Abstract
Garcinia dulcis (GD) extract possesses anti-hypertensive property that are poorly characterized. This study aimed to investigate an anti-inflammatory effect of GD flower extract in the 2-kidney-1-clip (2K1C) hypertensive compared to sham operative (SO) rat. Male Wistar rats were divided into 2 groups; the 2K1C group in which a silver clip was placed around renal artery to induce hypertension, and the SO normotensive group. Four weeks later, each group of rats were further divided into 2 subgroups, each subgroup was orally gavaged of either corn oil (vehicle) or 50 mg/kg BW GD extract daily for 4 weeks. The malondialdehyde (MDA) levels in serum, liver, and kidney were determined. Hematoxylin and eosin staining was carried out for histological examination, Periodic acid – Schiff staining for glomerular injury, Masson's trichrome staining for renal fibrosis, and immunohistochemistry for either tumor necrosis factor alpha (TNF-α) or endothelial nitric oxide synthase (eNOS) investigation. Taken together, our results demonstrated that GD flower extract decreased the MDA level in both serum and liver and kidney tissue and suppressed the expression of TNF-α in both liver and kidney of 2K1C hypertensive rats. Mesangial cell proliferation, expansion of mesangial matrix, widening Bowman's capsule space, congestion of glomerular capillary and vessel, cloudy swelling of renal tubular epithelial cell, and renal fibrosis were observed in the kidneys of 2K1C rats. Therefore, we concluded that GD flower extract can alleviate liver and kidney inflammation in which partially attenuates the glomerular injury in the 2K1C rat.
Introduction
Renovascular hypertension (RVH) is one of the most common causes of secondary hypertension. It is denoted as a stenosis of one or both renal arteries by more than 50% may lead to a reduction of renal perfusion pressure and activation of the renin angiotensin aldosterone system (RAAS). Prevalence of renal stenosis is high in patient with systemic atherosclerosis such as coronary artery disease (up to 18%) and peripheral artery disease (up to 25%). 1 Goldblatt 2-kidney-1-clip (2K1C), the most used experimental model of RVH, is induced by unilateral partial occlusion by placing a silver clip around a renal artery. A raising blood pressure (BP) is developed within 3 to 4 weeks after renal stenosis. 2 High levels of angiotensin II induce hypertension and several systemic alterations including vasoconstriction, increased sympathetic nerve activity, and increased aldosterone level which in turn causes water and sodium retention. These also cause an increase in synthesis of collagen type I and III by fibroblasts, leading to thickening of the vascular wall and myocardium, and fibrosis. In addition, this RVH model promotes the growth effect on renal cell, which has been implicated in the development of glomerulosclerosis and tubulointerstitial fibrosis. 3
Generally, chronic inflammation can trigger oxidative stress, which has been associated with hypertension. Inflammation is a complex immune response process to injury. First, inflammatory cells recognize the affected tissue, then the innate immune cells such as neutrophils and macrophages produce proinflammatory cytokines and reactive oxygen species (ROS) such as superoxide and hydrogen peroxide in order to eliminate pathogens. 4 It has been reported that inflammation also downregulates nitric oxide synthase (NOS) activity. Indeed, tumor necrosis factor alpha (TNF-α) attenuates NO production by destabilizing endothelial NOS (eNOS) mRNA, which in turn reduces eNOS protein expression. 5 In 2K1C model, it has been observed that angiotensin II promotes excess ROS production within the rostral ventrolateral medulla (RVLM), paraventricular nucleus of the hypothalamus, and in the ischemic kidney which consequently plays a major role in the maintenance of hypertension. 6
Garcinia dulcis (GD) Kurz., a tropical plant from the Guttiferae family, is mostly found in Southeast Asian countries with interesting properties to against human disease conditions. Its main bioactive compounds are morelloflavone and camboginol. Morelloflavone, biflavonoids comprising apigenin and luteolin, exerts various beneficial effects such as anti-inflammation, vascular remodeling inhibition, inhibition of HMG-CoA reductase activity to decrease cholesterol synthesis, and anti-atherosclerosis.7–9 On the other hand, camboginol (or garcinol), a compound belonging to benzophenone group, has potent antioxidative and anti-inflammatory effects.10,11 We recently described that morelloflavone and camboginol from either fruit or flower of GD possess anti-hypertensive, vasorelaxant, and diuretic effect.12–16 In addition, their other biological activities such as anti-inflammatory, anti-cancer, and altered gut microbiota have also been reported.17–19 Since we showed GD flower extract possesses anti-hypertensive effect and restores impairment of renal tubular function in the 2K1C rat, we chose to pursue our characterization by studying the effects of GD flower extract on inflammatory status and histological change of kidney and liver in the 2K1C hypertensive rat since no data were reported to date.
