Abstract
The study's objective is to conduct a comprehensive systematic review for assessing the safety and efficacy of Ayurvedic interventions in managing hemiplegia/Pakshaghata. The study involved a search across multiple online databases and online clinical trial registries. Additionally, major Ayurveda postgraduate institutes were contacted to acquire unpublished trial data related to hemiplegia/Pakshaghata. The review covered articles published until July 2023. Two reviewers independently performed data extraction and risk of bias assessment. The risk of bias assessment utilised the RoB 2 tool for randomised trials and the ROBINS-I tool for non-randomised trials. The screening process identified 28 articles from online databases and two dissertations from online repositories. However, practical challenges prevented access to grey literature from Ayurveda institutes. The 30 studies selected for this review, comprises nine randomised controlled trials (RCTs), eight non-randomised comparative trials, and thirteen pre-post studies. Quantitative analysis was unfeasible due to inadequate studies, leading to a qualitative analysis. All studies, except one, exhibited substantial bias upon risk of bias assessment. Moreover, most studies demonstrated methodological weaknesses attributed to a lack of masking, improper sampling techniques, non-validated outcome measurement tools, inadequate follow-up procedures, and confounding factors. The trials frequently did not document safety parameters, adverse events (AE), and adverse drug reactions (ADR). Current review could not definitively establish the efficacy and safety of Ayurvedic interventions in hemiplegia/Pakshaghata. Hence, the authors strongly advocate for good quality research incorporating proper methodology.
Background
The 2021 Global Burden of Disease (GBD) study shows stroke ranked the fourth leading cause of death after COVID-19, neonatal disorders and ischaemic heart disease. 1 In 2001, it was estimated that cerebrovascular diseases (stroke) accounted for 5.5 million deaths worldwide, equivalent to 9.6% of all deaths. 2 The World Health Organisation (WHO) definition of stroke is: “rapidly developing clinical signs of focal (or global) disturbance of cerebral function, with symptoms lasting 24 h or longer or leading to death, with no apparent cause other than of vascular origin”. 3 Stroke is one of the leading causes of hemiplegia, which significantly affects the patient's quality of life by reducing the ability to perform daily activities.4,5 Conventional stroke management includes three phases of treatment: acute management, rehabilitation, and secondary prevention. 6 A multidisciplinary team carries out stroke rehabilitation, and the goal of the treatment has changed from mere functional recovery to everyday living. 7 Stroke survivors were reported to have physical, cognitive, social, and emotional sequel persisting even a long time after the stroke incidence. 8 A survey conducted in the UK observed that nearly 50% of survivors reported one or more unmet needs concerning mobility, pain, fatigue, memory, and concentration. 8 So, there is a need to explore expertise from other disciplines to overhaul current lacunae.
Ayurveda views hemiplegia as a clinical entity, ‘Pakshaghata’, grouped under ‘Vatavyadhi’. 9 The central role of Ayurveda lies during the time of stroke rehabilitation. 10 It employs internal medicines along with procedures, including Panchakarma therapies. Evidence-based medicine (EBM) requires proof of effectiveness, efficiency, and safety for decision-making in patient care. 11 Integrating individual clinical expertise with the best external clinical evidence is the core of EBM practice. 12 Thus, empirical Ayurvedic knowledge accrued over the years can be tapped for integrated stroke rehabilitation after proper evaluation. Hence, the present study is carried out to document and evaluate currently available evidence regarding the safety and efficacy of Ayurvedic interventions in hemiplegia.
Materials and Methods
Inclusion Criteria
We selected studies fulfilling the following criteria for this review: 1. Type of Study-Randomised controlled trials, quasi-experimental trials, comparative clinical trials, and single group clinical trials on Ayurvedic management of hemiplegia were screened for data analysis. 2. Population- Cases diagnosed with hemiplegia (either by imaging or clinical symptoms) or Pakshaghata (as defined by Ayurvedic classics) who underwent Ayurvedic treatment were screened for this study. 3. Interventions- Ayurvedic treatment protocol with different procedural interventions (Shodhana) and non-procedural interventions (Shamana) with different dosage forms, types, schedules, and drugs as the intervention group in hemiplegia were included for data analysis. 4. Comparators/Control Group- Placebo or Shamana therapy and non-Ayurveda interventions in hemiplegia were screened as the comparative group to hemiplegia interventions.
