Abstract
Introduction
Pterocarpus marsupium, a medium deciduous tree known as Indian kino or Vijayasar, is used to treat diarrhea, ulcer, and skin diseases in traditional medicine. The aqueous extract of the heartwood is effective in controlling blood sugar levels. 1 Pterostilbene (trans-3,5-dimethoxy-4′-hydroxystilbene) is a bioactive phytochemical from the tree bark and is one of its many flavonoids and polyphenolic components. 2 It is a 3,5-dimethoxy analog of resveratrol and is synthesized by plants as a phytoalexin in response to stress, infection, and UV exposure.3–5
Pterostilbene is a low molecular weight molecule that has good absorption and distribution in the body and reasonable metabolic stability. 6 Despite structural similarity between pterostilbene and resveratrol pterostilbene is reported to have superior pharmacological activities compared to resveratrol.7–9 Additionally, dietary supplementation of pterostilbene was superior to resveratrol in improving cognitive function in experimental animals. Pterostilbene, could be detected in brain tissue, suggesting its ability to cross the blood-brain barrier. 8 Furthermore, it is not as readily converted to glucuronides or sulfates as resveratrol.9,10 The molecule has recently received significant attention, showing several therapeutic benefits in neurological, cardiovascular, metabolic, and hematologic disorders by acting on several signaling pathways.11–14 In a recent study, pterostilbene showed a positive effect on gut microbial composition, gut barrier disruption, insulin resistance, and obesity by enriching Akkermansia muciniphila. 15 Pterostilbene is a promising supplement sometimes referred to as “better resveratrol” but has not been extensively evaluated in human clinical studies as its parent compound.
The preclinical safety of chemically synthesized pterostilbene was studied in mice for 28 days at doses of 0, 30, 300, and 3000 mg/kg/day and showed no alteration in organ weight or clinical signs. 16 Synthetic pterostilbene at daily doses of 100 mg to 250 mg was well tolerated in hyperlipidemic adults. 17 Human safety data for P. marsupium tree bark extract containing high concentrations of pterostilbene is lacking. The present study was conducted to evaluate the clinical safety and short-term tolerance of a standardized extract of P. marsupium containing 90% pterostilbene at a given dose by assessing clinical and laboratory parameters and the occurrence of any adverse drug reactions associated with Silbinol®, in healthy volunteers.
Materials and Methods
Materials
The Sami-Sabinsa Group provided, a standardized extract containing 90% pterostilbene (Silbinol®) (Figure 1) from P. marsupium (PME) heartwood. The extract contained trace amounts of monomethyl and trimethyl resveratrol, polyphenols at 1% each, tannin, and coloring matters- which constituted 2%, and ash and moisture constituted 5% of the extract. Since the dose prescribed for the commercial product is 200 mg/day, the present study was carried out at a 100 mg dose twice a day.

Design and Ethics
A randomized, placebo-controlled, double-blind study was conducted on 60 healthy volunteers for 60 days from October 2020 to March 2021 at BGS Global Institute of Medical Sciences & Hospital, Bangalore. The sample size was derived based on incidence of gastrointestinal adverse events in a study conducted on pterostilbene by Riche et al. 17 In this study the incidence of gastrointestinal adverse events among pterostilbene subjects was approximately 4.00%. Assuming similar incidence in our study the sample size was determined as 60 by the study statistician. The details of the calculation are given in the supplementary section. The study included healthy male and female volunteers aged 18–49 years. The study investigator determined their health condition through medical history, physical examination, vital signs, laboratory tests, and an electrocardiogram (ECG) assessment. All the participants signed the informed consent and agreed to regular follow-up visits during the study.
The trial was carried out according to the Declaration of Helsinki, the International Conference on Harmonization Guidelines for Good Clinical Practice, and applicable local regulations. The study was approved by the Institutional Ethics Committee of BGS Global Institute of Medical Sciences & Hospital, Bangalore with the registration number CTRI/2019/08/020736. The study was coordinated and sponsored by ClinWorld, the clinical research division of. Sami-Sabinsa Group Limited., Bangalore. The study was designed and monitored by ClinWorld and executed by BGS Global Institute of Medical Sciences & Hospital. The statistical analyses were performed by the independent biostatistics team at Sanjeevani Bio Services, Hyderabad.
