Abstract
Liver cancer is the most common cancer among males in Africa. The disease has a poor prognosis and its treatment is associated with toxicity and resistance. For this reason, numerous herbal combinations are being subjected to anticancer screening to circumvent the shortcomings of the conventional anticancer drugs. In the current study, the in vivo anti-cancer effects of the chloroform root extract of the herb, Clausena excavata Burm were investigated. Liver cancer was induced in mice by a single intraperitoneal injection of diethylnitrosamine (DEN) followed by oral administration of the promoter of carcinogenesis, 2-aminoacetyl fluorine that was mixed with the mice feed. The cytotoxicity of the root extract of C. excavata on liver cancer cells was investigated using liver enzyme, histology, DNA fragmentation and caspases assays. Real time qPCR was conducted to evaluate the effect of the extract on apoptotic genes. The findings revealed that the extract of C. excavata significantly decreased the progression of hepatocarcinogenesis and the toxicity-induced production of the liver enzymes, alanine and aspartate aminotransferases. The histological analyses of the liver tissues revealed evidence of apoptotic cell death. The extract also provoked significant (p < .05) expressions of caspase 9 protein and gene as well as other apoptotic genes (P53, P27, Apaf-1, cytochrome C, bax and bid). Therefore, we postulate that the chloroform root extract of C. excavata induces apoptosis of liver cancer in mice.
Introduction
In 2020, Globocan estimated over 9.6 million cancer deaths with liver cancer as the second leading cause of premature cancer deaths and among the top 3 causes of cancer deaths globally.1,2 In Africa, it is the predominant form of cancer in males with very devastating consequences. Liver cancer presents as hepatocellular carcinoma (HCC), hepatoblastoma and cholangiocarcinoma, with very poor prognosis. The most common treatment method for liver cancer is oral dosing with the kinase inhibitor, sorafenib, which is associated with toxicity and resistance within the first 6 months of treatment. 3 This situation is worrisome and creates the need for further research on the identification of alternative options for the management of liver cancer.
Recent studies have shown that the consumption of whole plant extract helps to reduce the toxicity and resistance associated with conventional anti-cancer drugs. For this reason, some herbs including Radix isatidis, Pinellia ternate and Taxus chinensis were approved for clinical treatment of cancer patients in Asia. 3 Others such as Olgenlandia diffusia, Curcuma longa, Astralagus membranaceus are used in combination with the conventional drugs for the treatment of cancers. This combination therapy using natural herbs has been reported to improve the quality of life of cancer patients and the sensitivity of clinical chemotherapy. 3 In Africa, Clausena excavata (also known as agbasa or obuko in Nigeria, and cherek hitam in Malaysia) is a herb that has been consumed since time immemorial for the local treatment of cancer. Coumarins such as dentatin, nordentatin and clausenidin are examples of anticancer agents isolated from the roots of Clausena excavata. The in vitro anticancer effects of the compounds and extract of the roots of C. excavata has been reported.4,5 However, there is insufficient in vivo evidence to corroborate the in vitro anticancer properties of the extracts of C. excavata.
In the current study, the in vivo anti-cancer effects of the chloroform root extract of C. excavata was evaluated in diethylnitrosamine (DEN)-induced hepatocellular carcinoma mice.
Methods
Material
The fresh roots of Clausena excavata were collected from the jungle in Mambila, Taraba state, Nigeria by a certified botanist. The voucher number of the plant material, BCH/0222 was deposited in the herbarium and the material was air dried for 3 weeks and ground to fine powder prior to extraction.
Extraction
About 1.5 kg of the fine powder of the root of C. excavata was soaked successively in hexane, chloroform and methanol in a 2 L Erlenmeyer flask for 48 h each at room temperature. The respective solutions were sieved twice each with muslin cloth and filter paper and the extract collected was dried in a rotary evaporator set at 40 °C.
