Abstract
Background
Colorectal Cancer (CRC) is widely prevalent worldwide and its incidence is increasing. Chemotherapy is an important treatment method for colorectal cancer in addition to surgery, but it often causes physical and mental pain to patients due to its side effects. TCM emphasizes evidence-based treatment and a holistic concept, and the combination of TCM and chemotherapy can reduce chemotherapy side effects, improve chemotherapy efficacy, and enhance patients’ immunity. Yiqi Jianpi Kangai Decoction (YQJP) has been used clinically to treat patients with advanced colorectal cancer and may improve their immune function and prognosis. However, its mechanism has not been elucidated.
Objective
The study aims to explore the effect and mechanism of YQJP on enhancing the therapeutic effect of chemotherapy on spleen qi deficiency type CRC mice.
Methods
We used HPLC/MS to characterize the active substance components in YQJP. We established the spleen qi deficiency model induced by using the compound methods of diarrhea of bitter and cold, starvation, and full and excessive labor. and then inoculated CT-26 mouse colon cancer cells subcutaneously to establish the CRC Mice. We also evaluated the efficacy and mechanism of YQJP by using oral Yiqi Jianpi Kangai Decoction combined with an intraperitoneal injection of chemotherapeutic drugs (FOLFOX regimen). We evaluated the efficacy and safety of YQJP by observing the general condition (body weight, tumor size, food intake, hair condition, stool condition), HE staining, blood routine, and organ index of the mice. The expression of CD8+, CD4+ T cells, Th17 cells, and Treg cells in the tumor and spleen were detected by flow cytometry, and the levels of IL-17, IL-10, IFN-γ and TGF-β in the tumor were detected by ELISA; the expression of Ki-67, PCNA, RORγt and FOXP3 proteins in the tumor was detected by immunohistochemistry.
Results
YQJP contains 7 herbal compounds, which can effectively improve body weight, spleen condition, and bone marrow suppression in tumor-bearing mice inhibit tumor growth, and do not damage tissues and organs, which initially confirmed the anti-cancer effect and safety of YQJP. Further experiments showed that YQJP could elevate the proportion of CD8+, CD4+ T cells in the spleen, increase the proportion of Th17 cells in the tumor tissue of mice, and decrease the level of Treg cells. It can inhibit the expression of Ki-67 and PCNA. Meanwhile, it promotes the expression of IL-17 and IFN-γ and inhibits the expression of IL-10 and TGF-β. In addition, it can reduce the relative expression of FOXP3 and increase the relative expression of RORγt.
Conclusion
The combination of YQJP with chemotherapy had the effect of tumor suppression and enhanced chemotherapeutic efficacy in the spleen qi deficiency CRC mice. The related mechanism may be related to inhibiting proliferation, promoting apoptosis of tumor cells, increasing Th17 cells in tumor tissues, and decreasing Treg cell expression to improve the tumor microenvironment.
Keywords
Introduction
Colorectal cancer (CRC) is the third most prevalent malignancy, it is also the second leading factor in cancer-related deaths. 1 An estimated 1.9 million cases of morbidity and 900 000 deaths are reported to be induced worldwide in 2020. The incidence rate is higher in developed countries and increasing in developing countries compared to the previous incidence rate. With the popularity of colonoscopy, the incidence of early CRC is also increasing. The increasing number of CRC cases poses a significant burden on global public health. 2
Currently, surgery is still the best choice for treating CRC. However, most patients are found at an advanced stage with no chance of surgery. Adjuvant therapy for CRC includes chemotherapy, radiation therapy, immunotherapy, targeted therapy, Chinese medicine, and other therapeutic methods. Chemotherapy is the most common and primary treatment outside of surgery.3,4 The FOLFOX chemotherapy regimen is recommended by multiple guidelines as first-line chemotherapy for CRC, 5 which can inhibit tumor proliferation, delay disease progression, and prolong the survival cycle to a certain extent. However, while it kills tumor cells, it also kills normal cells of the body, reduces immunity, and is prone to gastrointestinal reactions, bone marrow suppression, and other adverse effects. The disadvantages of drug resistance, strong toxic side effects, and high cost of treatment have limited the use of chemotherapy drugs.
Traditional Chinese Medicine (TCM) is a splendid treasure of Chinese traditional medicine, and it has proven to be effective in the adjuvant treatment of malignant tumors. The combination and integrated use of TCM in post-operative conditioning of CRC with radiotherapy, biologic targeting, and immunotherapy are one of the characteristics of the clinical treatment of colorectal cancer in China. 6 The treatment protocol of Chinese medicine combined with chemotherapy is unique in improving patients’ quality of life, improving their immune function, preventing CRC recurrence and further development, prolonging overall survival, and reducing patients’ physical and mental discomfort due to the disease.7,8 In TCM, spleen deficiency is considered to be one of the most common symptoms in CRC patients. 9 Therefore, the method of benefiting Qi and strengthening the spleen is used throughout the treatment of CRC in TCM. Studies have suggested that the combination of Xiang Sha Liu Jun Zi decoction (a TCM prescription that benefits Qi and strengthens the spleen)with chemotherapy in the treatment of colorectal cancer patients can enhance the effect of chemotherapy while reducing chemotherapy toxicity, and enhance the immune function and improve the nutritional status of postoperative chemotherapy patients with colorectal cancer.10,11 Since TCM treatment emphasizes a holistic view not directly treating partial tumors, we speculate that it works maybe by improving the overall physical status and functions of patients and regulating cellular immunity to stimulate the immune potential of patients, and regulation of body immunity may be a potential key treatment modality for colorectal cancer treatment.12,13
The tumor microenvironment (TME) includes the external environment in which the tumor tissue is located and the surrounding T lymphocytes, macrophages, NK cells, and mature dendritic cells. 14 Specific cellular immunity in the tumor immune microenvironment is a major factor in tumor immunity, cellular immunity is a key part of tumor immunity, and infiltration of immune cells into the tumor microenvironment is an important step in a series known as the cancer immune cycle. 15 In cellular immunity, T lymphocytes are the most effective mediators of the adaptive anti-tumor immune response. Studies have demonstrated that helper T cells in TME are closely associated with carcinogenesis. T lymphocytes are effector cells that play an important role in TME, secreting relevant cytokines with direct anti-tumor effects and triggering specific cellular immune responses. Among T lymphocytes CD8+ T cells can be activated into cytotoxic T cells (CTL), which can enhance anti-tumor effects. CD4+ helper T cells are key regulators of immunity and inflammation, accounting for about 60% of T cells, and most of them differentiate into helper T cells after activation, which has anti-viral and tumor-killing effects. 16 Regulatory T cells (Treg) and helper T cells 17 (Th17) are important components of CD4+ T cells, and dysregulation of Th17/Treg cell homeostasis is associated with the development and progression of cancer. In tumors Treg cells mainly play a role in suppressing the tumor immune response, thus promoting carcinogenesis, while Th17 cells have the opposite role in tumors. 17 Enforced expression of IL-17 in tumor cells inhibits tumor progression by enhancing immune function in mice resulting in antitumor immunity. The transcription factor for Th17 cells in humans is RORC (RAR-related orphan receptor C), and RORγt (RAR-related orphan-receptor gamma) is a characteristic transcription factor for Th17 cells in mice. 18
In recent years, relevant studies have shown that TCM has a clear impact on immune cells that regulate the tumor microenvironment. It was proved that Ganoderma triterpenes and Ganoderma polysaccharides could reduce the proportion of Treg cells in the tumor tissue of H22 tumor-bearing mice and inhibit their secretion of FOXP3, IL-10, and TGF-β1, thus promoting anti-tumor immunity. 19 In addition, the researchers found that treatment of Lewis lung cancer tumor-bearing mice with Yanghe Tang reduced the levels of TGF-β and CD4+FOXP3+, increased IL-2 and CD3+CD8+ in the immune microenvironment of mice, and inhibited the expansion of lung cancer tissue in mice. 20