In this context, this study aimed to investigate the anti-inflammatory effect of GD flower extract in kidney and liver of 2K1C hypertensive compared with SO normotensive rats. The levels of malondialdehyde (MDA) in serum, liver, and kidney were measured. Histological studies were performed to investigate the characteristics of the liver and kidney, glomerular injury, and renal fibrosis. Both TNF-α and eNOS expression were also studied. We expect our novel findings described herein will subsequently lead to clinical application by using GD extract as anti-hypertensive and/or anti-inflammatory agent.
Materials and Methods
GD Extract Preparation
Preparation of GD flower extract was performed as previously described.15,16,20 Briefly, the GD flowers were collected at the Songkhla province (Thailand), deposited at the Herbarium No.0012625, and purified at the Faculty of Science, Prince of Songkla University, Thailand. The flowers were extracted with acetone and hexane, respectively, to yield several fractions. One of those fractions which composed of morelloflavone and camboginol was used in this study. The flowers, fruits, and chemical structures of morelloflavone and camboginol, the mainly bioactive compound from GD are shown in Figure 1.
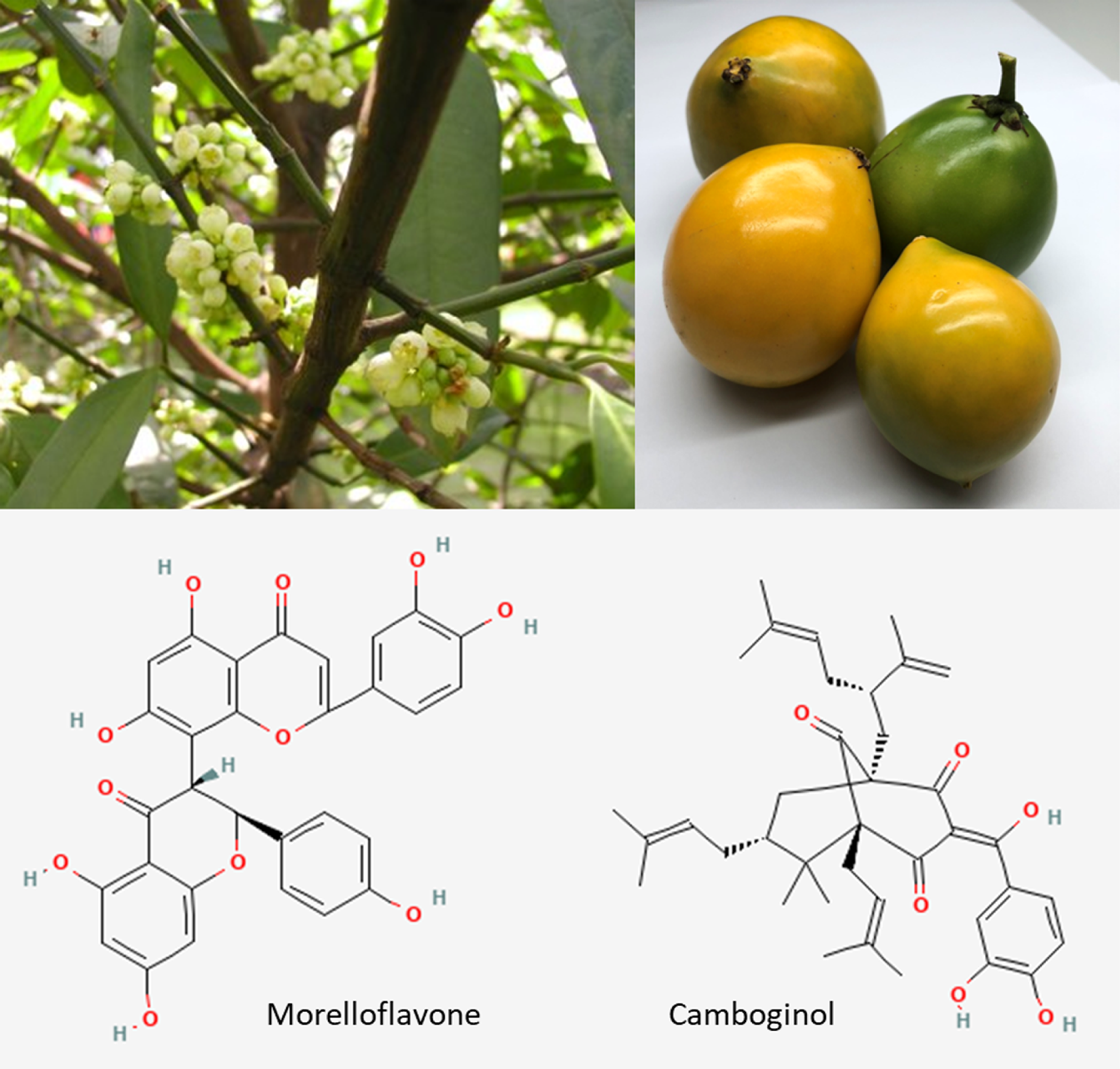
Flowers and fruits of Garcinia dulcis and its bioactive chemical substances, morelloflavone and camboginol (aka. garcinol). Chemical structures were downloaded from https://pubchem.ncbi.nlm.nih.gov.
