Abstract
Objective
To find out the effect of high-intensity interval training (HIIT) and mindfulness breathing on the levels of sex hormones, insulin resistance, lipid profiles, and tumor biomarkers in postmenopausal obese women.
Design
Seventy obese postmenopausal women (aged 55-65 years, with a body mass index (BMI) ranging from 35–44.9 kg/m2) were allocated into two groups for this prospective, randomized, controlled study: the experimental group, which received HIIT and mindfulness breathing in addition to their medications, and the control group, which only received medication. Serum sex hormone levels (estradiol, total testosterone [TT], free testosterone [FT], and sex hormone-binding globulin [SHBG]), insulin resistance assessment using the homeostasis model [HOMA-IR], lipids (total cholesterol [TC], triglycerides [TGs], high-density lipoprotein [HDL], and TC/HDL), and tumor markers (Carcino Embryonic Antigen [CEA] and Cancer Antigen [CA125]) were among the pre and post assessments conducted after 12 weeks.
Results
After 12 weeks of the study, the HIIT and mindfulness breathing group showed significant decreases in weight, BMI, FT, TT, estradiol, HOMA-IR, CEA, CA125, TC, TGs, and TC/HDL and increases in SHBG and HDL (all p < 0.001). In contrast, the control group had increases in weight, BMI, FT, TT, estradiol, HOMA-IR, CEA, CA125, TC, TGs, and TC/HDL and decreases in SHBG and HDL (all p < 0.001).
Conclusion
HIIT and mindfulness breathing were efficient approaches for lowering metabolic, hormonal, and tumor-related risk factors in obese postmenopausal women.
Introduction
The health of postmenopausal women is a global concern since it impacts both the economy and society in developed and developing countries. 1 Hormonal changes during menopause result in a variety of illnesses that negatively impact the quality of life for 80% of women. 2 A reduction in circulating estrogen level brought on by the onset of menopause could promote the formation of visceral fat. 3 This change in the distribution of fat points to a major role for estrogen in the development of postmenopausal obesity. 4
Since the pathophysiology of insulin resistance (IR) is impacted by fat distribution, the development of IR and postmenopausal obesity are strongly related. 4 Numerous causes and mechanisms caused by obesity may be corresponding to a high mortality risk and a negative impact on indicators related to tumor risk, 5 such as changes in adipokine production and alterations in sex hormones. 6
Hyperinsulinemia combined with abnormalities in cell proliferation and apoptosis promotes the formation of tumor cells with a high capacity for invasion. 7 The cancer incidence in Egypt is 157.0 per 100,000 women, and it could triple by 2050 due to the general increase in risk factors for the disease. 8
Therefore, routine screening for tumor biomarkers such cancer antigen (CA125) and carcino embryonic antigen (CEA) may help reduce the incidence and adverse consequences of cancer. 9 Additionally, studies have been conducted to evaluate the benefits of several therapies (medication, dietary habits, physical activity, and weight control) for reducing postmenopausal women's chance of developing cancer. 10
High-intensity interval training (HIIT) is one of the most recommended types of exercise because of its therapeutic benefits and accessibility in managing a variety of medical conditions. 11 It has been shown to reduce the risk of breast cancer in postmenopausal women by 23% by improving their inflammatory and adipokine profiles, insulin sensitivity, lipid metabolism, and cardiovascular function. 12
A simple mind-body exercise identified as mindful breathing can assist increase both the body's ability to relax and the mind's capacity to promote general wellbeing. 13 Given how difficult it may be to persuade patients to exercise and become more physically active, it might be an additional tool that therapists employ when dealing with their patients. 14
Consequently, including mindfulness into exercise therapy can enhance self-efficacy and exercise adherence, offer psychological and physiological advantages, 13 and lower stress. 15 All of which help postmenopausal women to lose weight and avoid several comorbidities.16,17
Relaxation-focused breathing methods, such as mindfulness-based breathing, are known to lower stress, which indirectly affects body weight and sex hormones via affecting the autonomic nervous system, sleep quality, and other hormonal and metabolic changes. 18
Reducing stress can affect the hypothalamic-pituitary-ovarian axis, which controls sex hormones, and indirectly promote a more balanced hormonal environment, which may eventually affect the levels of estradiol. 19
Therefore, we postulated that postmenopausal women might benefit from HIIT that incorporates mindfulness breathing. Thus, this study aims to demonstrate the effects of 12 weeks of HIIT and mindfulness breathing on metabolic and tumor markers that relate to cancer risk in postmenopausal obese women.
