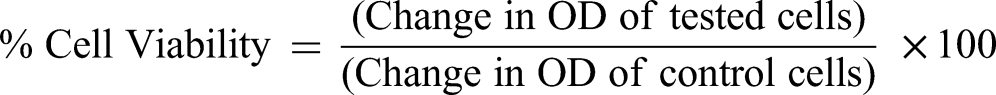Abstract
Diabetes mellitus, diagnosed as Madhumeho (sweet urine) in Thai traditional medicine, is believed to stem from imbalanced life elements. Ya That Opchoei mixture (YTO), a polyherbal mixture, is used to treat its symptoms. This study assessed the hypoglycemic potential of YTO and conducted qualitative and quantitative analyses of its bioactive constituents. YTO significantly inhibited α-glucosidase at IC50 0.05 ± 0.00 mg/mL and α-amylase activities at IC50 0.04±mg/mL. YTO at concentrations of 2.22 mg/mL significantly increased C-peptide secretion and stimulated glucose uptake. Liquid chromatography-mass spectrometry revealed constituents from Amomum testaceum, Cinnamomum bejolghota, C. burmanii, Syzygium aromaticum, and Glycyrrhiza glabra. High-performance liquid chromatography enabled quantification of bioactive compounds, including glycyrrhizic acid, eugenol, cinnamic acid, cinnamaldehyde, and coumarin. The coumarin content in 100 mL YTO was within the tolerable daily intake set by the European Food Safety Authority. This study confirms the hypoglycemic potential of YTO and presents its quality control process for safety assurance.
Introduction
Diabetes mellitus poses significant health and economic challenges. The worldwide occurrence of this disease among adults is predicted to reach 783.2 million or 12.2% by 2045 and costs associated with managing diabetes are expected to reach 1054 billion USD in the same year. 1 The primary characteristic of diabetes mellitus is hyperglycemia, which subsequently leads to macrovascular and microvascular complications. 2 In Thai traditional medicine, diabetes mellitus, diagnosed as Madhumeho (sweet urine), is attributed to imbalanced phlegm leading to bile dysfunction. Treatment involves careful elemental diagnosis based on the four elements (earth, water, wind, and fire), utilizing bitter, spicy, and hot-tasting medicines, tailored dietary adjustments, adequate hydration, sleep, and exercise. 3 Ya That Opchoei mixture (YTO) is formulated from a blend of spicy-hot tasting herbs such as the barks of Cinnamomum burmannii (Nees.) Blume. and C. bejolghota (Buch.-Ham.) Sweet, the young flowers of Syzygium aromaticum (L.) Merr. et Perry, a spicy-cool herb similar in taste to the Amomum testaceum Ridl. fruit, and the rhizome of the bitter-sweet tasting herb Glycyrrhiza glabra L. The YTO mixture is used in Thai community hospitals to treat symptoms associated with elemental imbalances. 4 Despite its widespread use, YTO faces two primary challenges: limited scientific validation of its claimed biological activity and batch-to-batch quality variability owing to inconsistencies in raw materials and preparation processes. Therefore, this study aimed to evaluate the hypoglycemic activity of YTO and to standardize its bioactive compounds to ensure safety through consistent quality.
Materials and Methods
Chemical Materials and Cells
Reagents and standards, including acarbose, acetonitrile, α-amylase, α-glucosidase, camphor, cinnamaldehyde, cinnamic acid, dexamethasone, dimethyl sulfoxide (DMSO), eugenol, formic acid, glucose, glutaMAX, insulin, 1-isobutyl-3- methylxanthine, methanol, menthol, p-nitrophenyl-α-D-glucopyranoside (PNPG), hydroxyethyl piperazine ethane sulfonic acid (HEPES), and resazurin sodium were bought from Sigma-Aldrich, USA. Glycyrrhizic acid monoammonium salt was obtained from Carlo Erba (Italy)
Herbal Materials Identification
The starting plant materials for YTO, including fruits of A. testaceum, dry barks of C. burmannii, dry barks of C. bejolghota, dry rhizomes of G. glabra, and dry buds of S. aromaticum were purchased from local traditional drugstores in Thailand (Figure 1). The plant species were authenticated using macroscopic and microscopic characters.

Morphological characteristics of the starting materials.
