Abstract
The aim of this study was to evaluate the effect of curcumin combined with
Introduction
Head and neck cancer is the sixth most common type of cancer worldwide and comprises a diverse group of tumors from the oral cavity and upper aerodigestive tract. 1 More than 90% of oral cancers are oral squamous cell carcinoma (OSCC). 1 The main risk factors for OSCC are the consumption of tobacco and alcohol, while other possible risk factors are high-risk human papilloma virus infection, poor oral hygiene, and chronic inflammation.1,2 Despite progress in the treatment of OSCC, the 5-year survival rate of later-stage OSCC (approximately 20-30%) has not improved.3,4 Hence, investigating better therapeutic and preventative methods for OSCC is needed. 4 Apoptosis (a form of programed cell death) is the main mechanism by which senescent or genetically altered cells are eliminated in multicellular organisms. 5 Emerging evidence suggests that carcinogens cause genetic alterations in molecules that play important roles during apoptosis in various cancers, including OSCC. 5 Downregulation of proapoptotic proteins (such as caspases, Bcl-2-associated X protein (Bax), Bcl-2-antagonist/killer (Bak)) and upregulation of antiapoptotic proteins (such as B-cell lymphoma 2 (Bcl-2), B-cell lymphoma-extra large (Bcl-xL), myeloid cell leukaemia-1 (Mcl-1)) may promote prolonged cell survival, apoptosis inhibition, chemoresistance, and poor prognosis in OSCC.6,7 Thus, finding targeted therapies that can inhibit antiapoptotic proteins and induce apoptosis to treat OSCC is crucial.5,7
Probiotics are nonpathogenic live microorganisms that, when administered in appropriate amounts in food or drink, exert beneficial effects on human health.
8
Curcumin, a major active constituent of turmeric (
To our knowledge, few studies have reported that LGG CFS is effective in the suppression of human CRC and GC cells.12,22 However, no study has determined the anticancer activity of LGG CFS in OSCC cells. Nevertheless, investigating the effects of the combination of LGG CFS with other anticancer natural products, such as curcumin, could offer a novel therapeutic approach to improve their activities. Thus, the aims of this study were to evaluate the effects of curcumin and LGG CFS administered alone or in combination on cell proliferation and apoptosis induction in SCC-9 OSCC cells.
Materials and Methods
Cell Culture
The human OSCC cell line SCC-9 and the primary human gingival fibroblast (HGF) cell line PCS-201-018 were purchased from The American Type Culture Collection (ATCC, Manassas, VA, USA). SCC-9 cells were cultured in a mixture of Dulbecco's modified Eagle's medium (DMEM) and Ham's F12 medium (1:1, DMF12) supplemented with 10% v/v foetal bovine serum (FBS), 100 units/ml penicillin and 100 µl/ml streptomycin. The HGF cells were maintained in DMEM supplemented with 10% v/v FBS, penicillin 100 units/ml, and streptomycin 100 µl/ml. DMEM, DMF12, FBS, and antibiotic reagents were purchased from Gibco (Waltham, MA, USA). SCC-9 and HGF cells were incubated at 37°C in a humidified incubator with 5% CO2.
Curcumin Reagent
To prepare a stock reagent, curcumin (Sigma‒Aldrich, Deisenhofen, Germany) was dissolved in dimethyl sulfoxide (DMSO) (Sigma‒Aldrich) at a concentration of 1 mg/ml and filtered through a 0.22 µM membrane. The curcumin reagent was stored in single-use aliquots at −80°C until use.
LGG Culture and LGG CFS Preparation
The probiotic LGG (ATCC 53103) was purchased from ATCC and grown on de Man, Rogosa, and Sharpe (MRS) agar (Becton, NJ, USA) at 37°C under 5% CO2 for 48 h. Forty-eight-hour colonies of LGG were inoculated in 5 ml of MRS broth and incubated at 37°C under 5% CO2 for 48 h. The bacterial solution was adjusted to an optical density of 2.0 at 600 nm, corresponding to bacterial numbers of 108 CFU/ml, as determined by plate counting on MRS agar. The bacterial solution was further diluted to concentrations of 107, 106, and 105 CFU/ml. All four bacterial dilutions (105-108 CFU/ml) were centrifuged at 1100 × g for 10 min at 4°C. The protein concentration of LGG CFS was measured by a Coomassie Plus (Bradford) assay kit (IL, USA) to standardize the CFS in every batch prepared. The protein concentrations of CFS collected from 105–108 CFU/ml LGG were 166.5 ± 2.4, 167.4 ± 3.6, 167.5 ± 2.8, 154.5 ± 3.6, and 151.5 ± 2.4 µg/ml, respectively. The 25% (v/v) LGG CFS used for cell treatment was prepared following the procedure reported in the study of Escamilla et al 11 Briefly, 2.5 ml of CFS from each 105–108 CFU/ml LGG was suspended in serum-free DMEM to a final volume of 10 ml. The final pH of each supernatant was adjusted to 7.3 with 1 N NaOH. The 25% LGG CFS was filtered through a 0.22 µM membrane for sterilization and to remove the remaining bacteria and debris before storage at −80°C until use.