Participants and Methods
Study Design and Participants
Between August 2020 and August 2021, obese postmenopausal women aged 55 to 65 were recruited for this prospective twelve-week randomized controlled study from (hidden for review). Before the intervention commenced, each participant filled out a consent form after being informed about the study's details and their rights as participants. With registration number (hidden for review), this study is registered on ClinicalTrials.gov and was authorized by the Ethics Committee Board of (hidden for review). The study was carried out in accordance with the Declaration of Helsinki.
The inclusion criteria were: 1) age between 55–65 years; 2) sedentary with a mean Physical Activity Questionnaire score 20 of 11.25 ± 1.38; 3) BMI of 37.67 ± 1.68 kg/m2; 4) dyslipidemia (total cholesterol TC > 200 mg/dl, high-density lipoprotein HDL < 35 mg/dl, and triglycerides TGs >150 mg/dl); 5) not engaged in any regular exercise training or diet program for at least a year before participating in this study; 6) at least five years without a menstrual cycle and have a first-degree relative with cancer in their family history.
The exclusion criteria were: 1) Diabetes; 2) long-term neurological or musculoskeletal illnesses with persistent motor deficits; 3) any other physical or medical conditions that might have an impact on the results of the study; 4) hormone therapy for at least six months before participation in this trial; 5) any cardiovascular or autoimmune condition, any current malignant or benign tumors, or any disorder associated with increased tumor markers. The fact that both groups took their drugs under a physician's supervision had no influence on the results of the study.
Out of the 85 women that were evaluated, 15 were excluded (n = 5 withdrew to participate, and n = 10 did not match the inclusion criteria). Out of 70 women, 35 were randomly assigned to the experimental group and another 35 to the control group. Ten women dropped out of the study for a variety of reasons, thus only the 60 women who completed the study were included in the data analysis. (Figure 1).

Consort Flow Chart of Study Participants.
Grouping
Seventy postmenopausal women were randomly allocated to participate using Minimpy® software, version 0.3, employing block size randomization (with sizes 2, 4, and 6) in a 1:1 way, according to a statistician who was not engaged in the study. Those in the experimental group (n = 35) received their prescribed drugs together with mindfulness breathing and HIIT, while those in the control group (n = 35) just received their prescription prescriptions. An independent researcher blindly concealed the group allocation in numbered envelopes, so neither the participants nor the result assessor knew. There was no communication between the participants of the experimental and control groups.
Sample Size
An independent t-test was used to determine between-group differences based on changes in the primary outcome (HOMA-IR), which was based on a pilot study with 12 participants. With an anticipated dropout rate of 15%, the anticipated required sample size was 60 participants for a moderate effect size (0.5), 80% statistical power and 0.05 alpha level. Accordingly, 70 participants were the total enrollment target based on this attrition.
Intervention
The initial physical interaction with the subjects was a familiarization session. Pre-test evaluations and a familiarization training session came next. The intervention phase, which lasted for the following weeks, ended with the post-test in the last week. Every pre- and post-test was carried out in the lab under the direction of the investigator (hidden for review), ensuring consistent conditions all the time.
Training Protocol
In accordance with Henke et al's protocol 21 participants in the experimental group underwent supervised cycling training (BC-6792G Exercise Bike, Maximum user weight: 120 kg, Body Sculpture-Egypt). To ascertain the peak heart rate (PHR), the participants engaged in cycle cardiopulmonary exercise test (CPET) on a bicycle spiro-ergometer (ZAN600, nSpire Health GmbH, Oberthulba, Germany). They were encouraged to perform a ramp technique with an increasing load of 20 W/min until they were physically exhausted. Heart rate, blood pressure, and workload (Watt and % of expected Watt reference) were recorded as well.