Animal Inductive Hypertension and Treatment Protocol
The experimental protocol in animal was performed as described in our previous study (Figure 2). 16 Briefly, male Wistar rats (5-week-old, n = 24) were housed under standard conditions at the Animal Laboratory Center, Thammasat University, Thailand. After one week of acclimatization, they were randomly assigned into 2 groups including 2K1C and sham operative (SO) which served as control (n = 12). On the day of inductive surgery, each rat was anesthetized with isoflurane inhalation and the left renal artery was exposed through a small retroperitoneal incision. A 0.2 mm-gap U shape silver clip was placed around the artery to reduce renal perfusion. Then, the muscle and skin layer were sutured separately. The SO rats underwent the same operating procedure except for renal artery clipping. Rats were allowed to recover and developed hypertension within four weeks.

Experimental design of study. Abbreviation: SO, sham operation; 2K1C, 2-kidney-1-clip; GD, Garcinia dulcis; BP, blood pressure; HR, heart rate; MDA, malondialdehyde; H&E, hematoxylin, and eosin; PAS, periodic acid – Schiff; IHC, immunohistochemistry; TNF-α, tumor necrosis factor alpha; eNOS, endothelial nitric oxide synthase.
Four weeks after the hypertensive induction phase, rats were further divided into 4 subgroups (n = 6, power analysis was used to calculate the sample size) including 1) SO + vehicle (SO), 2) SO + GD, 3) 2K1C + vehicle (2K1C), and 4) 2K1C + GD. Animals in each group were orally gavaged with either 50 mg/kg GD flower extract or corn oil (vehicle) daily for 4 weeks. The dose of treatment was based on our previously study showing that oral administration of GD flower extract at the dose of 50, 100, and 200 mg/kg for 2 weeks exhibited hypotensive action in rats. 15
Four weeks after the GD treatment phase, each rat was anesthetized with isoflurane inhalation and carotid artery was cannulated for blood sampling. Blood samples were centrifuged at 4000 RPM for 10 min, and serum was collected and stored at −80 °C until analysis. Next, the jugular vein was perfused with 0.1 M phosphate buffer saline. Liver and both kidney tissue samples were collected, a part of those tissues were stored at −80 °C for lipid peroxidation measurement, and the remaining tissues were fixed in 4% paraformaldehyde for the histological examination.
Lipid Peroxidation Measurement
Determination of serum MDA was performed as described by Costa et al (2006). 21 For MDA standard preparation, a 10−2 M MDA solution was diluted with 0.01 mmol/L hydrochloric acid (HCl) into 2.5, 5, 10, 20, and 30 μM to serve as standard solutions. Then, either 100 μL of serum or standard solution was mixed with 100 μL of thiobarbituric acid (TBA) cocktail (15% trichloroacetic acid, 0.38% TBA, 0.25 M HCl, Sigma-Aldrich, Darmstadt, Germany). The mixture was then heated at 95 °C for 30 min, cooled on ice for 2 min, and centrifuged at 10 000 RCF for 5 min. Finally, the absorbance of the supernatant was measured at 532 nm.
Determination of MDA from liver and kidney tissue was performed as described by Ohkawa et al (1979). 22 For MDA standard preparation, a 10−2 M MDA solution was diluted with deionized water into 2.5, 5, 10, 20, 30, and 60 μM to serve as standard solution. For tissue homogenate preparation, 1 g of tissue was mashed in 9 ml of 1.15% potassium chloride (kept cool on ice), centrifuged at 6000 RPM for 10 min, and the supernatant was collected for further analysis. Then, 100 μL of homogenate or working standard MDA solution was mixed with 100 μL of 8.1% sodium dodecyl sulfate, 750 μL of 20% acetic acid (pH 3.5), 750 μL of 0.8% TBA, and 300 μL of distilled water. The mixture was heated at 95 °C for 60 min, cooled on ice for 2 min, mixed with 2 ml of n-butanol, and centrifuged at 4000 RPM for 10 min. Finally, the absorbance of the supernatant was measured at 532 nm.