MTT Assay for Measurement of Cell Viability
The effects of curcumin and LGG CFS on the viability of SCC-9 cells were assessed by MTT assay to screen for the appropriate concentration for subsequent experiments. The dye 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) (Elabscience Biotechnology Inc, Texas, USA) was used to confirm the proliferative response. In brief, SCC-9 cells (5 × 103 cells/well, 10th – 15th passages) were seeded into 96-well plates and incubated at 37°C under 5% CO2 in a humidified incubator for 24 h. The culture medium was discarded, and the cells were separately treated with 5, 10, 20, 40, and 80 µg/ml curcumin and 25% (v/v) LGG CFS (at 105-108 CFU/ml) for 24 and 48 h. DMEM/FBS with 0.08% DMSO and 25% (v/v) MRS broth were used as the controls for the curcumin assay and LGG CFS assay, respectively. The culture medium was then discarded, and 100 µl of fresh DMEM without FBS was replaced in each well. The MTT dye was added to the wells (50 µl each), and the plate was incubated at 37°C for 4 h. The supernatant was removed, and 150 µl of DMSO was added to each well to dissolve the purple formazan product. The absorbance values were measured at a wavelength of 570 nm by a Synergy H1 hybrid multimode microplate reader (BioTek Instruments Inc., VT, USA). All cell viability assays were conducted in triplicate in three separate experiments. The percentage of living cells was calculated using the following formula: cell viability (%) = (absorbance of experimental well/absorbance of control well) × 100%. Based on the results from the MTT assay, the appropriate conditions were selected for use in the subsequent experiments.
The selected curcumin and LGG CFS concentrations were then examined with SCC-9 and HGF cells in single and combined treatments for 24 h to clarify whether the abovementioned concentrations did not affect the normal HGF cells. DMEM/FBS was used as the control. The effect of treatment on viability was evaluated using the MTT assay as described above.
Annexin V-FITC Apoptosis Staining Assay
SCC-9 and HGF cells (10th – 15th passages) were separately grown at a density of 105 cells/well in 6-well plates to detect apoptosis. The cells were incubated at 37°C in a humidified incubator under 5% CO2 for 24 h. Following incubation, cells were separately treated with curcumin, LGG CFS, and combined curcumin-LGG CFS for 24 h. DMEM/FBS was used as the control. The percentage of apoptotic cells was determined by flow cytometry using the FITC Annexin V Apoptosis Detection Kit with PI (BioLegend Inc., CA, USA). Briefly, cells were collected, washed with cold 1X phosphate-buffered saline (PBS), and then resuspended in Annexin V binding buffer. Subsequently, the cells were stained with 3 µl of FITC Annexin V and 6 µl of propidium iodide solution and incubated for 20 min at room temperature in the dark. Then, 400 µl of Annexin V binding buffer was added to each tube. A total of 10,000 stained cells were immediately analysed using a CytoFLEX S flow cytometer (Beckman Coulter Inc., IN, USA). Data were obtained from three separate experiments.
RNA Extraction and Real-Time RT‒PCR to Detect the mRNA Expression of Bax and Bcl-2 in SCC-9 and HGF Cells
SCC-9 and HGF cells (10th – 15th passages) were separately seeded in 6-well plates at a density of 106 cells/well and incubated at 37°C in a humidified incubator under 5% CO2 for 24 h. The culture medium was discarded, and the cells were separately treated with curcumin, LGG CFS, and combined curcumin-LGG CFS for 24 h. DMEM/FBS was used as the control. The culture medium was then discarded, and the cells were washed with 1X PBS. An RNeasy Plus Mini Kit (Qiagen, Hilden, Germany) was used to extract total RNA from SCC-9 and HGF cells following the manufacturer's instructions. RNA (approximately 1 µg) was used as the template to synthesize the complementary DNA using the Maxime RT Premix Kit (Intron Biotech Inc., Seongnam, Korea).