A 5-min warm-up and cool-down period at 50% of PHR denoted the beginning and end of the exercise program, which was conducted for 12 weeks in consecutive weeks with three sessions per week (36 sessions total), and an exercise prescription based on the PHR reached during the cycle CPET at baseline on the cycle ergometer. Ten cycling bouts for 60-s (85%–90% PHR) were interspersed with 75-s, low-intensity cycling (40% PHR), which corresponded to 8–10 points on the Borg scale (rated from 6-20). 22
Throughout the exercise, the participants’ heart rates were continually measured by wearing a pulse oximeter (made by Beurer, Germany). Throughout the 12-week period, the workload was adjusted to keep the exercise intensity (85%–90% PHR) between 15 and 19 points on the Borg scale. None of the patients suffered any adverse effects during the study period, which was routinely monitored by a member of the research team (hidden for review).
A physician (hidden for review) unaware of the study hypothesis) conducted a medical health interview with the participant during the baseline visit to determine their current state of health. The following precautions and contraindications were considered before a participant began an exercise training session: chest discomfort or a systolic blood pressure of more than 200 mm Hg and a diastolic blood pressure of more than 100 mm Hg.
Additionally, during an exercise training session, the following termination conditions were applied: indicators of poor perfusion (eg, confusion, nausea, lightheadedness, central cyanosis, asked to stop, and physical or verbal manifestations of acute exhaustion). Safety measures during exercise included wearing appropriate clothing and footwear, warming up and cooling down properly, monitoring heart rate to keep within the target heart rate zone, and drinking plenty of water.
Mindfulness Breathing
Mindful breathing is a mindfulness exercise that uses breathing as a target of attention. Prior to starting the HIIT, the experimental group engaged in a five-minute mindfulness breathing that was adapted from Beng et al 23 who provided a simplified version of this exercise. It involved the three steps listed below: A physical posture should be adopted, a mindfulness anchor should be established, and mindfulness should be maintained. Participants were assisted in sitting erect on a chair to establish a physical posture that supported their mental and physical health.
After that, participants learned how to develop the practice of mindfulness through figuring out to manage their thoughts so they could be in the present moment. Observing the breathing process is one of the simplest methods to accomplish this. In the last phase, participants were urged to stay mindful by focusing on their breathing and controlling their thoughts. Every time their thoughts wandered; participants were instructed to concentrate on their breathing. They were directed to spend five minutes concentrating on the breathing process.
Measures
After a 12-h fast, antecubital venous blood samples (8 ml) were collected from each participant before the trial began and after completion between 8:10
For all sex hormones, including estradiol, total testosterone (TT), free testosterone (FT), and sex hormone-binding globulin (SHBG), chemiluminescence hormonal analysis was used. By radioimmunoassay, tumor markers CEA and CA 125 were examined. An enzymatic approach was used to test the TGs and HDL. The following formula was used to determine the HOMA-IR index, which tested for the presence of IR: fasting glucose (mmol•L−1) _ fasting insulin (mU•L−1)/ 22.5. 24
BMI
The participants’ body mass (kg) and height (meters) were measured on a digital scale (TR-200 lP-China) with an accuracy of 0.1 kg and 0.1 cm and a capacity of 200 kg to determine their BMI using this formula [weight (kg)/ height (m2)].
Statistical Analysis
Data was analyzed using SPSS software, version 25, from IBM SPSS, Chicago, Illinois, USA. The Shapiro-Wilk test was used to determine whether the data distribution was normal, and continuous data were represented as mean ± standard deviation (SD).
Independent t-test was used to examine the differences between the two groups at baseline. Differences from pre-training to post-training within each group were analyzed using paired t-tests. A mixed repeated measures ANOVA (between-within-subject analysis of variance) was performed to identify the impact of the two different treatment programs across two time periods. Partial eta squared (ηp 2) was the effect size reported for the main effects (small (η² = 0.01), medium (η² = 0.06), and large (η² = 0.14)). The level of significance was set at a P-value less than 0.05.
Results
In the final data analysis, 60 postmenopausal women who finished the study were included. The mean age of all participants was 58.80 ± 2.78 years. The mean height of all participants was 159.73 ± 3.60 kg and ranged from 155.00 to 169.00 cm. The mean weight of all participants was 96.15 ± 5.81 kg and ranged from 87.00 to 117.50 kg. The mean BMI was 37.67 ± 1.68 kg/m2 and ranged from 35.10 to 44.80 kg/m2.
As illustrated in Table 1, there were no statistically significant differences between the experimental and control groups regarding age, height, BMI, weight, sex hormone concentration, HOMA-IR index, lipid profile, and tumor markers at baseline (P > 0.05).