Exclusion Criteria
1. Study Type- The review excluded pre-clinical trials, case-control studies, case series, case reports, and literature reviews from screening. 2. Population- current study did not consider cases involving systemic illnesses other than those affecting the nervous system or patients with significant comorbidities who underwent Ayurvedic treatment for hemiplegia (Pakshaghata). 3. Intervention- Studies that included non-Ayurvedic interventions in the study group were excluded from the review. 4. Outcome- Studies with outcome parameters different from those specified in the section ‘types of outcome measures’ were excluded from the study.
Types of Outcome Measures That Were Screened for the Review
Effectiveness/efficacy parameters, Response to treatment (Improvement in subjective and objective criteria of assessments), improvement in quality of life, Safety/morbidity parameters, and serious adverse events (resulting in death, disability, or incapacity) or complications that were life-threatening and led to hospitalisation or prolongation of hospitalisation are the significant outcomes evaluated in this review.
Search Methods for Identification of Studies
Electronic databases like PubMed, Cochrane Library, Google Scholar, AYUSH Research Portal (Govt. of India), DHARA, Ancient Science of Life, Shodhganga@INFLIBNET, and online clinical trial registers were scrutinised. Articles published until July 2023 were reviewed for the present study. Further, all the major Ayurveda postgraduate institutes were contacted through email to provide unpublished trial data related to hemiplegia/Pakshaghata.
Search Strategy (Keywords)
Ayurveda OR Ayurvedic OR Herbal OR AYUSH OR Indian traditional medicine OR hemiplegia OR Pakshaghata OR Pakshvadha AND stroke OR haemorrhagic stroke OR ischaemic stroke OR CVA as title, abstract, or keyword.
Data Collection/Synthesis
Two investigators independently screened the databases to get potential records. After removing duplicate records, the title and abstract were screened according to inclusion criteria for getting eligible articles. Full-text articles thus were again scrutinised and evaluated as per inclusion criteria. Selected studies were further undergone for data extraction. The data extraction table included 1. methods used in the study (randomisation/allocation concealment/blinding/sampling and sample size calculation/length of follow-up), 2. population under consideration, 3. interventions (treatment protocol administered/formulations used) and control, 4. outcomes measurements, 5. Outcomes (regarding safety /effectiveness /efficacy /improvement in quality of life).
Assessment of Risk of Bias and Overall Quality of Evidence
For each study, the risk of bias was assessed by the Cochrane RoB2 tool version 22 August 2019 (for randomised controlled trials) and the ROBINS-I tool version 19 September 2016 (for non-randomised trials). The disagreement between the reviewers was resolved through discussion.
Data Analysis
We have yet to analyse quantitative data due to the absence of suitable studies.
Results
Study Selection
Details of identification of eligible records for the review is depicted in PRISMA flow diagram (Figure 1).

PRISMA flow diagram showing steps followed during the study selection process. 13
Description of Studies
Randomised Controlled Trials
Out of nine randomised controlled trials (RCTs) selected for the review, one study 14 has double-blinding, and another study 15 has single-blinding in their design. All the remaining studies were open trials.16–20 One was a three-arm trial; 18 remaining studies had two arms. A precise subgroup analysis was conducted in one study, ie, seven sub-groups in the study group and 4 in the comparative group. 20 A total of 1202 participants from these nine studies diagnosed with Pakshaghata were randomly allocated into study and comparison groups. Among these, the study with subgroup analysis had a more significant number of samples, ie, 552 in the study group and 192 in the comparison group. 20 In contrast, MG Aggithaya et al 2014 reported his study with the least number of samples (nine in each group) in the study and comparison group. 14 The sampling method was not explained appropriately in any of these studies.