Histological Study
The liver and kidney tissues (4 samples from each group) were processed and embedded with paraffin, then 5 μm-thick paraffin sections were cut and mounted on glass slides. Sections were rehydrated with ethanol and distilled water as general procedure, then stained with hematoxylin and eosin (H&E, Sigma-Aldrich, Darmstadt, Germany) to evaluate general histology. For kidney tissue, sections were also stained with Periodic Acid-Schiff (PAS staining system; Sigma-Aldrich, Darmstadt, Germany) to detect glomerular injuries, and Masson's trichrome (Trichrome Stain AB solution, Sigma-Aldrich, Darmstadt, Germany) to detect renal fibrosis. After staining, sections were dehydrated and covered with cover slips. Characteristics of tissue were evaluated by a licensed pathologist. In addition, random 30 areas (400X magnification power, 1290 × 960-pixel size) were recruited from each animal for measurement of Bowman's capsule diameter (distance from a vascular pole to a tubular pole), renal corpuscle area, glomerular area, and Bowman's space (calculated by renal corpuscle area – glomerular area) using the Image J program (NIH Image, USA).
For immunohistochemical study (IHC), sections were subjected to heat mediated antigen retrieval with either 10 mM sodium citrate, pH 6.0 for TNF-α or Tris/EDTA buffer, pH 9.0 for eNOS using the microwave technique for 5 min, and then blocked with 10% normal horse serum for 1 h. Next, sections were incubated with the primary antibody (1:200, anti-TNF alpha, Abcam #ab220210 or 1:800, Recombinant Anti-eNOS antibody, Abcam # ab300071, Cambridge, United Kingdom) at 4 °C overnight. Next day, the secondary antibody [1:5,000, goat anti mouse IgG (H + L) cross-adsorbed secondary antibody, Biotin for TNF-α or 1:5,000, Goat anti-Rabbit IgG (H + L) Cross-Adsorbed Secondary Antibody, Biotin for eNOS, ThermoFisher Scientific, MA, USA] were incubated with the sections, followed by Streptavidin-Biotin complex (ThermoFisher Scientific, MA, USA) and DAB substrate kit (Abcam # ab64238, Cambridge, United Kingdom). Sections were dehydrated and covered with cover slip. The percentage positive area of TNF-α or eNOS was determined using Image J program (20 areas/n).
Statistical Analysis
Quantitative data were presented as mean ± standard error of the mean (SEM). Comparisons between the means among and between groups of the SO and the 2K1C were performed with one-way analysis of variance (ANOVA) and unpaired t-test using GraphPad Prism 8 (San Diego, CA, USA). A p-value of less than 0.05 was considered significant difference.
Results
Levels of MDA in Serum, Liver, and Kidney Tissue
Significantly differences in the MDA levels were observed in both serum, liver, right non-clipped (RK), and left clipped (LK) kidneys among all groups of animals (Figure 3). Indeed, the 2K1C group exhibited significantly higher MDA levels in serum, liver, RK, and LK than the respective values for the SO and the 2K1C + GD groups. While the MDA levels of serum and LK in the SO + GD group were significantly lower than the SO group, but not for liver and RK.

The effect of oral administration of 50 mg/kg BW Garcinia dulcis (GD) flower extract daily for 4 weeks on the levels of the malondialdehyde (MDA) in serum (A), liver (B), right kidney (C), and left kidney (D) in either sham operative (SO) or 2-kidney-1-clip (2K1C) hypertensive rat. *P < .05, **P < .01, ***P < .00, and ****P < .0001 in comparison between groups (unpaired t-test).
Histological Examination of the Liver
H&E staining presented a normal histology of the liver in all groups (Figure 4). Nevertheless, IHC showed a significant difference in the levels of TNF-α expression in liver samples among all groups. Positive areas for TNF-α in liver samples of the 2K1C group were significantly higher than those of SO and the 2K1C + GD groups, but no difference in those were observed between the SO and the SO + GD groups (Figure 5).

Hematoxylin and eosin (H&E) staining of livers from either sham operative (SO, A & B) or 2-kidney-1-clip (2K1C, C & D) hypertensive rat after oral administration of 50 mg/kg BW Garcinia dulcis (GD) flower extract daily for 4 weeks. Magnification power 200X.