Quality Assessment of the Starting Herbal Ingredients
Before YTO formulation, limit tests were performed on its herbal ingredients. These assessments included an evaluation of foreign matters, total ash, acid-insoluble ash, and water contents. The extractive values for each plant were determined.5–9
Formulation and Quality Assessment of YTO
After quality assessment, the plant materials were cleaned, milled, and sieved through a No. 40 sieve. YTO was formulated from 24 g each of C. burmannii bark, clove bud, C. bejolghota bark, A. testaceum fruit, and G. glabra root, and then boiled in 3 l of distilled water at 80 ± 5 °C for 15 min. The mixture was filtered to obtain a red-brown solution. The flavoring agents, camphor and menthol, were added when the solution cooled to 50 °C. Before further experimentation, YTO was tested to meet the physicochemical requirements (pH, viscosity, and refractive index). Screening for microbial contamination and pesticide residues was also performed. 10 YTO was freeze-dried to yield a homogeneous powder, which was preserved at 4 °C until further experiment.
Hypoglycemic Activity Assays
Evaluation of α-Glucosidase-Inhibitory Activity
YTO extract was examined for α-glucosidase inhibition activity with some modification from the method described by Bhatia. 11 Briefly, α-glucosidase (0.2 U/mL, 50 µL) was mixed with YTO at varying concentrations (0.01-2 mg/mL). The reactant was kept at 37 °C for 5 min. Next, 1 mM PNPG (50 µL) and 50 µL of 50 mM phosphate buffer (pH 6.9) were added. The reaction was then developed at 37 °C for 10 min. The concentration of free p-nitrophenol was determined by a microplate reader at 405 nm. Experiments were performed in triplicate.
Evaluation of for a-Amylase-Inhibitory Activity
YTO extract was tested for α-amylase-inhibitory activity using a method adapted from Luyen et al 12 The combination of YTO at varying concentrations (0.01-2 mg/mL) along with 50 μL potato starch in phosphate buffer solution (pH 7.0). The reaction was then incubated at 37 °C for 5 min. Next, 20 μL of 5 μg/mL α-amylase enzyme was added and incubated at 37 °C for 15 min. To terminate the reaction, 50 μL of 1 N hydrochloric acid was added. Iodine solution was added to produce the color. The mixture was then measured by a microplate reader at 650 nm. Experiments were conducted with three independent replicates.
Evaluation of % Cell Viability
To assess cell viability before investigating C-peptide secretion and glucose uptake, the cytotoxicity of the extract of YTO was assessed in 3T3-J2 cells. Briefly, 3T3-J2 cells were cultured at 5 × 10³ cells/well in a 96-well plate containing DMEM in an incubator (5% CO₂), 37 °C, 24 h. The cells were then incubated with YTO at varying concentrations for an additional 24 h. Subsequently, cells were rinsed 2 times with sterile PBS, treated with 0.15 mg/mL resazurin solution (20 µL) along with 100 µL of fresh medium, and kept in a 5% CO₂ incubator, 37 °C, 2 h. The viability of cultured cells was then assessed by measuring the fluorescence intensity of the resazurin reduction product at 560 nm excitation and 600 nm emission wavelengths. Three replicate experiments were performed.