MTT Assay (Cytotoxicity Assay)
The MTT (3-(4,5-dimethylthiazole-2-yl)-2,5-diphenyltetrazolium bromide) assay was performed to determine the IC50 of the 3 root extracts of Clausena excavata. Briefly, liver cancer (HepG2) cells were obtained from American Type Culture Collection (ATCC, Va, USA) and maintained in RPMI-1640 medium supplemented with 10% Fetal Bovine Serum (FBS). The cells were seeded for the assay in a 96-well plate at a density of 4 × 103 cells per well for 24 h and treated with increasing concentrations of either the hexane, chloroform or methanol extracts. Doxorubicin and 0.1% DMSO were used as positive and negative control respectively. The cells were treated for 48 h after which, 20 µl of MTT (5 mg/ml) was added to each well and the plates were incubated for 4 h in the incubator at 37 °C and 5% CO2. The reaction was terminated by the addition of 200 µl of DMSO to each well and the absorbance was read at 570 nm in an xMArk ELISA plate reader (Biorad, USA). The IC50 of each extract was calculated as described previously by Syama et al. 6
Animals
About 40 mice (BALB/c) weighing between 20 to 25 g were purchased for this study. The mice (male and female) were fed with standard chow and given free access to distilled water. The animals were housed in a metal cage in a controlled environment (12 h/12 h dark cycle). Animals were handled based on International protocols and guidelines. Ethical approval was also sought prior to the study (AEC/2023/001).
Experimental Design
The mice were allowed to acclimatize to the controlled environment for about 2 weeks prior to the commencement of the experiment. Animals were categorized into five groups of 4 mice each (Table 1). Hepatocellular carcinoma (HCC) was induced by a single weekly intraperitoneal injection of diethylnitrosamine (DEN) dissolved in olive oil at a dose of 200 mg/kg body weight (bw) for 2 weeks. To promote carcinogenesis, the animal feed was mixed with acetylaminofluorene (AAF) for another two weeks. Thereafter, the mice were treated with the chloroform root extract (dissolved in 1% DMSO) of C. excavata daily for 3 days in a dose dependent manner (Table 1). The positive control group (Group V) was treated with Doxorubicin (10 mg/kg bw) while the negative control group was treated with 1% DMSO. At the end of the treatment period, animals were anesthetized using chloroform and decapitated to collect the blood samples. Similarly, the organs were eviscerated, washed with phosphate buffered saline (PBS) and stored at −20 °C.
Experimental Design.
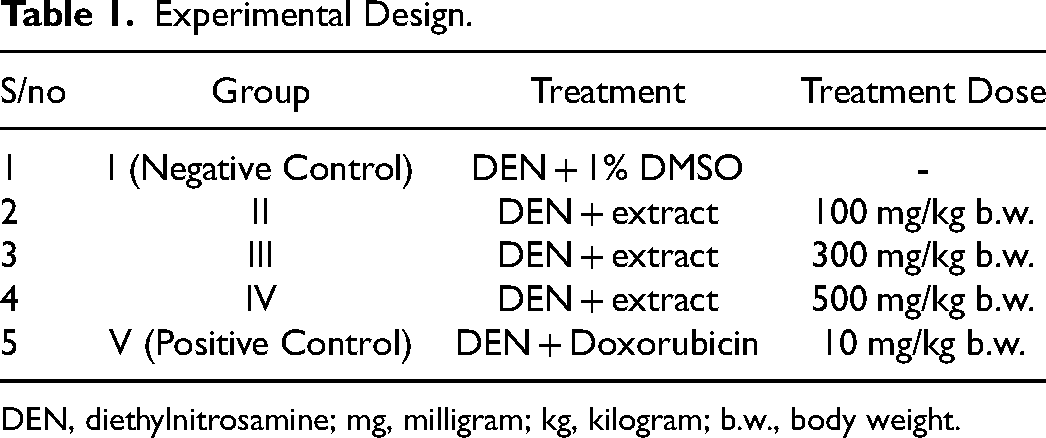
DEN, diethylnitrosamine; mg, milligram; kg, kilogram; b.w., body weight.
Liver Enzyme Assays
The liver enzymes, alanine aminotransferase (ALT) and aspartate aminotransferase (AST) assays were performed using the liver enzyme assay kits (Spectrum Inc, USA) following the manufacturer's protocol. The whole blood samples were centrifuged and serum was collected for the assay. The serum samples were mixed with the assay reagents and analysis was performed on a BK-200 Chemistry analyzer (Biobase, China). The concentration of the enzymes was expressed in International Units (IU).
