Abstract
Cancer mortality is a global concern. The current therapeutic approaches despite showing efficacy are characterized by several limitations. Search for alternatives has led to the use of herbal plants including C. edulis and P. capensis. However, there is limited research on antiproliferative effects of these medicinal plants. The study sought to evaluate antiproliferative effects of the plants against human breast and prostate cancers using cell viability, and gene expression assays to determine modulation of apoptotic genes. Further, Liquid Chromatography Mass Spectrophotometer (LC-MS) and Gas Chromatography Mass Spectrophotometer (GC-MS) analyses were performed to confirm phytocompounds in the extracts. The results indicated that ethylacetate extracts of C. edulis and P. capensis had the highest activity against cancer cells with IC50 values of 2.12 ± 0.02, and 6.57 ± 0.03 μg/ml on HCC 1395 and 2.92 ± 0.17 and 5.00 ± 0.17 μg/ml on DU145, respectively. Moreover, the plants extracts exhibited relatively less cytotoxic activities against Vero cell lines (IC50 > 20 μg/ml). The extracts also exhibit selectivity against the cancer cells (SI > 3). Further, mRNA expression of p53 in the treated HCC 1395 was increased by 7 and 3-fold, whereas by 3 and 2-fold in DU145 cells, upon treatment with ethylacetate extracts of C. edulis and P. capensis, respectively. Similarly, several-fold increases were observed in the number of transcripts of Bax in HCC 1395 and HOXB13 in DU145 cells. Phytochemical analyses detected presence of phytocompounds including flavonoids, phenolics, tocopherols and terpenoids which are associated with anticancer activity. Findings from this study provide a scientific validation for the folklore use of these plants in management of cancer.
Keywords
Introduction
Cancer is a global pandemic. Estimates show that in 2020, more than 10 million cancer deaths were reported. 1 Furthermore, the global cancer burden is anticipated to upsurge to 28.4 million cases by 2040, representing a 47% rise from the case load of 2020. 2 Much of the cancer burden is expected to occur in the developing countries due to poor access to early detection and treatment.3,4 Molecular understanding of the mechanisms behind various variables in cancer has advanced recently, whereby neoplasia is considered as a complex yet extremely coordinated multistep process characterized by alteration of genes controlling basic aspects of cellular functions such as apoptosis, cell proliferation and DNA repair. 5
Principal targets of genetic modifications include tumor suppressor genes such as p53 and HOXB13. Activated p53 targets downstream genes such as BAX and p21 involved in induction of various signaling pathway such as apoptosis and cell cycle arrest. 6 In addition, TP53 promotes impairment of P13/AKT pathway, which plays a crucial role in diverse cellular functions such as proliferation, through the induction of PTEN. 7 Notably, it has been reported that alteration of P13/AKT/ pathway contributes to the initiation and progression of cancer. 8 Additionally, the HOXB13 protein has been linked to diverse cellular processes, including cell division, apoptosis, cell differentiation, and migration. 9 Thereby, inactivation of these genes eliminates their negative regulations over cellular proliferation resulting in uncontrolled cell proliferation and eventually cancer. 10
Current treatments for cancers include radiation cryosurgery, chemotherapy, hormonal and radiation therapy. 11 Chemotherapeutic approaches target vulnerabilities in cancer cells to induce apoptosis. However, despite the fact that these treatments have showed efficacy, adverse effects have been observed in patients. These effects are attributed to the nonspecific nature of chemotherapeutic drugs, resulting in targeting of noncancerous cells in the body. 11 Moreover, belligerent metastatic cancers pose a risk of relapse after remission by chemotherapy treatments. 12 Chemotherapy resistance has also presented an emerging obstacle, bringing in capricious resistance and unsatisfactory outcomes to antineoplastic and cytotoxicity agents. 13 Surgery is only limited to localized tumors. 14
The complexity associated with current treatments, their limitations and high costs, necessitate the need to explore safe, affordable and efficacious chemotherapeutic drugs. 15 One such alternative is the use of phytotherapeutics.16-18 Furthermore, current research has identified a number of medicinal herbs with potential antineoplastic possibilities. Among these are medicinal plants including Prunus africana, Caranthus roseus, and Molinga oleifera.19,20 Besides, plant derived compounds such as vincristines, taxol and vinblastine have improved the effectiveness of chemotherapy against neoplastic cells. 21 Anticancer efficacy of medicinal plants is attributable to diversity of their chemical properties, structural nature and minimal toxicity. 15 Furthermore, herbal medicine is arguably available, affordable and cultural acceptability. 22
Carissa edulis and Pappea capensis are medicinal plants belonging to the families Apocynaceae and Sapindaceae, respectively. 23 Carissa edulis also known as Carissa spiranum 24 is a fast growing branched evergreen shrub, with a scrambling height of up to 5 m with a milky sap, forming a dense canopy. Young branches are green and smooth with short hair but as they mature they become light brown with cracks. Leaves which simple and opposite have an ovate to ovate-elliptic shape with a dark green color above and a pale green colour below. 25 The plant occurs in bushveld, more so in riverine and dry vegetation and in evergreen or deciduous woodland. It is well distributed from East Africa and Senegal in the north to Limpopo and Mpumalanga in the south. 25
Pappea capensis, commonly known as a wild plum, indaba tree or Jacket plum is a small to medium monoecious tree with a height of about 4 m. 26 Depending on environmental conditions; it may be deciduous or evergreen. The leaves are simple, hard textured and waxy and are usually crowded at the edge of branches. The fruits are furry green capsules that eventually split to release an edible red flesh containing black seed P. capensis is widely distributed in the southern part of Africa where its coverage runs from Northern Cape through Mozambique and northwards into Zambia and Botswana. 26
C. edulis and P. capensis are used among African communities to treat coughs, malaria, stomach ache, diarrhea ulcers, rheumatism and diabetes.27,28 In addition, they are claimed to have anticancer activities based on the folktale practices. However, there is a dearth of knowledge to scientifically validate their medicinal potential on breast and prostate cancers, and their modes of action. Hence, the present study aimed at investigating antiproliferative activities of C. edulis and P. capensis extracts on human breast and prostate cancer cell lines, HCC 1395 and DU145, respectively, along with their modulation on apoptotic-related genes, p53, BAX and HOXB13.
Materials and Methods
Collection and Preparation of Plant Extracts
Fresh leaves and stem barks of C. edulis and P. capensis were collected from Nthawa location, Mbeere North, Embu County, Kenya. The study area is located between Latitude 0o 35’39’20.816’'S and longitude 37o27’34.71’’E. Authentication of plants specimens was done by an acknowledged taxonomist and the voucher specimen deposited at Plant Science Departmental Herbarium. For future reference the samples were assigned voucher specimen number; C. edulis was assigned voucher specimen number 2018/CM1 whereas P. capensis, 2018/CM2. Fresh plants parts were shade dried at room temperature. The dried samples were pulverized into fine particles, followed by extraction using aqueous, ethylacetate (EtOAc) and dichloromethane (DCM) solvents. Extraction was carried out using protocols earlier described by Nelson et al 15 and Pendota et al 28 with slight modifications.