13
Based on the % cell viability, the IC₅₀ value (2.22 mg/mL) and its tenfold dilution (0.22 mg/mL) were chosen as the concentrations for determining C-peptide secretion and for the glucose uptake experiments. RIN-m5F cells were used for evaluating C-peptide secretion. Briefly, the RIN-m5F cells were cultured at 5 × 10⁴ cells/well in 48-well plates containing RPMI 1640 medium in an incubator (5% CO2, 37 °C, 24 h). The cells were then incubated with YTO at concentrations of 0.22 mg/mL and 2.22 mg/mL for 24 h. A vehicle was used as the negative control. After treatment, the cells were collected in centrifuge tubes, washed 2 times with sterile Krebs-Ringer bicarbonate-HEPES buffer (KRBH), and then suspended in 100 µL aliquots of either glucose-free KRBH, KRBH supplemented with 5.5 mM glucose, or KRBH containing 22 mM glucose. Each reaction was kept in an incubator (5% CO₂), 37 °C, 30 min. After 30 min, the supernatants were collected for C-peptide measurement using an ELISA kit. Total cellular DNA was extracted from the remaining cell pellets and quantified using a Qubit assay. C-peptide secretion data were adjusted based on the total DNA content. Three independent experiments were performed. 14
Evaluation of Glucose Uptake Activity of 3T3-L1 Adipose Cells
YTO extract was evaluated for its cellular glucose uptake inducing potential using a 2-NBDG florescence probe. For cell culture, 3T3-L1 adipose cells were differentiated in both transparent and black 96-well plates and maintained in an incubator (5% CO₂) at 37 °C. The test groups were pre-incubated with YTO (0.22 and 2.22 mg/mL, concentrations) for 24 h. The positive control group received a 10 min treatment with 50 nM insulin before the uptake study. All groups were then incubated with 100 μg/mL 2-NBDG (80 µL) for 25 min at 37 °C in a 5% CO₂ incubator. Then, cells were cleaned with cold buffer (200 μL, 2-3 times). A confocal microscope with 485 nm and 535 nm was used to determine the cellular uptake of 2-NBDG. Additionally, a microplate reader was used to measure the fluorescence intensity of 2-NBDG at the same excitation and emission wavelengths. The rate of glucose uptake was calculated as previously described:
15
Chemical Compound Identification Using LC-MS
The chemical constituents of YTO were analyzed using a Thermo Ultimate 3000 HPLC combined with a Bruker Amazon SL mass spectrometer. YTO (1 mg/mL) was separated on an ACE C-18 column (5 μm, 4.6 × 250 mm I.D.) with a mobile phase gradient system. The mobile phase was composed of solvent A (0.1% aqueous acetic acid) and solvent B (1% acetonitrile). The gradient eluted from 100% A to 0% A in 60 min. The flow rate was maintained at 0.7 mL/min, 35 °C. The wavelength of the UV detector was set at 270 nm. The EIMS instrument employed a quadrupole ion trap. The MS condition included a capillary voltage of 4500 V, the pressure of nebulizer gas was 2 bar, and the drying gas temperature was 220 °C with a flow rate of 7.0 l/min. The mass spectrometer was conducted in a positive ion mode with full-scan detection ranging from 70 to 1000 m/z. Characterization of phytochemical compounds in YTO was based on a combination of UV spectra, retention times, and MS fragmentation patterns.