YQJP is an improved Chinese herbal formula based on the classic formula Liujunzi Decoction. YQJP Combined chemotherapy can increase the curative effect of chemotherapy, but its molecular mechanism is not clear. 21 It is well known that CT-26 colon adenocarcinoma cells are often used in animal model studies of colorectal cancer.222324–25 In addition, spleen-deficient mice induced by a combination of three inducing factors, including limited feeding, fatigue, and diarrhea, were also widely accepted.262728–29 Therefore, in this study, we used the combination of YQJP and FOLFOX chemotherapy regimen to treat spleen qi deficiency CRC model mice, hoping to explore its efficacy and mechanism.
Materials and Methods
Materials
Cell Culture
The mouse colon cancer cells (CT-26) line was Purchased from the ATCC website (http://www.atcc.org). All cells were cultured in RPMI-1640 medium (Gibco, USA) containing 10% calf bovine serum (Gibco, USA), 100 U/mL penicillin, and 100 mg/mL streptomycin (Beyotime, Shanghai, China) at 37 °C with 5% CO2.
Animals
The animal use protocol listed below has been reviewed and approved by the ethical review committee of the Animal Experimentation Center of SOUTHWEST MEDICAL University (LACUC Issue No. swmu20220012), and the whole process followed the “3R” principle. Adult male BABL/c mice (6-8 weeks old) weighing 22–24 g were obtained from the Chengdu Dashuo Experimental Animal Co.
Chemotherapeutic Drugs
Calcium folate and Oxaliplatin were purchased from Jiangsu Hengrui Pharmaceutical Co. (Jiangsu, China). And the 5-fluorouracil was purchased from Shanghai Xudong Haipu Pharmaceutical Co. (Shanghai, China).
Chinese Medicine Granules Preparation
The YQJP formula granules is a combination of eight medicinal herbs: Pseudostelariae Radix (30 g, NO.21050116), Poria (15 g, NO.21100369), Atractylodes macrocephalae Rhizoma (12 g, NO.21100342), Pinelliae Rhizoma Praeparatum (12 g, NO.21100305), Citri Reticulatae Pericarpium (15 g, NO.21100120), Herba Hedyotis (15 g, NO.21100056), Cremastrae Pseudobulbus Pleiones Pseudobulbus (15 g, NO.21090024), Scutellariae Barbatae Herba (15 g, NO.21090168), Glycyrrhiza Radix et Rhizoma (10 g, NO.21100285). And Senna granules consist of senna leaves (100 g). The nine herbs were purchased from Chinese Medicine Pharmacy, Affiliated Hospital of Traditional Chinese Medicine, SOUTHWEST MEDICAL University (Luzhou, China), and were identified by two experienced pharmacists.
The YQJP formula granules and Senna granules were provided by the Sichuan Neo-Green Pharmaceutical Technology Development Co., Ltd (Sichuan, China). The production procedures of YQJP critically followed the standards of Good Agricultural Practices (GAP) and Good Manufactory Practices (GMP) and Chinese Pharmacopoeia. Briefly, crude herbal materials were fragmentised and then decocted in batches. The extract was filtered, concentrated, spay-dried, granulated, and packaged after extraction.
Both of these granules, after mixing and blending, with distilled water and left to stand, were stored in a −20°C refrigerator for backup.
Methods
Component Analysis of YQJP Powder with HPLC-MS
Standard Scutellarin, hesperidin, glycyrrhizic acid, Militarine, and Heterophyllin B were purchased from the National Institute for the Control of Pharmaceutical and Biological Products (Beijing, China). YQJP conjugates were determined using a Q-Exactive Plus Orbitrap high-resolution mass spectrometer coupled to a Vanquish HPLC (Thermo Fisher, USA). A gradient of 0.1% formic acid in water (A) and 0.1% formic acid in acetonitrile (B) was used. The optimal conditions for mass spectrometry were as follows: the analytes were separated using Hypersil GOLD™ (2.0 mm × 100 mm, 1.8 μm), the column temperature was set to 35 °C, the injection volume was 5 μl, the flow rate was 0.2 ml/min, and the mobile phases were 0.1% formic acid in acetonitrile (A) and 0.1% formic acid in water (B). Gradient elution was as follows: 0 min–5 min, 2% A; 5 min–8 min, 20% A; 8 min–36 min, 26% A; 36 min–65 min, 100% A.
The high-resolution mass spectrometry conditions of Q-Exactive Plus Orbitrap were as follows: HESI sources were used as ion sources, spray voltage: 3.2 kV (positive mode) and 3.0 kV (negative mode), sheath gas volume flow rate was 35 μl/min, and ion transfer tube temperature was 320 °C. The auxiliary gas flow was 10 μl/min, the auxiliary temperature was 350 °C. The scanning mode of Full MS/DD-MS2 mode was selected, and positive (scan range: 100-1500 m/ z) and negative (scan range: 100-1500 m/z) ions were collected simultaneously. The specific ion scanning mode was “OFF,” the spray gas was nitrogen, and the collision gas was high. Chemical composition data analysis: Compound Discoverer and Thermo Xcalibur Qual Browser were used for chemical composition data analysis.
Induction of Spleen qi Deficiency Model Mice
Eighteen mice were randomly selected as the Normal control group (Normal). According to the literature, the remaining mice have established animal models of spleen qi deficiency, modeling using multiple factors: diarrhea of bitter and cold, starvation, and full and excessive labor.27–32 Firstly, spleen qi deficiency groups were developed by daily gavage of senna solution to mice. Besides, mice swam daily to exhaustion for 21 days. At the same time, the mice were fed with half the amount of diet-restricted daily (standard rodent chow), while water was available ad libitum. The whole experiment process lasted 30 days. We measured and recorded the weight of mice before the experiment started, and measured the weight and growth status of mice every 3 days after the experiment started. The modeling was considered successful when the mice showed emaciation (weight loss), reduced feeding, withered and lusterless hair, huddled and lazy, and filthy anus.
Analysis of D-xylulose and Serum Amylase
On the 21st and 30th day after the last administration of the drug to the mice, respectively, the mice in each group were fasted for 12 h, given 10% D-xylose solution by gavage at 4 ml/pupil, and then blood was collected from the eyeballs after waiting for 1 h. After anaesthesia, the blood was anesthetized with 10% chloral hydrate and then killed, and the blood was centrifuged at 1500 rpm for 5 min after standing for one hour at 4°C, and then the blood serum was extracted. Serum D-xylose was detected by the resorcinol method according to the instructions of the mouse serum D-xylose test kit (Jiancheng, Nanjing, China); serum amylase was detected by the amylose-iodine colourimetric method according to the instructions of the mouse α-amylase test kit (Jiancheng, Nanjing, China).
Induction of Subcutaneous Tumor Model for Colon Cancer in Mice
Mice with a successfully induced spleen qi deficiency model (Model) were selected, and the mice were anesthetized and disinfected after removing the hair on the right side of the buttocks, and then the cell suspension of mouse CT-26 colon cancer cell line in logarithmic growth cycle was injected into the mice subcutaneously at a concentration of 2 × 106/0.2 ml. when a small skin mound was formed, and the growth of the tumor was observed after 5 days, and the modeling was considered successful when the volume of the subcutaneous tumor grew to more than 50 mm3.
Subsequently, the tumor-bearing mice were numbered according to their body weights and randomly divided equally into 4 groups. After the grouping, the body weight, tumor volume, and food intake were measured every other day. The tumor growth was measured by tumor diameters with a Vernier caliper and drawn tumor growth curves. The tumor's shortest diameter (d) and longest diameter (D) were used to calculate the tumor volume. Tumor volume (TV) = d2 × D × 1/2. Tumor suppression rate formula = [(control average tumor weight-average tumor weight of drug group)/average tumor weight of control group] × 100%. 33 At the end of the experiment, the mice were anesthetized and executed, and the tumors were removed and weighed.
Animal Treatment
The daily dose of herbal medicine given was 28.5645 g/kg. After grouping, the YQJP group and the YQJP + FOLFOX group used the herbal medicine solution for gavage once a day for 2 weeks. Chemotherapy regimens refer to previous reports, 34 in the FOLFOX group and YQJP + FOLFOX group were used 5-fluorouracil (15 mg/kg) and Calcium folinate (5 mg/kg) were by intraperitoneal injection for 5 days. On the first day, one hour after administration of 5-fluorouracil and Calcium folinate, Oxaliplatin (5 mg/kg) was then administered intraperitoneally. The mode of administration and drug dosage for each group are shown in Table S2.
Then, after 5 days of chemotherapy, the mice were allowed to rest for 2 days, which was one cycle. The chemotherapy was then repeated one more time according to this protocol. On the first day after drug administration, we collected information on the general condition of the mice, and on the second day, we concentrated on anesthetizing and executing the animals used, collecting blood from the mice, stripping the tumors, and collecting the heart, liver, spleen, kidney, and lungs. The flow of the whole experiment is shown in Figure 1.