Inclusion and Exclusion Criteria
Inclusion Criteria: Male and female volunteers in the age group of 18- 49 years, in good health as determined by the study investigator through medical history, physical examination, vital signs, laboratory tests, and electrocardiogram (ECG) assessment, willing to sign informed consent and attend regular follow-up visit were included into the study.
Exclusion Criteria: Participants who were on prescription or over-the-counter medications, including vitamins and minerals, who had a history or presence of hepatic or gastrointestinal disease or other condition that interferes with drug absorption, distribution, excretion, and metabolism and those not willing to stop smoking or alcohol consumption were excluded from the study. Other exclusion criteria included the history or presence of renal, pulmonary, epileptic, hematologic, cardiovascular, neurological, or psychiatric illness and immunodeficiency diseases. Individuals with a history of drug abuse or alcoholism and those with positive human immunodeficiency virus (HIV) tests and positive surface antigens for hepatitis B (HBsAg) or hepatitis C tests were also excluded from the study.
Randomization and Blinding
Eligible subjects were randomized to PME or placebo in a 1:1 ratio according to the randomization sequence generated by a statistician, independent of the sponsoring organization, who was not involved in the conduct or reporting of the study. An alpha code was generated for both active and placebo to improve the blinding of the study and concealment of allocations using computer-generated random allocation software (version 2.0). Block randomization (block size four) was followed. All study staff, investigators, and subjects were blinded throughout the study. The randomization codes were kept strictly confidential and were accessible only to authorized persons on an emergency basis as per the Sponsor standard operating procedures until the time of unblinding.
Intervention
The participants were instructed to take one capsule (100 mg) of PME or an identical placebo containing microcrystalline cellulose after breakfast and one after dinner (200 mg/day) for 60 days.
Outcome Measures
The primary end point was safety evaluation of Pterostilbene through the changes in vital signs, ECG, laboratory parameters and occurrence of adverse reaction from baseline to day 60. The secondary outcome was to evaluate the change in the antioxidant markers.
Analysis
Each participant was subjected to a detailed clinical examination. General physical examinations, including vital signs (body temperature, respiratory rate, resting pulse rate, and blood pressure) were carried out by a qualified medical practitioner. All participants were followed up for 15 days after completion of the study to check general well-being and safety.
The laboratory parameters analyzed included complete and differential blood count, lipid parameters, liver enzymes, kidney parameters, thyroid profile, routine urine analysis at the screening visit (baseline), day 30 and day 60. Fasting blood sugar (FBS) and glycosylated hemoglobin (HbA1C) were analyzed only at the screening and final visit. As an exploratory endpoint, glutathione (GSH), superoxide dismutase (SOD), and the extent of lipid peroxidation were analyzed at the initial and final visits. According to the regulatory requirement, all biochemical parameters were evaluated in a centralized lab with NABH accreditation (Elbit diagnostics, Bangalore). Total GSH was estimated using the Potassium Phosphate -EDTA) method, 18 malondialdehyde was estimated using the lipid peroxidation method, 19 and SOD using the Xanthine oxidase & Nitroblue Tetrazolium method. 20
Statistical Analysis
The statistical analysis was performed for all the subjects by STATA Software version 16.0. The data gathered was considered as either continuous or categorical variables. Continuous variables and normally distributed data within the group were compared using the paired t-test, and the data were presented as mean difference and P-value. The Wilcoxon Signed-Rank test was used to analyze the data not distributed normally, and the results are represented as median and P-value. For the comparative analysis between treatment groups, the Mann-Whitney test was used. The level of statistical significance was defined as P < .05.