Characteristics of Study Participants.

BMI: Body mass index; CA125: Cancer antigen 125; CEA: Carcinoembryonic antigen; FT: free testosterone; HDL: high-density lipoprotein; HOMA-IR: Homeostatic Model Assessment for Insulin Resistance; SHBG: Sex Hormone Binding Globulin; TC: Total cholesterol; TGs: triglycerides; TT: total testosterone.
Data represented as mean ± standard deviation (SD).
As shown in Table 2, the experimental group's weight and BMI decreased statistically significantly from pre-intervention (97.42 ± 6.18 kg and 37.88 ± 1.93 kg/m2) to post-intervention (75.49 ± 6.58 kg and 30.53 ± 1.65 kg/m2, respectively) (p < 0.001). On the other hand, the control group's weight and BMI increased significantly between before and after the intervention (p < 0.001), increasing from 94.87 ± 5.20 kg to 95.70 ± 5.77 kg and 37.47 ± 1.39 kg/m² to 38.25 ± 1.51 kg/m², respectively.
Comparison of Weight, BMI, Sex Hormones, HOMA-IR Index, and Lipid Profile Before and After 12 Weeks.
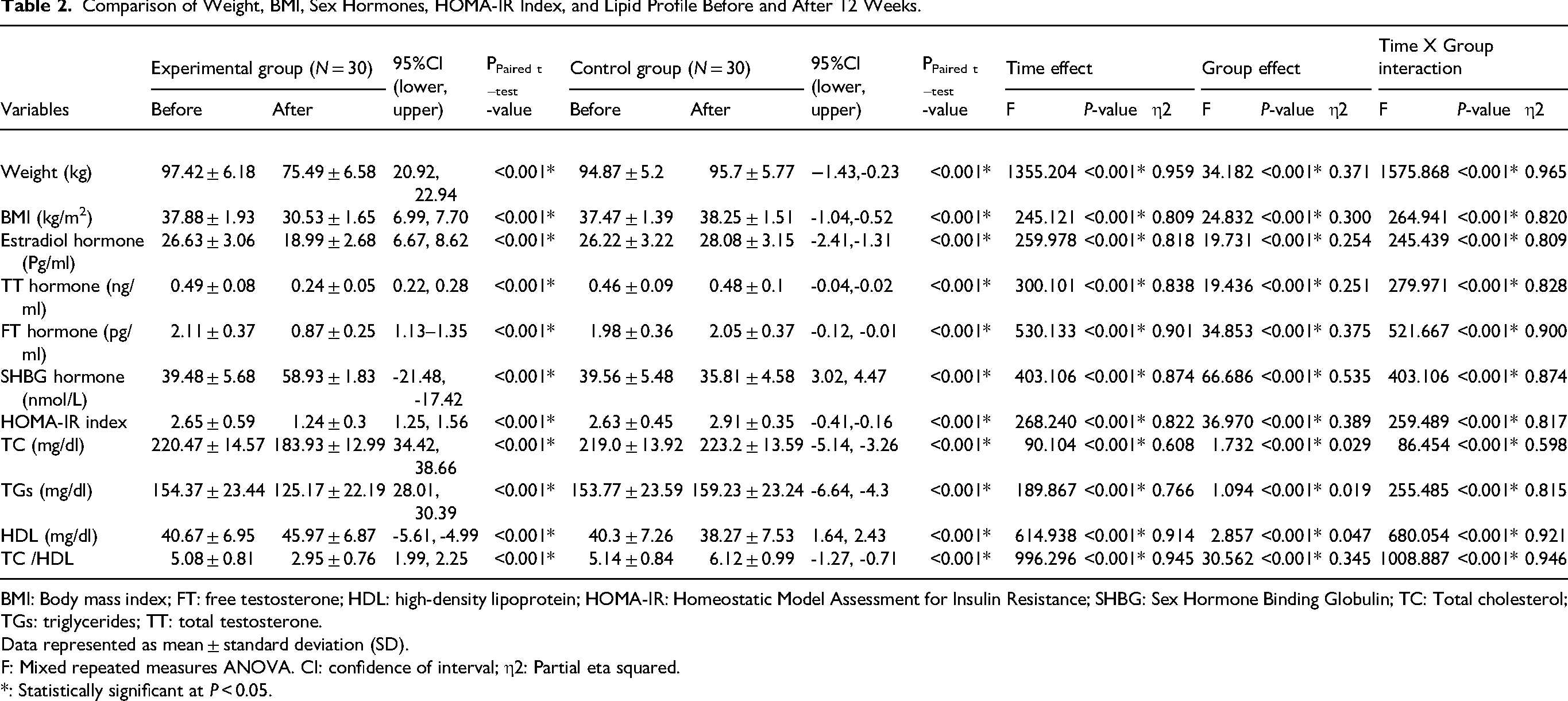
BMI: Body mass index; FT: free testosterone; HDL: high-density lipoprotein; HOMA-IR: Homeostatic Model Assessment for Insulin Resistance; SHBG: Sex Hormone Binding Globulin; TC: Total cholesterol; TGs: triglycerides; TT: total testosterone.