Histology Assay
The Histological assay was performed using methods described by Nie et al. 7 Briefly, liver tissues of the treated and untreated mice were fixed in 10% formalin at room temperature for about 48 h. The tissue sections were embedded in paraffin at 60 °C, cut into thin sections (3.5 µm) using a microtome (Reichert-Jung, UK) and treated successively with xylene and then 100, 95, 85 and 75% (v/v) of ethanol. Thereafter, the histological sections were stained with hematoxylin and eosin (H & E) at room temperature and sections were viewed under light microscope (Olympus, Japan).
DNA Fragmentation Analysis
The assay was performed using Apoptotic DNA assay kit (Roche, Germany) following manufacturer's protocol. The DNA was extracted from the liver tissues of treated and untreated mice. Briefly, 200 µl of the cell lysis buffer was added to each 20 mg of liver tissue sample, homogenized and incubated for 10 min at room temperature. Following incubation, 100 µl of isopropanol was added to 100 µl of the clear supernatant in a new tube, vortexed for 60 s and the mixture was transferred to a spin column attached to a collection tube. The tube was centrifuged for 1 min at 8000 rpm, and the flow through was discarded. The DNA pellet trapped in the filter tube was washed by the addition of 500 µl of wash buffer and centrifuged again for 60 s at 8000 rpm. Thereafter, the filter tube was attached to a microcentrifuge tube and DNA was eluted by the addition of 200 µl each of pre-warmed elution buffer. The purity and concentration of the extracted DNA was determined using UV/VIS Biophotometer (Eppendorf, Germany).
Thereafter, the DNA sample collected was mixed with the loading buffer in a ratio of 5:1 and run on 1% agarose gel (stained with Gel red) for 30 min at 80 V. The gel image was captured on an EGel Imager (Life Technologies, UK)
Caspase 3/8/9 Assays
Caspases 3, 8 and 9 assays were performed using the colorimetric assay kit (Biovision, USA) following manufacturer's protocol. For the caspase 3 assay, 25 mg of the homogenized liver tissue was suspended in 50 µl of chilled lysis buffer and centrifuged for 60 s at 1000 rpm to collect supernatant. About 50 µl of the 2x reaction buffer and 5 µl of the 4 mM LEHD pNA substrate was added to 50 µl of the supernatant and incubated for 2 h at 37 °C. Thereafter, the absorbance of the mixture was read on a microtitre plate reader (Biorad, USA). The same procedure was repeated for caspases 8 and 9 assays using their respective kits and the results were computed as fold change relative to the untreated control.
Gene Expression Studies
Primer Design
The primers used in this study were designed on the NCBI website using the primer-blast software and purchased from Integrated DNA Technologies (IDT, Singapore) (Table 2). Prior to primer synthesis, the quality of the primer was confirmed using the OLIGOCAL software.
Primer Sequences of Genes Amplified.

RNA Extraction
The mice RNA were extracted from the liver tissues using Total RNA extraction kit (Vivanties Tech., Malaysia) following the manufacturer's instruction. The purity and concentration of the extracted RNA was determined using UV/VIS Biophotometer (Eppendorf, Germany).
cDNA Synthesis and Real Time qPCR
Complementary DNA (cDNA) synthesis was performed using ReverTRA Ace qPCR RT Master Mix with gDNA remover (Toyobo, Japan) in a Thermocycler (Biorad, USA). The cDNA was synthesized via reverse transcription of the extracted RNA and the cDNA generated was used as template for the qPCR that was performed using the 2x SYBR green Master Mix (Bridgen, China). The PCR conditions were as follows: initial denaturation for 1 min at 95 °C, 40 cycles of 15 s denaturation, 30 s annealing at 60 °C and final extension for 1 min at 74 °C. In addition, the melting curve analysis of the reactions was performed and the β-actin gene was used for normalization. The ct (cycle threshold) values obtained were used to analyze the fold change of gene expression using the 2−ΔΔCt method.
Statistical Analysis
One way analysis of variance (ANOVA) was used to determine level of significance between groups using SPSS (IBM Inc, USA) version 24. The experiment was carried out in triplicates and values were reported as mean ± standard error of mean (SEM).
Result
MTT Assay
The IC50 of the hexane, chloroform and methanolic root extracts of C. excavata is shown in Table 3. Of the 3 extracts, the chloroform extract has the least IC50 (28.0 µg/ml) on liver cancer cells in vitro. Hence, the chloroform extract was selected for the in vivo bioassays.
In Vitro Cytotoxicity Assay.

