Abstract
Objective
Oral squamous cell carcinoma is the most common malignant tumor occurring in the head and neck region. Current treatment principles are based on radical surgery, supplemented by radiotherapy and chemotherapy. However, the 5-year survival rate for patients remains approximately 50%. Therefore, further research into the molecular mechanisms underlying oral squamous cell carcinoma development is needed to identify more effective treatment methods.
Methods
In this study, immunohistochemical techniques were used to quantitatively analyze the expression levels of E-cadherin, vimentin, CD206, programmed cell death receptor 1 (PD-1), and programmed cell death ligand 1 (PD-L1) in 45 pathological sections of oral squamous cell carcinoma. The results showed that the expression levels of vimentin, CD206, and programmed cell death ligand 1 were significantly associated with overall survival. Additionally, Cox multivariate analysis indicated that the M2 macrophage marker, CD206, is an independent risk factor for poor prognosis in oral squamous cell carcinoma. Furthermore, correlation analysis revealed that E-cadherin expression was negatively correlated with vimentin, CD206, and programmed cell death ligand 1 expression levels. Vimentin expression was positively correlated with programmed cell death receptor 1 and programmed cell death ligand 1 expressions.
Results
Immunohistochemical examination indicated that M2 macrophages are an independent risk factor for poor prognosis in oral squamous cell carcinoma and are closely associated with overall survival. Furthermore, they may influence the development and progression of oral squamous cell carcinoma tumors by inducing epithelial–mesenchymal transition in oral squamous cell carcinoma tumor cells. Third, programmed cell death ligand 1 expression has an adverse impact on oral squamous cell carcinoma prognosis and is significantly correlated with the expression levels of CD206, E-cadherin, and vimentin.
Conclusions
This study indicates that programmed cell death receptor 1/programmed cell death ligand 1 and CD206 expressions are independent risk factors for poor oral squamous cell carcinoma prognosis; however, whether programmed cell death receptor 1/programmed cell death ligand 1 expression influences the occurrence and development of oral squamous cell carcinoma through M2 macrophages requires further investigation.
Keywords
Introduction
Oral squamous cell carcinoma (OSCC) accounts for 90% of head and neck squamous cell carcinomas (HNSCCs). 1 More than 140,000 patients worldwide die from the disease annually. Despite great progress in surgery, radiotherapy, and chemotherapy, the 5-year survival rate of OSCC patients has remained approximately 50% over the past 30 years, with no significant improvement. 2 Therefore, further research on the molecular mechanisms of OSCC formation and development is the key to the establishment of more effective therapeutic methods.
Various molecular biology studies have shown that the malignant behavior of tumors is regulated by the tumor microenvironment (TME). 3 Tumor-associated macrophages (TAMs) are the most abundant nonspecific immune cells in the TME and play a crucial role in tumor progression. 4 They can be divided into specific but intersecting subpopulations based on their polarized needs, phenotype, and functions. 5 Studies have shown that CD206 is mainly positively expressed in M2 macrophages, and its increased expression is related to poor prognosis in lung, breast, bladder, prostate, and endometrial cancers as well as other malignant tumors. It can enhance the ability of tumor cell invasion and metastasis by promoting angiogenesis and immunosuppression.1,6
The occurrence and development of various cancers involve a series of complex processes, including the enhancement of the ability of cancer cells to invade and metastasize. Epithelial–mesenchymal transition (EMT) not only participates in cell skeleton remodeling but also plays a role in the interaction between the tumor and host. 7 Cells that undergo EMT exhibit decreased expression of epithelial genes such as E-cadherin (E-cad) and increased expression of mesenchymal genes such as vimentin.8,9 Additionally, studies on hepatocellular carcinoma and pancreatic cancer have demonstrated that M2 macrophages can promote EMT, thereby enhancing the invasive ability of tumor cells.10,11 However, research on the mechanism of M2 macrophages in EMT in OSCC is still insufficient.