Data represented as mean ± standard deviation (SD).
F: Mixed repeated measures ANOVA. CI: confidence of interval; η2: Partial eta squared.
*: Statistically significant at P < 0.05.
Significant main effects for time and group were found in weight [F (1,58) = 1355.204, p < 0.001, η2 = 0.959 and F (1,58) = 34.182, p < 0.001, η2 = 0.371] and BMI [F (1,58) = 245.121, p < 0.001, η2 = 0.809 and F (1,58) = 24.832, p < 0.001, η2 = 0.300] according to the mixed repeated measure ANOVA. Additionally, a statistically significant main impact of time and group interaction was identified [F (1,58) = 264.941, p < 0.001, η2 = 0.820 for BMI and F (1,58) = 1575.868, p < 0.001, η2 = 0.965 for weight].
Furthermore, Table 2 showed that the levels of the hormones FT, TT, and estradiol in the experimental group reduced statistically significantly before and after the intervention. On the other hand, TT, FT, and estradiol hormone levels in the control group increased statistically significantly. Furthermore, compared to the control group, the experimental group demonstrated a greater statistically significant improvement.
Moreover, the main effects of time and group were significant for the hormones estradiol [F (1, 58) = 259.978, p < 0.001, η² = 0.818 and F (1, 58) = 19.731, p < 0.001, η² = 0.254], TT [F (1, 58) = 300.101, p < 0.001, η² = 0.838 and F (1, 58) = 19.436, p < 0.001, η² = 0.251], and FT [F (1, 58) = 530.133, p < 0.001, η² = 0.901 and F (1, 58) = 34.853, p < 0.001, η² = 0.375]. Moreover, estradiol hormone, TT hormone, and FT hormone showed a significant interaction effect between time and group [F (1, 58) = 245.439, p < 0.001, η² = 0.809, F (1, 58) = 279.971, p < 0.001, η² = 0.828, and F (1, 58) = 521.667, p < 0.001, η² = 0.900].
As for SHBG hormone, the experimental group's level increased statistically significantly from pre- to post-intervention (p < 0.001 at 95% CI: −21.48, −17.42), but the control group's level declined (p < 0.001 at 95% CI: 3.02, 4.47) (Table 2). The SHBG hormone showed significant main effects of both group and time [F (1, 58) = 66.686, p < 0.001, η² = 0.535, and F (1, 58) = 403.106, p < 0.001, η² = 0.874, respectively]. Additionally, there was a significant interaction effect between group and time [F (1, 58) = 403.106, p < 0.001, η² = 0.874].
The experimental group's HOMA-IR index decreased statistically significantly (p < 0.001 at 95% CI:1.25, 1.56), while the control group's HOMA-IR index significantly increased (p < 0.001 at 95% CI: −0.41, −0.16) (Table 2). Also, the experimental group's mean HOMA-IR index (1.24 ± 0.3) was significantly lower than the control group's (2.91 ± 0.35).
Both time and group had significant main effects on the HOMA-IR index, according to the mixed ANOVA analysis [F (1, 58) = 268.24, p < 0.001, η² = 0.822 for time, and F (1, 58) = 36.97, p < 0.001, η² = 0.389 for group]. Furthermore, there was a significant interaction effect between group and time [F (1, 58) = 259.489, p < 0.001, η² = 0.817].