For aqueous extraction, a mass of 350 g of powdered C. edulis leaves and P. capensis stem barks was each weighed and soaked separately in 1L of double distilled water. Extraction was then performed in a water bath at 60°C for 2 hr. The extracts were allowed to cool after which they were decanted and re-soaked in a water bath at 60°C for a further 2 hrs. This was followed by filtration using filter paper and double layers of muslin cloth. The filtrates were thereafter freeze dried.
For the organic extraction, the powdered C. edulis leaves and P. capensis stem bark were each weighed and steeped separately in IL of EtOAc (98%) and DCM (98%) for 24 h. Thereafter, the extracts were decanted and re-soaked with the respective solvent (1000 ml) for a further 24 h. The extracts were filtered and concentrated under a vacuum rotary evaporator at 40°C.
Cell Culture and Treatment
The cell lines, Vero, human breast cancer (HCC 1395) and prostate cancer (DU-145) used in this study were obtained from America Type Culture Center (ATCC, Rockville, MD) through the Centre for Traditional Medicine and Drug Research (CTMDR), KEMRI. The cells were cultured in a T75 culture flask containing Dulbelcco's Modified Eagle Medium (DMEM) complemented with1% L-Glutamine, 10% Fetal Bovine Serum (FBS) and 1% streptomycin. Thereafter, they were incubated for 48 h at 5% CO2 and 37 °C to attain confluence, after which they were trypsinized to detach them from the flask. Cell count was performed by trypan blue exclusion test. To attain an optimal seeding density per well, (2 × 104), cell suspension was diluted accordingly. A 100 µl cell suspension was seeded in 96 well plates and incubated at 37 °C and 5% CO2 for 48 h.
Cell treatment was done by adding 15 μl of test sample to wells in rows. Maintenance media (DMEM) (35 µl) was added to make a volume of 150 µl in each well. A 3 fold serial dilution was performed from raw II to VII to give 7 concentrations; 100, 33.33, 11.11, 3.70, 1.23, 0.41 and 0.14 µg/ml. Row I of 96 well plates remained untreated and served as negative control. This procedure was done for each plant extract and in triplicate. Doxorubicin served as positive control (reference drug). The cells were incubated in a humidified environment for 72 h.
Cell Viability Assay
In vitro growth inhibitory effects of the plant extracts were colometrically evaluated using MTT assay. The assay relies on a principle whereby mitochondrial succinate dehydrogenase catalyzes the reduction of a water soluble tetrazoliums salt, MTT, into an insoluble blue formazan product, by cleavage of tetrazolium rings. Formation of formazan reflects cell viability which is measured spectrophotometrically. For this assay, MTT (5 mg) was dissolved in 1 ml phosphate-buffered saline. Thereafter, into each well, MTT dye (10 µl) was added followed by incubation for 2 h. Formazan formed was dissolved using DMSO. Absorbance was read at 540 nm. Percentage growth inhibition and the half maximal inhibitory concentration (IC50) for cancer cell lines and cytotoxic concentration (CC50) for normal cell lines were determined. The selectivity index values (SI) were computed by dividing the CC50 of normal cells by IC50 values of cancer cells. 29
RNA Extraction and Quantitative RT-PCR
The PureLink RNA Mini Kit (Life technologies, USA) was used to isolate RNA from cells pre-treated with the EtOAc extracts of C. edulis and P. capensis. The extracts were considered since they exhibited the highest in vitro antiproliferation activities as well as the highest selectivity index values against HCC 1395 and DU145 cells. The quantity and purity of the extracted RNA was analyzed using NanoDrop ND-2000 spectrophotometer (Thermofischer). The isolated RNA was transcribed into cDNA using FIREScript RT Complete Oligo-(dT) cDNA synthesis kit (Solis BioDyne, Estonia). Thereafter, expression of three genes (wild type TP53, BAX and HOXB13) was measured by HOT FIREPol EvaGreen qPCR mix kit (Solis BioDyne, Tartu, Estonia). Notably, HCC 1395 and DU145 both have mutant TP53 gene.6,30 In addition, the DU145 cell line has a mutant HOXB13 gene. 31 Conditions used for PCR amplification were; incubation step at 95 °C for 12 min, denaturation step at 95 °C for 15 s (50 amplification cycles), annealing steps for 30 s at 58 °C and finally extension step for 1 min at 72 °C. Melting curve analysis was carried out so as to verify specificity of the amplicon. Relative expression of gene was quantified as per standard 2-Δct calculation using GADPH as the housekeeping gene.
Quantitative Phytochemical Analyses
Liquid Chromatography-Mass Spectrometry Analysis
In this analysis, chromatographic separation was done using an ACQUITY UPLC BEH C18 column (Waters Corp., Wexford, Ireland). The oven temperature was set at 45 °C. Two solvent with 0.01% formic acid, A (water) and B (methanol), were the mobile phase. The solvent gradient system used was initiated at 5% B and a final 100% held for 20 min at a flow rate of 0.2 ml/min. The MS were equipped with a nano electrospray ion source in positive mode. The MS scan parameter included the m/z range 40–2000 and desolvation temperature of 150 °C and 120 °C. Drying gas (nitrogen) flow rate was set at 800 L/h. LC-MS data were controlled with MassLynx version 4.1 SCN 712 (Waters). Probable assignments of detected compounds were determined after generation of mass spectrum for each peak. In addition, compound identities were determined by establishing molecular ion peaks using adducts, common fragments and literature online databases (METLIN, ChemSpider) and where possible verified with authentic samples through co-injections.
Gas Chromatography-Mass Spectrometry Analysis
Analysis was undertaken with a GC-MS instrument equipped with a BPX5 non-polar capillary column, 30 m; 0.25 mm ID; 0.25 µm film thickness. Starting temperature was set at 35˚C (5 min); for 10.5 min, the temperatures were increased (10˚C /min to 250˚C). This was followed by a further increase for 29.9 min at 285˚C The injection temperature was set at 200˚C and the carrier gas, He, at a flow rate of 1.25 ml/min in splitless mode. An EI system with IE of 70 eV was used for detection. GC-MS analysis was conducted using Shimadzu QP 2010-SE with line temperature at 280°C and the ion source temperature was 230˚C in electron impact mode. MS scan parameters included temperature of the interface (250˚C), EI ion voltage of 70 eV, 3.3 min of solvent cut time, mass range of 50–600 m/z, and scan speed, 1666 µm/s.
Determination of Phytocompounds Concentration
Concentrations of phytocompounds were quantified using calibration curves of external standards. For LC-MS analysis, oleic acid was used as the external standard whereas hexadecanoic acid was considered for GC-MS analysis.