Immunotherapy has recently emerged as a promising therapeutic strategy in the treatment of various cancers. Studies have shown that programmed cell death receptor 1 (PD-1) and programmed cell death ligand 1 (PD-L1) are the main immune checkpoints in the development and progression of many human cancers. 12 Prognostic analysis of some cancers have shown that the PD-1/PD-L1 pathway can polarize M2 macrophages, resulting in EMT and affecting the invasion and metastasis abilities of malignant tumor cells such as pancreatic and renal cancer cells.13,14 Therefore, whether the PD-1/PD-L1 pathway can also affect OSCC invasion and metastasis through M2 macrophages warrants further research.
This retrospective cohort study aimed to investigate the expression characteristics and prognostic association of biomarkers, including E-cad, CD206, and PD-L1 in patients with OSCC.A total of forty-five patients with OSCC who had undergone surgery at the First Affiliated Hospital of Wannan Medical College from 2012 to 2014 were randomly selected. The expression levels of CD206, E-cad, vimentin, PD-1, and PD-L1 in each pathological section were quantified using immunohistochemical experiments, and the relationships among various markers were comprehensively analyzed to further understand the relationship between each index in OSCC and clinicopathological factors to identify novel strategies for the diagnosis and treatment of OSCC.
Materials and methods
Pathological specimens
From 2012 to 2014, we retrospectively and randomly enrolled 45 patients with OSCC who had undergone surgery at the First Affiliated Hospital of Wannan Medical College but had not undergone preoperative adjuvant therapy. The patients’ clinical data were collected and surgical pathological specimens were obtained from the pathology department of the hospital. Detailed information of all the selected patients has been removed. In this study, two experienced pathologists who were blinded to the patients’ clinical details and group assignments independently assessed all the slides. The tumor, node, metastasis (TNM) staging of OSCC followed the standards of the Union for International Cancer Control (UICC), and the pathological grading was based on the World Health Organization (WHO) 2005 classification, conforming to the principles of the 1975 Declaration of Helsinki (revised in 2024). The study was approved by the Ethics Committee of Wannan Medical College and followed the Enhancing the QUAlity and Transparency Of health Research (EQUATOR) guidelines. This study employs a cohort study design. 15 The reporting of this study complies with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Immunohistochemistry
After routine processing, the specimens were embedded in paraffin, sliced, dewaxed in xylene after being heated in an oven, hydrated with gradient ethanol, placed in a citrate buffer, and heated in a microwave oven to restore the antigens. Endogenous catalase was blocked with 3% hydrogen peroxide at room temperature. After blocking with goat serum, E-cad, vimentin, CD206, PD-1, and PD-L1 antibodies were added (USA Cell Signaling Technology) and incubated overnight at 4°C. All antibodies were sourced from Cell Signaling Technology, USA, as rabbit monoclonal antibodies, specifically, E-cad (3195S, 1:200 dilution), vimentin (5741S, 1:100 dilution), CD206 (24595S, 1:200 dilution), PD-1 (86163S, 1:100), and PD-L1 (13684S, 1:200). A secondary antibody was added to react with 3,3’–diaminobenzidine (DAB) in the dark for color development the next day. The color development time was 4 min for the mouse anti-E-cad antibody, 1 min for the rabbit anti-vimentin antibody, 3 min for the rabbit anti-CD206 antibody, 5 min for the rabbit anti-PD-1, and 5 min for the rabbit anti-PD-L1. After color development, they were counterstained with hematoxylin, mounted on slides, and scored under the microscope according to the scoring standards of each index.
Criteria for scoring the immunohistochemical results
E-cad and PD-L1 scoring criteria
According to the methods of Lima et al.,
16
E-cad was considered positive when the cytoplasm or membrane appeared clear yellow, and PD-L1 was considered mainly positive when there was deposition of brown-yellow substance in the cell membrane. The degree of immunostaining was determined according to the proportion of positively stained cells and the intensity of staining. Immune response score = intensity score × proportional score. The proportion of stained cells was scored as follows: 0 (no positively stained cells), 1 (<10% positively stained cells), 2 (10%–35% positively stained cells), 3 (35%–75% positively stained cells), and 4 (>75% positively stained cells). Staining intensity was graded according to the following scale: 0 (no staining), 1 (weak staining), 2 (moderate staining) and 3 (strong staining). The immune response score results were classified as 0, 1, 2, 3, 4, 6, 8, 9, or 12. An immune response score of 0 indicated negative expression, ≤4 indicated low expression, and >4 indicated high expression. The following criteria were used for defining high expression. Survival (Kaplan–Meier (K–M) method) and correlation analyses with clinical outcomes based on this cohort study data indicated that an immunoreactive score (IRS) >4 represented the optimal cutoff value for distinguishing prognostic differences among patients (log-rank
Vimentin scoring standard
When the cytoplasm appears brownish-yellow, vimentin expression is generally considered to be positive. 17 Four classifications of staining intensity were established based on the percentage of tumor cells that were favorably stained: 0 (0%), 1 (0%–5%), 2 (5%–20%), 3 (20%–50%), and 4 (>50%).