The flow of the whole experiment.
Animal Specimen Collection and Testing
During the experiment, the same person was appointed to conduct the measurement of the body weight and the size of the tumor of the mice. After drug administration of 12 h, 0.2 ml–0.3 ml of blood was collected from the mice for routine blood tests, and care was taken not to contact foreign objects such as mouse hair during the collection process to reduce the possibility of hemolysis. Then the tumor was stripped and weighed. The heart, liver, kidney, and spleen of mice were collected and weighed, and the organ indices of each organ were calculated; a portion of the tumor, heart, liver, kidney, lung, and spleen was taken and put into 4% paraformaldehyde tissue fixative for pathology testing. Some of the tumors were stored in the refrigerator at −80°C for ELISA, and fresh tumors and spleens were stored in PBS for flow cytometry.
TUNEL Assay
TUNEL staining was performed as described previously. 35 Briefly, after fixing, embedding, slicing, and dewaxing the mouse tumor tissue, add 20 ug/ml proteinase K solution (Thermo Fisher, Shanghai, China), react at 37 °C for 15 min, washed twice with PBS (Gibco, USA), add 50 ul of the prepared TUNEL assay solution (Beyotime, Shanghai, China), incubate at 37 °C for 1 h, wash 3 times with PBS; then restain the cell nuclei with DAPI staining solution (Beyotime, Shanghai, China) and wait for 10 min at room temperature. The cell nucleus was then restained with DAPI staining solution and left at room temperature for 10 min. The nuclei were rinsed with PBS for 5 min and washed 3 times to remove the unbound DAPI staining solution. Next, we wiped the edges of the sections, sealed them with a drop of sealing solution, and air-dried them. The fluorescence expression of tumor cells was observed using a Leica fluorescence microscope and images were taken. Finally, the expression of apoptotic cells in the tissue was analyzed using ImageJ image analysis software, and the %AREA was calculated as the percentage of positive cell expression.
Organ index
Then, we measured the heart, liver, kidney, and spleen weights of the mice to calculate each major organ index. The organ index is as follows: organ index (%) = (average organ weight (g)/average body weight × 100%. 36
Detection of Routine Blood Cells
Routine blood tests were done by the Department of Laboratory Medicine of the Affiliated Chinese Medicine Hospital of SOUTHWEST MEDICAL University. Using a fully automated hematology analyzer (SYSMEXN 9000), blood cell counts and hemoglobin levels of mice were performed.
HE Staining of Organs and Tumor Immunohistochemical Detection
The tumor, heart, liver, kidney, lung, and spleen tissues of mice were fixed with 4% paraformaldehyde tissue fixative (Biosharp, Hefei, China), placed in an embedding box after 24 h, rinsed with flowing water for 12 h, gradient dehydrated in anhydrous ethanol, and soaked in xylene; then soaked in paraffin solution for 2 h. The embedding was shaped and frozen for storage. The sections were sliced with a paraffin slicer at a thickness of 4–5 μm, placed in a baking machine to fully evaporate the water, and then dewaxed and rehydrated in xylene, gradient anhydrous ethanol, and double distilled water in turn. Hematoxylin solution was stained followed by eosin stain (H&E), and the tissue morphology was observed using a Leica fluorescence microscope.
For the immunohistochemical assay, according to the procedure of the endogenous biotin blocking kit, 1 drop of solution A (ovalbumin) was added to the section and incubated at room temperature for 10 min, and washed with PBS. Next, added solution B(D-biotin solution) and incubated at room temperature. Afterward, 1 drop of 5% BSA (Abcam, UK) was added and placed in a 37 °C incubator for 30 min; the primary antibody was added dropwise and left at 4 °C overnight, then the sections were removed, rewarmed for 20 min, secondary antibody was added dropwise and incubated at 37 °C for 60 min, washed and SABC magnifying agent was added dropwise for 50 μl, incubated at 37 °C for 20 min and then stained with DAB (DAKO, Denmark) and observed under the microscope. Finally, the slides were rinsed with hematoxylin staining in running water, then soaked in deionized water for 30 min and dried at room temperature overnight; the slides were sealed after dehydration and transparency. 37
The tissues were observed and photographed using a Leica fluorescence microscope, and the Image-Pro Plus software was used to analyze The expression of Nuclcar-associated antigen Ki-67(Ki-67), Proliferating cell nuclear antigen (PCNA), RAR-related orphan-receptor gamma (RORγt), Forkhead box protein 3 (FOXP3) in tissues was analyzed by calculating the ratio of IOD/AREA indicates the protein expression level.
Flow Cytometry Assay
The ratios of CD8+T cells,CD4+ T cells, T helper cell 17 (Th17), and Regulatory cell (Treg) in the tumor and spleen of mice were examined using flow cytometry. The whole process is divided into two steps. First and foremost, we have to prepare specimens. The mouse tumor tissues were rinsed and cut into small pieces with ophthalmic scissors, ground with a grinder, filtered, centrifuged and the supernatant discarded, and the remaining cells were washed twice with PBS, centrifuged, and collected as cell precipitate. Next, the cell suspension made from mouse spleen tissue was added to 3 times the volume of erythrocyte lysate, lysed at room temperature for 5 min, and washed twice with PBS, and the cell precipitate was collected by centrifugation. The cells were washed twice with PBS, and the cell precipitate was collected by centrifugation.
Besides, the cells were resuspended with 100 μLPBS, CD4 (0.5 μL/case), CD25 (1.25 μL/case), CD3-PE-Cy7, CD4-FIT, CD8-PerCP-Cy5.5, CD25-APC, CD45-eF506 and Live-Dye (Biolegend, USA), were added to each tube, incubated at 4°C for 30 min with light shielding, centrifuged at 300 g for 5 min, the supernatant was pipetted to the waste tank, the remaining cells were washed with PBS and centrifuged at 300 g for 5 min, the supernatant was discarded, 1x True Nuclear Fixation Concentrate (Biolegend, USA) was added. Add 1x True Nuclear Fixation Concentrate 500 μL, incubate at room temperature for 50 min, centrifuge at 350 g for 5 min, and discard the supernatant. Add 250 ml 1x True Nuclear Perm (Biolegend, USA) to each tube, wash twice, centrifuge at 350 g for 5 min, 100 μL 1x True Nuclear Perm to resuspend cells, add FOXP3 (5 μL/case) (Biolegend, USA) and IL-17A (5 μL/case) (Biolegend, USA) to each tube, incubate for 30 min at 4 °C, centrifuge at 350 g for 5 min, discard supernatant, add 250 μL 1x True Nuclear Perm, wash, centrifuge at 350 g for 5 min, discard supernatant, 300 μL 1x True Nuclear Perm Resuspend, and finally assay on the machine. 38
ELISA Assay
ELISA analysis of interleukin-17(IL-17), Interleukin-10 (IL-10), Interferon-γ(IFN-γ), Transforming growth factor-β (TGF-β) in the tumor of mice (before and after the treatment) was evaluated. In brief, we added protein lysate (Beyotime, Shanghai, China) to the tumor, put in steel beads, and grind them using an automatic grinder, then let them stand on ice for 30 min, take the supernatant, centrifuge at 12 000 rpm for 15 min using a centrifuge, and quantify the protein in the supernatant using the BCA method according to the steps in the instructions. Finally, the levels of IL-17, IL-10, IFN-γ, and TGF-β in the tumor were measured by ELISA, which was performed with the ELISA Kit (Boster, Wuhan, China) according to the manufacturer's instructions.
Statistical Analysis
The experimental data of this study were statistically analyzed using SPSS23.0 software. These experiment results are expressed as mean ± standard deviation (SD). T-test was used for within-group pre-post comparisons and the differences between groups. The comparison among multiple groups was analyzed by one-way analysis of variance (ANOVA). The significance level of all statistical analyses was α = 0.05. When p < .05, it means that the results of this experiment are statistically significant. Graphs were plotted using GraphPad Prism 8.0 and Photoshop software.
Results
YQJP Chemical Components Analysis
The YQJP extractions were obtained by mixing single herb granules and dissolving them in hot water. A total of seven chemical components were separated and identified on the basis of retention times and by comparing mass spectra and MS spectra with those reported in the literature and authentic standards. The detailed information is listed in Figure 2 and Supplementary Table S3.