TC, TGs, and TC/HDL levels in the experimental group decreased statistically significantly (after 12 weeks of the intervention). On the other hand, the control group's TC, TGs, and TC/HDL levels decreased statistically significantly. The TC [F (1, 58) = 90.104, p < 0.001, η² = 0.608), TGs (F (1, 58) = 189.867, p < 0.001, η² = 0.766), and TC/HDL (F (1, 58) = 996.296, p < 0.001, η² = 0.945] all showed significant main effects of time.
Furthermore, there was a significant group effect [F (1, 58) = 1.732, p < 0.001, η² = 0.029 for TC, F (1, 58) = 1.094, p < 0.001, η² = 0.019 for TGs, and F (1, 58) = 30.562, p < 0.001, η² = 0.345 for TC/HDL]. Additionally, TC, TGs, and TC/HDL showed a significant interaction effect between time and group [F (1, 58) = 86.454, p < 0.001, η² = 0.598; F (1, 58) = 255.485, p < 0.001, η² = 0.815; and F (1, 58) = 1008.887, p < 0.001, η² = 0.946, respectively].
In terms of HDL concentration, there was a significant increase in the experimental group versus a significant decrease in the control group (Table 2). Significant main effects for time [F (1, 58) = 614.938, p < 0.001, η² = 0.914) and group (F (1, 58) = 2.857, p < 0.001, η² = 0.047] were found according to mixed ANOVA analysis. Additionally, there was a significant interaction effect between time and group [F (1, 58) = 0.047, p < 0.001, η² = 0.921].
According to Table 3, the experimental group experienced statistically significant reductions in CEA and CA125 (p < 0.001 at 95% Cl:1.11, 1.23, and 95% Cl:5.04, 5.64, respectively). CEA and CA125, on the other hand, increased statistically significantly in the control group (p < 0.001 at 95%Cl −0.96, −0.87, and 95% Cl:-2.37, −2.13).
Change of Tumor Markers Before and After 12 Weeks, with or Without Intervention.

CA125: Cancer antigen 125; CEA: Carcinoembryonic antigen.
Data represented as mean ± standard deviation (SD).
F: Mixed repeated measures ANOVA. CI: confidence of interval; η2: Partial eta squared.
*: Statistically significant at P < 0.05.
Additionally, for CEA [F (1, 58) = 528.305, p < 0.001, η² = 0.901 and F (1, 58) = 246.737, p < 0.001, η² = 0.810] and CA125 [F (1, 58) = 3067.661, p < 0.001, η² = 0.981 and F (1, 58) = 4274.407, p < 0.001, η² = 0.987], significant main effects of time and group were found. It was observed that both CEA and CA125 had a significant interaction effect between time and group [F (1, 58) = 357.050, p < 0.001, η² = 0.860 and F (1, 58) = 3253.337, p < 0.001, η² = 0.982].
Discussion
This study aimed to assess the effects of a 12-week HIIT and mindfulness breathing intervention on postmenopausal obese women's metabolic and cancer-related risks (BMI, tumor markers, lipid profile, sex hormones, and HOMA-IR scores).
When comparing the experimental group to the control group, all evaluated outcomes revealed statistically significant improvements, including changes that were close to or higher than clinically significant different levels. This implies that the health risk profiles of obese postmenopausal women are positively affected.
The experimental group's BMI decreased significantly compared to the control group (mean reduction of 1.7 kg/m2, p < 0.001 for time impact, p < 0.001 for time x group interaction), exceeding the standard MCID (1 kg/m2). This suggests that there has been a clinically significant influence on body composition, which is linked to improvements in several of cardiometabolic risk factors. 25
Our findings are consistent with earlier studies26,27 showing how effective HIIT is at lowering body weight and fat mass in obese individuals, including postmenopausal women.
Taylor et al 26 state that HIIT is a safe and efficient way of quickly reducing body weight and total fat mass regardless of the population. Also, Dupuit et al 27 concluded that non-dieting overweight/obese postmenopausal women may benefit from HIIT or moderate-intensity continuous training (MICT) sessions to lose weight and fat mass, with HIIT showing more effective outcomes than MICT. Furthermore, in both pre and postmenopausal women, three months of HIIT decreased body weight (P < 0.01), waist circumference (P < 0.01), lean body mass (P < 0.001), and fat mass (P < 0.001), according to Mandrup et al 28
The results of this study corroborate those of Soleimaninanadegani et al, 29 who revealed that participants in mindfulness-based therapies, in addition to diet and exercise, experience suitable weight reduction results.