The mRNA expression levels of Bax and Bcl-2 (shown as the Bax/Bcl-2 ratio) were assessed by real-time RT‒PCR using primer sequences obtained from the study of Spampanato et al 23 The primer sequences were as follows: β-actin forward, 5'-GAC TAC CTC ATG AAG ATC CT-3’ and reverse, 5'-GCT TGC TGA TCC ACA TCT GC-3’; Bax forward, 5'-CCA GCT CTG AGC AGA TCA TG-3’ and reverse, 5'-TGC TGG CAA AGT AGA AAA GG-3’; and BCL-2 forward, 5'-GAC TTC GCC GAG ATG TCC AG-3’ and reverse, 5'-CAG GTG CCG GTT CAG GTA CT-3’. The reaction was performed in a total volume of 10 µl. A total of 0.1 µg of cDNA was added to the master mix composed of 5 µl (5 units/µl) of iTaq universal SYBR green SuperMix (Bio-Rad Laboratories Inc., CA, USA) and 0.5 µl (0.5 μM) of each forward and reverse primer. The reaction was performed using the QuantStudioTM 5 Real-Time PCR System (Thermo Fisher Scientific Inc., MA, USA). The protocol for real-time RT‒PCR was an initial denaturation step at 95°C for 3 min, followed by 95°C for 30 s, 55°C for 30 s, and 72°C for 30 s. The expression of ß-actin was used as an internal control to normalize the expression data. Each experimental condition utilized triplicate wells. The data were obtained from three separate experiments.
Western Blotting for Determination of Bcl-2, Bax and Caspase-3 Proteins
The SCC-9 and HGF cells treated with curcumin, LGG CFS, and combined curcumin-LGG CFS were lysed with 40 µl RIPA lysis buffer (Thermo Fisher Scientific Inc., MA, USA) containing PMSF protease inhibitor. Separation of protein (40 µg) was conducted on 12% SDS-PAGE gels and subsequently transferred to polyvinylidene difluoride (PVDF) membranes (Millipore, MA, USA). The membranes were blocked with 5% bovine serum albumin (Thermo Fisher Scientific Inc., MA, USA) for 2 h. Then they were incubated overnight with a primary antibody against bcl-2, bax, caspase-3 and β-actin (Cell Signalling Technology Inc., MA, USA, 1:4000 dilution for all antibodies). The membranes were then washed with TBS-T and incubated with a secondary antibody (Cell signaling Technology Inc., MA, USA, 1:8000 dilution). The bands were developed with ECL western blotting detection reagents (Thermo Fisher Scientific Inc., MA, USA). The chemiluminescent signal was measured by Amersham ImageQuantTM 800 Western blot imaging systems (Cytiva Inc., MA, USA). The band density was then analysed using ImageJ software (US National Institutes of Health, MD, USA). The data were obtained from three separate experiments.
Analysis of the Apoptotic and Anti-Apoptotic Protein Levels in SCC-9 and HGF Cells Using Bio-Plex Pro RBM Apoptosis Assays Panel 3
For apoptotic and antiapoptotic protein measurements, SCC-9 and HGF cells treated with curcumin, LGG CFS, and combined curcumin-LGG CFS were harvested and lysed with lysate dilution buffer containing phenylmethylsulfonyl fluoride (PMSF) protease inhibitor. The quantitative expression of a proapoptotic protein (caspase 3) and antiapoptotic markers (Bcl-xL/Bak dimer and Mcl-1/Bak dimer) were analysed by Bio-Plex Pro RBM apoptosis assays Panel 3 (Bio-Rad Laboratories Inc, CA, USA). The reagents were prepared according to the manufacturer's instructions. Ten microlitres of blocking buffer, 30 µl of lysate samples, and 10 µl of capture beads were added to a 96-well plate and mixed well using a shaker at 850 rpm for 1 h at room temperature. The plate was washed 3 times with 1x assay buffer using a Bio-Plex Pro Wash Station (Bio-Rad Laboratories Inc., CA, USA). After washing, 40 µl of detection antibodies were added, and the samples were incubated using a shaker at 850 rpm for 1 h. Twenty microlitres of streptavidin-phycoerythrin was added for 30 min of incubation at room temperature. Bead-conjugated samples were washed, resuspended in 100 µl of assay buffer, and investigated using a Bio-Plex 200 Array Reader (Bio-Rad Laboratories Inc., CA, USA). Data were then analysed with Bio-Plex Manager Software Version 6.0 (Bio-Rad Laboratories Inc, CA, USA). The experiments were performed in triplicate wells. The results are shown as the observed concentration (ng/ml).
LGG CFS Analysis Using Liquid Chromatography‒Tandem Mass Spectrometry (LC‒MS/MS)
Only limited data describing possible types of biological compounds in LGG CFS are available. To characterize the biological compound present in LGG CFS, the supernatant at a concentration of 106 CFU/ml was sent for analysis using the LC‒MS/MS technique at The Institute of Molecular Biosciences, Mahidol University, Thailand. Separation was conducted by LC‒MS/MS, TripleTOF 6600+, Sciex, USA. The injection volume was 5 µl. Separation was performed at a 1 ml/min flow rate under a gradient program in which mobile phase A was 0.1% formic acid (v/v) in water and mobile phase B was 0.05% formic acid (v/v) in acetonitrile. LC gradient elution was performed starting with 10% B increasing to 30% B over 20 min and then further increasing to 30% B over 30 min. The eluent was monitored by an electrospray ion mass spectrometer (ESI-MS) in positive ion mode and scanned from m/z 100 to 1250. ESI was performed using a needle voltage of 2300 V under an optimum collision energy level of 65%. Nitrogen was used as the drying gas at a flow rate of 7 l/min. The pressure of the nebulizer gas (nitrogen) was 40 psi. The gas temperature was kept constant at 350°C. Raw data were subjected to a Scanning SWATH Acquisition and OneOmicsTM system for peptide identification.