(A) TIC diagram of YQJP extract analyzed by HPLC-MS. positive mode and negative mode. (B) TIC diagram of standards extract analyzed by HPLC-MS. positive mode and negative mode.
Effect of Combining Chemotherapy with YQJP on the State of Survival and Tumors of Mice
Successful Establishment of Spleen qi Deficiency Model
During the modeling period, two mice died due to choking and the instillation of medicine into the trachea. The difference in body weight between the two groups before modeling was not significant (P > .05), but after 30 days of modeling, the difference in body weight between the two groups was statistically significant (P < .01), indicating that the difference in body weight between the two groups after modeling was significant, and the body weight of the Spleen qi deficiency model group was significantly lower than that of the normal group, and the mice in the Spleen qi deficiency model group showed hugging and laziness, reduced feeding, anal filth, and dry hair, indicating that the spleen qi deficiency model was successfully established (Figure 3A and B, and Table 1).

(A) Spleen qi deficiency model mice with filthy anus and dry hair. (B) Changes in body mass curves in the Spleen qi deficiency group and normal groups during modeling. (C) D-xylulose Serum amylase levels at 21 and 30 days in each group of mice. (D) Serum amylase levels at 21 and 30 days in each group of mice.
Weight Changes Before and After Spleen qi Deficiency Model (g).