Quantitative Analysis of Bioactive Markers Using HPLC
The experiment was performed following the ICH Q2 (R1) guidelines. 16 Freeze-dried YTO powder (50 mg) was homogenously mixed with methanol 10 mL and sonicated in an ultrasonicator at 40 °C for 30 min. Standard stock solutions (50 mL) of coumarin (100 μg/mL), cinnamic acid (50 μg/mL), glycyrrhizic acid (500 μg/mL), cinnamaldehyde (50 μg/mL), and eugenol (500 μg/mL) were prepared in methanol. Working solutions were then prepared by diluting the standard solutions to generate calibration curves with at least five different concentrations of each compound. All prepared solutions were stored at 4 °C until further experiments.
Data Analysis
IC50 values were determined by GraphPad Prism software (v.6.0). The three independent experimental data were reported as mean ± standard error of the mean. One-way analysis of variance (ANOVA) followed by the Tukey's post-hoc test or the Games-Howell post-hoc test were used to evaluate statistically significant differences (SPSS software, version 2020). A P-value ≤.05 was considered statistically significant.
Results
Quality Assessment of the Staring Herbal Materials and YTO
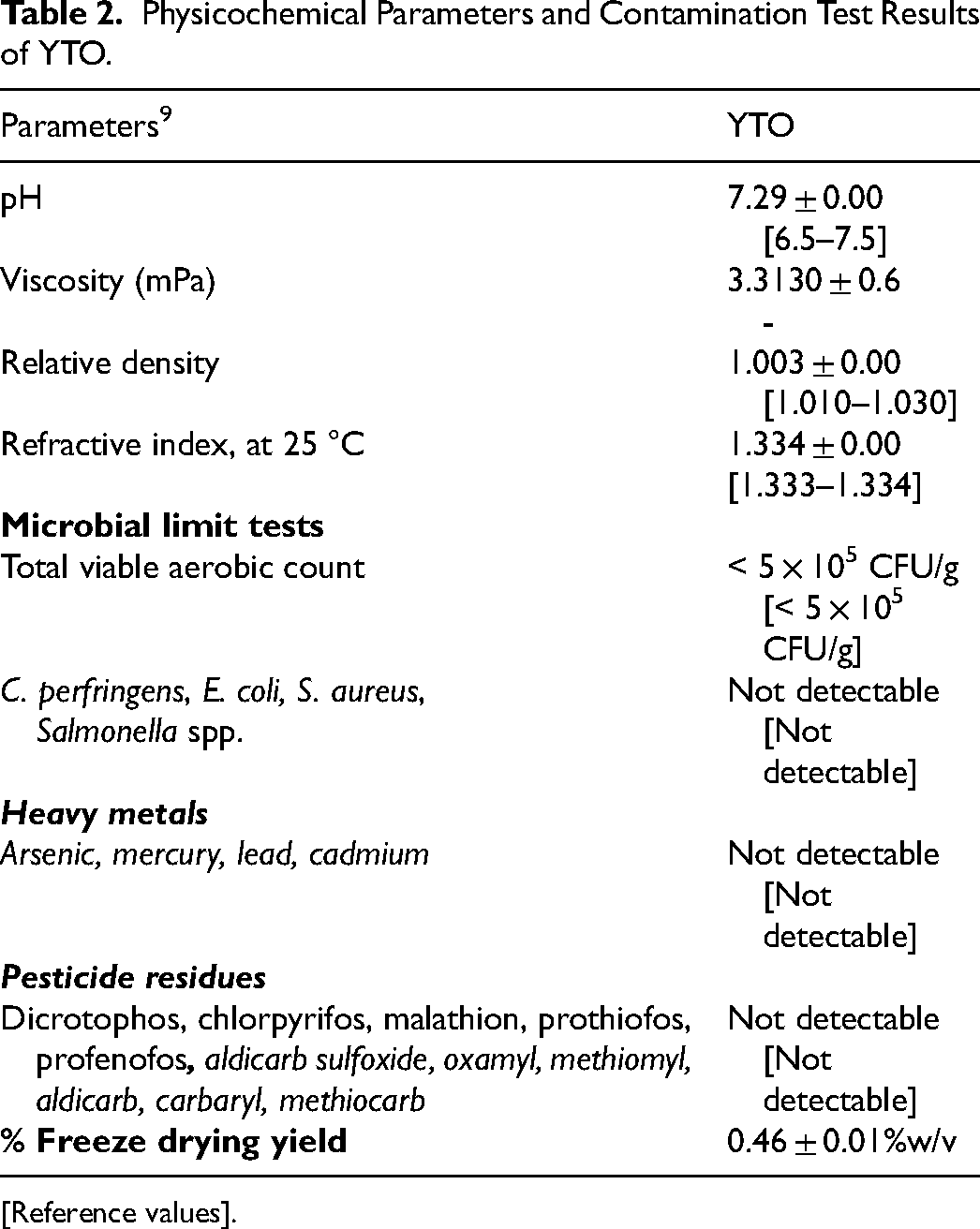
The physical appearance of the material is shown in Figure 1. The microscopic characteristics of the raw materials are presented in Supplemental Data 1. Five herbal materials were analyzed for their limit tests and physicochemical properties. The water content ranged from 4.86–11.40%, and foreign matter contamination ranged from 0.01–0.43%. The materials showed ethanol extractive values of 3.01–31.80% and water-soluble extractive values of 9.90–33.87% (Table 1). The physicochemical parameters of the YTO mixture were determined. The pH of YTO was 7.29, the viscosity was 3.3130 mPa, and the refractive index was 1.0030. The microbial tests exhibited the absence of C. perfingens, E. coli, S. aureus, and Salmonella spp. No organophosphate and carbamate residues were found in the pesticide analysis of YTO (Table 2).
Limit Tests and Physicochemical Properties of the Herbal Materials Used.

[Reference Values].
Physicochemical Parameters and Contamination Test Results of YTO.
[Reference values].
Inhibition of α-Amylase and α-Glucosidase by YTO
At concentrations of 2, 1, 0.5, 0.25, 0.13, 0.06, and 0.03 mg/mL, YTO exhibited α-amylase activity by 83.77 ± 0.81%, 75.85 ± 1.04%, 71.70 ± 1.18%, 66.41 ± 3.37%, 62.61 ± 1.02%, 57.67 ± 4.50%, and 46.10 ± 0.52%, respectively (Figure 2). YTO exhibited a lower IC50 (0.05 ± 0.00 mg/mL) compared to that of the positive control acarbose (0.01 ± 0.00 mg/mL). Similarly, the extracts at concentrations of 2, 1, 0.5, 0.25, 0.13, 0.06, and 0.03 mg/mL inhibited α-glucosidase activity by 97.61 ± 1.38%, 97.30 ± 0.85%, 97.23 ± 0.45%, 97.13 ± 0.82%, 91.97 ± 1.02%, 59.50 ± 6.79%, and 33.39 ± 0.84%, respectively (Figure 3). The IC50 for α-glucosidase inhibition was 0.04 ± 0.00 mg/mL, which was higher than that of acarbose (0.24 ± 0.01 mg/mL).