Categorical variables are presented as frequency and percentage of the population. A descriptive comparison is provided to differentiate the treatment effect between and within the groups.
Results
Baseline Demographics
A total of 92 subjects were screened, of which 60 subjects (27 males and 33 females) were eligible and randomized, N = 30 in each PME and placebo group. Of the 60 subjects, fifty-nine completed the study (N = 29 in PME and N = 30 in placebo), and one was lost to follow-up (Figure 2). The demographic details are given in Table 1, which were comparable between the study groups. The mean age was 31.92 years in overall subjects, 30.45 years in the PME group, and 33.33 years in the placebo group. The average BMI was 23.43 ± 2.91 kg m−2, and the average weight of the population studied was 61.02 ± 9.82 kg.
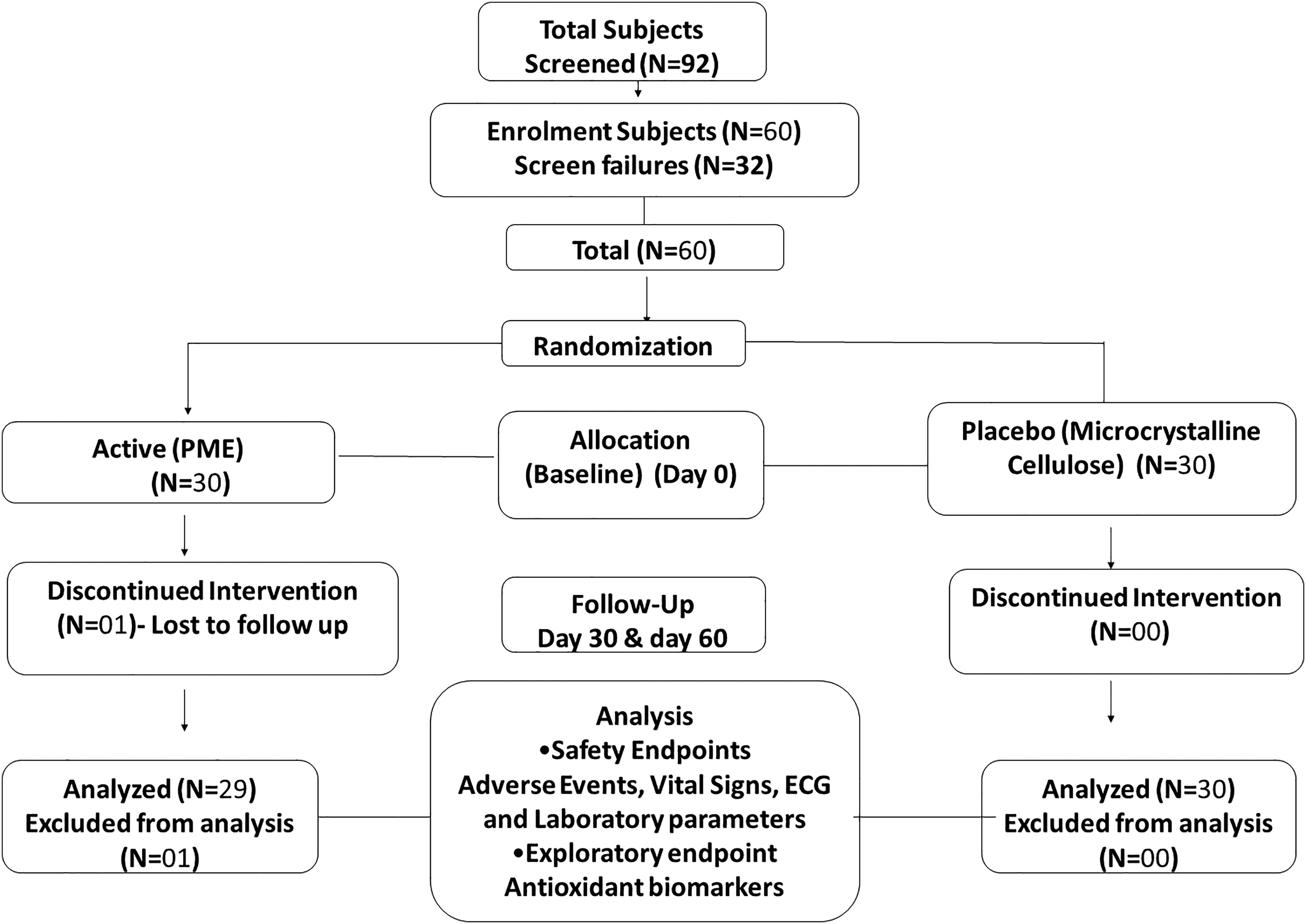
Consort diagram.