Statistical Analysis
Data are presented as the mean ± standard deviation (SD). Differences between groups were analysed using one-way analysis of variance (ANOVA) (for normally distributed data) or the Kruskal‒Wallis test (for nonnormally distributed data), followed by Dunn's multiple comparison test. GraphPad Prism version 9.0 for Windows (GraphPad Software Inc., CA, USA) was used for statistical analysis.
Results
Effects of Curcumin on the Viability of SCC-9 Cells
Curcumin decreased the viability of SCC-9 cells in a dose-dependent manner after 24 and 48 h. Treatment with curcumin at doses of 5, 10, and 20 µg/ml did not significantly affect the viability of SCC-9 cells. However, curcumin at 40 and 80 µg/ml significantly reduced SCC-9 cell viability (
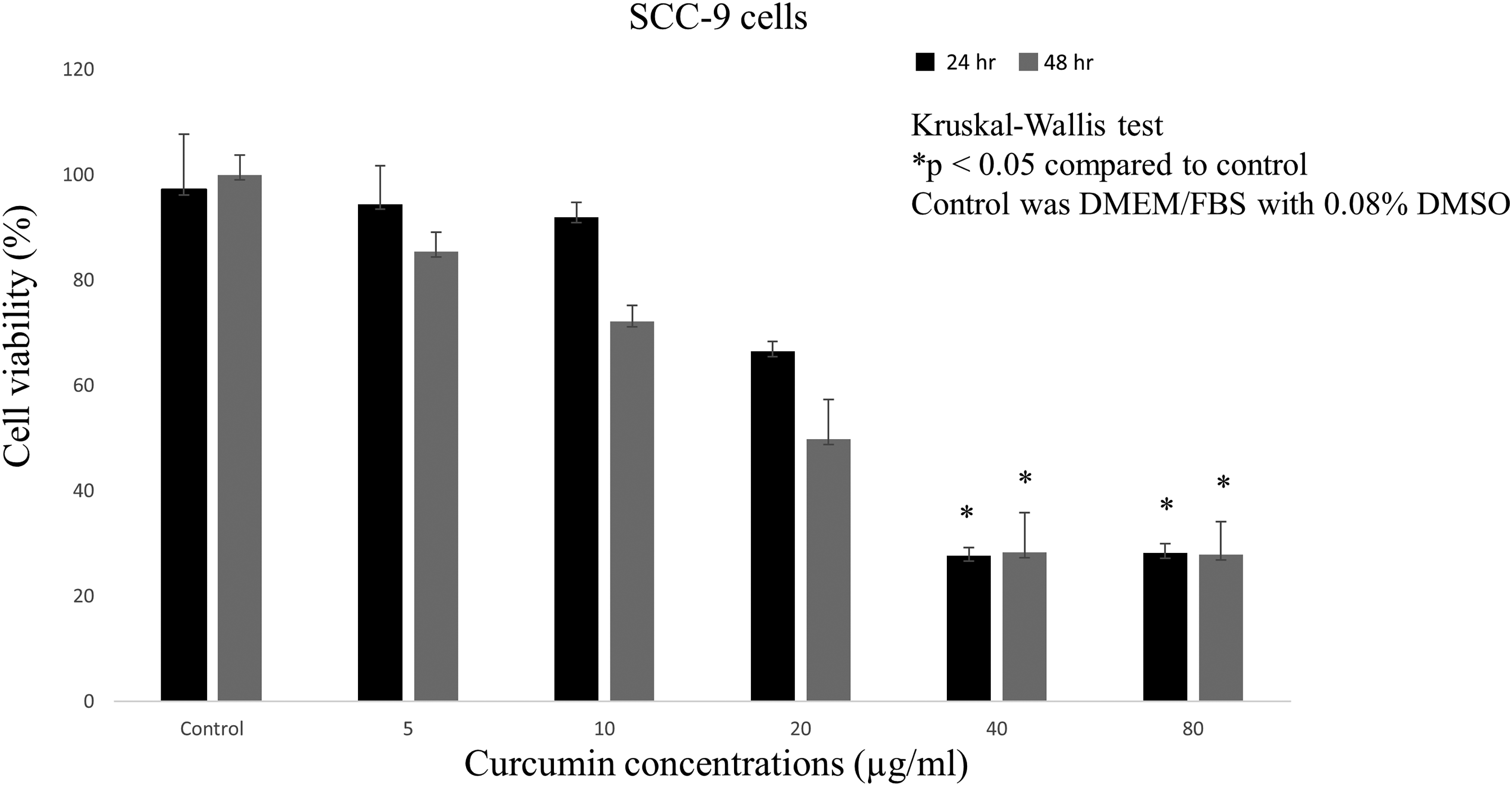
Effects of curcumin on the viability of SCC-9 cells. SCC-9 cells were treated with various concentrations of curcumin for 24 h (black) and 48 h (grey). Cell viabilities were determined by MTT assays. The data represent the result of three separate experiments (mean ± SD).