Values are presented as mean ± standard error of mean (SEM), (*) indicates statistical significance (p < .05).
Liver Enzyme Assay
The activity of the main liver enzymes, alanine and aspartate aminotransferases were determined in the blood samples of the treated and untreated mice. The treatment with the chloroform extract of C. excavata caused a significant (p < .05) decrease of blood AST and ALT level (Figure 1). The decrease observed was significantly lower than that of the untreated control samples. Also, the treatment with Doxorubicin caused a significant (p < .05) decrease in the level of blood AST.

ALT and AST level of the treated and untreated mice (Groups I, Untreated control; II, Normal control; III, 100 mg/kg bw, IV, 300 mg/kg bw; V, 500 mg/kg bw and VI, Doxorubicin; bw, body weight; IU, International Unit; L, litre). *Means are significantly (p < .05) different from control, n = 4.
Histology Assay
The chloroform extract of C. excavata did cause nuclear polymorphism and disorganization of the mice tumor (Figure 2). As the treatment dose increased to 500 mg/kg body weight (Figure 2D), the mice tissue showed prominent and condensed nucleoli which is a major feature of apoptosis. Doxorubicin (positive control) caused apoptosis of the tumor tissues and prevented nuclear damage and disorganization of non tumor hepatocytes (Figure 2E).

Hematoxylin and Eosin stained micrographs of untreated mice liver tissues (A) and treated mice liver (B) 100 mg/kg bw; (C) 300 mg/kg b.w. and (D) 500 mg/kg bw; (E) doxorubicin (10 mg/kg bw), bw – body weight). The arrows show regions of high nuclear polymorphism, non linear arrangement of tumors in A, and moderate nuclear polymorphism, disarrayed arrangement of hepatic plates and mitotic figures in B, C and D (treated mice tissues). D (high dose treatment) showed more prominent nucleoli (typical feature of apoptosis) (x400 Magnification).
DNA Fragmentation Assay
