Abstract
Background
Multiple sclerosis (MS) is a demyelinating disease of the central nervous system. Caffeine, as the most widely consumed psychoactive substance, has been suggested to have potential effects on the clinical course and disability levels of MS patients. This study aimed to review the current evidence on the effects of coffee/caffeine in patients with MS.
Methods
This study followed the Joanna Briggs Institute (JBI) Manual for Evidence Synthesis and PRISMA 2020 statement. Clinical evidence regarding the effects of caffeine/coffee in MS patients was considered. A systematic search was performed in PubMed, Scopus, Web of Science, and Embase in October 2023, and updated via handsearching in March 2024. JBI's critical appraisal tools were utilized to scrutinize the risk of bias.
Results
Out of 297 screened records, eight studies were eventually found to meet our inclusion criteria. The sample size of the studies varied between 12 and 1372 and the study designs were retrospective cohort, RCT, single-blind crossover trial, single-arm pilot study (each one study), and cross-sectional (four studies). No significant association between the level of disability and coffee/caffeine intake has been reported, although it was reported to be associated with cognitive improvements.
Discussion
Evidence indicates an association between coffee/caffeine consumption, and improved cognitive outcomes in patients with MS, while there is no considerable relationship with the disease disability. Considering the limitations of the evidence, such as the small number of studies, and great diversity in study designs, the findings of this study should translate to clinical practice with caution.
Introduction
Multiple sclerosis (MS) is a chronic inflammatory and demyelinating disease of the central nervous system (CNS) that typically appears in early adulthood. Based on the current knowledge, MS is most likely an autoimmune condition with demyelinating processes that affect the white and gray matter of the CNS. 1 About 85% of patients initially experience relapsing-remitting MS (RRMS), which can progress to secondary progressive MS (SPMS).2,3 The symptoms of MS are caused by the loss of myelin and depending on the location of the plaques, these symptoms can include numbness, visual abnormalities, brainstem symptoms, bladder dysfunction, paresis, ataxia, and a gradually escalating cognitive impairment. 4 Although there is no definitive cure for MS, pharmacological treatment protocols along with supplementation such as vitamins are minerals5–8 have been found to have favorable impacts on the symptoms and progression of MS. Caffeine, the most widely consumed psychoactive substance, is one of the suggested supplements with beneficial effects in MS. 9 After 20-30 min following intake, caffeine's plasma concentration reaches its peak and it acts on brain receptors because of its hydrophobic nature, which allows it to cross the blood-brain barrier. 10 It naturally occurs in coffee, tea, and cocoa, and improving exercise performance and cognitive status are suggested as the benefits of caffeine supplementation.11,12 Despite the scattered reports, the possible effects of caffeine/coffee on the disease course and symptoms in MS are not exhaustively reviewed. This study aims to comprehensively review the evidence concerning the impacts and safety of caffeine/coffee in patients with MS.
Methods
This systematic review was carried out in accordance with the techniques outlined in the Joanna Briggs Institute (JBI) Manual for Evidence Synthesis and the Preferred Reporting Item for Systematic Review and Meta-analysis (PRISMA 2020) standards.13,14
Eligibility Criteria
Specific criteria were defined to choose eligible articles for inclusion. The following were the inclusion criteria: (1) Clinical studies of any design (including both observational and interventional studies) that investigated the effect of coffee consumption or caffeine supplementation on MS, regardless of the disease phenotype, with a physician diagnosis, participation in an MS clinical trial, or enrollment from an MS specialty clinic that presupposes a physician diagnosis; (2) The study needs to be an empirical study using unique data. (3) The articles must be peer-reviewed and published in peer-reviewed journals. (4) Articles published in English. Conference papers, publications focused on the effects of coffee or caffeine on animal models of MS, and in vitro investigations were not considered for inclusion.
Information Sources, Search Strategy
In October 2023, an electronic search was conducted in PubMed, Web of Science, Scopus, and Embase. No filters were applied for the search, and a manual search based on the included references and citations was also conducted for a comprehensive coverage of the published studies in March 2024. The full search strategies for all databases are presented in Table 1.
Search Strategies.