Effects of 25% LGG CFS on the Viability of SCC-9 Cells
Treatment of SCC-9 cells with 25% (v/v) LGG CFS at 108 CFU/ml significantly decreased the viability of SCC-9 cells after 24 and 48 h of incubation (

Effects of LGG CFS on the viability of SCC-9 cells. SCC-9 cells were treated with various concentrations of LGG CFS for 24 h (black) and 48 h (grey). Cell viabilities were determined by MTT assays. The data represent the result of three separate experiments (mean ± SD).
Effects of the Combined Treatments on the Viability of SCC-9 and HGF Cells
Curcumin-H (40 µg/ml) and LGG CFS-H (25% (v/v) LGG CFS 108 CFU/ml) administered alone significantly inhibited the viability of SCC-9 and HGF cells (

Effects of curcumin and LGG CFS on viability of SCC-9 and HGF cells. SCC-9 cells (dark blue) and HGF cells (light blue) were treated with curcumin at the concentration of 5 µg/ml (curcumin-L) and 40 µg/ml (curcumin-H) or with 25% v/v LGG CFS at the concentration of 106 CFU/ml (LGG CFS-L) and 108 CFU/ml (LGG CFS-H) or with the combination treatments (curcumin-L + LGG CFS-L, curcumin-L + LGG CFS-H, curcumin-H + LGG CFS-L, and curcumin-H + LGG CFS-H) for 24 h. Cell viabilities were determined by MTT assays. The data represent the result of three separate experiments (mean ± SD).
Effects of the Combined Treatments on SCC-9 and HGF Cell Apoptosis
To determine the type of cell death (apoptosis or necrosis) induced by curcumin-L + LGG CFS-L treatment in SCC-9 cells, an Annexin V-FITC kit and flow cytometry were used. Our results showed that the curcumin-L + LGG CFS-L caused a significant increase in the number of SCC-9 apoptotic cells after 24 h of incubation when compared with the control and the single treatments of curcumin-L and LGG CFS-L (

Effects of curcumin and LGG CFS on apoptosis of SCC-9 (A) and HGF (B) cells and the percentage of apoptotic SCC-9 and HGF cells (C) analysed by flow cytometry. Effects of curcumin at the concentration of 5 µg/ml (curcumin-L) or 25% v/v LGG CFS at the concentration of 106 CFU/ml (LGG CFS-L) or their combinations (curcumin-L + LGG CFS-L) on the apoptosis of SCC-9 (A) and HGF cells (B) after 24 h treatment (shown in the representative scatter plots). The percentage of the total (early and late) apoptotic SCC-9 and HGF cells were summarized in graph C. The data represent the result of three separate experiments (mean ± SD).
Bax/Bcl-2 mRNA Expression Ratio in SCC-9 and HGF-Treated Cells
Bax regulates cell death, while Bcl-2 promotes cell survival. Thus, we compared the Bax/Bcl-2 ratio in SCC-9 and HGF cells exposed to curcumin and LGG CFS. A higher ratio indicates more cell death. The single treatments of curcumin-L and LGG CFS-L did not raise the Bax/Bcl-2 mRNA expression ratio in either SCC-9 or HGF cells compared to the control (

Bax/Bcl-2 mRNA expression ratio in SCC-9 and HGF cells treated with curcumin and LGG CFS. Effects of curcumin at the concentration of 5 µg/ml (curcumin-L) or 25% v/v LGG CFS at the concentration of 106 CFU/ml (LGG CFS-L) or their combinations (curcumin-L + LGG CFS-L) on the Bax/Bcl-2 mRNA expression ratio after 24 h treatment. The data represent the result of three separate experiments (mean ± SD).