The DNA fragmentation assay was conducted to confirm if the extract caused apoptosis of the liver tumors. Following extraction of the DNA from the liver tumors, the sample was run on 1% agarose gel and the image is shown in Figure 3. DNA fragmentation was observed in the treated samples (lanes B to D) and positive control (lane F) unlike the untreated control (lane E).
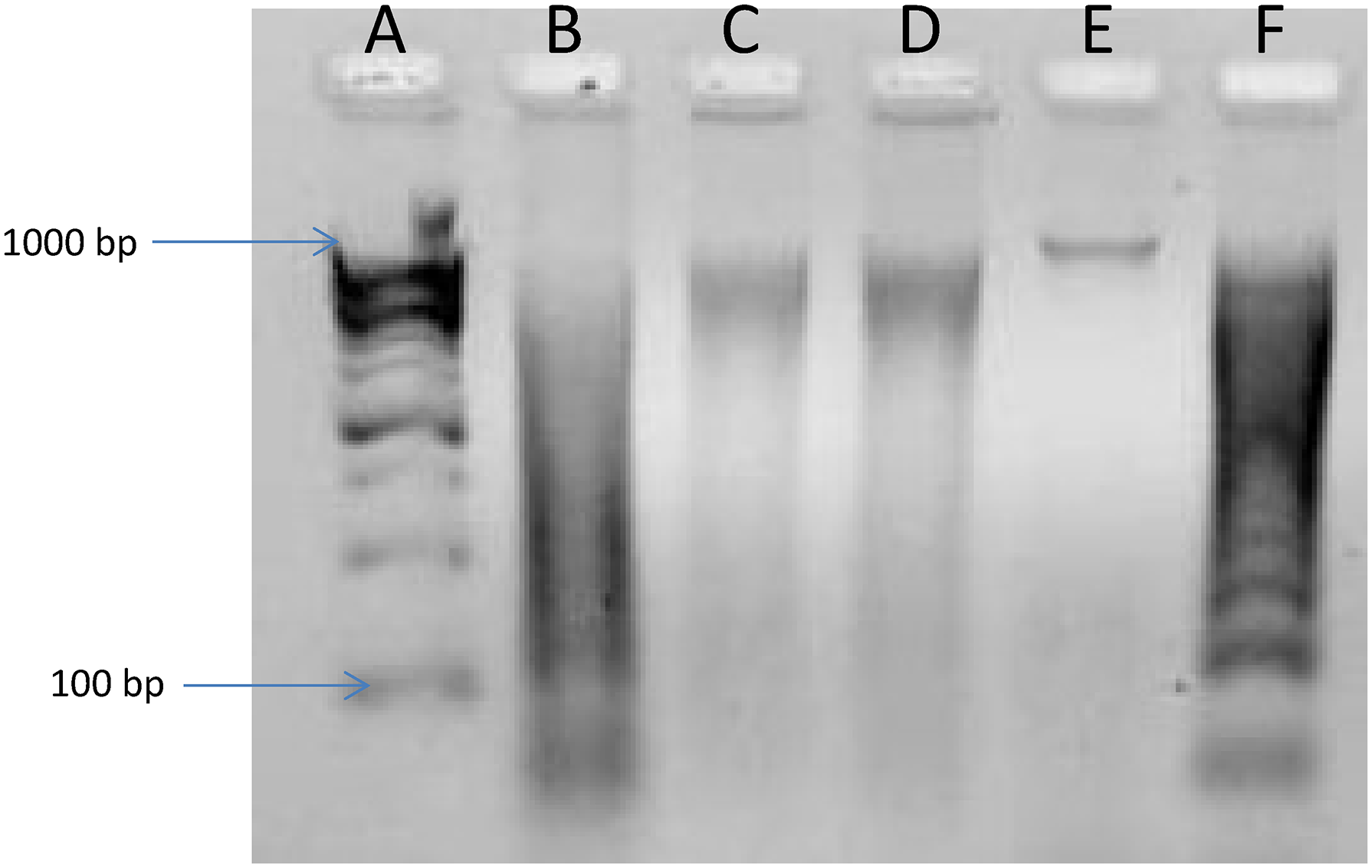
Agarose gel image of treated and untreated mice DNA (Lane (A) DNA ladder; (B), 100 mg/kg body weight (bw), (C)300 mg/kg bw, (D) 500 mg/kg bw; (E) untreated control; (F) positive control).
Caspase Assay
Caspases-3, −8 and −9 levels of the treated and untreated mice are shown in Figure 4. Treatment of mice with the extract of C. excavata caused a significant fold increase in the level of caspases-3, −8 and −9. The level of caspases in the treated mice was significantly (p < .05) higher than that of the untreated control.
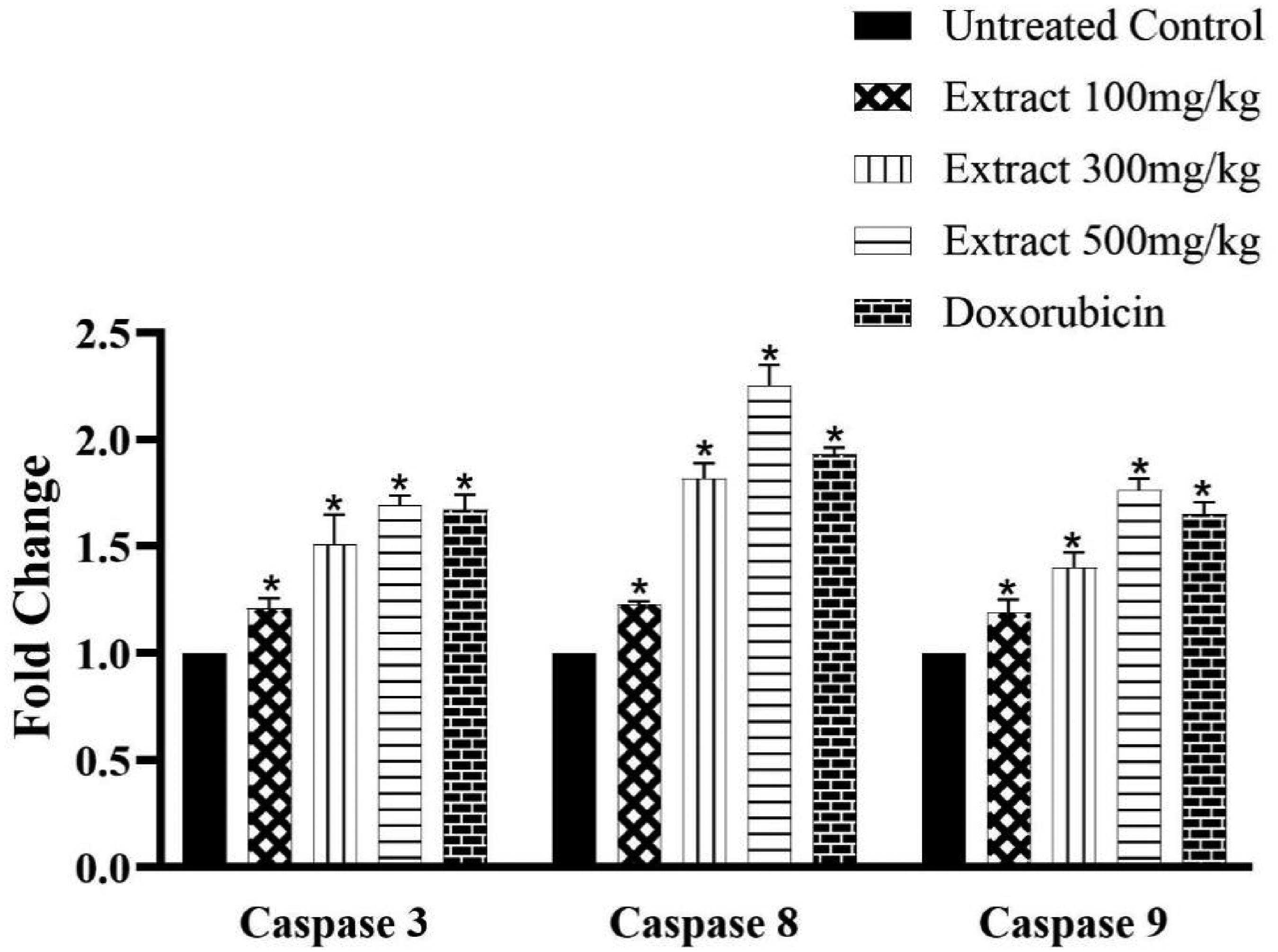
Caspases 3, 8 and 9 protein level of treated and untreated mice (Groups I – Untreated control, II – 100 mg/kg bw, III – 300 mg/kg bw, IV – 500 mg/kg bw and V – Doxorubicin; bw – body weight). *Means are significantly (p < .05) different from control, n = 4.
Gene Expression (qPCR) Analysis
Following treatment with the chloroform root extract of C. excavata, the expression of the genes encoding caspase-3 and −9 increased significantly (Figure 5A). However, the highest dose (500 mg/kg) of treatment did not cause any significant (p > .05) change. The expressions of Bax, Bid, P53 and cytochrome C genes increased significantly only at the highest treatment dose of 500 mg/kg (Figure 5A and B). The extract caused a significant (p < .05) decrease in the expressions of Bcl-2 and P27 genes (Figure 5C). Significant expressions of cyclins A & D and catalase genes were observed only at the medium dose of treatment (300 mg/kg body weight).