Immunohistochemistry for tumor necrosis factor alpha (TNF-α, brown color) in the livers of either sham operative (SO, A & B) or 2-kidney-1-clip (2K1C, C & D) hypertensive rats after oral administration of 50 mg/kg BW Garcinia dulcis (GD) flower extract daily for 4 weeks. Negative control (E) and the % relative TNF-α positive area (F) were shown. Magnification power 200X. ****P < .0001 in comparison between groups.
Histological Examination of the Kidney
H&E (Figure 6) and PAS staining (Figure 7) presented normal glomerular and renal tubular characteristics in the SO and SO + GD groups, while both kidneys of the 2K1C group showed obviously increased mesangial cell proliferation, widen Bowman's capsule space, congestion of glomerular capillaries and vessels, and cloudy swelling of renal tubular epithelial cells. An expansion of mesangial matrix was also observed in the glomeruli of the 2K1C group. The GD treatment partially attenuated these impairments since a slightly increased mesangial cell proliferation and congestion of glomerular capillary were still observable in the 2K1C + GD group.

Hematoxylin and eosin (H&E) staining of right (RK) and left kidneys (LK) from either sham operative (SO, A − D) or 2-kidney-1-clip (2K1C, E − H) hypertensive rats after oral administration of 50 mg/kg BW Garcinia dulcis (GD) flower extract daily for 4 weeks. Increased mesangial cells proliferation (black arrow), increased Bowman capsule's space (star), cloudy swelling of tubular epithelial cell (black arrowhead), and vascular congestion of glomerular capillaries and vessels (white arrow) were indicated. Magnification power 400X.

Periodic acid–Schiff (PAS) staining of right (RK) and left kidneys (LK) from either sham operative (SO, A − D) or 2-kidney-1-clip (2K1C, E − H) hypertensive rats after oral administration of 50 mg/kg BW Garcinia dulcis (GD) flower extract daily for 4 weeks. Increased mesangial cells matrix (black arrow) and increased Bowman capsule's space (star) were indicated. Magnification power 400X.
Measurement of the renal corpuscle component (Figure 8) showed both kidneys of the 2K1C group exhibited a significantly decreased Bowman's capsule diameter, renal corpuscle area, glomerular area, and Bowman's capsule space in comparison to the SO group. The LK of the 2K1C + GD group had a significantly lesser Bowman's capsule diameter, renal corpuscle area, and glomerular area compared with those of the 2K1C group.

Bowman's capsule diameter (A), renal corpuscle area (B), glomerular area (C), and Bowman's space (D) of right (RK) and left kidney (LK) from either sham operative (SO) or 2-kidney-1-clip (2K1C) hypertensive rat after oral administration of 50 mg/kg BW Garcinia dulcis (GD) flower extract daily for 4 weeks. *P < .05, **P < .01, and ***P < .001 in comparison between groups (unpaired t-test).
Masson's trichrome staining (Figure 9) exhibited normal glomerular and renal tubular characteristics in the SO and SO + GD group. Renal fibrosis, as shown in blue color, was observed in glomerular and renal tubular area of both kidneys from the 2K1C group. The GD treatment did not alleviate the renal fibrosis in both kidneys of the 2K1C group.

Masson's trichrome staining of right (RK) and left kidneys (LK) from either sham operative (SO, A − D) or 2-kidney-1-clip (2K1C, E − H) hypertensive rats after oral administration of 50 mg/kg BW Garcinia dulcis (GD) flower extract daily for 4 weeks. Renal fibrosis areas (blue color) were indicated with a black arrow. Magnification power 200X.
The IHC for TNF-α (Figure 10) demonstrated that both kidneys of the 2K1C group had an increased TNF-α expression compared with the ipsilateral kidney of the SO group. The GD treatment lowered an TNF-α expression in both kidneys of the 2K1C + GD group. There was a significant difference in the positive area of TNF-α expression in kidney among all groups. For the RK samples, TNF-α expression in the 2K1C group were significantly higher than those of SO and the 2K1C + GD group. For LK samples, TNF-α expression in the 2K1C group was also significantly higher than those of SO and the 2K1C + GD group.