Bcl-2, Bax and Caspase-3 Protein Expression in SCC-9- and HGF-Treated Cells Analysed by Western Blot Analysis
A western blot confirmed a significant declining level of Bcl-2 protein in SCC-9 cells following treatment with curcumin-L + LGG CFS-L compared to that in the control and single curcumin-L and LGG CFS-L groups (
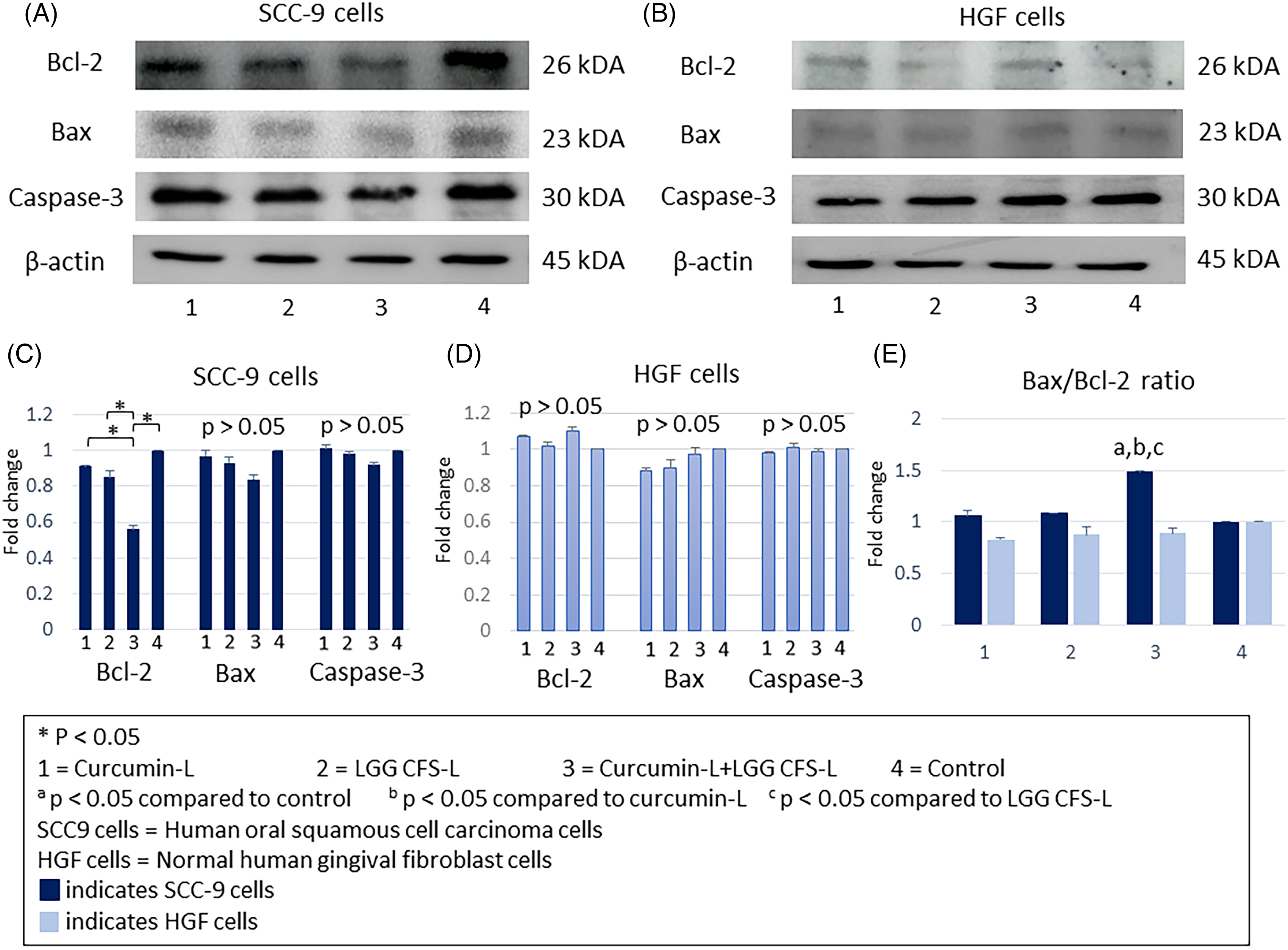
Western blot analysis of Bcl-2, Bax, and caspase-3 protein expression and Bax/Bcl-2 protein expression ratio in SCC-9 and HGF cells. SCC-9 and HGF cells were incubated with curcumin at the concentration of 5 µg/ml (curcumin-L) or 25% v/v LGG CFS at the concentration of 106 CFU/ml (LGG CFS-L) or their combinations (curcumin-L + LGG CFS-L) for 24 h. β-actin was used as a loading control. Western blotting results were quantified with ImageJ software. The data represent the result of three separate experiments (mean ± SD).