Statistical Analysis
Data on absorbance of in vitro antiproliferative activities were subjected to descriptive statistics. Values were expressed as Mean ± SEM. Statistical differences among treatments were tested with One-way ANOVA, followed by Tukey's post-hoc test for multiple comparison tests at significant level of p ≤ 0.05. IC50 values were computed using linear regression analysis (GraphPad Prism 5 (San Diego, US). Gene expression fold change was calculated using QuantstudioTM Design and analysis software (Thermo Scientific, USA). Interpretation on mass spectra LC-MS and GC-MS was performed based on reference published by Adams and Chemical mass spectral databases together with the library-mass spectral database (NIST library).
Results
Antiproliferative Effects of C. edulis and P. capensis Extracts
Antineoplastic effect of C. edulis and P. capensis extracts was estimated by MTT assay. Results shown in Figures 1, 2, 3 and 4, demonstrated specific antiproliferative activities of extracts toward HCC 1395 and DU145 cells. Additionally, all the tested extracts elicited dose-dependent inhibition of the cancer cells. Growth inhibitory activities of C. edulis and P. capensis were also quantified in terms of IC50 (Table 1). The National Cancer Institute (NCI) criterion was used whereby an IC50≤ 20 µg/ml was considered highly active whereas IC50 value of 21–100 µg/ml was considered moderate active and above IC50 ≥ 100 µg/ml was considered inactive (Boik, 2001). Aqueous, EtOAc and DCM extracts of C. edulis were highly active against HCC 1395 with IC50 values of 8.49 ± 0.23 2.12 ± 0.02, and 5.77 ± 0.30 μg/ml, respectively. The extracts were also potent on the DU145 cell line. Similarly, all the P. capensis extracts showed cytotoxic effects on DU145 cell lines with IC50 values of 12.67 ± 0.46, 5.00 ± 0.17, and 10.09 ± 0.38 for aqueous, EtOAc and DCM extracts, respectively. Moreover, the HCC1395 cells were susceptible to P. capensis extracts. All the extracts exhibited relatively less cytotoxic activities against Vero cell lines

In vitro antiproliferative activities of C. edulis extracts on HCC 1395. Bar with no asterisks within the same concentration are significantly different (p < 0.05) by one-way ANOVA followed by Tukey's post hoc tests. Key: HCC 1395-breast cancer cell line; EtOAC-ethyl acetate; DCM-dichloromethane.

In vitro antiproliferative activities of P. capensis extracts on HCC 1395. Bar with asterisks within the same concentration are not significantly different (p > 0.05) by one-way ANOVA followed by Tukey's post hoc tests. Key: HCC 1395-breast cancer cell line; EtOAc-ethyl acetate; DCM:dichloromethane.

In vitro antiproliferative activities of C. edulis extracts against DU145. Bar with no asterisks within the same concentration are significantly different (p < 0.05) by one-way ANOVA followed by Tukey's post hoc tests. Key: DU145-prostate cancer cell line; EtOAc-ethyl acetate; DCM-dichloromethane.
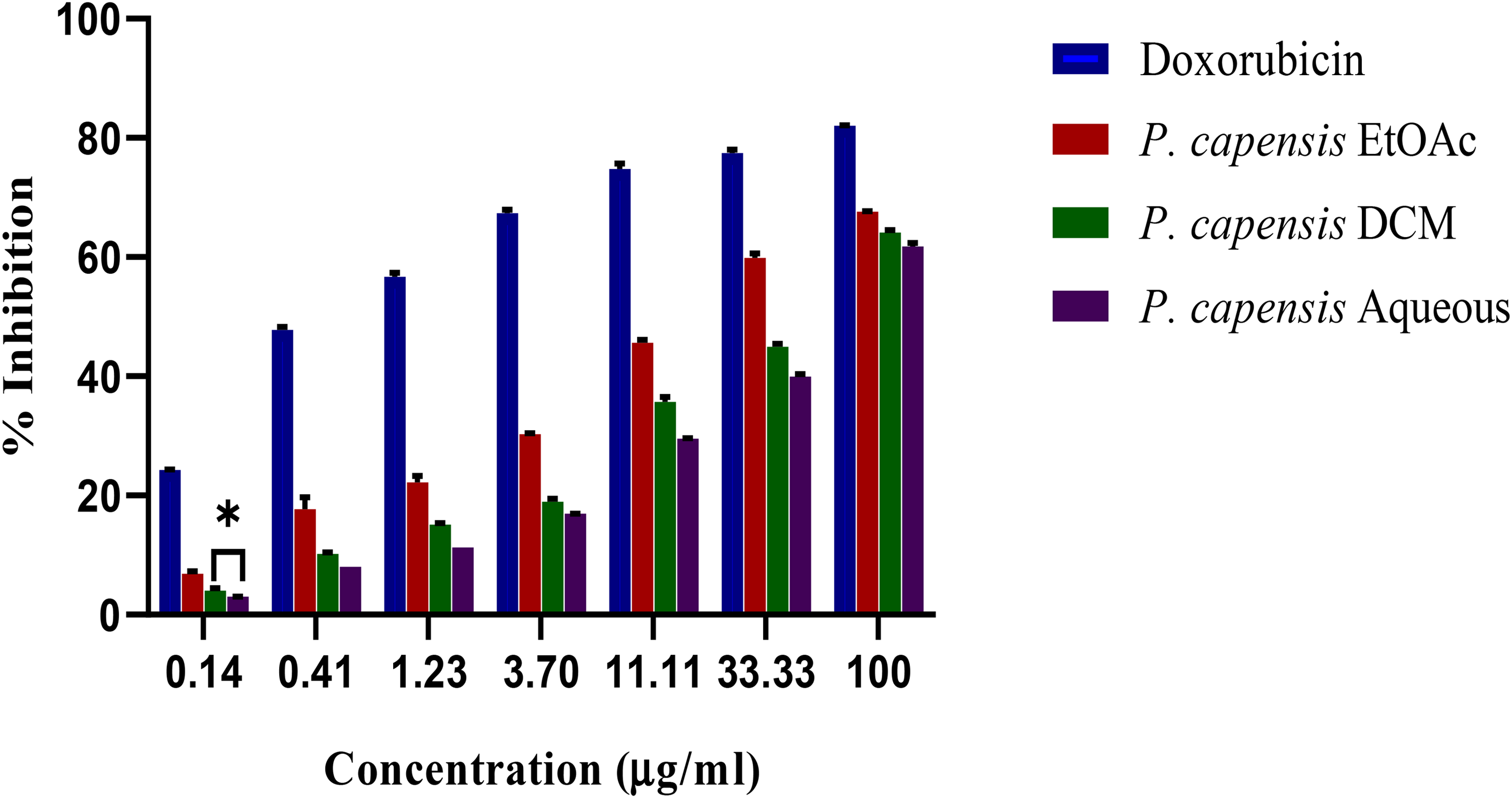
In vitro antiproliferative activities of P. capensis extracts against DU145. Bar with asterisks within the same concentration are not significantly different (p > 0.05) by one-way ANOVA followed by Tukey's post hoc tests. Key: DU145- prostate cancer cell; EtOAC-ethyl acetate; DCM-dichloromethane.