Criteria for grading CD206 and PD-1
According to the evaluation techniques reported by Hu Y et al. 18 and Naruse T et al., 19 CD206 is primarily positive when the cell membrane is stained with brownish yellow. To determine the number of positive cells, five high-power fields were chosen at random (400 times). The final result value for each sample was determined as the average of five fields of view. The samples were then classified into groups with high and low expressions, with the median of all samples’ expression numbers serving as the cutoff value.
Statistical data analyses
To investigate the link between E-cad, vimentin, CD206, PD-1, and PD-L1 expressions and the prognosis in OSCC, chi-square test was used to assess all raw data using Statistical Package for Social Sciences (SPSS) software (version 23.0). The K–M survival curve was then created, and Cox regression analysis was performed.
Results
Basic conditions of clinical patients
Of the 45 OSCC patients who were enrolled, 17 (37.78%) were female and 28 (62.22%) were male. The average patient age was 64 (range: 39–81) years. None of the patients had undergone preoperative adjuvant chemoradiotherapy. Primary tumors were located in the tongue in nine cases (20.00%), in the gingiva in 11 (24.44%), in the cheeks in eight (17.78%), and in the palate and lips in 17 (30.78%). The tumor had metastasized to the lymph nodes in 16 patients (35.56%). The WHO classification (2005) was used for pathological grading. The follow-up duration was 5–103 months (median: 76 months and mean: 64.5 months) (Tables 1 and 2).
Relationship between E-cad and vimentin expression levels and clinicopathological factors in oral squamous cell carcinoma.
E-cad: E-cadherin.
The relationship between the expression levels of CD206, PD-1, and PD-L1 in oral squamous cell carcinoma and clinicopathological factors.

PD-L1: programmed cell death ligand 1; PD-1: programmed cell death receptor 1.