Caspase-3 levels, Bcl-xL/Bak Ratio and Mcl-1/Bak Ratio in SCC-9- and HGF-Treated Cells Analysed by Bio-Plex Pro RBM Apoptosis Assays Panel 3
The level caspase-3, the frequently activated death protease, increased in SCC-9 cells compared to HGF cells after treatment with curcumin-L + LGG CFS-L, but the difference was not significant (

Expression of caspase-3 level (A), Bcl-xL/Bak ratio (B), and Mcl-1/Bak ratio (C) in SCC-9 and HGF cells treated with curcumin and LGG CFS using Bio-Plex Pro RBM apoptosis assays Panel 3. SCC-9 and HGF cells were incubated with curcumin at the concentration of 5 µg/ml (curcumin-L) or 25% v/v LGG CFS at the concentration of 106 CFU/ml (LGG CFS-L) or their combinations (curcumin-L + LGG CFS-L) for 24 h. The protein was extracted and detected with the Bio-Plex Pro RBM apoptosis assays panel 3. The experiments were performed in triplicated wells.
Bcl-xL and Mcl-1 are among the cell survival-promoting proteins in the Bcl-2 family, while Bak regulates cell death similarly to Bax. Thus, the Bcl-xL/Bak and Mcl-1/Bak ratios indicate the extent of cell survival. The higher the Bcl-xL/Bak and Mcl-1/Bak ratios are, the more cells survive. However, there were no significant differences in the Bcl-xL/Bak and Mcl-1/Bak ratios between the treatment and control groups in both SCC-9 and HGF cells (
Protein Profiling of LGG CFS Identified by LC‒MS/MS
A total of 30 peaks were annotated in ESI positive mode. The biological compounds were classified into two major groups: metabolic enzymes and proteins (26 types of proteins) and surface layer proteins (4 types of proteins). These results are summarized in Figure 8.

Protein profiling of LGG CFS at the concentration of 106 CFU/ml through LC–MS/MS. The biological compounds were classified into two groups: metabolic enzymes and proteins (26 types of proteins) and surface layer proteins (4 types of proteins).
Discussion
Here, we explored the ability of curcumin and LGG CFS to inhibit SCC-9 OSCC cells
LGG (ATCC 53103) has been proclaimed to be the first probiotic studied in cancer.
26
This bacterium has been used in several experimental designs to test its usefulness in anticancer therapeutic practice.
9
In this study, we found that 25% v/v LGG CFS at a concentration of 108 CFU/ml significantly decreased SCC-9 cell viability. Some previous studies have reported that LGG (live, lyophilized, and in its supernatant form) has anticancer activities in various cancer cells.
9
Both live and lyophilized LGG at a concentration of 108 CFU/ml could increase chemokine (c motif) ligand (XCL1) in bladder tumor-bearing mice, suggesting that XCL1 may act as a chemoattractant for T and NK cells and induce tumor regression.
27
Furthermore, synthesized silver-LGG nanoparticles significantly inhibited CRC cell proliferation, suggesting that LGG treatment could inhibit CRC cell proliferation by inducing apoptosis.
28
In comparison, viable LGG at a low concentration of 103 CFU/ml did not inhibit the viability of HSC-3 OSCC cells.
13
LGG CFS could decrease MMP-9 but increase ZO-1 protein levels, resulting in an antimetastatic effect in CRC cells.
12
Taken together, these results suggested that 25% (v/v) CFS from LGG (108 CFU/ml) in serum-free DMEM had anticancer potential against SCC-9 OSCC cells
Recently,
The experiments in the present study attempted to evaluate the effects of curcumin and 25% v/v LGG CFS on cell proliferation and apoptosis in cancer cells using SCC-9 as a model. Our study demonstrated that when high concentrations of either curcumin or 25% v/v LGG CFS were administered alone to SCC-9 and normal HGF cells, significant inhibition of proliferation was observed after 24 h of treatment, suggesting potential side effects to normal cells. High curcumin concentrations (≥ 20 µM) were previously found to induce a decrease in the viability of normal dermal fibroblast cells, 31 which is in line with our study. In contrast, the present study showed that curcumin and 25% v/v LGG CFS did not inhibit cell proliferation in either cell line at low concentrations. However, the combination of low concentrations of curcumin and 25% v/v LGG CFS showed a significant inhibitory effect on SCC-9 cell viability but not on HGF cell viability. Combining curcumin and 25% v/v LGG CFS at reduced doses might induce an additive antiproliferative effect with limited toxicity. 32 The precise mechanism of combining curcumin and 25% v/v LGG CFS at low doses in OSCC cells remains to be investigated in future studies.