Most studies used Ayurvedic symptoms as the primary criteria for diagnosing Pakshaghata, whereas one study used contemporary techniques and the Ayurvedic approach. 17 Participants’ age groups ranged from 18 to 80 in these trials. Disease chronicity was mentioned only in five studies,14,17–19,21 from 30 days to 2 years. Only one study reported follow-up after the treatment, 18 and the remaining studies were either not mentioned or mentioned as nil. Three studies reported the absence of adverse events (AE).14,18,20 Other studies were not reported regarding AE. Similarly, data regarding the dropout of trial subjects are available only in three studies.17,18,22
Details of Study Interventions in RCTs
In the intervention trials, seven studies were given the study drug and Panchakarma procedure in both groups.14,16,18–22 In contrast, S.S Jaideep et al 17 gave conventional modern treatment, including antiplatelet agents, oral anticoagulants, anti-hypertensives, and medications for diabetes mellitus/hyperlipidaemia, along with physiotherapy and speech therapy as required. Then, it was compared with groups receiving conventional, Ayurvedic, and speech therapy. One study has analysed add-on physiotherapy and Panchakarma treatment with standalone Panchakarma therapy. 15
Non-Randomised Comparative Trials
There were eight non-randomised comparative studies selected according to the inclusion criteria.23–30 These studies were carried out with 417 participants with Pakshaghata, with the sample size ranging from 6 (Kandarp Desai) 24 to 56 (Madhavan Pillai et al) 26 in each group. Participants were recruited based on the signs and symptoms of Pakshaghata in six studies.23–26,28,30 In contrast, the other two studies used modern parameters and imaging techniques to confirm the cerebrovascular accident.27,29 The age specification of participants was available only in four studies.23,25,27,29 Disease chronicity is described in five studies ranging from ten days to two years.25–27,29 Out of eight studies, only one reported follow-up after treatment. 29 Two studies reported the absence of AE during the study period.,25,28 but dropout patient data were unavailable with any of the trials.
Details of Study Interventions in Non-Randomised Comparative Trials
Among non-randomised comparative trials, three studies examined the effect of Panchakarma in Pakshaghata,25,26,30 another two compared an Ayurveda drug with Panchakarma therapy,23,27 one study evaluated the efficacy of a drug with different ingredients, 24 one study was analysed internal usage of different medicated oils in Pakshaghata, 28 and the eight one was to evaluate the effect of Ayurvedic protocol in Pakshaghata with conventional treatment. 29 Out of these, two trials had three groups for comparison.23,28 Two of the eight studies used placebo medicines for comparison.23,26 The first study compared a standalone Ayurvedic medicine to a group having add-on medicine with panchakarma therapy and to another group having placebo medicine with Panchakarma therapy. 23 The second study compared the efficacy of a classical medicine with placebo for administering Panchakarma therapy. 26
Single Group Pre-Post Trials
Thirteen single group pre-post trials were selected in this study.9,31–42 Among them, two studies36,41 selected patients by Pakshaghata signs and symptoms and eligibility for Panchakarma, eight studies9,31,33,37–40,42 used both clinical features of Pakshaghata and modern diagnostic techniques, and three studies32,34,35 only used modern diagnostic criteria for patient selection. Of these 13 trials, four studies specifically mentioned ischaemic/infarction stroke as an inclusion criterion for trial subjects.31,33,38,40 Patients from 16 to 80 years of age were selected in these thirteen trials. Stroke duration was considered a selection criterion in three trials.31,33,36 Those studies selected patients from 15 days to more than five years of chronicity. The sample size of these studies ranged from 20 to 60 and collectively enrolled 412 stroke/Pakshaghata patients in them. Only four studies reported follow-up periods after treatment.31,33,41,42 However, six trials34,37,39–42 reported data regarding dropout during the trial period. Only four studies31,37,39,40 reported the absence of AE during the trial.