The relationship between clinicopathological factors and the expression levels of the indices
Data presented in tables 1 and 2 show the relationship of these five indicators, including E-cad, vimentin, CD206, PD-1, and PD-L1 with clinicopathological factors, including sex, age, tumor location, lymph node metastasis, pathological grade, and death.
Pathological grades II and III indicated statistically poorly differentiated OSCC; aged >60 years was a clinical factor. E-cad, vimentin, CD206, PD-1, and PD-L1 expression levels were not significantly associated with patient sex, age, or tumor location
Expressions of E-cad, vimentin, CD206, PD-1, and PD-L1
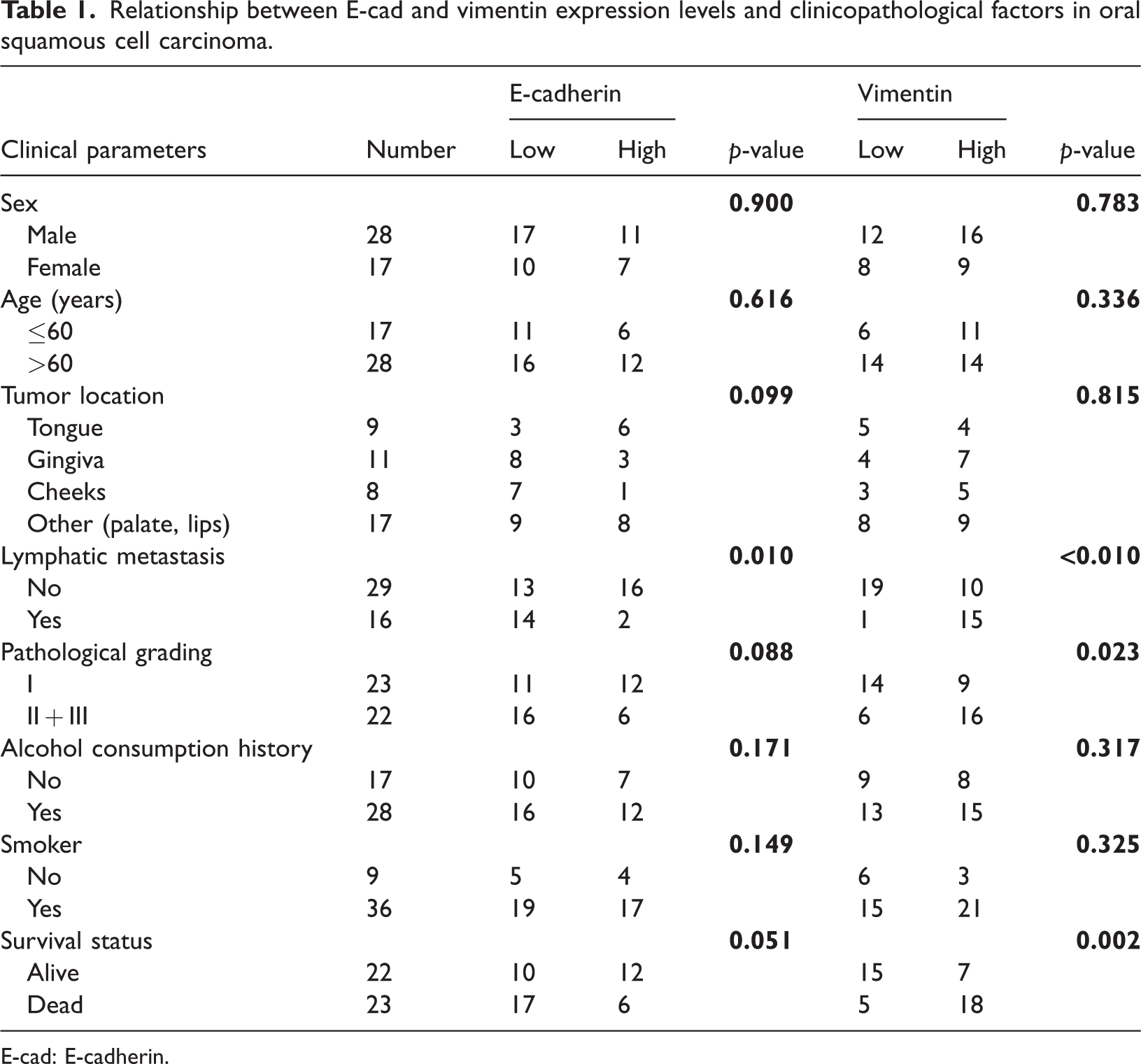
E-cad is mainly expressed on the membrane of epithelial cells and appears yellow (Figure 1). In the specimens, the E-cad expression level was lower at the invasive front of the tumor, with 27 (60.00%) of the 45 cancer tissue specimens showing low expression. Vimentin is mainly expressed in the cytoplasm of epithelial cells, appearing brownish yellow in color (Figure 2). Higher the pathological grade, higher the vimentin expression level in the tumor epithelium; 25 (55.55%) of the specimens exhibited high expression of vimentin (Table 1). CD206 is mainly expressed on the cell membranes of spindle-type macrophages and dendritic cells in cancer nests and interstitium, appearing dark brownish yellow (Figure 3). CD206 was highly expressed in 28 (62.22%) of the 45 specimens. PD-1 was mainly expressed on the cell membrane of tumor-infiltrating lymphocytes, appearing yellow-brown in color (Figure 4); 25 (55.56%) specimens had high PD-1 expression. PD-L1 was mainly expressed in the cell membrane or cytoplasm of tumor cells, appearing yellow-brown in color (Figure 5). PD-L1 was highly expressed in 24 (53.33%) specimens (Table 2). Both vimentin and CD206 expression levels were higher at the invasive front of the tumor when PD-L1 expression levels were higher (Figure 7).