Baseline Demographics (Mean ± SD).

The values are represented as Mean ± SD. BMI: Body mass index
Anthropometric and Vital Parameters
At the end of the study, both groups showed no significant change in body weight, BMI, blood pressure, body temperature, pulse rate, and respiratory rate (Table 2) and no change was observed after 30 days of supplementation (Table S1, S2). The ECG records were normal in all patients at both the screening and the final visit (Table 2).
Blood Pressure, Pulse Rate and Other Vital Signs.
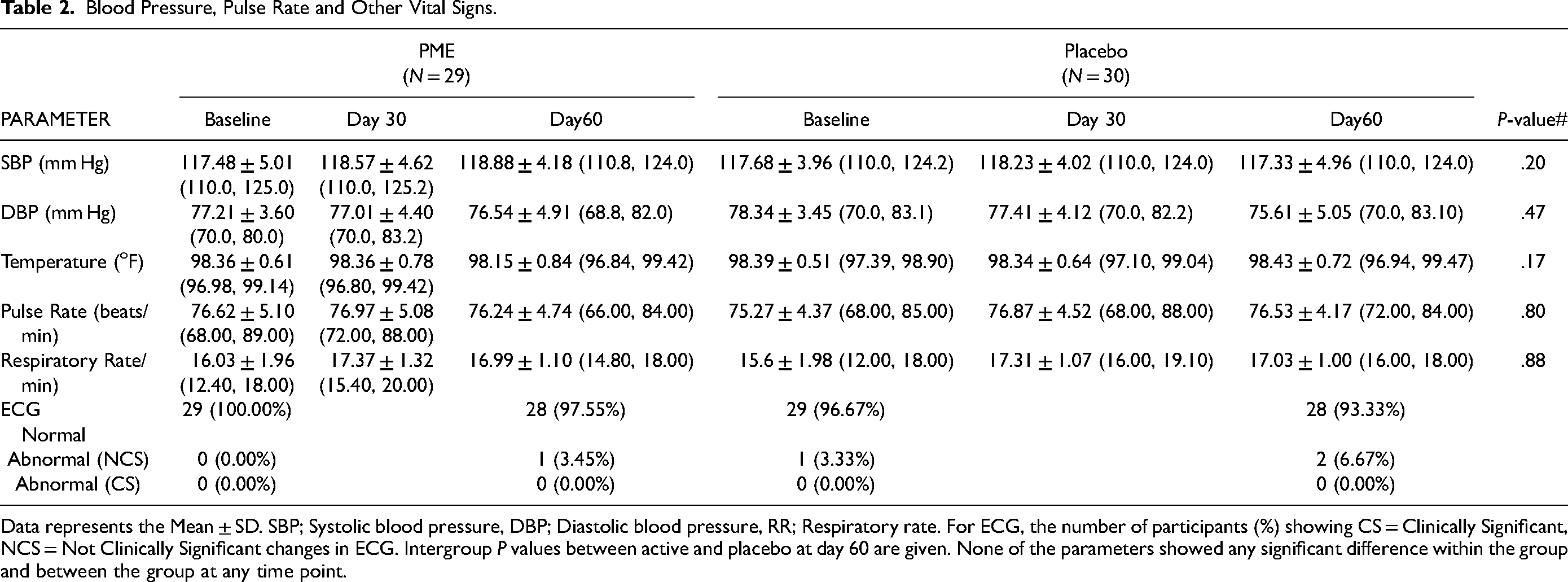
Data represents the Mean ± SD. SBP; Systolic blood pressure, DBP; Diastolic blood pressure, RR; Respiratory rate. For ECG, the number of participants (%) showing CS = Clinically Significant, NCS = Not Clinically Significant changes in ECG. Intergroup P values between active and placebo at day 60 are given. None of the parameters showed any significant difference within the group and between the group at any time point.