Details of Study Interventions in Pre-Post Trials
Out of 13 single group pre-post trials, seven were about the effect of individual therapeutic procedures,9,32,36,37,40–42 and six were drug/formulation studies.31,33–35,38,39
Risk of Bias in Included Studies
Randomised Controlled Trials
All selected randomised trials were deemed highly biased upon overall bias assessment, except one trial (Aggithaya et al), 14 which was found to have ‘some concern’ in the overall bias assessment (Figure 2). The same study 14 extensively discussed randomisation and allocation concealment, concluding an absence of bias within the domain of randomisation procedures. Two studies16,17 had ‘some concern’ in the randomisation procedure due to lack of information. The remaining six studies15,18–22 were found to have poorly described randomisation procedures, which caused high bias in the randomisation domain. Except for one study, 14 all selected RCTs were deemed highly biased in the domain of ‘deviation from intended intervention’ due to the lack of proper blinding and absence of data concerning non-protocol interventions or non-adherence to the assigned intervention. In the Aggithaya et al, 2014 study, both the participants and the assessing physiotherapists were masked. Additionally, patient selection and treatment supervision were independently carried out by two separate Ayurvedic physicians. Since the Panchakarma therapists (treatment providers) were not blinded in the study, one of the Ayurvedic physicians supervised all treatment sessions to minimise potential biases. However, due to the lack of a published protocol, the bias assessment in the domain of ‘deviation