E-cad is mainly expressed on the cell membrane in oral squamous cell carcinoma. (a) High expression. (b) Low expression. (a1 and b1: 100×; a2 and b2 200×; and a3 and b3: 400×). E-cad: E-cadherin.
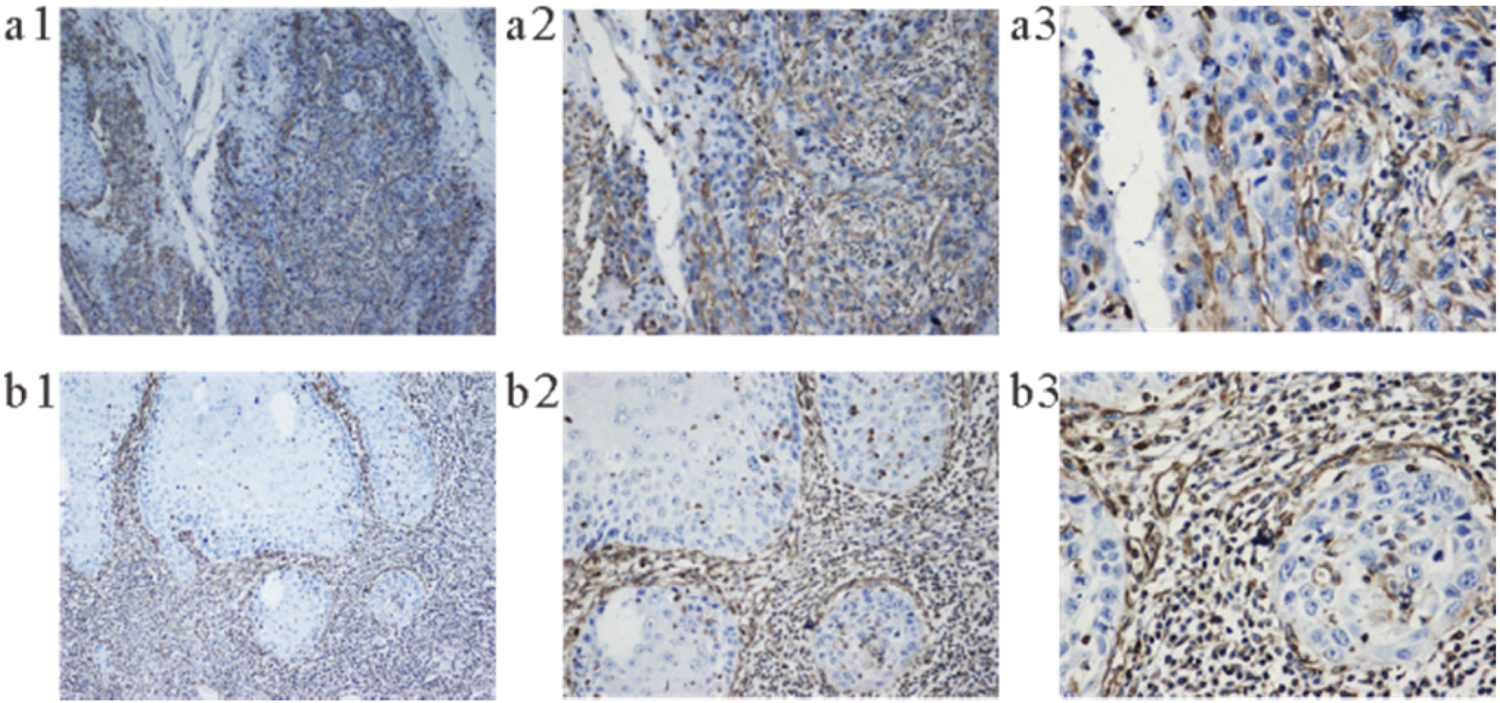
The expression of vimentin in oral squamous cell carcinoma is mainly in the cytoplasm of the tumor-infiltrating leading edge. (a) High expression. (b) Low expression. (a1 and b1: 100×; a2 and b2 200×; and a3 and b3: 400×).