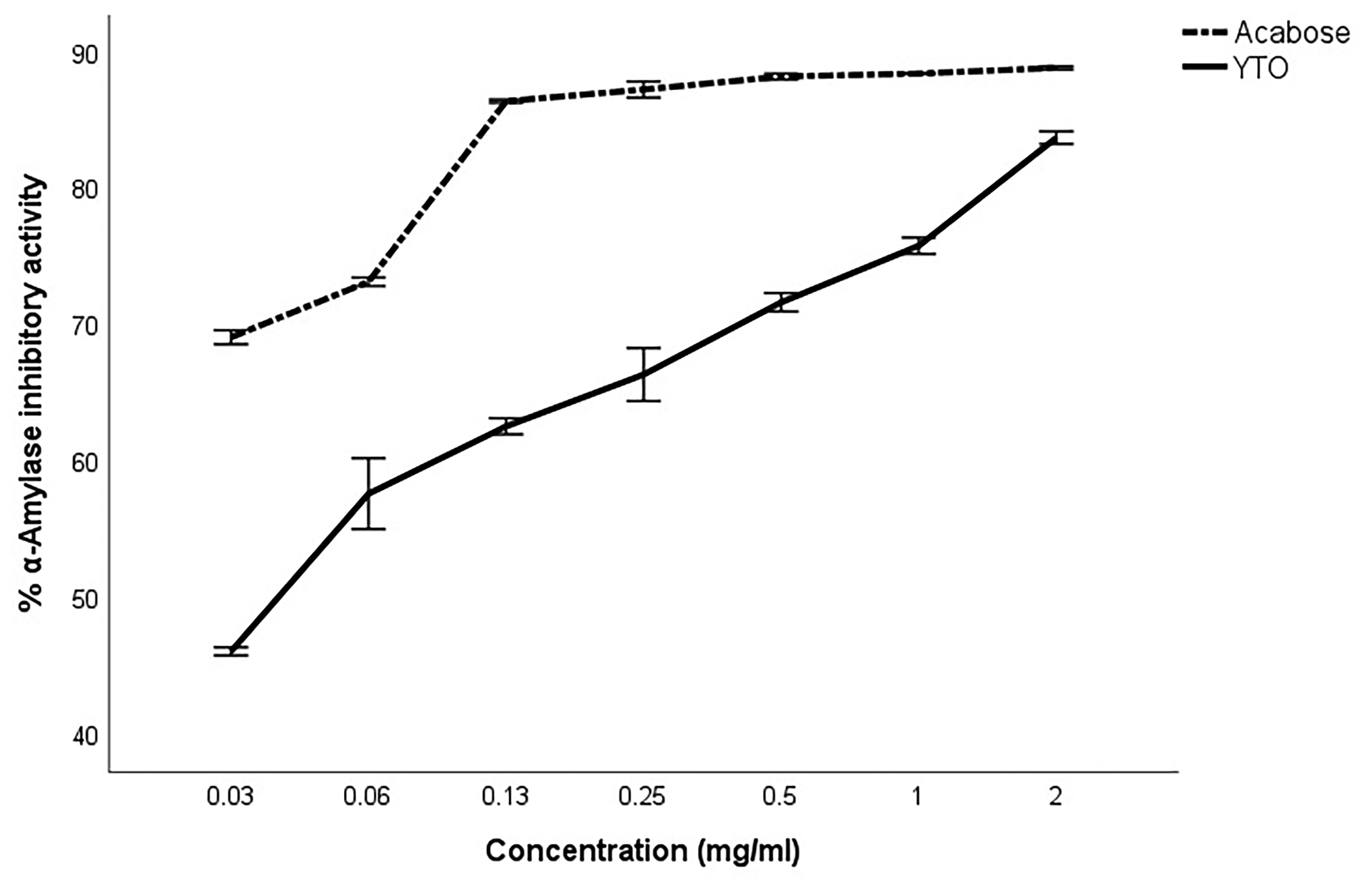
% α-Amylase inhibitory activity of YTO.