from intended intervention’ was concluded as ‘Some Concern’. Out of nine studies, two studies13,16 were found to be highly biased due to missing outcome data, and the rest of the studies were unbiased. Similarly, in outcome measurement, only one study 14 was found unbiased owing to the proper blinding of the assessor, and the rest of the studies were highly biased. Due to the absence of published protocols or trial registry data, all eight selected studies were noted to have some concerns regarding biases arising from the selection of reported results. In the P.R. Nair et al, 1985 study, although physiotherapy functional ability assessment and hemiplegia clinical improvement assessment were identified as outcome measures in the methodology, the former was not reported in the result section of the article. Consequently, the study 15 was deemed highly biased in this specific domain due to selective reporting.
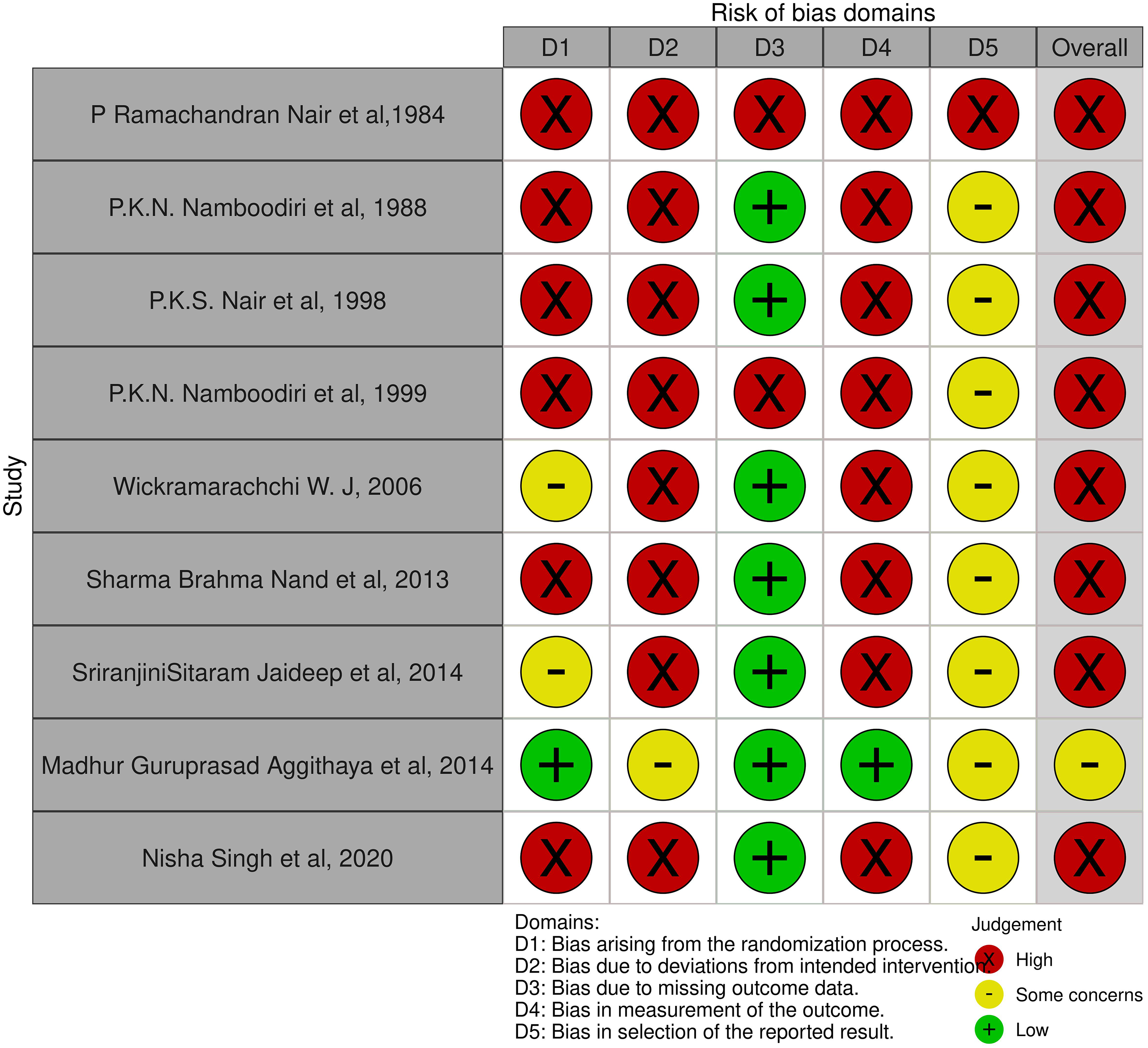
Risk of bias of randomised trials. 43
Non-Randomised Trials
Even though no apparent biases were observed in the domains, like the selection of participants, classification of intervention, deviation from intended intervention, and missing data, all of the selected studies were highly biased in overall assessment due to biases in the other domains (Figure 3). Although two studies26,28 measured confounding variables such as comorbidities and stroke severity, they did not control for these factors either through study design or statistical analysis. Hence all the selected studies were biased due to confounding. Since no data regarding blinding is available, all the included studies have high chances for biases