The data presented are the mean ± SE. *Versus normal group mice, #Versus each group before and after. *P < .05, **P < .01, #P < .05, ##P < .01.
D-Xylulose and serum Amylase in Each Group of Mice
The results of D-xylulose and serum amylase tests showed that compared with the normal group, the serum D-xylulose and amylase contents of the spleen qi deficiency group after 21 and 30 days of gavage were significantly reduced, and the differences were statistically significant (P < .05); this indicated that the intestinal absorption ability of mice was weakened after senna gavage, and the model of spleen qi deficiency was successfully established; and the spleen qi deficiency group after 30 days of gavage showed a trend of reduction in serum D-xylulose and amylase contents when compared to the normal group after 21 and 30 days of gavage. Compared with the spleen qi deficiency group after 21 days of gavage, the serum D-xylose content and amylase content also showed a decreasing trend, and the difference was significant (P < .05), but the difference was not statistically significant (P > .05) between the normal group after 21 and 30 days of gavage, which indicated that with the prolongation of senna gavage, the intestinal absorption capacity of the mice was weakened further (Supplementary Table S5, Figure 3C and D).
Body Weight of Tumor-Bearing Mice Before and After Drug Intervention in Each Group
There was no significant difference in the body mass of each group before drug administration (P > .05). After 14 days of drug intervention, there was no death in each drug group, and the tumor-bearing mice in the control group, the YQJP group, and the YQJP + FOLFOX group had good survival status, could exercise and ate normally, and had shiny hair. The mice in the control group had a significant increase in body weight after treatment, and the difference was significant (P < .01); the mice in the FOLFOX group had a poor survival status, and the mice had a poor mental state during the drug administration, and the body mass increased slightly after the drug administration, but the difference was not statistically significant (P > .05) (Figure 4A and Table 2)

(A) Body weight change curve of each group during drug intervention. (B) Tumor volume change curve. (C) Tumor size of each group of tumor-bearing mice. (D) Tumor weight of each tumor-bearing mouse.
Changes in Body Mass Before and After Treatment (g).
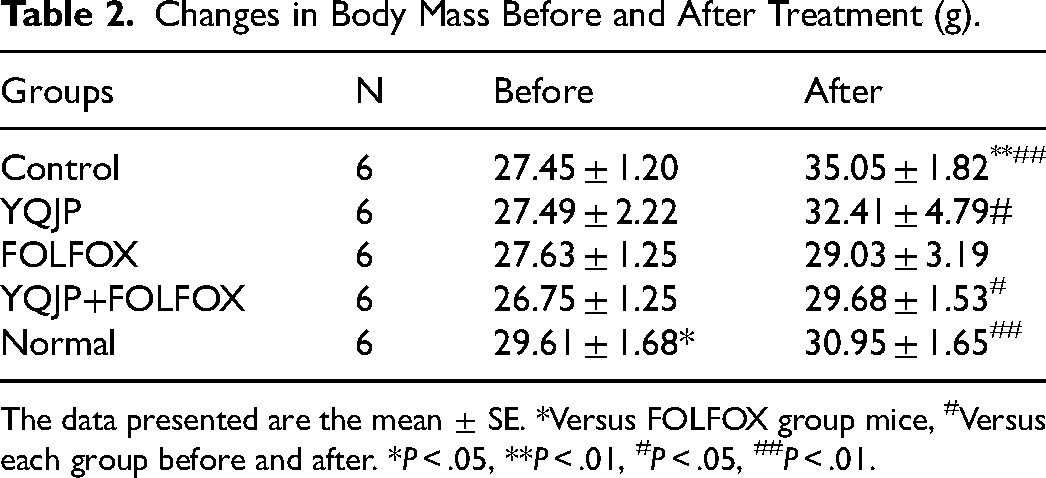
The data presented are the mean ± SE. *Versus FOLFOX group mice, #Versus each group before and after. *P < .05, **P < .01, #P < .05, ##P < .01.
Tumor Growth in Different Groups of Mice
There was no significant difference in the tumor volume of each group before drug administration (P > .05). After 14 days of drug intervention, the tumor volume increased significantly in all groups, and compared with the control group, the tumor volume was significantly smaller (P < .01) and the tumor mass was reduced (P < .05) in the YQJP group, FOLFOX group, and YQJP + FOLFOX group, and the differences were statistically significant (Figure 4 and Table 3). The tumor volume and tumor mass were lower in the YQJP + FOLFOX group compared with the FOLFOX group, but the difference between the two groups was not statistically significant (P > .05). The tumor suppression rates of each group were 23. 56% in the YQJP group, 48.98% in the FOLFOX group, and 61.91% in the YQJP + FOLFOX group, with the best effect in the YQJP + FOLFOX group (Table 4).
Changes in Tumor Volume Before and After Treatment (mm3).