% α-Glucosidase inhibitory activity of YTO.
% Cell Viability of YTO
Treatment with YTO at concentrations of 0.00025–2.5 mg/mL displayed a dose-dependent effect on cell viability (Table 3). Only the highest concentration, 2.5 mg/mL, showed a significant difference in viability from that in the control. The IC50 of YTO was 2.22 mg/mL.
% Cell Availability of Control and YTO.

* Significantly different compared to the control at P ≤ .05.
C-Peptide Secretion Activity of YTO
This study evaluated the antidiabetic potential of YTO by determining its effects on C-peptide secretion from insulin-secreting RIN-m5F cells. C-peptide, a co-secreted product of insulin, is an indirect marker of insulin release. Thus, C-peptide levels can be used to indirectly assess pancreatic β-cell function and insulin secretion ability. YTO at concentrations of 0.22 and 2.22 mg/mL significantly increased C-peptide secretion from RIN-m5F cells compared to that from the control group (P < .05) in the presence of various glucose concentrations. However, these two concentrations did not differ significantly in their ability to stimulate C-peptide release (Figure 4).

Effect of YTO on stimulating insulin release from RIN m5F cell.
Glucose Uptake Ability of YTO
A glucose uptake assay with 2-NBDG was used to investigate the capability of YTO to improve insulin sensitivity by assessing its activity on glucose transport into 3T3-L1 cells (Figure 5). While YTO induced 2-NBDG uptake in 3T3-L1 cells at both concentrations of 0.22 and 2.22 mg/mL compared to that in the control group, only the higher concentration exhibited a significant difference compared to that of insulin (Figure 6). This suggests that YTO may possess insulin-sensitizing properties, particularly at the higher concentration, by enhancing glucose transport into adipocytes.

Effects of YTO in stimulating 2-NBDG uptake by 3LT3 adipocytes.

% 2-NBDG uptake in 3LT3 adipocytes induced by YTO.
Identification of Phytochemical Compounds by LC-MS
The LC-MS chromatogram (Figure 7) showed various compounds in YTO. The constituents were characterized by comparing their mass fragmentation profiles and retention times with those described in previous publications and available online databases, including SDBS and PubChem (Table 4).

Total ion chromatogram based on the MS2 chromatogram of YTO.
Identification of the Compounds in YTO.

Quantitative Analysis of Bioactive Markers Using HPLC
Bioactive markers of YTO were analyzed using a validated HPLC method. The HPLC chromatogram revealed peaks corresponding to coumarin, cinnamic acid, glycyrrhizic acid, cinnamaldehyde, and eugenol (Figure 8). Among the compounds analyzed, glycyrrhizic acid exhibited the highest yield, whereas cinnamaldehyde showed the lowest yield (Table 5).
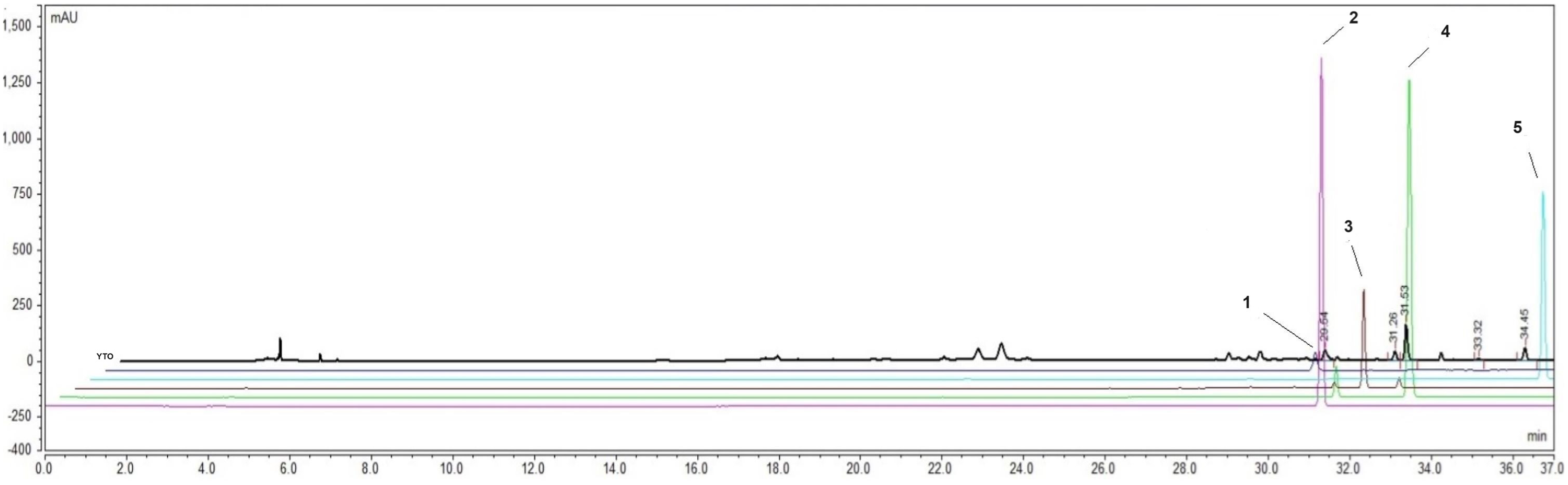
HPLC chromatogram of YTO compared to the standard peaks.