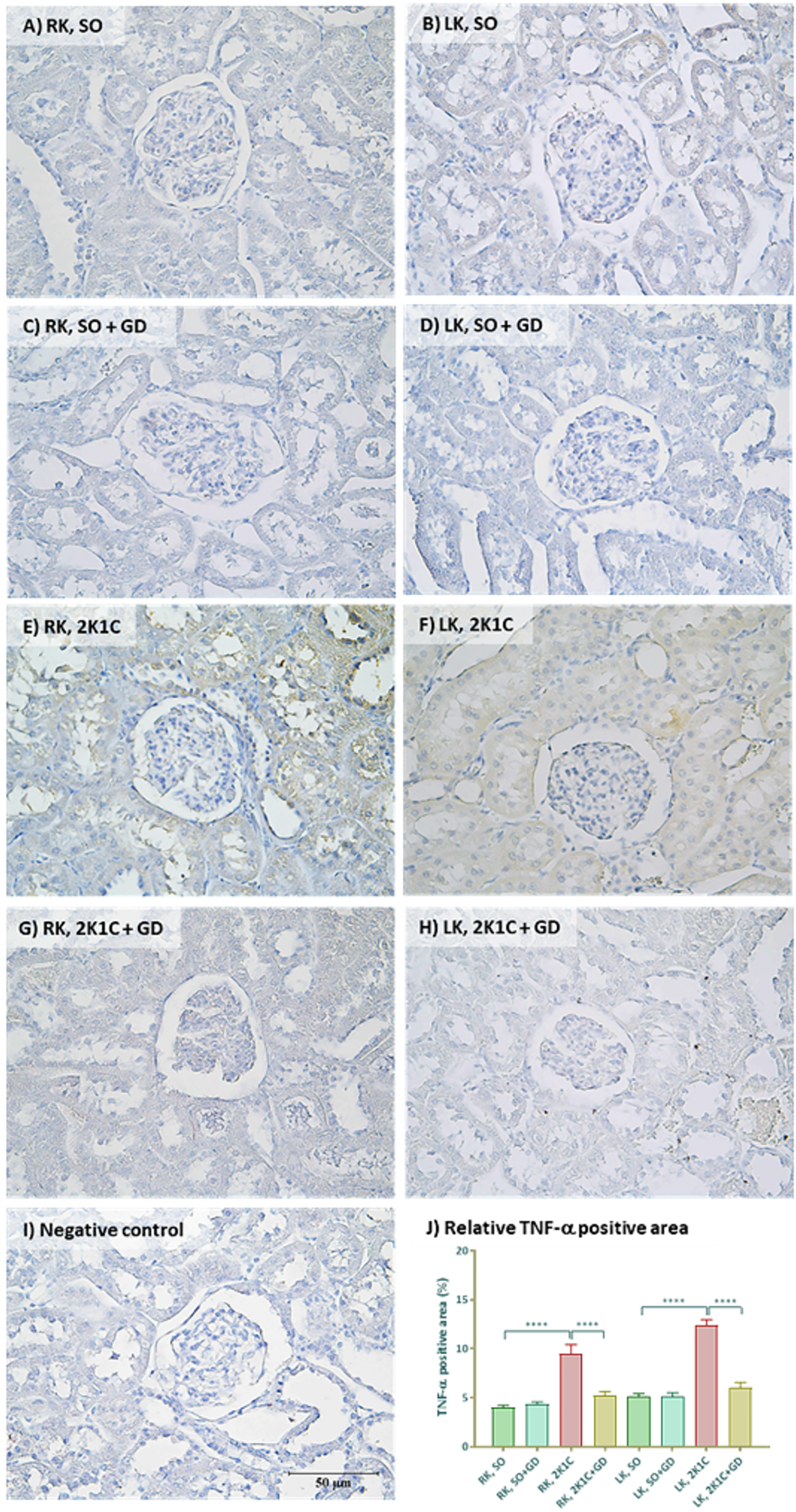
Immunohistochemistry for tumor necrosis factor alpha (TNF-α, brown color) in right (RK) and left kidneys (LK) of either sham operative (SO, A − D) or 2-kidney-1-clip (2K1C, E − H) hypertensive rats after oral administration of 50 mg/kg BW Garcinia dulcis (GD) flower extract daily for 4 weeks. Negative control (I) and the % relative TNF-α positive area (J) were shown. Magnification power 400X. ****P < .0001 in comparison between groups (unpaired t-test).
The IHC for eNOS (Figure 11) showed that eNOS expression in both kidneys of the 2K1C group was lower than that of the ipsilateral kidney of the SO group. Quantitatively, the positive areas of eNOS expression in kidney among all groups were significantly different. For the RK samples, eNOS expression in the 2K1C group was significantly lesser than the SO group. For the LK sample, eNOS expression in the 2K1C group was also significantly lower than the SO group. The GD treatment did not restore the observed impairment in eNOS expression in both kidneys of the 2K1C group.

Immunohistochemistry for endothelial nitric oxide synthase (eNOS, brown color) in right (RK) and left kidneys (LK) of either sham operative (SO, A − D) or 2-kidney-1-clip (2K1C, E − H) hypertensive rats after oral administration of 50 mg/kg BW Garcinia dulcis (GD) flower extract daily for 4 weeks. Negative control (I) and the % relative eNOS positive area (J) were shown. Magnification power 400X. ****P < .0001 in comparison between groups (unpaired t-test).