According to our perspective, HIIT-induced increases in metabolic rate and lipolysis, 30 as well as mindfulness-induced behavioral modifications (controlling obesity and excessive eating episodes), 31 are suggested mechanisms for weight loss.
The lipid profile showed significant improvements as well in the current study. There were statistically significant decreases in TC (36 mg/dL, p < 0.001), TGs (26 mg/dL, p < 0.001), and the TC/HDL ratio (p < 0.001) within the experimental group concurrent with significant increase in HDL level (3.7 mg/dL, p < 0.001). A decrease in cardiovascular risk is the main clinical objective; however, the generally accepted MCID values for specific lipid components are less standardized than for functional outcomes.
Reductions in TC and TGs of at least 20–30 mg/dL and an increase in HDL of at least 5 mg/dL are frequently suggested in the literature as beneficial clinical changes. Our findings show significant improvements in the lipid profile that are clinically significant. 32
These findings contrast with previous research by Sawyer et al, 33 Smith-Ryan et al, 34 and Batacan et al 35 that found no significant changes in lipid profiles following HIIT in obese people. Their conclusion, considering our findings, may consider differences in participant characteristics, study duration, and design.
Nonetheless, the HIIT program counteracts dyslipidemia reflected by TGs and TC levels reduction together with HDL level increase in dyslipidemic women, according to supportive results by Alvarez et al 36 and Ouerghi et al 37 (P < 0.01). To improve inflammatory, anthropometric, and lipid levels in physically inactive women, Zaer et al 38 observed that an eight-week HIIT might be an effective alternative for other time-consuming exercises. This conclusion was drawn from the nature of HIIT, which increased fat oxidation to counteract dyslipidemia. 37 Furthermore, Chen et al 39 demonstrated that mindfulness training altered metabolic and lipid indicators by upregulating the metabolism of glycerophospholipids.
The experimental group's HOMA-IR score in the current study decreased by 1.4 units, which is clinically significant (p < 0.001). A reduction that shifts individuals from an insulin-resistant state (>2.0) to normal sensitivity (<1.5) is generally regarded as clinically meaningful in lowering the risk of diabetes, even though the exact MCID for HOMA-IR is still up for debate. A clinically significant shift is exceeded by the observed 1.4-unit reduction, which places the average score considerably below the insulin resistance threshold. 40
The findings of this study are in line with those of Martins et al 41 (12 weeks of HIIT among high-risk postmenopausal women with type 2 diabetes mellitus) and de Matos et al 42 (eight weeks of HIIT among obese sedentary individuals), who noticed a significant decrease in HOMA-IR (P = 0.056 and p < 0.0001, respectively).
Other studies, like Arad et al, 43 showed no change in insulin sensitivity in obese African American women after 14 weeks of HIIT, which contrasts with our findings. This discrepancy could be explained by variations in the participants’ training regimens and physical characteristics.
The beneficial effects of HIIT on IR could be attributed to post-exercise adaptations that change other associated parameters, such as boosting the oxygen supply to adipose tissue through various vascular adaptations, without affecting changes in body fat. 33
Interventions based on mindfulness may help people with IR, by lessening depression and improving the capacity to successfully control attention, stress, mental health, and behaviors that are related to IR, such as emotional eating. 44
Additionally, compared to the control group, the experimental group produced a statistically significant improvement in sex hormone levels, lowering estradiol, TT, and FT while raising SHBG (p < 0.001). It's crucial to keep in minds that the literature does not yet provide standardized MCID values for sex hormones in relation to exercise interventions and disease risk reduction. However, because of their links to better metabolic profiles and a lower risk of breast cancer, decreases in circulating estrogen and testosterone and elevations in SHBG are typically regarded as physiologically and clinically significant.
Therefore, these statistically significant changes in our study reflect clinically relevant hormonal adaptations, which is consistent with the direction of effects observed in studies such as McTiernan et al 45 which examined the effects of a 12-month moderate-intensity exercise program on sex hormones in postmenopausal, overweight, and sedentary women. They found that women who lost 2% of their body fat had significantly lower levels of estrogen and testosterone.