IC50 and CC50 of Different Solvents Extracts of C. edulis and P. capensis.

Values not sharing a lowercase alphabet superscript column-wise are significantly different (p < 0.05) (mean ± SEM; n = 3).
Key: DCM-Dichloromethane, IC50 –Half−maximal Inhibitory Concentration; CC50–50% cytotoxic concentration; HCC 1395−Breast cancer cell line; DU145−Prostate cancer cell line.
Selectivity index
The degree of selectivity of plants extracts and doxorubicin was expressed by selectivity index (SI). SI was computed based on the ratio of CC50 to IC50 values. A value greater than 3 was considered highly selective. As shown in Table 2, the EtOAc extract of C. edulis displayed the highest degree of selectivity against HCC 1395 and DU145 cell lines with SI values of 13.49 and 9.79 respectively. The aqueous extract of C. edulis had the least SI value (2.94 for HCC 1395 and 2.48 for DU145). Similarly, P. capensis extracts induced selective cytotoxicity towards the cancer cell lines, whereby the EtOAc extract exhibited the highest SI value for HCC 1395 (4.8) and DU145 (6.40). The least SI value was exhibited by the aqueous extracts of P. capensis. Doxorubicin displayed a significantly lower SI value compared to those P. capensis and C. edulis extracts towards the cancer cell lines (p < 0.05).
Selective Index of C. edulis Extracts.

Selective Index of P. capensis Extracts.

Key: HCC 1395- Breast cancer cell line DU145-Prostate cancer cell line; DCM-Dichloromethane.
Gene Expression Analysis
To determine possible mechanisms of apoptotic induction, gene modulation changes of pro-apoptotic genes were investigated. Isolated RNA from all the extracts demonstrated high purity and quality. Results in Figure 5 show that the mRNA expression of p53 in the treated HCC 1395 was significantly increased by 7-fold and 3-fold after treatment with EtOAc extracts of C. edulis and P. capensis, respectively (p < 0.05). Further, the extracts were able to modulate the expression of BAX in HCC 1395 cells in a significant manner. A significant increase of about 3-fold and 2-fold in p53 expression was also observed in DU145 cells with C. edulis and P. capensis extracts treatment, respectively, compared to the untreated cells (Figure 6). Similarly, several-fold increases were observed in the number of transcripts of BAX of DU145 cells in response to treatment with both extracts. Results also indicated that treatment of DU145 with extracts led to a marked increase in the expression of HOXB13 gene. However, there were no significant changes in the expression levels of the gene upon treatment with doxorubicin as compared to the untreated cells (Figure 6; p > 0.05).

Upregulation of p53, bax in HCC 1395 cells by ethylacetate extracts of C. edulis and P. capensis. Bars with asterisks per gene are not significantly different (p > 0.05) by one way ANOVA followed by Tukey's post hoc test. Key: HCC 1396-breast cancer cell line; EtOAc-ethyl acetate.

Upregulation of p53, bax and HOXB13 in DU145 cells by ethylacetate extracts of C. edulis and P. capensis. Bar with different lowercase letters per gene are significantly different (p < 0.05) by one-way ANOVA followed by Tukey's post hoc tests. (Bars equals to mean ± SEM; n = 3). Key: DU145-prostate cancer cell line; EtOAc-ethyl acetate.
Phytochemical Analysis
Phytochemical analysis of aqueous extract of C. edulis and P. capensis by LC-MS identified various phenolics and flavonoids in varying quantities (Table 3). In the C. edulis extracts, the most abundant organic acid and phenolic compounds were citric acid (0.12 µg/mg), dicaffeoylquinic acid (0.113 µg/mg), and epicatechin (0.066 µg/mg). The main flavonoids were quercetin-3-O-glycosyl-xyloside (0.1 µg/mg), lutelion (0.09 µg/mg) and apigenin (0.071 µg/mg). In the analysis of the aqueous extract of P. capensis, polyphenols including p-coumaric acid (0.098 µg/mg), catechin (0.021 µg/mg), epichatechin (0.020 µg/mg) were in major concentration. Findings also showed that rutin was the most abundant flavonoids, with a concentration of 0.093 µg/mg. Other detected flavonoids included quercetin and quercetin-3-O-rhamnoside each with abundances of 0.038 µg/mg, lutelion (0.014 µg/mg), apigenin (0.011 µg/mg) and kaempferol (0.005 µg/mg). The LC-MS spectra of aqueous extract of C. edulis and P. capensis with different retention time are shown in figures 7 and 8, respectively.

LC-MS chromatogram of aqueous extract of C. edulis.

LC-MS chromatogram of aqueous extract of P. capensis.
Phytochemical Profile of Aqueous Extracts of C. edulis and P. capensis.

Key: Aq. C. edulis- Aqueous C. edulis; Aq. P. capensis-Aqueous P. capensis; min-Minute.
GC-MS analyses of EtOAc extract of C. edulis confirmed 18 phytocompounds whereas that of P. capensis revealed 23 compounds (Table 4). The compounds were categorized into 5 chemical classes: terpenoids, phytosterol, fatty acids and their derivatives, and phenolics. The terpenes/terpenoids present in the extracts were further classified into 4 groups; monoterpenes, sesquiterpenoids, diterpenoids and triterpenoids. As shown in Table 2, five triterpenoids were detected in the C. edulis extract, whereby α-amyrin, squalene, β-amyrin were found in highest concentration of 9.14 µg/mg, 4.0 µg/mg, 0.55 µg/mg respectively. Furthermore, phytosterols including β-sistosterol (3.2 µg/mg), campesterol (0.68 µg/mg), and stigmasterol (0.68 µg/mg) were detected in the EtOAc C. edulis extract. The results also revealed presence of fatty acids and their derivatives, namely palmitic acid, stearic acid, oleic acid, tetradecanoic and benzenepropanoic acid, 3, 5-bis (1, 1-dimethylethyl)-4-hydroxy-methyl ester, in the order of their abundances. α-Tocopherol (2.97 µg/mg) and β-Tocopherol (0.15 µg/mg) forms of vitamin E were also present in the extract.
Phytochemical Profile of Ethylacetate Extracts of C. edulis and P. capensis.

Key: EtoAC- Ethylacetate; min-Minute.
In turn, the analysis of EtOAc extract of P. capensis detected presence of monoterpenoids including P-cymene and camphor (Table 4). β-Amyrin (1.70 µg/mg), betulin (0.61 µg/mg) and α-Amyrin (0.38 µg/mg) were the triterpenoids present in ethylacetate extract of P. capensis. Furthermore, GC-MS analysis showed presence of three phytosterols whereby stigmasterol (2.00 µg/mg), stigmast-7-en-3-ol (1.96 µg/mg) and stigmast-4-en-3-one (0.42 µg/mg) were found in major concentration. Three forms of vitamin E were also detected in the extracts including β-tocopherol (1.28 µg/mg) α-tocopherol (0.27 µg/mg) and γ-tocopherol (0.25 µg/mg). GC-MS spectra of ethylacetate extract of C. edulis and P. capensis confirmed presence of various compounds with difference retention time (Figure 9 and 10).