The expression of CD206 in oral squamous cell carcinoma is mainly on the macrophages. (a) High expression. (b) Low expression. (a1 and b1: 100×; a2 and b2 200×; and a3 and b3: 400×).
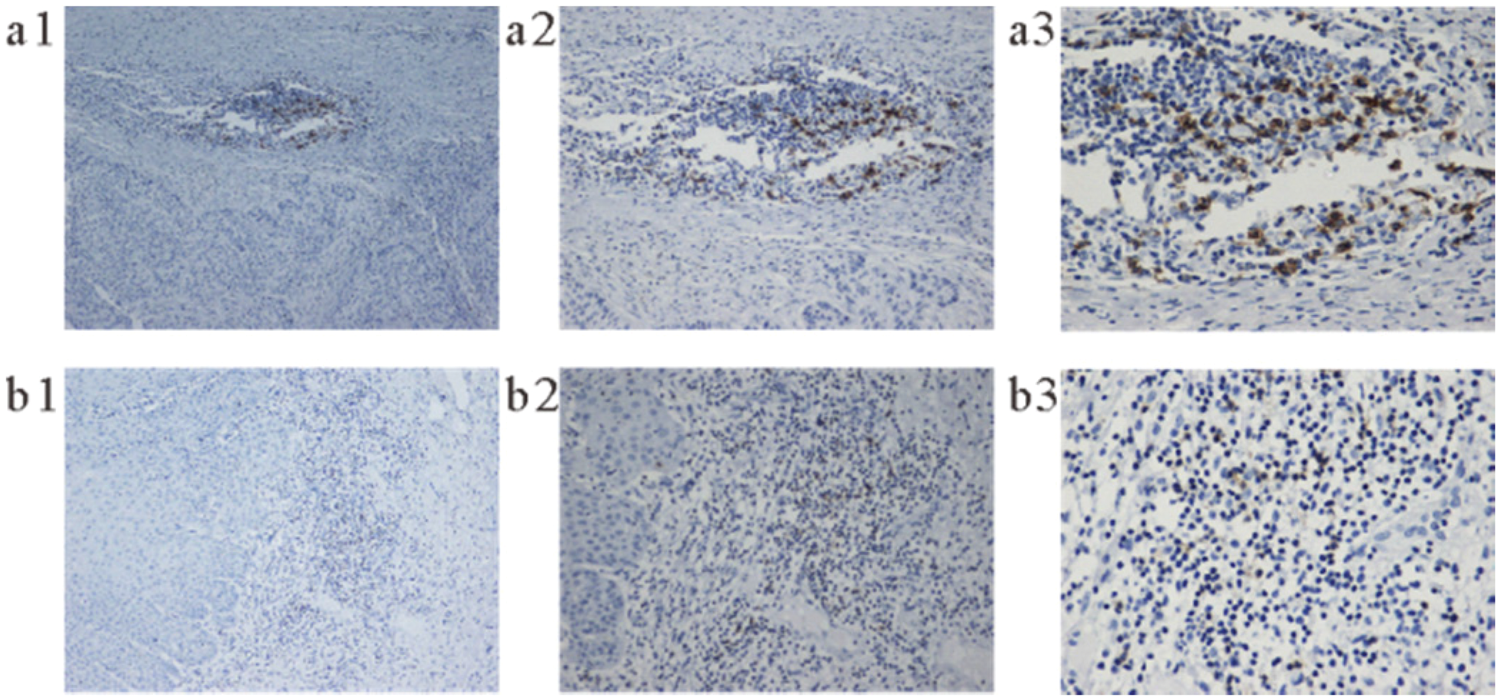
The expression of PD-1 in oral squamous cell carcinoma is mainly on TILs. (a) PD-1 high expression. (b) PD-L1 low expression. (a1 and b1: 100×; a2 and b2 200×; and a3 and b3: 400×). PD-L1: programmed cell death ligand 1; PD-1: programmed cell death receptor 1: TILs: tumor-infiltrating lymphocytes.

The expression of PD-L1 in oral squamous cell carcinoma is mainly on tumor cells. (a) High expression. (b) Low expression. (a1 and b1: 100×; a2 and b2 200×; and a3 and b3: 400×). PD-L1: programmed cell death ligand 1.