Selection and Risk of Bias Assessments
EndNote software version 21 was used to remove duplicate records and management of the citations. Two researchers (S.S. and M.B.) independently reviewed the titles and abstracts for eligibility based on inclusion criteria using Rayyan.ai. 15 The final articles were chosen by two authors (S.P. and A.N.) after a second screening process based on the full texts. Any discrepancies were resolved through discussion between the two reviewers. In cases where a consensus could not be achieved, a third author (S.S.) was consulted at each stage. In addition, two authors (S.P. and A.N.) independently assessed the quality of the studies included in the analysis using the JBI critical appraisal tools. 16 Discrepancies were resolved through discussion between the two authors or by a third opinion (M.T.). The JBI critical appraisal checklists are utilized to assess the quality and risk of bias in different study designs. The checklist for cohort studies 17 evaluates 11 key items, including the similarity of compared groups, validity of exposure and outcome measurements, identification and handling of confounding factors, completeness of follow-up, and appropriateness of statistical analysis. A higher number of “Yes” responses indicates a lower risk of bias and higher quality. For randomized controlled trials, the 13-item JBI critical appraisal checklist 16 focuses on aspects such as the randomization process, allocation concealment, blinding, baseline similarities, completeness of follow-up, and statistical analysis. JBI critical appraisal checklist for analytical cross-sectional studies 17 assesses 8 criteria related to the study population, exposure and outcome measurements, confounding factors, and statistical analysis. Finally, the JBI critical appraisal checklist for quasi-experimental studies 13 evaluates 9 items, including the clarity of cause and effect, similarity of comparison groups, presence of a control group, and reliability of outcome measurements. These tools provide a structured approach to critically appraise the methodological quality and risk of bias in the included studies.
Data Extraction and Method of Synthesis
One of the authors (S.B. or M.B.) extracted the data from each article using a designated data collection table in Microsoft Excel 2019. The extracted data was then reviewed by another author (E.G-K or S.P). The information obtained from each included first author and year of publication, study design, study setting (eg, hospital, specialty clinic, community-based), sample size, participant characteristics (age, sex (percentage of female participants), disease phenotype), disability level based on the Expanded Disability Status Scale (EDSS), intervention or exposure details, control methods, outcome assessment scales used (eg, cognitive tests, fatigue scales, disability measures, serum markers), and main findings of the study. This detailed information extracted from the included studies was used to narratively synthesize the results and draw conclusions about the effects of caffeine and coffee consumption on different aspects of the disease. Considering the significant diversity between the studies, meta-analysis was not feasible.
Results
Search Results
The initial search yielded 605 articles, of which 297 remained for further progress after duplicate removal. Sixteen studies were primarily included through the title/abstract screening process, and eight articles were ultimately included based on the full-text evaluation.18–25 One of the excluded studies used a combination of histamine and caffeine, 26 two studies contained no caffeine or coffee-related variables,27,28 four studies did not evaluate the effects of caffeine/coffee supplementation on MS patients,29–32 and one study had not provided the required information regarding efficacy and safety, 33 so it was excluded in the full-text stage (Figure 1).