in measuring outcomes. Among the eight selected studies, two studies26,28 were found to be highly biased in selecting the reported results from the available information. Without a pre-registered protocol or statistical analysis plan, it took much work to judge biases due to the selection of the reported results for the remaining studies.
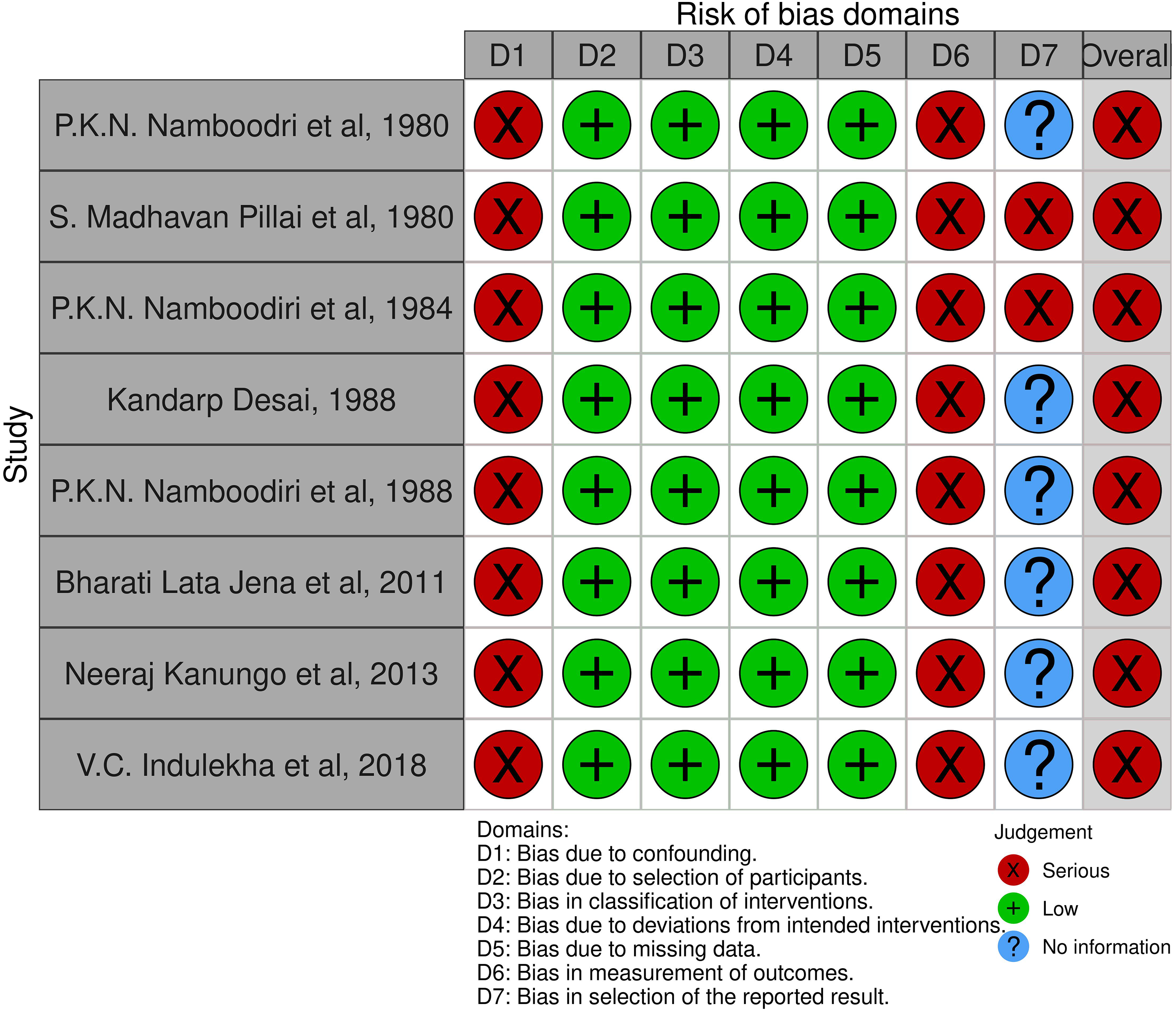
Risk of bias of non-randomised trials. 43
Effects of Intervention
Among RCT trials, Wickramarachchi W. J., 2006 16 took the cardinal symptoms of Pakshaghata, ie, loss of function, pain, headache, swelling, heaviness, loss of sensation, dizziness, and difficulty in walking, for the assessment. In addition to this activity test, feeding, dressing, personal hygiene, transfer, and mobility were included in scores from 0 to 3 in one study. 16 Muscle strength (MRC score), muscle tone (the modified Ashworth scale), tendon reflexes, range of movements, and functional abilities (Berg balance scale) were the additional parameters in one study. 14 A unique 100-mark hemiplegia assessment scale was used in four studies for the assessment.15,18–20 In one study, apart from this, modern evaluation parameters like cardiac autonomic function tests, which included heart rate variability (HRV), blood pressure variability (BPV), and baroreflex sensitivity (BRS), were the assessment criteria. 17 Two studies administered the Pakshaghata clinical features grading scale for assessment.21,22 Statistical methods and significance levels are not mentioned in the two studies.19,20 The statistical analysis used was the Fischer test, Wilcoxon signed-rank test, Z test, and t-test in the selected studies.