Hematological Profile
Hematological parameters such as hemoglobin, erythrocyte count, platelet count, packed cell volume (PCV), mean corpuscular volume (MCV), mean platelet volume (MPV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), total and differential leucocytes counts were within the normal limits, 21 and relative change from screening to final visit between PME and placebo group was not statistically significant. All hematological parameters were comparable between the PME and placebo groups at baseline and at the end of the study and after 30 days (Table 3 and Table S3).
Hematological Analysis.

Data represents the Mean ± SD. Inter group P values between active and placebo at day 60 are given. The normal range was defined as hemoglobin (12.0 to 16.0 g / dl), platelet count: (150-350×106/L), Erythrocytes(4.1-5.9 ×106/L) PCV-Packed Cell Volume (37%-52%) MCV-Mean Cell Volume (80-100 fl), MCH-Mean corpuscular hemoglobin (26.0 to 34.0 pg / cell), MCHC-Mean corpuscular hemoglobin concentration (31.0 to 37.0 g / dL), MPV-Mean platelet volume 8 to 12 fl), total leukocytes (4.5 to 11.0 × 109/L), lymphocytes (22%-44%), Eosinophils (0% to 8%), Monocytes (4% to 10%), Neutrophils- (4% to 70%), Basophils- (0% to 3%)*P < .05 statistically significant difference within the group at day 60 compared to baseline. # P value between PME and Placebo at day 60.
Blood Biochemical Analysis
Lipid Profile
Lipid levels showed some fluctuations but were within the reference range in both PME and placebo. The changes in total cholesterol (TC), triglycerides (TG) and very low-density lipoprotein cholesterol (VLDL-C) were within the normal reference range (TC: < 200 mg/dL, TG < 150 mg/dL and VLDL-C: < 50 mg/dL) throughout the study. The median Low density lipoprotein cholesterol (LDL-C) levels were 112 (96.3,133.3), and 98.2 (84.8,126.8) mg/dL in PME and 109.3 (95.8,121.3) and 106.2 (86.8, 115.9)mg/dL, in placebo at baseline and day 60, and high density lipoprotein cholesterol (HDL-C) levels were 41 (34.5, 47.5) and 38 (33,42.5) mg/dL in PME and 41 (35,49) and 40 (30.5,46) mg/dL in placebo at baseline and day 60,. Both LDL and HDL levels were at borderline (LDL-C:<100 mg/dL and HDL-C:40-60 mg/dL) at the baseline and remained in the same range at day 60. 22 No statistically significant differences were observed between the PME and placebo groups (Table 4 and Table S4).
Blood Biochemical Parameters.

Median and interquartile values for lipid parameters are given. The normal range were TG < 150 mg dL−1, TC < 200 mg dL−1, LDL <100 mg dL−1, VLDL < 30 mg dL−1, HDL 40-60 mg dL−1, FBS was < 100 mg/dL and Hb1AC was < 5.7%, ALT- 7 to 55 IU/L, AST- 8 to 48 IU/L, ALP- 40 to 129 IU/L, GGT- 8 to 61 IU/L, Bilirubin- 0.1 to 1.2 mg dL−1, urea-6-24 mg dL−1, uric acid- 2.4-7 mg dL−1 creatinine- 0.7-1.3 mgdL−1, eGFR→60, sodium-135-145 mmol L−1 potassium-3.6-5.2 mmol L−1, chloride-96-106 mmol L−1, T3-0.8-2 ng/ml, T4- 5.0–11.0 ug/dL, TSH- 0.40. Low-Density Lipoprotein-Cholesterol, HDL-C: High-Density Lipoprotein-Cholesterol, VLDL-C: Very low-density lipoprotein C, FBS: Fasting blood sugar, HbA1C: Glycosylated Hemoglobin, AST- Aspartate Amino Transferase, ALT -Alanine aminotransferase, ALP- Alkaline Phosphatase, GGT-Gamma-glutamyl transferase, eGFR-Estimated Glomerular Filtration Rate; T3: Serum-triiodothyronine, T4: Serum-Tetra-iodothyronine, TSH: Serum thyroid stimulating hormone,
*P < .05, ** P < .01 statistically significant difference within the group at day 60 compared to baseline.