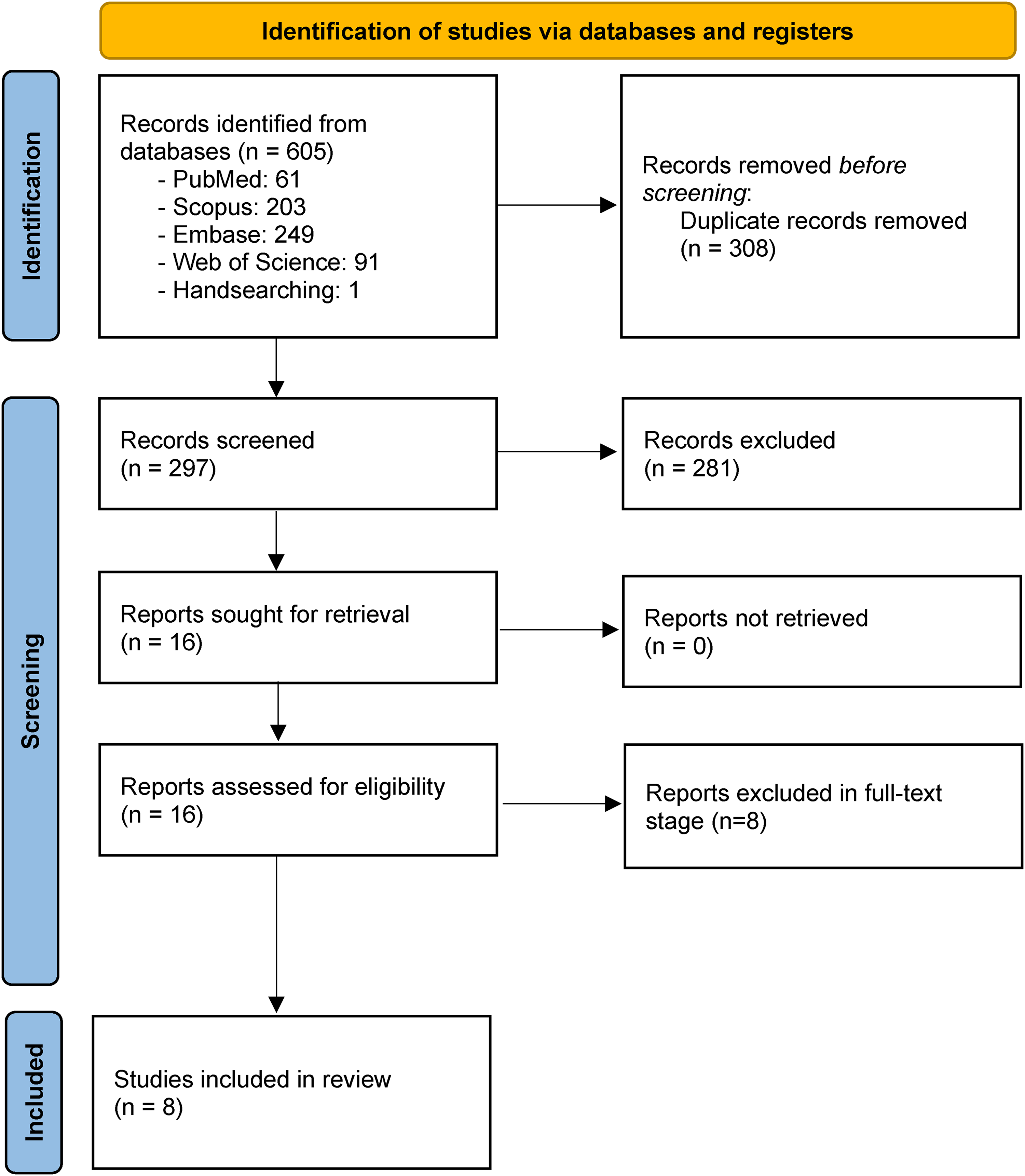
PRISMA flow diagram. From: Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021;372:n71. doi: 10.1136/bmj.n71. For more information, visit: http://www.prisma-statement.org/.
Characteristics of Included Studies and Risk of Bias Assessments
The selected studies comprised one RCT, one retrospective cohort study, four cross-sectional studies, one single-blind crossover trial, and one single-arm pilot study. The included studies reported either outcomes of coffee/caffeine supplementation compared to placebo groups and their respective baseline status, or its association with the disease status. Table 2 is a summary of the included studies and the detailed results of risk of bias assessments are presented in Supplemental material 1. Identification and dealing with confounders were found to be the most common possible source of bias in the included studies.
The Characteristics and Findings of the Included Studies.