Fold change of apoptotic genes of treated and untreated mice using 2−ΔΔCt method (Groups I – untreated control, ii – 100 mg/kg bw, III – 300 mg/kg bw, IV – 500 mg/kg bw and V – Doxorubicin; bw – body weight). *Means are significantly (p < .05) different from control, n = 4.
Discussion
Plants are the mainstay of traditional medicine and have been used since time immemorial for the treatment of cancers. Although chemotherapy is the commonest method for the treatment of liver cancers, it is associated with undue side effects and resistance. In recent past, the consumption of whole plant extracts has increased tremendously because it is associated with little or no side effects. In some instances, anticancer drugs are used in combination with the plant extracts because they improve the sensitivity of chemotherapy and also reduce the incidences of drug resistance.
C. excavata extract is a common recipe in some developing countries of Africa for the treatment of cancers. 5 The extract of C. excavata alongside some of the compounds were used to treat liver cancer cells in vitro.
The current study investigates the in vivo anti-tumor properties of the chloroform extract of C. excavata in BalB/c mice. The chloroform extract of C. excavata root was used in this study because it demonstrated the lowest IC50 (28.0 µg/ml). Following the successful induction of liver tumors in the mice, the animals were treated with low, medium and high doses of the chloroform extract of the root of C. excavata. The liver tumors caused an increased expression of the enzymes, ALT and AST in the blood of the mice. However, the treatment with C. excavata significantly decreased the release of the enzymes in a dose dependent manner. Hepatoprotective agents are known to decrease the production of the liver enzymes.8–10 The Hematoxylin and Eosin stained micrographs of the treated mice tissues showed visible signs of apoptosis of tumor cells that includes nuclear disorganization and condensation of nucleolus. Doxorubicin and the extract of C. excavata further prevented the non tumor cells from nuclear damage.