On the other hand, a study by Monninkhof et al 46 on sedentary postmenopausal women that included aerobic and strength training programs revealed no significant reduction in estrogen and androgen levels. The disparities between the current study's results and the Monninkhof et al study may be explained by differences in the training protocols used.
Although there is no clear evidence, mindfulness-induced weight loss and stress reduction may have an indirect impact on hormone levels.
Additionally, the experimental group's effects on tumor markers were statistically significant (CA125 decreased from 11.2 ± 4.4 U/ml to 5.9 ± 3.4 U/ml, p < 0.001) and CEA decreased from 1.84 ± 0.61 ng/mL to 0.6 ± 0.06 ng/mL, p < 0.001). As with sex hormones, the literature does not establish standardized MCID values for tumor markers such as CEA and CA125 in relation to exercise treatments for cancer prevention. However, reductions in these indicators are usually considered favorably, indicating a beneficial biological impact that may eventually have therapeutic significance regarding cancer risk, confirming the broader claim that exercise helps prevent cancer according to Friedenreich 47 supportive study.
Esfahbodi et al 48 demonstrated only a reduction in CEA and CA125 levels without statistical significance after 8 weeks of aerobic exercise among breast cancer women and recommended further studies to assess the impact of exercise on tumor markers in obese postmenopausal women which our findings support. The differences in training protocols, evaluating methods, participant age, and the degree of obesity in each participant could all be responsible for these disparities in outcomes.
Mindfulness-based interventions has improved many psychological and physiological outcomes in patients with cancer, such as cortisol slopes, blood pressure, and length of telomere. 49
Importantly, there were no reported negative effects of the intervention, suggesting that this HIIT and mindfulness breathing program can be safely used with obese postmenopausal women, even if they already have cardiometabolic issues. The thorough enhancements seen in metabolic, hormonal, and tumor markers indicate that this integrated strategy has a great deal of potential to produce clinically significant health benefits and lower the risk of metabolic disorders and possibly some types of cancer in this susceptible group.
Limitations
While this study provides valuable evidence, several limitations should be considered. These include: 1) the difficulty in isolating the precise impact of mindfulness breathing alone, as existing research primarily examines its effects in combination with other interventions (eg, exercise, diet, medication); 2) the limited generalizability of the findings due to the specific characteristics of the study sample (exclusively postmenopausal women); 3) the potential for unmeasured confounding factors, such as dietary habits and stress levels; 4) the uncertainty regarding participant adherence to the intervention outside of the supervised study setting; 5) there is a paucity of data on long-term adherence and sustained results, which limits its practical application. Future studies should examine long-term effect and
Conclusions
This 12-week study demonstrated that regular participation in HIIT and mindfulness breathing significantly and clinically improved body mass, dyslipidemia, and sex hormone profiles, alongside reducing several cancer-related markers in sedentary, obese, postmenopausal women. These findings underscore the profound impact of combining HIIT and mindfulness breathing on physical health, extending well beyond simple weight loss. Future research should investigate the effects of this intervention on other tumor markers and explore its applicability across diverse age groups and genders.
Footnotes
Acknowledgments
With special thanks to Hassan M. Habib and Ebthall Mohamed for their assistance with data analysis and interpretation, we sincerely thank all study participants for agreeing to participate.
Ethical Approval
The study was carried out in accordance with the Declaration of Helsinki and approved by the Faculty of Physical Therapy Ethical Committee board (no: P.T.REC/012/002723), in addition to clinical trial.gov registration (NCT04471506), after adequate explanation of the study's details.
Authors’ Contributions
Ebtesam N. Nagy: Conceptualization, Methodology, Software. Marwa S. Saleh: Data curation, Writing- Original draft preparation. Hend A. Elsheimy: Visualization, Investigation. Hussein G. Mogahed: Supervision. Marwa M. Elsayed: Software, Validation, Writing- Reviewing and Editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Trial Registration
Trial registry name: Effect of Interval Aerobic Training and Mindfulness Breathing on Hormonal, Metabolic, and Tumor Markers in Postmenopausal. Trial registry URL: https://clinicaltrials.gov/study/NCT04471506. Trial registry number: clinical trial.gov registration (NCT04471506).