GC-MS chromatogram of ethylacetate extract of C. edulis.
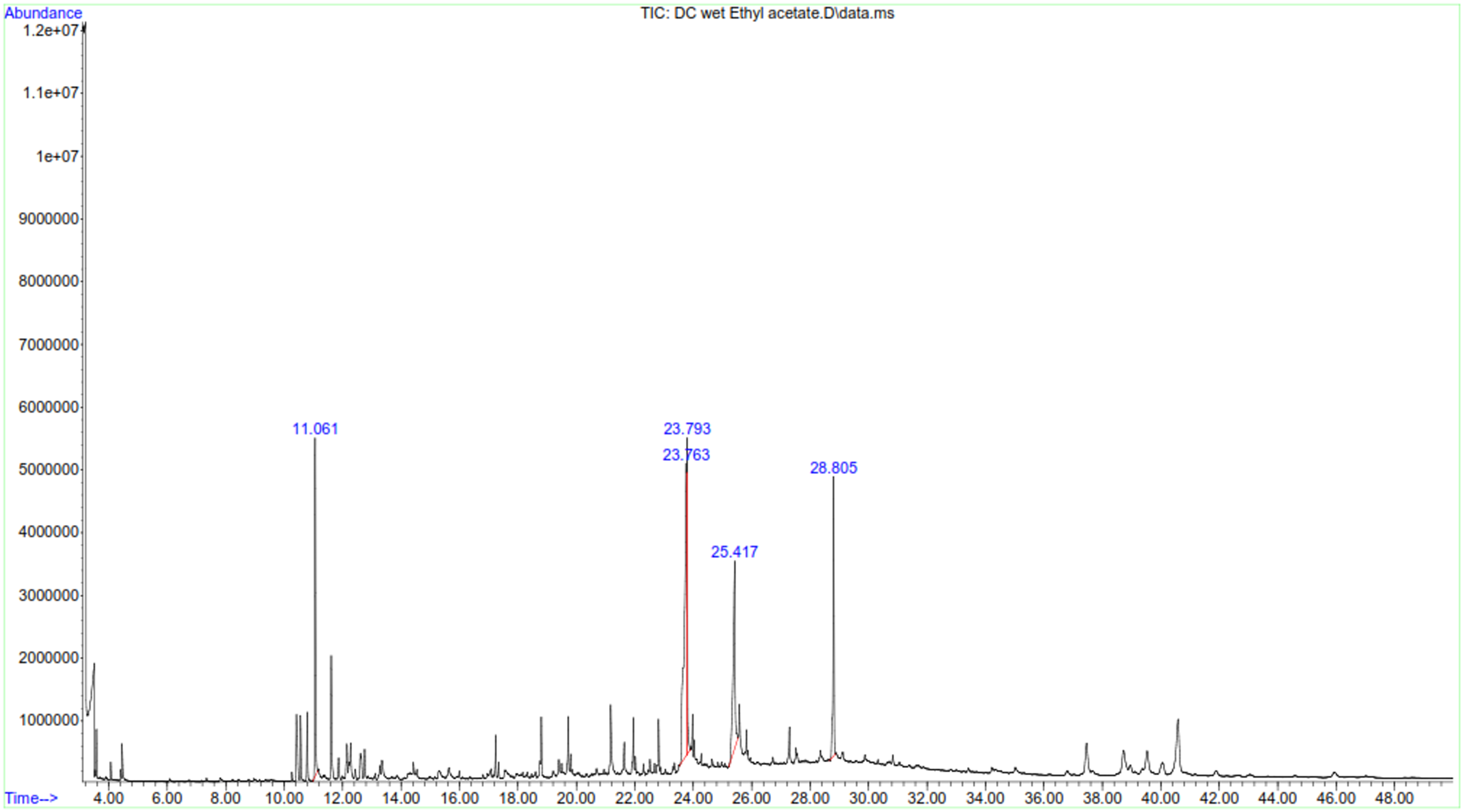
GC-MS chromatogram of ethylacetate extract of P. capensis.
Similarly, GC-MS analysis showed presence of 23 phytochemicals in the DCM extract of C. edulis including terpenoids. Analysis of terpenoids showed that sesquiterpenoids, α-copaene and caryophyllene had similar concentrations (0.1 µg/mg) as tabulated in Table 5. Squalene (9.9 µg/mg) had the highest concentration among triterpenoids, followed by β-Amyrin (7.0 µg/mg), α-amyrin (0.9 µg/mg) and betulin (0.6 µg/mg). Taraxasterol was the most abundant sterol (12.7 µg/mg), whereas stigmasterol was the least abundant sterol in the DCM extract of C. edulis (1.0 µg/mg). Results also indicated presence of various Vitamin E forms with varying concentrations, including α-tocopherol (9.6 µg/mg), β-tocopherol (0.7 µg/mg) and γ-tocopherol (0.4 µg/mg).
Phytochemical Profile of Dichloromethane Extracts of C. edulis and P. capensis.

Key: DCM-dichloromethane; min-Minute.
As shown in Table 5, the DCM extract of P. capensis was shown to contain various phytocompounds. Campor, α-copaene and phytol were the only monoterpeniod, sesquiterpenoid and diterpenoids compounds present in the extract, respectively. α-Amyrin was the most abundant (5.09 µg/mg) triterpenoid whereas betulin was the least abundant (0.42 µg/mg). Ergosta-5, 22-dien-3-ol was the most abundant phytosterol, whereas β-sitosterol was the least abundant sterol in dichloromethane extract of P. capensis (0.21 µg/mg). δ-Tocopherol and α-Tocopherol were present in the extract albeit in varying concentration. Palmitic acid showed the highest abundance (42.9 µg/mg) among the fatty acids, whereas tridecanoic had the least abundance (0.42 µg/mg). Various phenolic compounds were also reported in dichloromethane extract of P. capensis with varying concentrations including Vanillin (2.56 µg/mg), a methoxy phenol, homovanillyl alcohol (1.13 µg/mg), 2,6-dimethoxy Phenol (0.28 µg/mg) and 3-(1-methylethyl)-phenol (0.28 µg/mg) (Table 3). GC-MS spectra of dichloromethane extract of C. edulis and P. capensis confirmed presence of various compounds with difference retention times (Figure 11 and 12).