Factors affecting the survival and prognosis in OSCC patients
Out of the 45 patients, 23 (51.11%) died during the follow-up period, and the 5-year survival rate was comparatively low. We used pertinent statistical methods, such as the K–M curve and Cox regression analysis, to examine the impact of different indicators and clinicopathological factors on the prognosis and survival of OSCC patients.
The overall survival rate was notably higher for patients with low expressions of vimentin, CD206, and PD-L1 than for those with high expressions, as shown by the K–M curve (Figure 6). Cox univariate regression analysis (Table 3) showed that the survival prognosis was significantly correlated with the lymph node metastasis status as well as the expression levels of vimentin, CD206, and PD-L1. OSCC patients with lymph node metastasis (

Correlation of E-cad, vimentin, CD206, and PD-L1 in oral squamous cell carcinoma. (a) Low expression of E-cad. (b) High expression of vimentin. (c) High expression of CD206. (d) High expression of PD-L1. (a1, b1, c1, and d1: 100×; a2, b2, c2, and d2: 200×; and a3, b3, c3, and d3 400×f). E-cad: E-cadherin; PD-L1: programmed cell death ligand 1.
Results of univariate regression analyses.

HR: hazard ratio; CI: confidence interval; PD-L1: programmed cell death ligand 1; PD-1: programmed cell death receptor 1.
Results of multivariate regression analyses.

HR: hazard ratio; CI: confidence interval; PD-L1: programmed cell death ligand 1.
The relationship between E-cad, vimentin, CD206, PD-1, and PD-L1
According to the correlation analysis of E-cad, vimentin, CD206, PD-1, and PD-L1 shown in Table 5, E-cad expression was significantly associated with the expressions of vimentin, CD206, and PD-L1. E-cad expression was negatively associated with vimentin, CD206, and PD-L1 expressions, as indicated by the negative Pearson correlation coefficients (−0.639, −0.299, and −0.327, respectively) (Figure 7). At the same time, the expression of vimentin is positively correlated with those of CD206 and PD-L1, with values of 0.133 and 0.282, respectively. This implied a positive correlation between vimentin expression and CD206 and PD-L1 expressions.
Results of the correlation analyses of E-cad, vimentin, CD206, PD-1, and PD-L1.

E-cad: E-cadherin; PD-L1: programmed cell death ligand 1; PD-1: programmed cell death receptor 1.

Survival rates of 45 patients. High expressions of vimentin, CD206, and PD-L1 were associated with decreased patient survival. PD-L1: programmed cell death ligand 1.
Discussion
The relationship between CD206+ M2 macrophages and OSCC prognosis
Among the immune cells in the TME, TAMs are the most abundant and significant stromal cells. They are linked to the invasion, metastasis, survival, and proliferation of tumor cells and are crucial for tumor development.
In various cancers, CD206+ M2 macrophages are considered to exert immunosuppressive and tumor-promoting effects, and CD206 is one of their markers. 6 In the present study, strong CD206 expression was associated with low survival in OSCC patients. In addition, multivariate regression analyses revealed that CD206 was an independent risk factor for poor prognosis in OSCC patients, suggesting a substantial role of CD206+ M2 macrophages in worsening the prognosis in OSCC patients.