Discussion
We previously reported that serum angiotensin II levels in the 2K1C, but not SO group increased by 4.75 folds at 4 weeks after induction of hypertension. The BP in the 2K1C group were significantly higher than those of the SO normotension, and 50 mg/kg GD flower extract could decrease the augmented BP in the 2K1C rats. Although the renal blood flow and the glomerular filtration rate (GFR) in the 2K1C group were not different from the SO, renal vascular resistance, urine flow rate, osmolar clearance, negative free water clearance, and fractional excretion of sodium and potassium significantly increased suggesting remarkable alterations of renal tubular function in the hypertensive rats. In addition, treatment with GD flower extract (50 mg/kg) restored those renal tubular impairments in the 2K1C group, which suggesting its renoprotective effect that may involve some anti-inflammatory properties. 16
In this study, we further demonstrated that MDA levels in serum, liver, and both kidneys of the 2K1C hypertensive group were significantly higher than those of SO, which suggesting the occurrence of oxidative stress and inflammation in the 2K1C hypertensive group. In general, chemical reaction between ROS and unsaturated fatty acid or lipid peroxidation finally results in MDA formation, therefore, the level of MDA reflects the oxidative stress and inflammatory status. 23 Overactivation of RAAS system in 2K1C is likely to trigger a production of ROS. 6 Moreover, there is also supportive evidence that angiotensin II activates angiotensin type 1 receptors to promote an inflammatory response and organ damage. 24 Our recent findings accordingly showed serum angiotensin II levels in the 2K1C group were significantly higher than observed in the SO normotensive group. 16 Therefore, the high level of angiotensin II may induce oxidative stress and tissue inflammation in our hypertensive model. Since GD flower treatment can lower the MDA levels in both serum and the kidney and liver tissue of 2K1C rats, we reasonably propose this extract has anti-inflammatory effect in both systemic and tissue responses. The mechanism of anti-inflammatory action may involve the NF-κB signaling pathway and subsequently attenuate the releasing of pro-inflammatory cytokines such as TNF-α, interleukin-6, interleukin-1, prostaglandin E2 and nitric oxide from the immune response cells. 17
In the same way, we found that expression of TNF-α in liver and kidney tissues of the 2K1C hypertensive group were significantly higher than those of the SO group. This finding confirmed the local inflammation in those tissues of hypertensive rats. TNF-α is a proinflammatory cytokine released by macrophages and monocytes during acute inflammation. 25 It plays a role in the maintenance of inflammation through the induction of adhesion molecules, chemical factors, and cytokines, besides acting on the filtration and activation of macrophages. Recently, there are accumulated evidence reported that TNF-α plays a crucial role in inflammatory renal tissue injuries induced by hypertension. Since TNF-α would be able to promote renal vascular constriction and reduce GFR and induces diuresis and natriuresis, 26 it is conceivable that TNF-α caused the alteration of renal function in our animal hypertensive model. Previous study also reported the increased TNF-α levels in RVLM of 2K1C rats which may play an important role in raising of sympathetic vasomotor tone and BP in this hypertensive model. 27 From our present study, the treatment with GD extract is able to reduce the TNF-α expression in the liver and kidney tissues of the 2K1C group. This finding not only supports the anti-inflammatory property of the GD flower extract but also revealed the anti-hypertensive and the renoprotective effect.
In addition, IHC study demonstrated that eNOS expression in both kidney samples of the 2K1C group are lower than that of the ipsilateral kidney of the SO group. In general, eNOS is expressed in both vasculature and tubular epithelial cells of the kidney. eNOS exerts several important roles in controlling renal function such as increased GFR, prevention of nephropathy, and blunting the myogenic response, tubuloglomerular feedback, tubular reabsorption, and oxidative stress. 28 It was reported that TNF-α attenuates NO production by destabilizing eNOS mRNA and reduced eNOS protein expression. 5 Moreover, the reduction of eNOS protein levels was reported in the renal medulla of both clipped and contralateral kidney of the 2K1C rat. 29 In this study, the GD extract could not restore the eNOS expression in the kidney of 2K1C rat, despite increasing eNOS mRNA levels were observed in the isolated thoracic aorta from those 2K1C rat. 16 Thus, it is possible that our renovascular hypertensive animal model retained a severe and chronic damage of the kidney.