MS: multiple sclerosis; FSS: Fatigue Severity Score; ESS: Epworth sleepiness scale; EDSS: Expanded Disability Status Scale; GSC: guarana, selenium and L-carnitine, MMSE: mini-mental state examination, CVLT-II: California Verbal Learning Test, BDI-II: Beck Depression Inventory-II, 9-HPT: Nine-hole peg test, T25-FW: Timed 25-foot walk, SDMT: Symbol digit modalities test; RSVP: Rapid Serial Visual Presentation; oxDNA = oxidized DNA; IL-1β = interleukin-1 beta; IL-6 = interleukin-6; TNF-α = tumor necrosis factor-alpha; IFN-γ = interferon-gamma; IL-10 = interleukin-10; RCT: randomized controlled trial; MSWS-12: 12-item Multiple Sclerosis Walking Scale, BBS: Berg Balance Scale, TUG: Timed Up-and-Go, MSIS-29: Multiple Sclerosis Impact Scale, PGIC: Patients’ Global Impression of Change; QoL: Quality of life. Ø as one of the secondary outcomes of the study, this study investigated the effects of coffee consumption for 12 weeks.
Results of Individual Studies
A study by Herden & Weissert 18 involved a cohort of 124 MS patients sampled from a specialty MS clinic in Germany over 6 months. Participants’ ages ranged from 18 to 80 years-old and the majority were females (63.7%). Coffee intake was assessed based on the number of cups consumed per day, with the average intake being 2.5 cups. Fatigue levels were evaluated using the Fatigue Severity Scale (FSS). EDSS was utilized to assess the disability levels, sleep quality was investigated using the Epworth Sleepiness Scale (ESS), and cognitive abilities was assessed through subjective evaluations. The results found no difference in coffee consumption between patients who reported fatigue (FSS ≥ 4) compared to those without fatigue. Coffee intake also did not differ based on disability levels assessed by EDSS, sleep quality measured by ESS, presence of bladder voiding disorders, or even late coffee consumption. However, higher coffee intake was positively associated with improved subjective concentration and attention span, particularly in patients with milder disability stages (EDSS 0-4). Overall, in this study, coffee consumption did not appear to have any negative effects and was linked to better perceived cognitive abilities.
In a single-blind crossover trial, Sharma et al 19 investigated the effects of caffeine (100 mg caffeine tablets, with no milk, and without sweetener) abstinence on cognitive performance in 12 MS patients (including 7 females, 55-70 years old) from the North Bristol NHS Trust's MS service over 9 days. Cognition was assessed using choice reaction time, Stroop test, and Rapid Serial Visual Presentation (RSVP) paradigm at baseline and after 7 days of caffeine abstinence. The results showed caffeine abstinence was associated with an increased number of errors made on the Stroop task. However, there was no effect on response latency. No impact of caffeine abstinence was seen on the RSVP task performance. Interestingly, performance improved across testing sessions, which could be due to practice effects or caffeine abstinence. Additionally, analyses found no changes in performance on simple reaction time and Stroop tasks after abstaining from caffeine for a week, suggesting that short-term caffeine abstinence does not affect performance on basic cognitive tasks in MS. Results of this study also indicated that caffeine may benefit patients under conditions requiring high attention, such as the dual-task version of RSVP and suggested the long-term positive impacts of caffeine on cognition in MS could become evident after prolonged periods of abstinence.
Ivashynka et al, 20 in a cross-sectional study, investigated the association between coffee consumption and MS severity in 208 MS patients (including 66.8% female patients and 87.0% RRMS) from the University Hospital in Novara, Italy, with a median age of 42.8 years. MS severity was assessed using the EDSS and Multiple Sclerosis Severity Score (MSSS). Coffee intake was measured based on number of cups consumed per day. The results found no correlation between MSSS scores and whether patients were coffee consumers. MSSS scores were also not related to the number of cups consumed per day or total cup-years of coffee intake. The odds ratios for having higher MSSS scores in the upper tertile of the distribution were 1.30 for those drinking 1-3 cups of coffee daily and 1.14 for those with 4-8 cups daily, compared to non-consumers.
In a cross-sectional survey by M. B. D’hooghe et al, 21 the association between coffee consumption and disability progression was investigated in 1372 MS patients from the Flemish MS Society in Belgium over 3 months. The sample included 893 patients with RRMS and 479 with progressive MS. Mean participant age was 50 (17-85) for RRMS and 59 (23-89) for progressive MS. Also, the female ratio was 75.6% (675) and 62.2% (298) at RRMS and progressive MS, respectively. Patients with EDSS of higher than 6 were 321 (35.9%) and 383 (80%) in Relapsing and Progressive MS, respectively. Disability status was assessed using EDSS scores. Coffee intake was categorized as non-drinking, less than one drink weekly, one to seven drinks weekly, and two or more drinks daily. The results found that in the RRMS group, regular coffee drinkers had a lower risk of reaching an EDSS score of 6, compared to non-drinkers. However, in the progressive MS group, no association was observed between coffee consumption and the risk of reaching an EDSS score of 6. This study shows coffee intake may help slow the worsening of disability for RRMS patients.