The data presented are the mean ± SE. *Versus FOLFOX group mice, #Versus Control group mice. *P < .05, **P < .01, #P < .05, ##P < .01.
Mean Tumor Weight and Tumor Suppression Rates of Each Group.

The data presented are the mean ± SE. *Versus FOLFOX group mice, #Versus Control group mice. *P < .05, **P < .01, #P < .05, ##P < .01.
Effect of Combining Chemotherapy with YQJP on the Proliferation and Apoptosis of Tumors
Detection of Ki-67 and PCNA Expression in Tumors by Immunohistochemistry
The immunohistochemical results showed that the relative expression of Ki-67 and PCNA proteins in tumor tissues of the FOLFOX group and YQJP + FOLFOX group were significantly lower compared with the Control group, and the difference was statistically significant (P < .01); meanwhile, the decrease was more obvious in YQJP + FOLFOX group compared with FOLFOX group, and the difference was statistically (P < .05), indicating that the antiproliferative effect of YQJP + FOLFOX group was better than that of FOLFOX group (Figure 5A).

(A) Immunohistochemical expression of Ki-67 PCNA protein in tumor samples of each group after medication (200×). (B) TUNEL staining detected apoptosis in tumor specimens after drug use in each group (200×).
Detection of Apoptosis in Tumors by TUNEL Staining
The results of the TUNEL staining method showed that the expression of apoptosis-positive cells in tumor tissues was significantly increased in the YQJP group, FOLFOX group, and YQJP + FOLFOX group compared with the control group, and the difference was statistically significant (P < .01), indicating that YQJP group, FOLFOX group, and YQJP + FOLFOX group could promote apoptosis of tumor cells; compared with the FOLFOX group, the YQJP + FOLFOX group had more apoptosis-positive cells in the tumor tissues(P < .01), and the apoptosis phenomenon was the most obvious. (Figure 5B). It indicates that YQJP combined with chemotherapy can promote apoptosis of tumor cells.
Safety and Toxicity of Combining Chemotherapy with YQJP
Organ Indices in Different Groups of Mice
The organ indices of each group showed that compared with the Normal group, the spleen indices of the Control group, YQJP group, FOLFOX group, and YQJP + FOLFOX group were increased to different degrees (P < .01); the differences in liver indices were not statistically significant in all groups except the FOLFOX group (P > .05); the kidney indices were decreased in all groups (P < .01); the cardiac index was reduced in Control group and YQJP group and YQJP + FOLFOX group compared with Normal group (P < .01); spleen index and liver index were reduced in YQJP + FOLFOX group compared with FOLFOX group (P < .05) (Figure 6A, Table 5). The YQJP + FOLFOX group improved the spleen morphology significantly (Figure 6C).

(A) Histograms of the results of organ indices for each group of mice. (B) Histograms of the results of the quantification of erythrocytes, leukocytes, hemoglobin, and platelets in each group of mice. (C) Histomorphology of spleen of mice in each group after drug administration. (D) Liver, heart, kidney, and lung tissue morphology of each group after medication (200×).
Organ Index (mg/g).

The data presented are the mean ± SE. *Versus FOLFOX group mice, #Versus Normal group mice. *P < .05,**P < .01, #P < .05, ##P < .01.
Various Blood Components in Different Groups of Mice
The results of routine blood tests suggested that compared with the Normal group, hemoglobin was reduced to different degrees in all groups (P < .05), and white blood cells were reduced significantly in the FOLFOX group (P < .05); compared with FOLFOX, red blood cells were elevated in the YQJP + FOLFOX group (P < .05), with statistically significant differences, and hemoglobin and white blood cells were elevated, but the differences were not significantly statistically significant (P > .05) (Figure 6B, Table 6). It indicates that the combination of YQJP with chemotherapy can increase the level of hemoglobin, red blood cells, and white blood cells in mice to a certain extent and attenuate the toxicity of chemotherapy.
Blood Routine Results for Each Group of Mice (n = 6).

The data presented are the mean ± SE. *Versus FOLFOX group mice, #Versus Normal group mice. *P < .05, **P < .01, #P < .05, ##P < .01.
HE Staining of major Organ Tissues in Different Mice Groups
HE staining showed no significant degeneration and organ damage in the liver, kidney, and lung tissues in all groups. Heart tissues of mice in the FOLFOX group showed myocardial fiber destruction transverse stripe loss, and cell nucleus consolidation (Figure 6D).
Effect of QYJP Combined with Chemotherapy on Tumor Microenvironment
Effect on Systemic Immunity in Mice
The results of flow cytometry suggested that the proportion of CD4+ T cells in the spleen tissues of mice in the YQJP and YQJP + FOLFOX group was significantly higher compared with the control group, and the difference was statistically significant (P < .01), while the proportions of Th17 cells, Treg cells, and Treg/Th17 in the spleen tissues of each group were not significantly different (P > .05) (Figure 7A, Table 7). While in spleen tissues, the proportion of CD8 + cells was reduced in the remaining four groups compared with the Normal group. Compared with the Normal group, the YQJP and Control groups were statistically significant (P < .05), but the comparison between the remaining groups was not statistically significant (P > .05) (Figure S1). It indicates that YQJP strengthens the spleen to enhance the systemic immunity of mice.

(A) Flow charts and histograms of CD4+ T cells, Th17 cells, treg cells, and treg/Th17 ratio in the spleen of each group. (B) Flow chart and histogram of CD4+ T cells, Th17 cells, Treg cells, and Treg/Th17 ratio in tumor tissues of each group. (C) Expression of FOXP3 and RORγt in tumor tissue after medication in each group (200×) and histogram. (D) Histogram of cytokine expression in tumor tissues of each group. (E). Histogram of cytokine expression in each group before administration.
Flow Cytometry Detects the Proportion of CD4+ T Cells, Th17 Cells, Treg Cells, and Treg/Th17 Cells of Spleens.

