and Their Weights. (D) Immunohistochemistry Analyses of MnSOD, CaMKII, and AMPK Expression in Tumor Tissues from Each Group of Mice (n = 6 per Group). Scale bar, 50 μm. Data are Presented as the Mean ±SEM of Each Group of Mice from Three Separate Experiments, *P < .05, **P < .01 Versus the Control.
Discussion
This study explored whether and how QRYLD treatment modulated the malignant behaviors of MCF-7 cells. First, we identified bioactive compounds in the QRYLD aqueous extracts by HPLC and they mainly included high concentrations of chlorogenic acid, caffeic acid, quercetin, rutin, ferulic acid, and luteolin. The synergistic and addictive effects of these bioactive compounds may contributed to their biological functions. Biological analyses indicated that QRYLD attenuated the MnSOD/CaMKII/AMPK signaling to inhibit the Warburg effect and malignant behavior in MCF-7 cells. Evidently, transcriptomic analysis indicated that QRYLD treatment modulated the expression of several genes, such as MnSOD, in MCF-7 cells. Bioinformatic analysis unveiled that the DEGs mainly participated in metabolism-related signaling pathways, such as the MnSOD/MAPK signaling pathway, and biological processes, including the Warburg effect. Furthermore, QRYLD treatment significantly inhibited the malignant behaviors of MCF-7 cells by inhibiting their proliferation, migration, invasion, spontaneous apoptosis in vitro and tumor growth in vivo. In addition, QRYLD treatment decreased the expression of MnSOD, CaMKII, and AMPK in MCF-7 cells and in the implanted tumors in mice. Interestingly, the therapeutic effects of QRYLD treatment were less than that of MnSOD silencing in MCF-7 cells. These data support the notion that QRYLD may suppress MnSOD expression and attenuate the MnSOD/CaMKII/AMPK signaling to inhibit the Warburg effect and malignant behaviors in MCF-7 cells. Our novel findings suggest that QRYLD may target MnSOD expression and may provide new pharmacological mechanisms by which QRYLD treatment inhibits the malignancy of breast cancer (Figure 6).

A Diagram Illustrates the Molecular Mechanisms by Which QRYLD Treatment Attenuates the MnSOD/CaMKII/AMPK Signaling and Warburg Effect in Breast Cancer.
MnSOD expression is usually upregulated in cancer cells, and MnSOD can promote the release of H2O2 in mitochondria and upregulate the expression of CaMKII, which can phosphorylate and activate AMPK. AMPK is an important energy receptor in cells that can enhance glycolysis and the Warburg effect.42–46 AMPK activation is also crucial for cellular responses to stress, autophagy, survival, and cancer cell growth.47–51 By markedly scavenging mitochondrial superoxide, MnSOD overexpression reshapes the ROS landscape, modifying its composition and spatial dynamics. Consequently, the outcome transcends a mere decrease in ROS, constituting a fundamental reprogramming of the cellular redox status.52,53 This reprogramming activates transcription factors sensitive to oxidative changes, such as HIF-1α, ultimately promoting the activation of a genetic program that enhances proliferation, migration, invasion, and glycolysis. Accordingly, MnSOD is a potent antioxidant and an oncogenic factor that supports the survival of cells under hypoxic conditions. MnSOD can shift glucose metabolism from oxidative phosphorylation to glycolysis in human triple-negative breast cancer cells by transmitting the glucose signal and changing the oxygen transmission pathway.54,55 Consistently, we found that MnSOD overexpression enhanced glycolysis in MCF-7 cells by increasing CaMKII and AMPK expression and promoted malignant behaviors, such as proliferation, migration, wound healing, and invasion, in MCF-7 cells. 56 Conversely, treatment with QRYLD to reduce MNSOD expression or MnSOD silencing dramatically decreased glucose uptake, lactic acid production, and Glut-1 expression, which are hallmarks of suppressed glycolysis, extending previous findings.57,58