A double-blinded randomized placebo-controlled clinical trial by Teixeira et al 22 examined the effects of guarana-supplemented cappuccino (GSC) on biomarkers of oxidative stress and inflammation in 28 RRMS sclerosis patients from Brazil over 12 weeks. Patients were randomly assigned to receive either GSC (combination of 200 mg of L-carnitine L-tartrate, 150 mg of dry guarana extract, and 25 μg of chelated selenium, mixed with 20 g of industrially produced cappuccino coffee) (n = 15) or placebo (containing cappuccino coffee without the supplement) (n = 13) supplementation. Patients consumed one sachet dissolved in hot water/milk daily. Disability was assessed using EDSS scores ranging from 0 to 5.5. As one of the secondary outcomes of the study, the authors reported an appropriate safety profile; however, no significant improvement was reported for the group receiving cappuccino coffee without the supplements.
Another pilot study by Segela et al 23 explored the relationship between caffeine intake and cognitive performance in 63 MS patients at the University of Miami. Participants were categorized into two groups based on their daily coffee consumption: zero to one cup (n = 30) and two or more cups (n = 33). Disability levels were assessed using the EDSS score and cognitive abilities were measured using Mini-Mental State Examination (MMSE) and, California Verbal Learning Test (CVLT-II) and Beck-Depressions- Inventory (BDI-II) scale was utilized for psychiatric assessments. The results showed that MS patients consuming two or more cups of coffee per day outperformed those having zero to one cup in terms of total learning score on the CVLT-II test of verbal memory. However, no differences were detected between groups on measures of processing speed or self-reported depressive symptoms.
A cohort study by Duarte et al 34 conducted in Porto, Portugal over 11 months, involved a total of 126 participants with an average age of 45.3 years, with a female ratio of 60.3%. Among the participants, 76 individuals (60.3%) reported consuming coffee. The disability progression and cognitive function were assessed by multiple scales, including EDSS, 9-Hole Peg Test (9-HPT), Timed 25-Foot Walk (T25-FW), and Symbol Digit Modalities Test (SDMT). Coffee consumption was recorded in terms of cups per day. The results found no significant association between EDSS scores and high coffee consumption or total coffee intake. Additionally, coffee consumption levels did not differ in performance outcomes in the 9-HPT, T25-FW, and SDMT cognitive assessments, suggesting that coffee intake may not influence disease progression or disability in MS.
A single-arm pilot clinical trial by Afsoon Dadvar et al, 25 consisted of a 2-week placebo run-in and a 12-week caffeine treatment (200 mg/day) in Zahedan, Iran, and 30 MS patients (including 23 RRMS patients, EDSS < 6, aged between 20-55 years, weight > 40 kg) have participated. The study measurements were assessed at baseline as well as after 2, 4, 8, and 12 weeks. 12-item Multiple Sclerosis Walking Scale (MSWS-12), Berg Balance Scale (BBS), Timed Up-and-Go (TUG), Multiple Sclerosis Impact Scale (MSIS-29), Patient's Global Impression of Change (PGIC) were assessed in this study. This study confirms the beneficial effects of caffeine supplementation in enhancing balance and functional mobility. Finally, this initial study suggests caffeine is an effective intervention in boosting MS patients’ ambulatory performance and quality of life which suggests future definitive placebo-controlled RCTs with larger sample sizes and longer follow-ups.
Results of Synthesis
Disease Severity and Disability Progression
A retrospective cohort study 18 and a cross-sectional study 20 found no association between daily coffee/caffeine intake and disability scores or overall disease severity in MS patients. However, a large cross-sectional survey 21 reported that regular coffee drinking was linked to a lower risk of reaching high disability scores in patients with RRMS, but not progressive MS. A cohort study 34 also did not find any effects of coffee/caffeine consumption on disability progression.