In three RCT studies, although both groups showed significant improvement in scores within the group, there was no statistically significant difference between the groups in any domain.14,16,19 Three studies emphasised the efficacy of Panchakarma therapy over the comparator.18,20,22 One study has shown add-on physiotherapy procedure has better benefits when compared with the Panchakarma group. 15 In SS Jaideep et al 2014 study, cardiac autonomic parameters were statistically improved in the adjuvant Ayurveda treatment group. 17 Out of the chosen RCTs, one study did not perform statistical analysis for comparing between groups. 21
In non-randomised comparative trials, the assessment was based on the different types of symptomatic scoring in 5 studies.23,24,26–28 A 100-mark grading score was used in two studies23,28; and activity of daily living and the Barthel index were used in another study. 29 Panchakarma-related studies concluded that both groups significantly improved the assessment parameters. However, these studies failed to establish a significant difference between the groups.25,26 The study and control groups had a similar effect in two drug-related studies.23,24 In another study, no precise data about between-group comparison analysis was available. 27 Studies related to the effect of Tailas (medicated oil) also concluded a non-significant effect between groups. 28 In contrast, one study 29 specifically mentioned the significance of Ayurvedic treatment against the conventional modern treatment in Pakshaghata. The study by P.K.N. Namboodri et al found that the treatment protocol effectively managed Pakhaghata after a comprehensive analysis, yet it did not present an analysis between the groups. 30
In the case of the single group before-after trials, the main assessment criteria were subjective parameters of Pakshaghata, wherein the muscle bulk, power, and reflexes were assessed. Few studies administered assessment criteria like MRC (Medical Research Council) grading for muscle strength, 40 SSQOL (Stroke Specific Quality Of Life),9,37 MBI (Modified Barthel Index), 37 and NIHSS (National Institute of Health Stroke scale)38–40 for outcome measurement. Neurological mapping was used to assess the improvement in two studies.41,42 Panchakarma procedures-related studies reported significant improvement in all the parameters.9,32,36,37,40–42 Significant results were observed in all parameters in two drug-related studies,38,39 whereas in three studies,33–35 the statistical assessment was not mentioned. Along with the parameters mentioned above, the study related to Rasayana 31 assessed PSV (Peak systolic velocity) and EDV (end-diastolic velocity) ratios of ICA/CCA (Internal carotid artery / common carotid artery). It stated that significant results were observed in all these parameters.
Safety of Intervention
The analysis revealed that 30% of the studies (n = 09) reported adverse events (AEs) data.14,18,20,25,28,31,37,39,40 Among these, one study (P.K.N. Namboodiri et al) 18 documented a serious adverse event (SAE), specifically a death due to cardiac arrest during the one-year follow-up. Notably, details regarding patient intervention group and causality assessment of the SAE were not reported. The remaining eight studies indicated no AEs during the study period. Similarly, 33% (n = 10) of the studies included information on participant dropouts,17,18,22,34,37–42 with six studies reporting dropouts17,18,22,34,38,40 and four studies reporting no dropouts.37,39,41,42 In Sreeranjini et al, 2006, the dropout was due to the participant's inability to remain hospitalised for the entire study period. The remaining five studies did not provide explicit reasons for participant attrition.
Discussion
Summary of Main Results
The minimal divergence in efficacy between the study and control groups in most RCTs and non-randomised comparative trials suggests a lack of substantial differences in the interventions used. While single-group pre-post trials demonstrated encouraging outcomes with Panchakarma therapy or drug intervention, the genuine practical significance of these interventions can only be determined through proper RCTs. Many studies lacked methodological rigour, potentially impacting the reliability of their findings. A major challenge identified in the analysis was the lack of follow-up data, with 80% of the studies (n = 24) omitting this critical information. Among the six studies that did include follow-up data, four had follow-up periods of only 15 to 30 days, which is insufficient for evaluating a chronic condition like stroke. Additionally, some studies overlooked vital variables that could influence the outcomes, such as the stroke type (77%, n = 23) and stroke duration (63%, n = 19) in the inclusion criteria. Seventy per cent of the studies (n = 21) omitted crucial information regarding safety parameters like adverse drug reactions or adverse events. Similarly, sixty-seven per cent of studies (n = 20) did not include the data regarding dropout/withdrawal from the study. Another critical observation was using non-validated tools in 77% of studies (n = 23) for outcome measurement, highlighting the importance of employing validated tools to ensure the validity and reliability of the outcomes obtained. Hence, based on these published documents, arriving at a definitive conclusion regarding the overall effectiveness and safety of Ayurvedic treatments or protocols for Pakshaghata would be difficult.