# P value between PME and Placebo at day 60.
Blood Sugar Profile
Fasting blood sugar was 87 [72.20 to 132.20] mg/dL in PME and 86 [70.90 to 106.65] mg dl−1 at the end of the study. Similarly, no significant change was observed in glycosylated hemoglobin percentages 5.50 [5.00 to 6.08] g% and 5.50 [5.00 to 5.90] g% for PME and placebo, respectively. Both groups’ values were within the normal range at screening and the final visit (Table 4).
Liver and Renal Function
Parameters of liver function such as total bilirubin, Aspartate transaminase (AST), Alanine transaminase (ALT), and Gamma-glutamyltransferase (GGT) were within the standard reference ranges in subjects supplemented with PME, comparable to the placebo group at the screening, day30 and final visit. Renal function parameters like urea, uric acid, creatinine, glomerular filtration rate, sodium, potassium, and chloride levels were comparable between placebo and PME at all time points (Table 4 and Table S5, S6).
Thyroid Function
The mean serum-triiodothyronine (T3), serum-tera-iodothyronine (T4), and serum-thyroid stimulating hormone (TSH) were 1.40 ± 0.18(μg/dL), 8.49 ± 1.98(μg/dL), and 3.02 ± 2.19(μIU/ mL), respectively, at the screening visit. T3, T4 and TSH at the final visit were 1.44 ± 0.2(ng/mL), 9.20 ± 2.23(μg/dL), and 2.62 ± 1.37(μIU/mL), respectively. No statistically significant changes were observed in the thyroid function test outcomes for the PME group compared to the placebo at all time points (Table 4 and Table S7).
Urine Analysis
Urine analysis for physical parameters like color, appearance, pH, specific gravity, protein, glucose, ketone bodies, bilirubin, nitrites, urobilinogen, and microscopic parameters, were observed within the normal reference range and were comparable between PME and placebo groups at all time points (Table S8).
Antioxidant Biomarkers
Glutathione was 128.25 ± 28.32 (nM/mL) and 132.46 ± 25.55 (nM/mL) respectively in the PME and placebo arms at the initial visit which changed to 142.28 ± 39.46 nM/mL) and 134.45 ± 34.42 (nM/mL) respectively, at the final visit. The mean change from baseline to final visit in the PME arm was 14.41 (increase of 11.3% from baseline), comparatively higher than in the placebo arm. Malondialdehyde remained unchanged at the final visit compared to baseline values in both the PME and placebo groups. Superoxide dismutase decreased in both the PME and placebo groups at the end of the study (Table 5).
Antioxidant Biomarkers (Mean ± SD).

# P value between PME and Placebo at day 60 *P < .05 statistically significant.
Adverse Events
Adverse events were recorded throughout the study by self-reporting from the subjects and clinically significant laboratory parameters and vital signs changes. Three participants in the placebo group reported mild adverse events of fever and fatigue, which were mild and resolved (Table 6). None of the participants reported other adverse events during the study period of 60 days, and no clinically significant biochemical changes were observed in the PME and placebo groups (Supplementary section Table 1-9).
Summary of Adverse Events Reported During the Study.