An abnormal histology of the kidney was observed in both kidneys of the 2K1C group along with an increase in mesangial cell proliferation, expansion of mesangial matrix, widen Bowman's capsule space, congestion of glomerular capillary and blood vessel, cloudy swelling of renal tubular epithelial cell, and increased renal fibrosis. Measurement of renal corpuscle components demonstrated that both kidneys of 2K1C rats showed a reduced Bowman's capsule diameter, renal corpuscle area, glomerular area, and Bowman's capsule space in comparison to the ipsilateral kidney of SO rats. Data from our previous study showed that the weights of LK decreased, while those of RK of the 2K1C rats increased when compared with ipsilateral kidney of control group. 16
Those above abnormalities of 2K1C kidneys are likely associated with the overactivation of RAAS-induced glomerular injury, tubular inflammation, and renal fibrosis. 3 The reduction in renal corpuscle size may involve angiotensin II-induced mesangial cell contraction and proliferation. Angiotensin II exerts several effects on mesangial cells such as cytosolic calcium stimulation, inositol phosphate formation, prostaglandin production, cell contraction, and stimulation of cell growth and synthesis of a variety of proteins, essentially the components of the extracellular matrix including collagen and fibronectin. 30 Moreover, the previous study reported that TNF-α level is increased in renal tubule and interstitial cells of the kidney injury rat model suggesting that TNF-α may associate with renal interstitial fibrosis.31,32
In this study, the GD treatment partially restores those impairments since the slight increased mesangial cell proliferation, slight congestion of glomerular capillary, and renal fibrosis were stilled observed in 2K1C + GD rats. However, the GD treatment has the interesting property to preserve the glomerular and renal tubular functions in 2K1C rats. 16 In addition, LK of 2K1C + GD rats had a significantly lesser Bowman's capsule diameter, renal corpuscle area, and glomerular area compared with the ipsilateral kidney of 2K1C rats, while those parameters did not differ significantly between the kidney of SO and SO + GD rats. These data suggested the severity of renal stenosis in the LK of 2K1C + GD rats is higher than for 2K1C rats.
Previous studies also reported the corresponding abnormalities of 2K1C rat's kidneys, however, there was a difference in some extent. It has been shown that clipped kidneys showed cortical interstitial fibrosis, abnormalities of renal corpuscle and renal tubule, glomeruli lesion, thickening of basement membrane, and increased collagen in perivascular, peritubular, and pericapsular area. Moreover, the expression of renin, laminin, interleukin-6, transforming growth factor-β and TNF-α increased in clipped kidneys of 2K1C rat.29,33 The weight of clipped kidneys decreased but non-clipped kidneys remained unaltered compared with ipsilateral kidneys of the control group. In non-clipped kidneys of 2K1C rats had higher mesangial cell expansion, interstitial fibrosis, glomeruli macrophage infiltration, interstitial macrophage infiltration, vascular wall thickness, intrarenal angiotensin II expression than the clipped kidneys and ipsilateral kidneys of the control group. 34 These data suggested that the degree of kidney damage may depend on the severity and duration of renal stenosis development.
A limitation of this study is the lack of information about the molecular signaling pathway(s) involved in the anti-inflammation properties of GD flower extract. In addition, clinical application of GD flower extract as anti-hypertensive and/or anti-inflammatory agents should be investigated further.
Conclusion
Daily oral administration of 50 mg/kg GD flower extract for 4 weeks demonstrated an anti-inflammatory effect by lowering the MDA levels in serum and liver and suppressed an expression of TNF-α in liver and both kidneys of 2K1C hypertensive rats. Kidney damage occurred in both clipped and non-clipped kidney of 2K1C rats. Indeed, the GD flower extract partially diminished the glomerular injury but could not be able to restore the renal fibrosis or the eNOS expression impairment occurred in 2K1C rats.
Footnotes
Acknowledgements
The GD flower extract was kindly supported by Assoc.Prof.Dr. Wilawan Mahabusarakam, Faculty of Science, Prince of Songkla University, Thailand.
Author Contribution
NT − conceived and designed the experiments, performed the experiments, analyzed, and interpreted the data, contributed reagents & materials, analysis tools or data, wrote the manuscript; RP − performed the experiments, contributed reagents & materials, wrote the paper; WH, PM, PC, PS, KS, and SH − analyzed and interpreted the data, analysis tools or data, wrote the manuscript. All authors have read and agreed to the published version of the manuscript. All authors have read and agreed on the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Thammasat University (Fast track Grant No. TUFT 19/2021).
Ethical Approval
All the reagents used in this study were prepared, used, and disposed of according to the set laboratory guidelines. Animal study was approved by the Thammasat University Animal Care and Use Committee under Protocol No. 031/2018.