The data presented are the mean ± SE. *Versus FOLFOX group mice, #Versus Control group mice. *P < .05, **P < .01, #P < .05, ##P < .01.
Effects on the Immune Microenvironment of Tumor in Tumor-Bearing Mice
The results of flow cytometry in the spleen suggested that YQJP affected the systemic immunity of the tumor-bearing mice, so we further examined the levels of CD8+ cells, CD4+ T cells, Th17, and Treg cells in the tumor microenvironment. The results showed that compared with the control group and FOLFOX group, the proportion of CD4+ T cells in the tumor tissues of the YQJP group and YQJP + FOLFOX group was significantly higher, and the increase was most obvious in the YQJP + FOLFOX group, and the difference between the two groups was statistically significant (P < .05) compared with the FOLFOX group, and the tumor immune response was stronger in the YQJP + FOLFOX group. Secondly, the proportion of Th17 cells in tumor tissues was significantly higher in the YQJP + FOLFOX group compared with the FOLFOX group, and the difference was statistically significant (P < .01); and the Th17 cells were lower in the FOLFOX group compared with the control group, and the difference between the two groups was statistically significant (P < .05). In addition, compared with the FOLFOX group, Treg cells were lower in the YQJP + FOLFOX group, and the ratio of Treg/Th17 was significantly lower (P < .01), and the difference was statistically significant (Figure 7B, Table 8). In addition, compared with the control group, the proportion of CD8+ cells in tumor tissues was elevated in the YQJP group (P > .05); compared with the FOLFOX group, the proportion of CD8+ cells in tumor tissues was reduced in the YQJP group and the YQJP + FOLFOX group, and the differences were not statistically significant (P > .05) (Figure S1).
Flow Cytometry Detects the Proportion of Th17, Treg, Treg/Th17 and CD4+T Cells in Each Group of Tumors (n = 3).

The data presented are the mean ± SE. *Versus FOLFOX group mice, #Versus Control group mice. *P < .05, **P < .01, #P < .05, ##P < .01.
Immunohistochemical Expression of RORγ-t, FOXP3 in Tumor Specimens from Different Groups of Mice
The immunohistochemical results showed that the relative expression of FOXP3 protein in tumor specimens in the YQJP group and YQJP + FOLFOX group were lower than that in the control group and FOLFOX group, and the difference was statistically significant (P < .01); secondly, compared with the control group and FOLFOX group, the relative RORγ-t protein in the YQJP group and YQJP + FOLFOX group expression was higher in the YQJP and YQJP + FOLFOX groups compared with the control and FOLFOX groups, and the difference was statistically significant (P < .01) (Figure 7C).
ELISA to Detect the Expression of Cytokines IL-17, IL-10, IFN-γ and TGF-β in Mouse Tumors
The ELISA results prior to drug administration showed no difference in IL-17, IFN-γ, IL-10, and TGF-β expression among the groups, and the results were not statistically significant (P < .05). The ELISA results showed that the contents of IL-17 and IFN-γ in the tumor tissues of the YQJP group and YQJP + FOLFOX group were significantly higher compared with the FOLFOX group, and the differences were statistically significant (P < .01); compared with the FOLFOX group, the contents of IL-10 in the YQJP group and YQJP + FOLFOX group were significantly lower, and the differences were statistically significant (P < .01). Compared with the control group, the TGF-β content in the YQJP group and YQJP + FOLFOX group was significantly reduced, and the difference was statistically significant (P < .01); compared with the FOLFOX group, the TGF-β content in the YQJP + FOLFOX group was reduced, and the difference was statistically significant (P < .05) (Figure 7D and E, Table 9, Table S1). It indicates that YQJP elevates the expression of IFN-γ and IL-17 in the tumor microenvironment and promotes the low expression of TGF-β and IL-10.
Cytokines in Tumor Specimens After Medication in Each Group (n = 6, pg/mg).