To confirm if the chloroform extract caused apoptosis, DNA fragmentation analysis was conducted and it was observed that the treatment caused a fragmentation of mice DNA. The fragmentation of DNA is caused by caspase-3-activated DNases and it is a typical feature of apoptotic cell death.11–13 It was also observed that the treatment caused a significant increase in the expression of caspases-3 and −9 proteins. The gene expression assay confirms this as we observed increased expressions of caspase-3 and −9 mRNAs. Caspase-9 is the key initiator of the mitochondrial pathway of apoptosis while caspase-3 is an executioner of apoptosis. One of the ways through which caspase-3 executes apoptosis is through the activation of DNases that brings about fragmentation of DNA and eventual cell death. Cell death is a prerequisite for development and homeostasis of many multicellular organisms. 14
Similarly, the treatment with C. excavata caused significant increase of P-53 and −27 mRNAs. P-53 and −27 are tumor suppressors that help to regulate cell birth and cell death.15–17 P53 is the most studied tumor suppressor because the loss of P53 function is responsible for over 50% of cancers. In fact, tumors become resistant to chemotherapy without P53 function.18,19 We observed over 30-fold increase in the expression of P53 mRNA, which is among the highest of all genes investigated in this study. Among the functions of P53 are regulation of cell cycle, provision of genome stability and the induction of apoptosis.20,21 Apoptosis is controlled by the ratio of pro- to anti-apoptotic genes and proteins. Higher expressions of the pro-apoptotic proteins trigger apoptosis while higher amounts of the anti-apoptotic proteins prevent apoptosis.
We observed that the treatment caused increased expression of the pro-apoptotic mRNAs, bax, bid, cytochrome C, Apaf 1 and caspase 9. P53 is one of the main activator of Bax that in turn triggers the release of cytochrome C. The liberated cytochrome C combines with dATP and Apaf 1 to form the apoptosome complex. The assembly of the apoptosome complex then stimulates the apoptosis of tumor cells. 22 We postulate that the apoptosis of tumor cells observed in this study may have occurred through the above mechanism.
Caspase 9 is the main regulator of the intrinsic pathway of apoptosis while caspase 8 regulates the extrinsic pathway of apoptosis.23,24 The intrinsic pathway of apoptosis is a product of mitochondrial activity and the assembly and formation of the apoptosome complex. 22 Caspase 9 is an initiator caspase while caspase 3 is an executioner of apoptosis.24,25 The increased expressions of caspase-3 and −9 mRNAs suggest that the extract of C. excavata may have caused apoptosis of the mice tumor cells via the intrinsic mitochondrial pathway.
Conclusion
The extract of C. excavata caused significant upregulation of the P53 gene above 30 folds. More so, we postulated that the extract caused apoptosis of the mice tumor cells via the intrinsic mitochondrial pathway. Therefore, the chloroform extract of C. excavata is a potential anti-liver cancer agent.
Footnotes
Acknowledgements
The authors are grateful to Dr Godwin I Ayuba and the staff of Molecular Biology Lab, Kaduna State University.
Author Contributions
Peter M Waziri: Manuscript preparation/ Research Design
Richard Auta: In vivo assay
Mustapha U Imam: Gene expression studies
Ben A Chindo: In vivo assay
Zakari Ladan: Plant collection/ Extraction
Zainab Mohammed: In vivo assay
Samson Wayah: Statistical analysis
Ja'afar Mohammed: Gene expression studies
Mohammed I Tahir: Gene expression studies
Abdurrahman E Ahmad: In vivo assay
Yusuf Alhassan: Statistical analysis
Daniel Tyoapine: In vivo assay
Abel S Agbaji: Research design/protein assays
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The ethical approval for this research was granted by the Research Ethics Committee (KASU/AEC/2023/001), Kaduna State University.
Funding
This research project was supported and funded by NRF TetFund grant (TETF/DR&D/CE/NRF2020/SETI/19/VOL 1), Nigeria.