Linearity, Range, LOD, LOQ, and Contents of the Bioactive Markers in YTO.

Abbreviations: LOD, Limit of detection; LOQ, Limit of quantitation.
Discussion
α-Amylase in saliva and the pancreas, along with α-glucosidase in the small intestine, hydrolyze carbohydrates into glucose, impacting blood sugar levels. 24 Based on the results, YTO significantly inhibited α-amylase and α-glucosidase activities, which could be associated with its herbal composition. A study by Ervina (2019) showed that an aqueous extract of C. burmannii exhibited α-glucosidase inhibitory and antioxidant activities. 25 Another study reported that C. burmannii and C. bejolghota extract effectively inhibited the activities of both pancreatic α-amylase and intestinal α-glucosidase. 26 G. glabra extract was found to inhibit both enzymes. 27 Compounds from S. aromaticum, including eugenol, maslinic acid, and oleanolic acid, were found to decrease the activities of carbohydrate-digestion enzymes in diabetic rats.28,29 Besides inhibiting carbohydrate-digestion enzymes, YTO exhibited the abilities of stimulating insulin release and enhancing cellular glucose uptake. Considering the herbal ingredients of YTO, C. burmannii extract has been shown to modulate insulin signaling, glucose transport, and adiponectin secretion.30,31 C. burmanii bark, similar to that of other Cinnamomum species, contains cinnamic acid, cinnamaldehyde, and coumarin. Cinnamic acid protects against diabetes-induced DNA damage, regulates the expression of glucose uptake transporters in L6 myotubes, and induces insulin release.32–34 Cinnamaldehyde exhibits both hypoglycemic and hypolipidemic effects in vivo and shows α-amylase inhibitory activity.35–37 Coumarin possesses antidiabetic properties by stimulating insulin secretion, lowering blood glucose, and inhibiting protein glycation. 38 Additionally, a methanolic extract of C. burmanni containing 0.07% w/w of coumarin demonstrated no significant acute or subchronic toxicity in vivo (500, 1000, and 2000 mg/kg, doses). 39 Type A proanthocyanidins from C. burmannii, C. cassia, and C. verum, have been shown to exhibit insulin-like effects. 40 The extract of C. bejolghota bark was reported to improve glucose tolerance, decrease blood sugar levels in rats, enhance antioxidation enzymes, and reduce the malondialdehyde content in streptozotocin-induced diabetic animals. 41 G. glabra extract and its isolated isoflavones, genistein, daidzein, and formononetin, displayed various mechanisms of hypoglycemic action, including PPAR-γ receptor binding, which led to decreased blood sugar levels, stimulated insulin secretion, and elevated glucose tolerance.19,42,43 Moreover, glycyrrhizic acid possessed the ability to protect cells from diabetic complications. 44 S. aromaticum powder was shown to lower blood glucose levels in diabetic rats. 45 Its isolated compounds, dehydrodieugenol, dehydrodieugenol B, and oleanolic acid, showed hypoglycemic activity in KK-Ay type 2 diabetic mice. 46 Ellagic and gallic acids were also detected in this plant. 17 Ellagic acid has been reported to reduce fasting blood glucose levels, stimulate insulin release, modulate RS1 expression, regulate GLUT4 gene expression, suppress lipid accumulation, and decrease inflammation in the adipose tissue. 47 Gallic acid showed antidiabetic activity in Db/db mice by upregulating pAkt, PPAR-γ, and Glut4. 48 A meta-analysis further highlighted the benefits of eugenol for antihyperglycemic activity, including increased glucose uptake and prevention of late diabetic complications. 49 These findings imply that the synergistic effects of plant constituents in YTO may contribute to its antihyperglycemic effect through various mechanisms.