The data presented are the mean ± SE. *Versus FOLFOX group mice, #Versus Control group mice. *P < .05, **P < .01, #P < .05, ##P < .01.
Discussion
In TCM, It was found that “spleen qi deficiency” was the most common type of colorectal cancer. 39 In this study, the “spleen qi deficiency” model was established with a compound factor method involving an irregular diet, 31 excessive fatigue, and senna folium with the bitter-cold property. 40 Meanwhile, body weight, behavior, D-xylose, and serum amylase, which were closely related to the spleen qi deficiency model, were used to evaluate the success of model construction. 41 The results showed that the compound factor modeling method could reduce the body weight of mice, change the behavior of mice, reduce the D-xylose content and serum amylase, and weaken the absorption function of the small intestine.
Chinese herbal formula, Chinese herbs, and Chinese medicinal compounds all show good anti-tumor effects. 39 Chinese medicines are complex in composition and their pharmacological effects are also characterized by “multi-component-multi-target”. 42 TCM emphasizes the holistic concept, which has similarities with the holistic nature of the organism's immunity.
Some researchers used the “spleen-strengthening and bowel-clearing method” to treat colorectal cancer patients with intestinal mucosal dysfunction caused by chemotherapy, and found that this experienced formula could enhance immunity and improve quality of life. 43 It was found that the use of a Chinese herbal medicine compound with “Jianpi” in combination with chemotherapy for colorectal cancer patients could improve patients’ survival status, enhance chemotherapy efficacy, reduce the incidence of toxic side effects, and improve the physical and mental discomfort caused by chemotherapy.44,45 In clinical trial studies, YQJP is believed to enhance the effect of chemotherapy and strengthen the immunity of the patient's organism. In this study, we found that YQJP could improve the body condition of mice, increase their body weight, and improve their survival status with no damage to their tissues and organs. Blood cell assays showed that YQJP increased hemoglobin in mice, which can improve bone marrow suppression after chemotherapy. In conclusion, YQJP is a safe and effective anti-cancer formula.
Th17 cells have an independent transcription factor, and they can participate in the regulation and development of autoimmune diseases, allergic reactions, tumors, and other diseases. After the differentiation and maturation of Th17, it mainly relies on the secretion of IL-17-based cytokines to play a role.46,47 Th17 has a bidirectional regulatory effect on tumors. 18 In the early stages of CRC, Th17 promotes an inflammatory response by secreting inflammatory factors, thereby promoting tumorigenesis, while in established colorectal tumors, Th17 has anti-tumor effects. 48 Th17 cells do not have direct mediation of toxic activity against tumor cells, and polarized Th17 cells mediate tumor regression in an IFN-γ-dependent manner in mice. 49 IFN-γ has an important anti-proliferative and pro-apoptotic role in tumor cells. 50 Treg cells are another important component of T cells, which mainly secrete suppressive cytokines such as IL-10 and TGF-β to suppress the malignant immune response of the body. 51 In TME, there are often large numbers of Treg cells, which often have opposite roles to Th17 cells in tumor immunity and mediate immunosuppressive signaling pathways, and their differentiation is largely dependent on the transcription factor FOXP3. 52 During the development of cancer, Treg mainly manifests itself by suppressing the antitumor immune response and contributing to tumor immune escape, which in turn promotes the development of malignancy. 53
In tumor immunotherapy, both Th17 cells and Treg cells do not act directly, but through the secretion of different cytokines by the body in different diseases, states to promote the differentiation of CD4+ T cells in different directions and regulate the ratio of Th17 cells to Treg cells. 54 In the early stages of CRC development, elevated Th17 promotes inflammatory responses and Treg, which suppresses inflammatory responses, decreases, while the proportion of Th17 cells decreases and, conversely, the proportion of Treg cells increases gradually with further tumor progression. 55 During early tumorigenesis, IL-17 promotes tumor growth, whereas, in established tumors, IL-17 production by Th17 cells enhances anti-tumor immunity. the most potent tumor-protective properties of IL-17 arise from an indirect immune-mediated phenomenon that allows the immune system to become cytotoxic.
Results from clinical trials have shown that increased Th17 cells are associated with improved prognosis in patients with oropharyngeal squamous carcinoma and salivary gland tumors.56,57 In addition, Th17 cells are largely present in the tumor microenvironment of cancer patients, and in comparison, patients with melanoma, early-stage ovarian cancer, and malignant pleural effusions with increased Th17 cells have better survival rates. 58 The results of our study showed that YQJP could increase the levels of cytokines IL-17 and IFN-γ, decrease the levels of IL-10 and TGF-β in tumor tissues, and decrease the relative levels of FOXP3 and increase the levels of RORγt, thus increasing the proportion of Th17 cells and decreasing the proportion of Treg cells. Meanwhile, our results showed that YQJP combined with FOLFOX regimen chemotherapy could promote apoptosis while inhibiting further proliferation of tumor cells. A significant decrease in Ki-67 and PCNA was seen in the YQJP group and YQJP + FOLFOX group, and TUNEL staining showed a significant increase in apoptotic cells. This indicates that YQJP may work by promoting apoptosis and inhibiting proliferation. However, we have only conducted in vivo experiments, and in the next experiments, we will further validate the effectiveness of YQJP in vitro, as well as the synergistic effect with chemotherapeutic agents. At present, only a preliminary judgment of its effectiveness and safety can be made, but further evidence is needed to support our conclusion.
In conclusion, YQJP combined with FOLFOX regimen chemotherapy can enhance the chemotherapy effect and improve the anti-tumor effect, and we speculate that the mechanism may work by regulating Th17 cells and Treg cells and regulating cellular immunity in the tumor microenvironment.
Conclusions
In this study, we reported that YQJP in combination with chemotherapy inhibited the progression of CRC, as evidenced by changes in body weight, biological behavior, tumor load, and T lymphocytes and cytokines in mice. Specifically, we demonstrated that YQJP may exert its anticancer effects by modulating the tumor immune microenvironment, as evidenced by the inhibition of tumor cell proliferation and the promotion of apoptosis by increasing Th17 cells and related cytokines and decreasing Treg cells and their cytokines in mouse tumor tissues. This finding helps us to better understand the anticancer effects of YQJP in regulating immunity and inhibiting and delaying tumor development. In future studies, we will further optimize the construction of the spleen deficiency model and the drug ratios in YQJP to determine which drugs play major anticancer roles in it, and conduct further in vitro experiments to verify whether they have synergistic effects. We will pave the way for the wide application of YQJP in clinical treatment.
Supplemental Material
sj-docx-1-chp-10.1177_2515690X241313097 - Supplemental material for Yiqi Jianpi Kangai Decoction Enhances the Chemotherapy Effect by Inducing Apoptosis and Regulating Treg and Th17 Cells in Colorectal Cancer Mice Model with Spleen Qi Deficiency
Supplemental material, sj-docx-1-chp-10.1177_2515690X241313097 for Yiqi Jianpi Kangai Decoction Enhances the Chemotherapy Effect by Inducing Apoptosis and Regulating Treg and Th17 Cells in Colorectal Cancer Mice Model with Spleen Qi Deficiency by Qinsha Wang, Shichao Li, Yao Fan, Weidong Chen, Qingfeng Jiang, Xin Sun, Qijun Lv, Wusheng Li and Yingtian Jia in Journal of Evidence-Based Integrative Medicine
Footnotes
Acknowledgments
The authors thank the Animal Experiment Center of SOUTHWEST MEDICAL University for their support in animal experiments. We thank the Sichuan Provincial Administration of Traditional Chinese Medicine and the Luzhou Science and Technology Bureau for their support of this study.
Author Contributions (Roles)
Qinsha Wang and Shichao Li provided the study background, study objectives, and hypothesis, performed the primary study, collected data, analyzed and interpreted the results, and prepared and submitted the manuscript.
Yao Fan, Yingtian Jia, and Weidong Chen constructed the spleen qi deficiency CRC mice model, collected data, and analyzed and interpreted the results.
Qingfeng Jiang, Xin Sun, and Qijun Lv performed pharmacological interventions on mice, collected specimens, and performed experiments.
Yingtian Jia and Wusheng Li designed and approved the protocol, discussed and summarized the main ideas, and revised the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Animal care and handling were performed in accordance with the International Guide for the Use and Maintenance of Laboratory Animals. Animal ethical approval was granted by the Laboratory Animal Ethics Committee of SOUTHWEST MEDICAL University (LACUC Issue No. swmu20220012).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Luzhou Science and Technology Bureau Project, Scientific Research Project of the Sichuan Administration of Traditional Chinese Medicine (grant number 2022-SYF-48, 2022-SYF-51, 2020JC0137).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