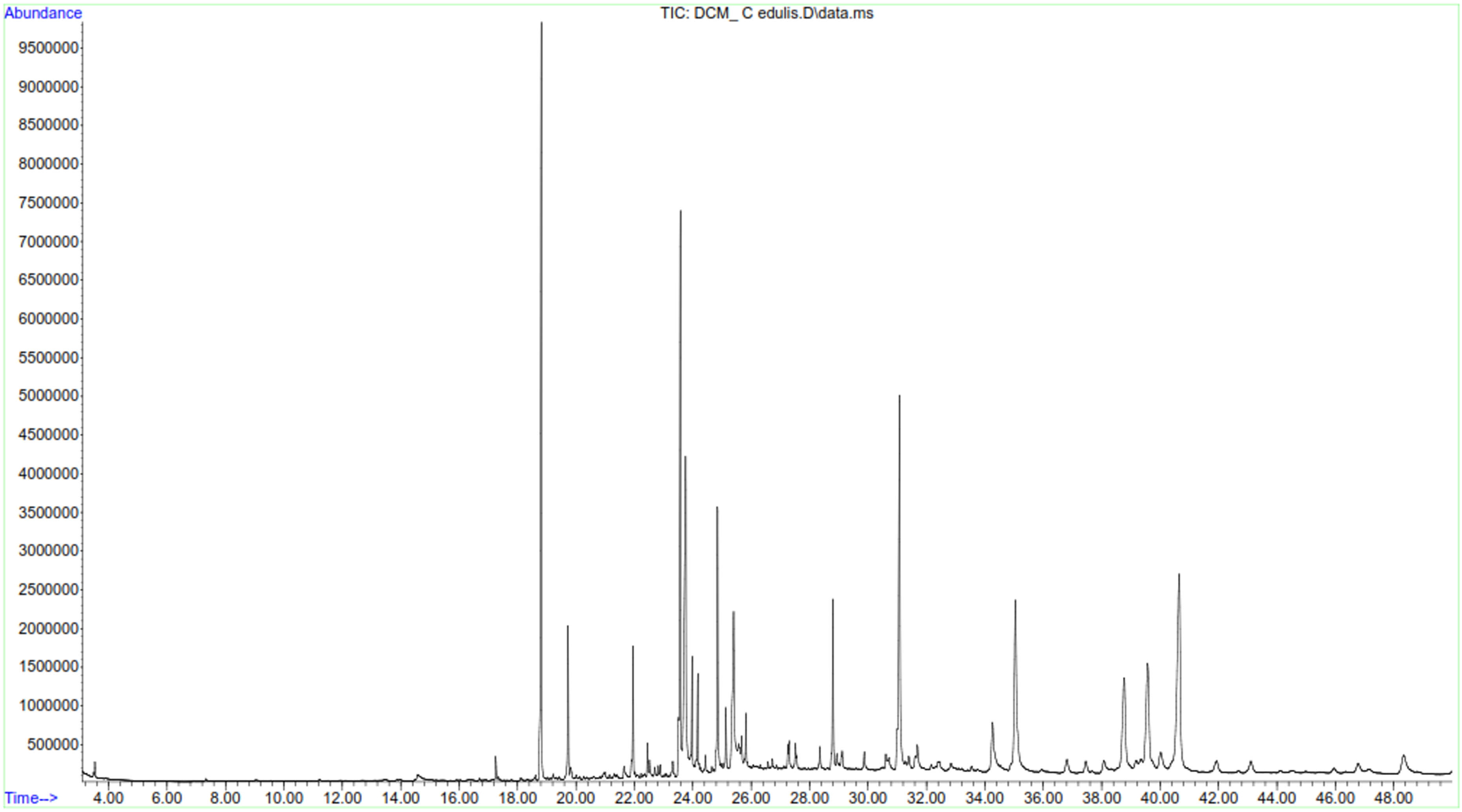
GC-MS chromatogram of dichloromethane extract of C. edulis.
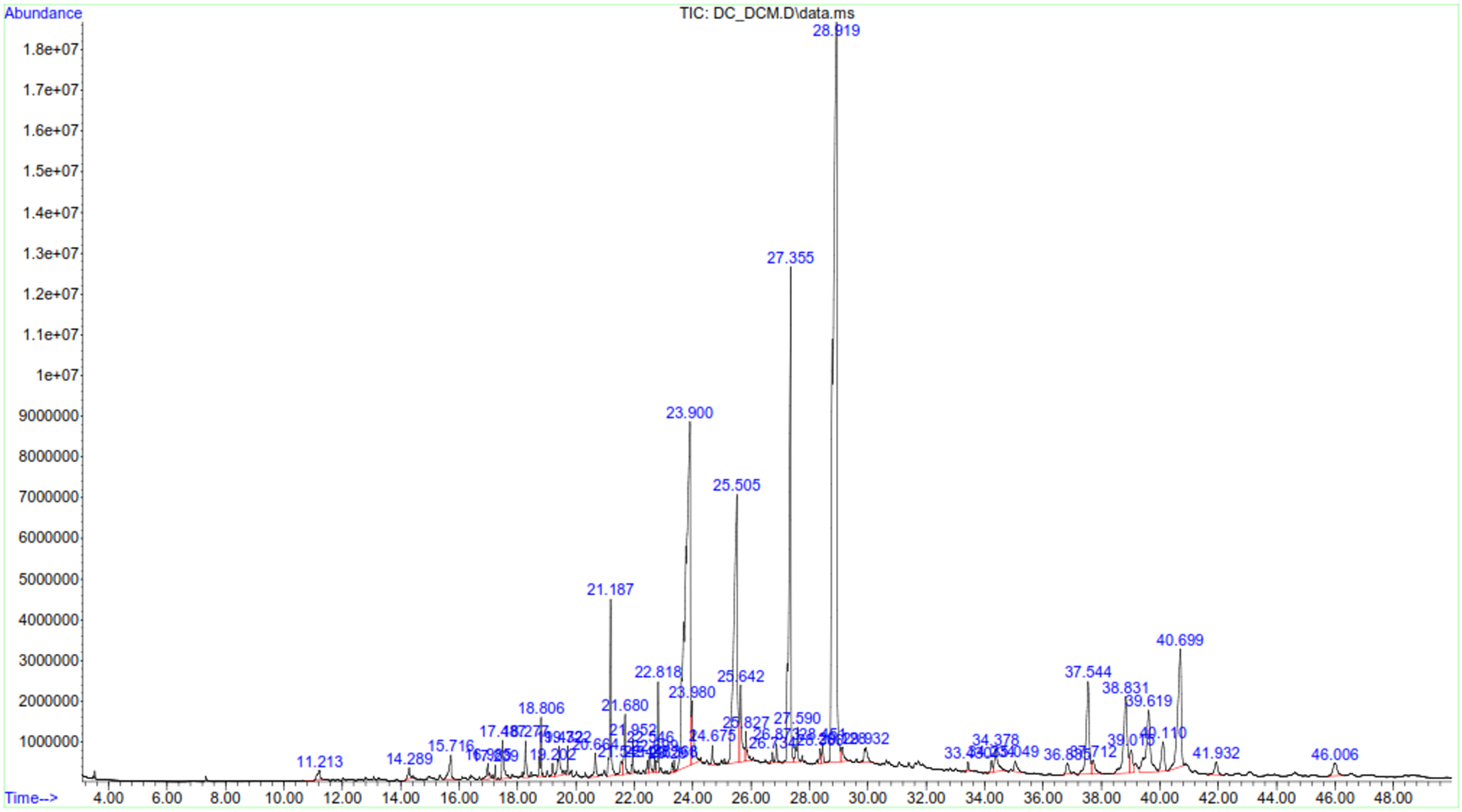
GC-MS chromatogram of dichloromethane extract of P. capensis.