Several studies have reported that curcumin can exert its antitumor effect by inducing cell cycle arrest, leading to apoptosis.15,17 Flow cytometry analysis revealed that curcumin significantly induced apoptosis in OSCC cells in a dose-dependent manner in previous studies, in which higher concentrations favored late apoptosis over early apoptosis.33,34 Live LGG and LGG CFS have been reported to play a role in decreasing the viability of cancer cells and inducing cancer cell apoptosis. 9 Dehghani et al reported that LGG CFS at a concentration of 0.25 mg/ml significantly enhanced the number of HT29 CRC apoptotic cells compared to untreated cells. 35 Here, we found that treating SCC-9 cells with the combination of low concentrations of curcumin and 25% v/v LGG CFS significantly increased the number of early and late apoptotic cancer cells compared to their single treatments. Moreover, the combination treatment did not increase the number of apoptotic HGF cells. These findings suggested that combined curcumin and LGG CFS treatment at reduced doses might trigger apoptosis in SCC-9 cells but have no effect on HGF cells.
Levels of apoptosis-related proteins are altered in several cancers, including OSCC.
7
Induction of apoptosis has appeared to be a beneficial approach to overcome oral cancer by targeting and eliminating OSCC cells.5,7 Several studies have shown promising results regarding oral cancer cell apoptosis induced by various biological and chemical interventions.36,37 Tumor cells induce the upregulation of antiapoptotic proteins to acquire resistance to apoptosis.38,39 Bcl-2 binds to Bax, thereby inhibiting apoptosis.
40
The effects of apoptosis induction are more dependent on the Bax/Bcl-2 ratio than on the quantity of Bax or Bcl-2 alone.
41
The antiapoptotic proteins Bcl-xL and Mcl-1 heterodimerise with Bak at the mitochondrial outer membrane, thus inhibiting cytochrome c release from the mitochondria.
42
Therefore, the ratios of Bcl-xL/Bak and Mcl-1/Bak dimers could represent cell survival.
42
Here, we found that treating SCC-9 cells with the combination of curcumin and 25% v/v LGG CFS at low concentrations significantly increased the Bax/Bcl2 ratio compared to their single treatments. In contrast, this combination did not increase the Bax/Bcl2 ratio in HGF cells, suggesting a selective effect on the tumor and less toxicity on normal gingival fibroblast cells. Cellular uptake of curcumin and LGG CFS may be higher in tumor cells than in normal cells.
43
Additionally, curcumin probably modulates the growth of tumor cells by regulating multiple cell signaling pathways, such as the cell proliferation pathway, cell survival pathway, caspase activation pathway, tumor suppressor pathway, death receptor pathway, mitochondrial pathways, and protein kinase pathway.
43
Various curcumin concentrations significantly upregulate the expression of the Bax protein, downregulate the expression of the Bcl-2 protein, and increase the Bax/Bcl-2 ratio in HNSCC and OSCC cell lines.34,44,45 Live LGG treatment could inhibit CRC cell proliferation by inducing apoptosis by increasing the Bax/Bcl-2 ratio and the number of apoptotic cells after 48 h of treatment.
10
Moreover, live LGG treatment has significantly reduced CRC incidence, multiplicity, and volume
Escamilla et al reported a possible bioactive compound in LGG CFS using CFS size fractionation. Treating human colorectal carcinoma cells (HCT-116) with 50–100 kDa and > 100 kDa fractions of LGG CFS resulted in a significant decrease in cancer cell invasion. The authors suggested that the bioactive component within the LGG CFS might be a large molecular weight compound such as a protein, nucleic acid, or polysaccharide.
12
In this study, protein profiling of LGG CFS at a concentration of 106 CFU/ml was carried out using LC‒MS/MS. The main components of the LGG CFS were metabolic enzymes and proteins and surface layer proteins. Previously, a
Finally, the effects of curcumin combined with chemotherapeutic drugs have been reported in several
Conclusions
In the present study, the antiproliferative and apoptosis-inducing effects exerted by the coadministration of curcumin and LGG CFS against SCC-9 OSCC cells were observed. Moreover, the combination of curcumin and LGG CFS at low concentrations did not reduce the viability of normal gingival fibroblasts. Further studies with additional OSCC cell lines and experimental models are needed to confirm our preliminary results. Moreover, the underlying molecular mechanisms of curcumin and LGG CFS in anti-cancer activities need to be further investigated.
Supplemental Material
sj-docx-1-chp-10.1177_2515690X241258369 - Supplemental material for A Combination of Curcumin and Lactobacillus rhamnosus GG Inhibits Viability and Induces Apoptosis in SCC-9 Human Oral Squamous Cell Carcinoma Cells
Supplemental material, sj-docx-1-chp-10.1177_2515690X241258369 for A Combination of Curcumin and
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Author Contributions
C.U. performed the laboratory experiments, analysed, and interpreted the data. P.R. conceptualized and designed the study, and did funding acquisition and project administration. CU and PR drafted and revised the manuscript for intellectual content. All authors read and approved the final manuscript.
Ethical Approval
Not involvement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