Quality of Evidence
Except for one study, 14 of the included RCTs displayed methodological flaws, resulting in compromised quality of evidence. Most trials lacked sufficient details regarding randomisation, allocation concealment, and blinding, resulting in a high risk of bias. Only one study 16 was sourced from outside India, limiting the generalizability of the review findings. The absence of published protocols and clinical trial registry data hindered the risk of bias assessment. Similarly, all chosen non-randomised comparative trials were deemed ‘seriously biased’ due to confounding variables, lack of blinding, and the absence of a registered protocol or statistical plan.
Limitations
Despite our efforts to obtain unpublished trial data related to Hemiplegia/Pakshaghata from major Ayurveda postgraduate institutes, they could not provide any such information for our review. Consequently, we were unable to screen these grey literature sources. The generalizability of our review findings is constrained by the absence of data from populations outside of India and Sri Lanka. During the risk of bias assessment, we noted a lack of published protocols/trial registry data, which made it challenging to evaluate deviations from the intended intervention and selective reporting. Another significant limitation was our inability to conduct a Meta-analysis of the selected trials due to their methodological diversity and disparate outcomes, rendering quantitative analysis unfeasible.
Conclusion
Implication for Practice
This systematic review revealed a scarcity of high-quality studies addressing the treatment of Hemiplegia/Pakshaghata within Ayurvedic science. Even though Ayurveda physicians have been treating this disease, this review fails to produce robust and valid evidence to prove its efficacy and safety due to the dearth of quality research. Hence, the study outcomes described in the trials have limited external validity. From the above observations, the authors strongly recommend proper documentation of the efficacy and safety of current Ayurvedic practices in Hemiplegia/Pakshaghata.
Implication of Research
Evidence-based medicine (EBM) emphasises the necessity for substantiating effectiveness, efficiency, and safety in patient care, highlighting the considerable journey ahead for Ayurvedic researchers. This review has identified a significant knowledge gap in good quality evidence regarding the Ayurvedic management of Hemiplegia/Pakshghata. To address this gap, researchers should prioritise conducting high-quality RCTs focusing on Hemiplegia/Pakshaghata management. Essential elements for these studies include robust methodologies encompassing proper randomisation, adequate blinding, allocation concealment, validated and reliable outcome measurements, and comprehensive reporting. Ayurvedic researchers must adopt a multidisciplinary approach in stroke rehabilitation research. Evidence generated through these meticulous methods will enhance the scientific community's acceptance and justification of Ayurvedic treatment for Hemiplegia/Pakshaghata.
Footnotes
Acknowledgements
We would like to thank Vd. Prof. K. S. Dhiman, former Director General of CCRAS, for his guidance and support.
Author Contributions
NS and BCR conceptualised the project. ALM and PPN drafted the protocol, ALN, PPN, and DRN retrieved the literature, extracted, evaluated the risk of bias, and analysed the data. ALM wrote this original draft. PPN, and DRN edited the article. AA, BC, NS, and RA reviewed and edited the article. NS and RA have done project administration and Supervision. All authors have read and approved this manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This review was funded by the Central Council for Research in Ayurvedic Sciences (CCRAS), Ministry of AYUSH, Government of India (Grant No. 3-13/ 2019-CCRAS/Admn/IMR/Systematic Review/Hq 1).
Ethics Approval and Consent to Participate
Not applicable.
Availability of Data and Materials
The datasets used and analysed during the current study are available from the corresponding author upon reasonable request.
Patient and Public Involvement
Patients or the public were not involved in the design, conduct, reporting, or dissemination plans of our research.
Consent for Publication
Not applicable.