Discussion
Cancer remains a global health problem and the current therapeutic approaches despite showing efficacy are not devoid of intrinsic problems. Nevertheless, medicinal plants have received attention as novel chemotherapeutic agents owing to their biocompatibility, availability, minimal toxicity and affordability. 16 This drives the need to pursue medicinal plants as potential candidates for antineoplastic drugs. The study revealed the antiproliferative activities of C. edulis and P. capensis on HCC 1395 and DU145 cancer cell lines by upregulating apoptotic genes. 6
Findings showed that the extracts elicited a concentration-dependent growth inhibitory effect. This could be taken to suggest that there was an accumulation of biologically active principles in the cells with increase in dosage levels, resulting to an increase in efficacy. This is in tandem with Chu et al 32 study, whereby it was noted that efficacy of a drug is directly affected by its intracellular concentration. Further, there could be a possible correlation of receptor and intracellular signalizing with different pharmacological mechanism. 33 In regard to this, Burow et al 34 showed that an increase in concentration of glyceollins, isolated from soy, correlated with a greater suppression of ER signaling resulting to a decrease in proliferation of MCF-7 cells.
That effectiveness of plants extracts was dependent on extract concentrations corroborates earlier studies of Bhagat et al, Ghout et al and Mellado et al35-36 Cell growth inhibition activity of dichloromethane extracts of Ephedra chilensis against breast and prostate cancer cell demonstrated a concentration dependent activity. 36 Bhagat et al 35 also reported concentration dependent antiproliferative activities of Eucalpyptus citriodora extracts against prostate and ovary cancer. Further, Ghout et al 35 reported that antiproliferative effects of organic extracts of Euphorbia dendroides against rat brain tumor (C6) and cervical (HeLa) cell lines increased with increasing extracts concentration.
Findings of IC50 values of C. edulis and P. capensis extracts demonstrated that the extracts were highly active against HCC 1395 and DU145 cell lines according to NCI criterion. These findings indicated that a low concentration of extracts was required to inhibit cell proliferation in cancer cells by half. This corroborates previous findings of previous studies.37-39 In any chemotherapeutic drug, selectivity is highly desirable, whereby the drug targets cancer cell and displays insignificance toxicity towards normal cells. 40 Notably, this feature is one of the limitations of the current chemotherapeutic agents. 41 As observed in the current study, C. edulis and P. capensis extracts demonstrated maximal cytotoxicity towards the cancerous cells without significant effects on normal cells. 42 Previous studies have also reported high selectivity indices of herbal extracts. For examples, leaves methanol extracts of Sarcocephalus probeguinii demonstrated SI ranging from 3.15–18.28 on HeLa, MCF-7, colorectal cancer cell lines which was higher compared to that of reference drug. 39
To further investigate mechanism of action underlying antiproliferative activities of C. edulis and P. capensis, the study evaluated modulation of expression levels of apoptotic related genes by ethylacetate extracts of C. edulis and P. capensis. Treatment of cancer cells with the extracts led to an upregulation of wild type p53 gene. TP53 acts as a transcription regulator of several targets genes involved in apoptosis, DNA repair, cell proliferation and cell cycle regulation. Therefore, upregulation of p53 could have led to an increase in the expression level of p21 protein, a CDK inhibitor, leading to cell cycle arrest. Further, the tumor suppressor gene could have targeted p53 upregulated modulated apoptosis (PUMA) which is localized to mitochondria and signals apoptosis through the activation of executioner caspases. 43
In addition, upon activation, p53 physically interacts with pro-apoptotic proteins through its DNA-binding domain leading to sequestration of BAX/BAK, and subsequent activation of caspases thereby, inducing intrinsic mitochondrial mediated apoptosis. 44 Activation of p53 also promotes impairment of AKT activation through induction of PTEN. The gene encodes a phophatase that dephosphorylates P13 K, an activator of AKT pathway. 45 Therefore, in this study, an increase in p53 could have resulted in growth inhibition of DU145 and HCC 1395 cells by similar mechanisms upon treatment with C. edulis and P. capensis extracts.
Moreover, C. edulis and P. capensis extracts could have targeted p53-MDM2 pathway, anatagonized MDM2, resulting to concurrent transcription activation of p53. As one of the downstream targets of AKT pathway, MDM2 promote cell growth and proliferation. Upon AKT mediated activation, MDM2 localizes to the nucleus and forms a complex with p53 protein by binding to N-terminal part of transactivation domain. 46 Hence, inhibiting p53 mediated transcription activities and enhancing its degradation. Since cellular homeostasis and function of p53 protein are regulated by MDM2, overproduction of MDM2, as reported in many tumors, inhibits functions of p53. 47 Therefore, targeting interaction between AKT-MDM2-P53 pathway, in favor of p53 protein, could lead to its activation and gain of function. 46
The observations are in agreement with other studies which reported antiproliferative activities of plants extracts through upregulation of p53. For instance, Pyracantha angustifolia and Paullinia cupana extracts induced antiproliferative activities in breast and hepatocellular carcinoma cells by upregulating p53 consequently inducing PUMA-mediated apoptosis. 43 Rahal and Simmen 48 demonstrated that exposure of soy isoflavone to breast cancer cell line induced functional interaction between p53 and PTEN, thereby inducing apoptosis. Similarly, ethanol extract of Euryale inhibited the AKT pathway thereby; activating intrinsic apoptosis by in lung cancer cells. 49 Further, Vernonia amygdalina extracts inhibited viability in breast cancer cells by upregulating p53 that subsequently caused desphosphorylation of AKT at Thr308 residue. 50
In this study, C. edulis and P. capensis extracts increased expression levels of BAX gene in the treated cancer cells. Bax is directly activated by p53 and increases apoptosis in cells. 51 Upon activation, Bax forms a homodimer with BAK causing mitochondria outer membrane permeabilization which result in the efflux of cytochrome c and other intermembrane proteins from mitochondria. Consequently, apoptosome is formed from cytochrome c, dATP, APAF-1 and procaspase-9. Procaspase-9 is then activated to caspase-9 within the apoptosome. 52 Caspase-9 activates caspase-3 and −7, executioner caspases, which eventually break down proteins resulting in apoptosis. 35 Therefore, in this study, cell inhibition of HCC 1395 and DU145 cancer cell, could have been due to activation of BAX by p53 therefore, contributing to progression of apoptosis. Induction of apoptosis via p53-bax pathway has also been reported by other studies.53,54
The studied extracts upregulated expression levels of HOXB13 in the DU145 cells, suggesting a growth suppressive function of HOXB13. 55 Similar to the findings of the current study, Liu et al 31 noted that tretinoin induced growth arrest in DU145 by upregulating expression levels HOXB13 proteins by inhibiting the activity of DNA methyltransferases, an epigenetic modification. Therefore, in this study, a similar mechanism could be attributed to upregulation of HOXB13 genes in DU145 cells. 31