R. No: Randomization number, AE: Adverse event, SAE: Severe adverse event
Discussion
The current study evaluated the clinical safety of Silbinol®, a standardized extract containing 90% pterostilbene from P. marsupium (PME) at a dose of 200 mg per day in healthy human volunteers. The clinical and biochemical parameters did not show significant changes, during the study duration of 60 days suggesting the extract's safety for human consumption. Pterostilbene is gaining importance as a dietary supplement for various therapeutic benefits, as it offers higher bioavailability than resveratrol. 6 Although there are numerous preclinical studies on the pharmacological effects of P. marsupium extracts, their safety has not been systematically studied in healthy humans. Preclinical studies on antioxidant and antidiabetic activities have been carried out with aqueous, methanolic and ethyl acetate extracts of the bark of P. marsupium tree.1,23,24 Silbinol, extracted from the heartwood of the tree has been found to be safe and effective in preclinical models of diabetes, chronic kidney disease, CVD and cancer.25–27 Chemically synthesized pterostilbene was not found to be toxic even at very high oral doses at 3000 mg/kg in mice. 16 In another recent study, intragastric administration of pterostilbene up to 500 mg/kg indicated no special concerns for toxicity. 28 Previously, Riche et al reported the efficacy and safety of chemically synthesized pterostilbene at 125 mg twice daily for 52 days in 80 hypercholesteremic patients [16]. A short-term safety of P. marsupium extract containing pterostilbene was established for 450 mg once daily or 225 mg twice daily dose in five subjects in each arm for 14 days. 29 In contrast to these two reports, the current study used P. marsupium extract, standardized for 90% pterostilbene, to establish the clinical safety of the natural product. Besides regular hepatic, renal, and glucose markers, hematology, thyroid, lipid profiles, complete urine analysis, clinical parameters, and ECG were assessed as safety markers.
We observed some fluctuations in lipid levels in the present study, although the values were still within the normal reference range, for TC, TG and VLDL as suggested by American heart association, 30 while, LDL-C and HDL-C levels were in the borderline in both groups from the baseline to the end of the study. In a recent analysis based on two studies, synthetic pterostilbene was reported to increase total cholesterol and LDL-C.31–33 One of the studies was conducted in combination with nicotinamide riboside in elderly subjects 60–80 years old, while the other was conducted in patients with hypercholesterolemia. The LDL increase was reported to get attenuated in the presence of a baseline cholesterol medication in hypercholesterolemic patients, and this increase did not influence the atherosclerotic cardiovascular disease risk scores. 33 In the second study in geriatric subjects, the baseline cholesterol levels were significantly different in the treatment groups compared to the placebo, which could have been the confounding factor in the interpretation of the results. In addition, the study used a combination of pterostilbene with nicotinamide riboside.
Contrary to these reports, our study showed a slight decrease in LDL-C and total cholesterol at day 60 compared with the initial levels in both groups. Lipid levels are influenced by eating habits and have a wide biological variation in humans. Biological variability in measurement of cholesterol and HDL-C was reported to be 6–7%, while it was 9% for LDL-C, and 28% for triglyceride. 34 Therefore, the variations observed in the present study were only natural variations in lipid levels, which were comparable for both the placebo and the active arms. Another contributing factor to the fluctuations in lipid levels could be the study time during the festival season in India (the last quarter of 2020).
The etiology of several diseases is often related to inflammation and oxidative damage. The antioxidant activity of pterostilbene has been implicated in the multiple health benefits observed in animal and human studies.33,35 Several cell-based studies have shown a decrease in reactive oxygen species by pterostilbene. 36 We observed an 11% increase in total glutathione (GSH) levels in subjects consuming PME, which was comparatively higher than placebo, although not statistically significant, perhaps due to the considerable variation within the participants. However, SOD levels declined in all subjects, and lipid peroxidation remained unaffected. Antioxidant defense systems are highly complex and subject to intricate control. In healthy subjects, the system generally remains in homeostasis. In an earlier study, short-term oral supplementation of GSH did not improve glutathione status nor reduce markers of oxidative stress in healthy adults, 37 substantiating our results.