Quality assurance of YTO was performed according to the WHO guidelines.6,7 The starting materials were authenticated by their morphological and microscopic characteristics. Sclereids of different sizes and shapes were found in the fruits of A. testaceum, the barks of C. bejolghota, and C. burmanni. Pollen grains were used to characterize the S. aromaticum flowers. The rhizome of G. glabra contained vessels and fibers (Supplementary Data 1). The limit tests of the raw materials did not exceed the previously reported limits.4–8 The physicochemical and contamination test results of YTO met the requirements of the Thai herbal preparation pharmacopoeia standards. 9
LC-MS analysis of YTO revealed 21 compounds.17,18,20–23 The result indicated that YTO contained compounds in the following order: G. glabra > C. burmannii > S. aromaticum > C. bejolghota = A. testaceum. Hydrophilic compounds, such as proanthocyanidins, phenolic acids, triterpenoid glycosides, and flavonoid glycosides, were preferentially extracted over lipophilic compounds because of the preparation method for YTO (decoction). Five easily detected and quantified compounds with hypoglycemic activity were chosen as the bioactive markers of YTO. The quantification result demonstrated that glycyrrhizic acid was the most abundant compound (6.80 ± 0.15% w/w), followed by eugenol, coumarin, cinnamic acid, and cinnamaldehyde (Table 4). Although coumarins show hypoglycemic activity, their potential hepatotoxicity requires careful consideration. A tolerable daily intake (TDI) of 0.1 mg/kg bodyweight for coumarin has been recommended by the European Food Safety Authority (EFSA).50,51 The coumarin content in YTO was 0.052% w/v (based on the freeze-dried weight of 0.46 ± 0.1 g/100 mL). Therefore, the consumption of 100 mL YTO/day by adults remains within the TDI established by the EFSA.
Conclusion
In the present study, YTO was prepared using standardized raw materials, and the mixture was subsequently evaluated for its hypoglycemic activity and quality. The hypoglycemic results indicated that this mixture inhibited the activity of α-amylase and α-glucosidase. YTO induced insulin release and increased glucose uptake in cell-based diabetes mellitus assays. LC-MS analysis demonstrated that YTO contained flavonoids, phenolic compounds, lignans, proanthocyanidins, and terpenoids. The bioactive markers glycyrrhizic acid, eugenol, coumarin, cinnamic acid, and cinnamaldehyde were analyzed using HPLC. The coumarin content in 100 mL of YTO complied with the TDI set by the EFSA for use in adults. These findings indicated the therapeutic potential of YTO as a safe and effective herbal medicine for managing hyperglycemia, a critical factor in diabetes mellitus and its associated complications. Nonetheless, this study was based on cell culture analyses, limiting its applicability to humans. Future work should include in vivo studies to verify the efficacy and safety of YTO. If positive results are obtained, clinical trials can be used to assess its effectiveness and adverse effects in human patients. Altogether, this research provides a significant foundation for further studies on YTO use in diabetes mellitus management.
Supplemental Material
sj-docx-1-chp-10.1177_2515690X251324810 - Supplemental material for Evaluating the Hypoglycemic Efficacy and Quality Assurance of Ya That Opchoei Mixture
Supplemental material, sj-docx-1-chp-10.1177_2515690X251324810 for Evaluating the Hypoglycemic Efficacy and Quality Assurance of Ya That Opchoei Mixture by Thaweesak Juengwatanatrakul, Kusuma Jitsaeng and Rawiwun Kaewamatawong in Journal of Evidence-Based Integrative Medicine
Footnotes
Authors’ Contributions
Conceptualization: Thaweesak Juengwatanatrakul (TJ), Kusuma Jitsaeng (KJ), Rawiwun Kaewamatawong (RK). Investigation: TJ, KJ, RK. Formal analysis: KJ, RK. Project administration: TJ. Writing–original draft: TJ, KJ, RK. Writing–review and editing: RK.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Ubon Ratchathani University [grant number N/A].
Data Availability Statement
Voucher specimens with reference numbers TOB-UBU-1–10 are deposited at the Faculty of Pharmaceutical Sciences, Ubon Ratchathani University. All data generated in the current research work have been included in the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.