Antiproliferative activities of C. edulis and P. capensis could be attributed to effects of bioactive constituents detected by LC-MS and GC-MS analyses. This study postulates that flavonoids including quercetin, lutelin and apigenin contributed to the antiproiferative activities of C. edulis and P. capensis extracts. These compounds have been demonstrated to be promising antineoplastic compounds owing to their ability to inhibit tumor-driving signaling pathways. In vitro experiments have demonstrated that quercetin exhibits antitumor activities by stimulating expression of pro-apoptotic genes including p21 and BAX genes. 56
Additionally, antiproliferative activities of luteolin against liver, breast, lung and prostate cancers have been documented. 57 luteolin exerts antineoplastic effects via various molecular mechanisms including targeting epigenetic targets such as DNA methylation and histone deacetylases enzymes. Further, apigenin induce autophagy, apoptosis and suppress invasion in tumor cells. 58 Apigenin modulates P13 K/AKT, signaling pathway associated with cell survival and proliferation. 59
Presence of phenolics compound including catechin and epichaten could be associated with antiproliferative activities of C. edulis and P. capensis extracts. Catechin has been demonstrated to possess anticancer activities through its upregulation effect of the wild type TP53 gene. 60 Similarly, epicatechin rich extracts have been shown to increase levels of p53 protein resulting in growth inhibition. 61 As an effective anticancer agent, rutin exerts its activity by modulating p53 and bax genes activities in various cancer cells.62,63
Further, terpenoids present in the extracts including phytol, betulin, β and α- amyrin, squalene, camphor, copaene, betulin and caryophelllene have been associated with antiproliferative activities. The potential of phytol, a diterpene in modulating anticancer activities has been demonstrated in MCF-7 and PC-3 cell lines. 64 Phytol confers cell death by activating intrinsic mitochondria pathway. 65 Similarly, betulin possess antitumor activities through the activation of intrinsic mitochondrial pathway in various cancer cells.53,66
Previous researches have reported antitumor activities of triterpenes, β and α- amyrin in pancreatic, breast, stomach and ovarian cancers. 67 Squalene has also been reported to elicit significant cell growth inhibition proliferation activity on cancer cells. 68 As an active principle of A. crassna, caryophellene has been shown to induce caspase 3, an executioner in the caspase-dependent apoptosis cascade in selected cancer cell lines. 69
Presence of potent phytosterols including β-sitosterol, stigmasterol, campesterol and lanosterol could have been involved in the antiproliferative activities in C. edulis and P. capensis extracts. β-sitosterol exerts anticancer activities against prostate, breast and ovarian cancer through interference of several cell signaling pathways including P13 K pathways.70,71 Stigmasterol has been demonstrated to repress human ovarian, breast carcinoma (PC-3) and prostate adenocarcinoma (DU145) cells.42,72 Stigmasterol stimulated action of pro-apoptotic proteins including Bax and repressed Bcl-2, thus supporting apoptosis. 72 Scholtysek et al 73 also reported that stigmasterol and β sitosterol inhibited cell growth in DU145 cells by upregulating expression levels of p53 thereby, triggering an increase in the intracellular force generation. Studies on biological activities of methanolic and aqueous extracts of Morinda lucida showed that the main compound campesterol exhibited anticancer activity. 74
Tocopherols were also revealed in the extracts of C. edulis and P. capensis. Therapeutic effects of tocopherols in tumor therapy has been thoroughly reviewed. 75 Tocopherols have been demonstrated to stimulate extermination of human prostate carcinoma cells (LNCaP and DU-145) by upregulating of p21. 76 Further, their effects on caspase signaling pathways in prostate cancer in animal model has been demonstrated. 77
Fatty acids including oleic, palmitic, stearic, dodecanoic and tetradecanoic acids reported in C. edulis and P. capensis have been shown to confer apoptotic effects against breast cancer. 36 In vitro experiment provide proof that oleic acid (omega-9 fatty acid) hinders proliferation in cancer cells through the obstruction of p-AKT signaling pathways and significantly upregulated p21 and p53 proteins in tongue carcinoma. 7 Palmitic acid targeted the DNA topoisomerase 1 as one of the molecular target in induction of apoptosis in cancer cells. 78
Lauric acid (dodecanoic acid) was also demonstrated to elicit inhibitive effect on breast cancer cells by modulating p53 gene. 79 N-hexadecanoic acid isolated from Kigelia pinnata was demonstrated to stimulate apoptosis in cancer cells by interacting with DNA topoisomerase. 80 Similarly, stearic acid, a saturated fatty acid exerts significant inhibition of mammary tumor development by inducing pro-apoptotic genes. 81
In this study, cytotoxicty of doxorubicin against Vero cell lines was higher compared to C. edulis and P. capensis extracts. Compelling studies have reported that due to lack of specificity and selectivity, toxic effects of doxorubicin are multidirectional with cardiotoxicity being the most common. Other common sites for doxorubicin induced-apoptosis and tissue damage is in brain, liver and kidneys. 82 Therefore, findings in this study serve as more evidence of adverse effects against normal cells.
Conclusion
The current study concludes that the extracts of C. edulis and P capensis demonstrated potent antiproliferative effects with desirable characteristics including selectivity and minimal toxicity against normal cell lines. It was established that one of the possible mechanism of action was through the upregulation of pro-apoptotic genes. The bioactivities of the plant extracts could be attributed to presence of phytochemical compounds which were detected, and have been previously reported to possess anticancer activities. This study, therefore, provides a scientific rationale of using C. edulis and P. capensis to manage breast and prostate cancer in traditional medicine. Nevertheless, there is a need for further purification and characterization of phytochemicals associated with antiproliferative activities in this study.
Footnotes
Acknowledgements
The Authors acknowledge the Director of Centre of Traditional Medicine and Drug Research- Research (KEMRI) for allowing the use of the centre laboratory to perform efficacy studies. The authors also wish to acknowledge Reginah Mbugua and Jecinta Ndung’u (KEMRI), Mr Ibrahim Waweru and Gitonga Mwaniki (Department of Biochemistry, Microbiology and Biotechnology laboratories-Kenyatta University) for their technical support.
Author Contributions
Carolyn Muruthi performed the experiments and drafted the manuscript. Mathew Piero Ngugi, Steven Runo and Peter Mwitari coordinated and supervised the study. All authors read, and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The experimental protocols and procedures used in this study were approved by the Scientific and Ethics Review Unit- KEMRI, Kenya.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