Our data disagreed with a previous observation that MnSOD deficiency does not significantly alter the levels of glycolysis in breast cancer cells. The difference between these findings may stem from the use of different breast cancer cell lines with varying metabolic states. In fact, MnSOD silencing promoted the occurrence of oxidative phosphorylation. 59 In breast cancer cells, MnSOD may maintain a stable increase in H2O2 levels, thus maintaining the activation of AMPK and the transformation of intracellular metabolism into glycolysis, whereas MnSOD silencing or AMPK inhibition can reduce the growth of cells. 60 Hence, the MnSOD/CaMKII/AMPK axis may be crucial for supporting the bioenergetics of cancer cells, and it can promote the progression of breast cancer. 61
In our experiment, treatment with QRYLD or induction of MnSOD silencing down-regulated CaMKII and AMPK expression in MCF-7 cells. Furthermore, QRYLD treatment or MnSOD silencing significantly reduced the expression of HK-2, PFK-1, LDH-A, and PKM-2, the key metabolic enzymes in the Warburg effect in MCF-7 cells. Consequently, QRYLD treatment or MnSOD-silencing suppressed the malignant behaviors of MCF-7 cells in vitro and in vivo. By contrast, MnSOD overexpression significantly enhanced the expression of CaMKII, AMPK, which are key metabolic enzymes and critical regulators of proteins such as HIF-1α, Glut-1, and c-Myc expression, in MCF7 cells, and these changes were accompanied by enhanced malignant behaviors of MCF-7 cells in vitro and in vivo. These parallel data suggest that the MnSOD/CaMKII/AMPK axis may be a key factor regulating the progression of breast cancer, and this axis may represent a therapeutic target for breast cancer treatment.
In conclusion, our data indicated that QRYLD contained several bioactive components. However, we did not validate which compound(s) and their interactions are responsible for the biological functions of QRYLD treatment in MCF-7 cells. We are interested in further investigating how these bioactive compounds contribute to the biological functions of QRYLD treatment in MCF-7 cells in the future. More importantly, we found that treatment with QRYLD significantly modulated the expression of many genes, including the metabolism-related gene MnSOD. QRYLD treatment, similarly as MnSOD silencing, significantly suppressed malignant behaviors and the Warburg effect of MCF-7 cells as well as tumor growth in vivo. These effects were associated with inhibition of the MnSOD/CaMKII/AMPK signaling in MCF-7 cells. Our findings may provide new insights into the pharmacological actions of QRYLD in inhibiting breast cancer growth and unveil new therapeutic targets for designing therapies for breast cancer.
Abbreviations
Qing-Re-Yi-Liu decoction
Manganese superoxide dismutase
Calmodulin-dependent kinase II
AMP-activated protein kinase
Glucose transporter-1
Hypoxia-inducible factor 1α
Hexokinase-2
Phosphofructokinase 1
Lactate dehydrogenase A
Pyruvate kinase isozyme 2
Multiplicity of infection
Differentially expressed genes
Protein–proteininteractions
Footnotes
Ethics Approval and Consent to Participate
All experiments were performed following the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health (NIH Publication No. 85-23, revised 1996). The study was conducted in accordance with the Declaration of Helsinki. The study was approved by the Ethics Committee of the First Affiliated Hospital of Xi’an Jiaotong University (No. 2017 lunshenkezi No. 44, dated March 2, 2017).
Author Contributions
Yuan Cao performed statistical analysis. Lianqing Sun provided the plant materials. Junxia Wang performed phytochemical analysis. Jing Du performed the in vivo and in vitro experiments. Ying Zhang and Ying Yuan conceived and supervised the study. Zhe Zhang drafted the manuscript.
All authors read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by grants from the National Natural Science Foundation of China (no. 81703899), Shaanxi Provincial Natural Science Basic Research Program (no.2017JM8068), Shaanxi Provincial Administration of Traditional Chinese Medicine Project (no.15-JC021), Xi'an Jiaotong University First Affiliated Hospital General Project (no. 2021ZXY-06), The second batch of provincial-level Chinese medicine young and middle-aged scientific and technological backbone projects (no. 2023-ZQNY-008). Shaanxi province traditional Chinese medicine research and innovation team (TZKN-CXTD-07).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
The data included in this manuscript are available from the corresponding author.