Cognitive Function
A single-blind crossover trial 19 showed short-term caffeine abstinence increased errors on the Stroop cognitive test in MS patients. A retrospective cohort study 18 found higher coffee intake was associated with subjectively better concentration and attention in MS patients. A small cohort study 23 reported better verbal learning and memory performance among MS patients with higher daily coffee consumption. However, two studies19,34 did not find significant effects of coffee/caffeine intake on other cognitive test measures.
Inflammation and Oxidative Stress
As one of the secondary outcomes, a randomized controlled trial 22 showed that coffee consumption is not associated with improved oxidative stress and inflammation markers.
Fatigue and Sleep Quality
A retrospective cohort study 18 found no relationship between daily coffee consumption and fatigue levels or sleep quality in MS patients.
Ambulatory Performance and Quality of Life
A primary single-arm pilot clinical trial 25 found enhancive effects of caffeine for functional mobility and quality of life in MS.
Discussion
Improving patients’ quality of life as well as preventing the progression of the disease, are the main goals of intervention in MS. 35 In addition to some non-pharmacological methods,36–39 the management of MS is mainly based on pharmacological interventions.40–42 In addition, integrative and complementary medicine methods such as acupuncture, diet therapy, and herbal medicine are also found to be associated with positive effects on MS patient's clinical condition.43–46 Based on the common belief regarding the -at least- safety of integrative medicine methods, even in case of lack of considerable benefits, there are concerns regarding the appropriate adherence to evidence-based practice principles in this regard, which may harm the effects of the disease management.8,47 This study was a try to review the latest evidence regarding the possible effects of coffee/caffeine in MS patients. Based on the findings of this study, there is limited evidence on this topic and the studies have a great level of diversity regarding the assessment methods and study design. The small sample size in the included studies is also another considerable limitation of the evidence. Despite some observed benefits, the recommendation for coffee/caffeine consumption in MS patients is not fully supported by clinical evidence and there is a need for future studies to reach an evidence-based conclusion in this regard.
Considering the great prevalence even in mildly disabled patients,48–51 and lack of effective pharmacological interventions for MS-related cognitive impairment, 52 this study shed light on the possible role of caffeine as a cognitive enhancer in MS patients which suggested future well-designed RCTs in this regard. These effects have previously been reported in other neurological conditions such as Alzheimer's Disease. 53 The anti-oxidative properties make caffeine an interesting topic for research studies. in this condition, more clinical research is needed to completely understand how and to what extent coffee and caffeine consumption might be considered as a potential treatment method. In addition, studies suggested that genetic polymorphisms, physical fatigue, and mental energy may influence the cognitive response to caffeine, 54 which may be the reason for conflicting results between the studies. A study by Herden & Weissert, 18 as the only available evidence regarding MS-related fatigue, could not detect a significant association in this regard. Considering the negative impact of fatigue in the daily practice of MS patients, 55 there is a need for more attention to conclude this correlation.
Coffee and caffeine are found to lower the risk of developing neurological conditions such as Parkinson's Disease. 56 The relationship between coffee consumption and the risk of developing MS has been investigated in several studies. A case-control study by Hedström et al examined data from two large independent studies and the results showed that compared to participants who reported no coffee consumption, the odds of MS were substantially reduced among those who reported high coffee consumption exceeding 900 ml daily. 57 Additionally, a case-control study by Dastoorpoor et al found that drinking coffee, as well as black and green tea, was associated with a reduced risk of developing MS, 58 providing further evidence for the potential protective effects of polyphenol-rich beverages like coffee and tea against MS. Pekmezovic and colleagues reported that coffee consumption was significantly more frequent among MS patients compared to healthy controls. 59 Moreover, a dose-response relationship was observed, with both the number of cups of coffee consumed per day and the duration of coffee consumption in years being positively associated with the risk of developing MS. 59 Assessment of the possible link between coffee/caffeine consumption and the risk of developing MS was not the goal of the current study and it is suggested to be investigated in more depth in future studies.