The relationship between CD206+ M2 macrophages and EMT
Clinical studies have shown that EMT promotes the transformation of the surface of tumor cells to the mesenchymal form, reducing the adhesion between cells and enhancing the tumor’s invasion and metastasis abilities. EMT is marked by low E-cad expression and high vimentin expression. 12 Our immunohistochemical analysis showed that 87.5% of the specimens with lymph node metastasis had low E-cad expression, and 94.0% had high vimentin expression. Higher the pathological grade of OSCC, higher the vimentin expression level. Furthermore, overall survival was poor for OSCC patients with strong vimentin expression. High vimentin expression accounted for 78% of all deaths that occurred during the follow-up period. There was no statistically significant relationship between overall survival and E-cad expression level, most likely because of the limited sample size of this study. Each index’s association indicated a negative relationship between the expression levels of vimentin and E-cad. Consistent with the features of EMT, specimens exhibiting high E-cad expression had lower vimentin expression levels. Thus, we believed that OSCC patients’ tumor cells had undergone EMT. Additionally, the expression level of the M2 marker, CD206, was negatively correlated with the E-cad expression level and positively correlated with vimentin expression, indicating that the number of CD206+ M2 macrophages also increased in OSCC tumor specimens that had undergone EMT. Since the results showed that EMT was more pronounced in OSCC patients with lymph node metastasis, and CD206+ M2 macrophages were closely related to poor prognosis in OSCC patients, we infer that CD206+ M2 macrophages promote the invasion and metastasis abilities of OSCC tumor cells via EMT induction, resulting in poor prognosis in OSCC patients.
Relationship between PD-1, PD-L1, CD206+ M2 macrophages, and EMT
Accumulating evidence from research on immune escape in tumorigenesis and development has shown that the PD-1/PD-L1 pathway is a crucial immune checkpoint in these processes. Research is still ongoing to determine the mechanism of tumor immune escape. According to some studies, PD-1/PD-L1 expression may influence tumor cells’ capacity to invade through TAMs and alter the epithelium of tumor cells, thereby increasing the tumor’s ability to metastasize.20,21 Therefore, we also measured the PD-1 and PD-L1 expressions in samples from OSCC patient. Statistical analyses revealed that the short overall survival of OSCC patients was significantly linked to high PD-L1 expression, which was also positively correlated with the expression level of the M2 macrophage marker, CD206. In contrast, E-cad expression was negatively correlated with OSCC but positively correlated with vimentin expression. Consequently, PD-L1 expression and the quantity of M2-type macrophages both increased when the tumor cells of OSCC patients underwent EMT. However, PD-1 expression showed no significant correlation with clinicopathological factors; however, it exhibited an association with the expression of each index, potentially owing to the small sample size of this study. Thus, whether PD-1/PD-L1 expression affects the invasion and metastasis abilities of OSCC tumors by affecting CD206+ M2 macrophages needs further evaluation.
Conclusion
This study mainly quantified the expression of M2 macrophage markers CD206, E-cad, vimentin, and PD-1/PD-L1 markers through immunohistochemical experiments. Data analyses revealed that CD206 is an independent risk factor for poor prognosis in OSCC patients. In addition, according to our analyses, M2 macrophages may promote the invasion and metastasis abilities of OSCC tumor cells by inducing EMT in them, resulting in poor prognosis in OSCC patients. Considering the relatively small sample size of this study, further research is needed to definitely conclude whether PD-1/PD-L1 expression can affect the invasion and metastasis abilities of OSCC tumors via their influence on M2 macrophages.
Footnotes
Acknowledgments
We extend our gratitude to all personnel involved in this study for their contributions.
Author contributions
Conceptualization, Zheng-yi Lai and Jie Ma; Methodology, Zheng-yi Lai, Jie Ma, De-tao Tao, and Xiu-an Zhu; Validation, Jie Ma; Formal analysis, Zheng-yi Lai and De-tao Tao; Investigation, Zheng-yi Lai and Jie Ma; Resources, Zheng-yi Lai; Data Curation, Jie Ma and Jia-yi Yin; Writing-Original Draft, Jie Ma and Zheng-yi Lai; Writing-Review & Editing, Zheng-yi Lai; Visualization, Zheng-yi Lai, Jie Ma; Supervision, De-tao Tao; and Funding acquisition, Jing-ping Zhou and De-tao Tao.
Zheng-yi Lai and Jie Ma contributed equally to this work.
Data availability statement
All data in this study were derived from clinical specimens, with informed consent obtained from the participants. Database data were sourced from public repositories.
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, or publication of this article.
Funding
Anhui Natural Science Foundation (No. 22080-85QH247), Anhui Key University Natural Science Research Project (No. KJ2021A0845), Talent Introduction Program of Yijishan Hospital of Wannan Medical College (YR202108, YR202105), and Middle-aged and Young Research Fund of Yijishan Hospital of Wannan Medical College (XJ2021002303)