Safety is of utmost importance for any product consumed by humans. Scientific evaluation of the safety of a standardized herbal extract in healthy individuals authenticates its use as a supplement for the general population. In the present study, none of the subjects reported any serious adverse events during the study period of 60 days, strengthening the preclinical data on the safety of pterostilbene. The clinical, and biochemical information projected a relatively safe and normal profile of the herbal supplement in healthy individuals at a dose of 100 mg bid (200 mg per day) within the trial period and further supported the safety profile of PME containing 90% pterostilbene.
A few limitations of the study could be that the study was conducted at a single site, which limits the ethnicity of the population studied. The study did not include special populations such as pediatrics, geriatrics, pregnant and lactating women, and use in patients suffering from renal, hepatic, cardiac diseases. This study was not a large cohort study, although the sample size was sufficient to meet the 0.8 power, which could be another limitation. Future studies in special population for long duration and studies related to drug-supplement interaction would be valuable to ascertain the long -term human safety of this supplement.
Conclusions
Although there are limited clinical studies, pterostilbene is being pursued as a more effective supplement with similar therapeutic benefits as resveratrol. This randomized, placebo-controlled clinical study in healthy adults highlights the safety of natural PME at a dose of 200 mg per day (100 mg, bid). Our results pave the way for further therapeutic studies on the multiple health benefits of PME and its use as a dietary supplement.
Supplemental Material
sj-docx-1-chp-10.1177_2515690X231198312 - Supplemental material for A Short-Term Safety Evaluation of Silbinol®- an Extract from Pterocarpus marsupium in Healthy Adults- a Randomized, Double-Blind, Placebo-Controlled Study
Supplemental material, sj-docx-1-chp-10.1177_2515690X231198312 for A Short-Term Safety Evaluation of Silbinol®- an Extract from Pterocarpus marsupium in Healthy Adults- a Randomized, Double-Blind, Placebo-Controlled Study by Muhammed Majeed, Kalyanam Nagabhushanam, Shaji Paulose and Lakshmi Mundkur in Journal of Evidence-Based Integrative Medicine
Supplemental Material
sj-doc-2-chp-10.1177_2515690X231198312 - Supplemental material for A Short-Term Safety Evaluation of Silbinol®- an Extract from Pterocarpus marsupium in Healthy Adults- a Randomized, Double-Blind, Placebo-Controlled Study
Supplemental material, sj-doc-2-chp-10.1177_2515690X231198312 for A Short-Term Safety Evaluation of Silbinol®- an Extract from Pterocarpus marsupium in Healthy Adults- a Randomized, Double-Blind, Placebo-Controlled Study by Muhammed Majeed, Kalyanam Nagabhushanam, Shaji Paulose and Lakshmi Mundkur in Journal of Evidence-Based Integrative Medicine
Footnotes
Acknowledgments
The authors thank Dr Balachandra and his group of doctors for their contributions and the participants, without whom the study would not have been possible. The authors thank all members of the clinical research group for their help and assistance. The statistical analysis by the biostatistics team from Sanjeevani Bio Services, Hyderabad, is gratefully acknowledged.
Authors’ Contributions
MM: Conceptualization, Resources and review
KN: Study Design, Editing and Review,
SP: Clinical study protocol, data interpretation, review
LM: Data interpretation, original writing, review and editing
Data Availability Statement
All data from the trial are included in the manuscript. The corresponding author can be contacted for further information.
Declaration of Conflicting Interests
All authors are affiliated with Sami-Sabinsa Group Limited or Sabinsa Corporation.
The product Silbinol®, which contains natural Pterostilbene from P. marsupium, is marketed by Sabinsa Corporation.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
The study was approved by the Institutional Ethics Committee of BGS Global Institute of Medical Sciences (Bangalore).
Clinical Trial Registration
Patient Consent Statement
All participants signed an informed written consent to participate in the study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