It should be noted at this point that coffee drinking should not be confused with caffeine use. A cup of coffee has considerably more substances than just caffeine. Even while caffeine has received the greatest attention, other substances have been found to have a major impact on the body. 60 Furthermore, the soothing impact of taking a coffee break may have a larger influence than previously thought. Coffee can be used to create a more ordered daily routine, and research into the psychological impact of a hot cup of coffee might be exciting. In this regard, investigations should compare the effects of caffeine, as caffeine pills have the same effect as coffee's caffeine content.
While the current systematic review provides a discussion of the existing evidence on the effects of coffee and caffeine in MS, the limitations of the available data highlight the need for a more robust research agenda in this field. To establish the causal effects of caffeine on specific MS-related outcomes, such as cognitive function, fatigue, and physical function, well-powered, placebo-controlled RCTs are needed. These studies should investigate the optimal dose, timing, and formulation (eg, coffee, tea, supplements) of caffeine that may confer benefits in MS patients. Moreover, the mixed findings across the included studies underscore the need for high-quality, longitudinal cohort studies that can prospectively evaluate the relationship between coffee/caffeine intake and the risk of developing MS, as well as the impact on disease course and disability progression. These studies should utilize standardized, objective assessments of caffeine exposure and comprehensive adjustment for potential confounding factors. Given the potential variability in individual responses to caffeine, studies should investigate the role of genetic polymorphisms, as well as other host factors (eg, sex, age, disease phenotype, comorbidities) that may modify the relationship between caffeine and MS-related outcomes.
The included studies in this systematic review may be subject to several potential sources of bias that should be considered when interpreting the findings. Several studies relied on self-reported coffee/caffeine intake, which is susceptible to recall bias. Participants may not accurately report their true consumption levels, introducing measurement errors. More objective assessments of caffeine exposure, such as plasma or urinary caffeine levels, would provide a more robust measure. Confounding factors are other important source of bias in the included studies. Many did not adequately control for factors that could influence both coffee/caffeine consumption and MS outcomes, such as lifestyle factors, comorbidities, and medication use. Residual confounding may have obscured true associations. Finally, the small sample sizes and heterogeneity in study designs, populations, and outcome measures limit the ability to draw firm conclusions and synthesize the evidence. The paucity of high-quality, large-scale studies on this topic underscores the need for more robust research to clarify the potential role of coffee and caffeine in MS.
Conclusion
The evidence suggests that individuals with MS should not experience any negative effects from consuming a moderate amount of caffeine and it can be associated with cognitive improvements in these patients. Further research, including well-designed prospective studies, is needed to clarify the potential role of coffee and other dietary factors in the management of MS.
Supplemental Material
sj-docx-1-chp-10.1177_2515690X241293114 - Supplemental material for The Effects of Coffee/Caffeine in Patients with Multiple Sclerosis; A Systematic Review
Supplemental material, sj-docx-1-chp-10.1177_2515690X241293114 for The Effects of Coffee/Caffeine in Patients with Multiple Sclerosis; A Systematic Review by Shabnam Salekzamani, Saman Baharomid, Sina Pakkhesal, Maryam Balafkandeh, Elnaz Gholipour-Khalili, Mahnaz Talebi, Sarvin Sanaie and Amirreza Naseri in Journal of Evidence-Based Integrative Medicine
Footnotes
Acknowledgments
The research protocol was approved and supported by Tabriz University of Medical Sciences (grant number: 73003). This study is registered in the International Prospective Register of systematic reviews (PROSPERO ID: CRD42023482359). We would like to thank the Clinical Research Development Unit of Tabriz Valiasr Hospital, Tabriz University of Medical Sciences, Tabriz, Iran for their assistance in this research.
Author Contributions Statement
S.S., S.B., S.P., M.B., E.G-K., and A.N.: systematic search; study selection, data extraction, risk of bias assessment, preparing the figures, drafting the manuscript; S.S. and M.T.: conceptualization; supervision and critically editing the manuscript. All authors approved the final version for submission.
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research protocol was approved and supported by Tabriz University of Medical Sciences (grant number: 73003).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
