Abstract
Recent studies indicate that erectile dysfunction (ED) and prostate cancer share common potential risk factors such as chronic inflammation, prostatitis, cigarette smoking, obesity, a high animal fat diet, sedentarism, and depression. There is great interest in knowing if ED is associated with prostate cancer. This study aimed to investigate if men afflicted with ED harbor an increased risk of prostate cancer, utilizing two concurrent comparison groups, constructed from the Taiwan NHIRD, with up to 8 years’ follow-up. Among men with no preexisting prostate cancer, an ED group of 3,593 men ≥ 40 years of age and two non-ED comparison groups of 14,372 men from the general population, 1:4 matched by age and index date (GENPOP); and 3,594 men with clinical benign prostatic hyperplasia (BPH), matched by similar criteria were assembled. A Cox model was constructed to calculate the adjusted hazard ratio (aHR) after controlling for age, socioeconomic factors, and various medical comorbidities. During the 11,449 person-year follow-up for the ED group, 24 incident prostate cancer developed. During the 44,486 and 11,221 person-year follow-up for the GENPOP and the BPH group, respectively, there were 33 and 25 incidents of prostate cancer. The ED group demonstrated a 2.6-fold greater risk of prostate cancer than that by the GENPOP with an aHR of 2.63 (95% confidence interval [CI] [1.51, 4.59], p < .001). There was no significant difference in risk between ED and BPH group (aHR = 0.83, 95% CI [0.46, 1.48]). This concurrent, double comparison, longitudinal study revealed a positive association between ED and subsequent prostate cancer incidence.
Sexual health is an integral part of overall health and quality of life. Erectile dysfunction (ED) is a common condition in older men. The classical causes of ED include diabetes mellitus (DM), hypertension, lifestyle factors, and lower urinary tract symptoms (Shamloul & Ghanem, 2013). Age is the primary risk factor for ED (Shamloul & Ghanem, 2013), and men with comorbidities have worse erectile function than those without comorbidities (Lewinshtein et al., 2006). There is great interest in knowing if ED is associated with other major diseases, such as prostate cancer.
The prevalence of ED has been investigated in both Western and Eastern populations. The prevalence of ED is 77% in the general population of Finnish men aged 50–75 (Shiri et al., 2003). A similar prevalence was reported in Singaporean men, increasing from 43% for men in their forties to 77% for men in their sixties (Tan, Hong, Png, Liew, & Wong, 2003).
Many men are afflicted with ED for some time, prior to diagnosis of prostate cancer. A study designed to estimate the prevalence of ED in men screened for prostate cancer reported a prevalence of 37.5% (Bianco et al., 2009). In a cohort of Brazilian men undergoing routine screening for prostate cancer, 40% had moderate or severe ED (Paranhos, Antunes, Andrade, Freire, & Srougi, 2009). ED, classified according to the International Index of Erectile Function score before prostate cancer treatment, was assessed within a prostate cancer screening population. The results revealed a high prevalence of baseline ED (approximately 50%) in the study cohort, leading the authors to conclude that ED was common before prostate cancer treatment (Walz et al., 2008).
In the last 25 years, studies indicate that ED and prostate cancer share common potential risk factors such as chronic inflammation (Nguyen, Li, & Tewari, 2014; Wang, Chao, Lin, Tseng, & Kao, 2016), prostatitis (Dennis, Lynch, & Torner, 2002; Lee et al., 2008), cigarette smoking (Bacon et al., 2003; Islami, Moreira, Boffetta, & Freedland, 2014; Weber et al., 2013), obesity (Allott, Masko, & Freedland, 2013; Bacon et al., 2003; Derby et al., 2000; Weber et al., 2013), a high animal fat diet (Giovannucci et al., 1993; La Favor, Anderson, Dawkins, Hickner, & Wingard, 2013), sedentarism (Derby et al., 2000; Morote et al., 2014), and depression (Chang et al., 2015; Weber et al., 2013).
Few studies have evaluated the risk of developing prostate cancer in men with ED. A recent two-arm cohort study directly compared the incidence of prostate cancer between a newly diagnosed ED cohort and a non-ED cohort. The investigators concluded that those with organic ED had a 1.27-fold higher risk of prostate cancer (Lin, Chang, Lin, Kao, & Wu, 2017). However, when considering the effect of membership bias where patients in the ED cohort likely received more frequent workups to screen for prostate cancer, it would be premature to draw definitive conclusions as to potential relationships between ED and prostate cancer risk. Many if not most urologists and oncologists, at this writing, consider the relationship of ED and prostate cancer is, just as described in an expert commentary, “nothing more than the expression of a simple coexistence of two different conditions over the same age range” (Capogrosso, Montorsi, & Salonia, 2017).
The authors conducted a population-based longitudinal study, with 8 years’ follow-up to examine this potential association, and to evaluate the magnitude of the risk if statistically significant. In this longitudinal study, it was designed to estimate the magnitude of risk, if any, in comparison to the general population who do not have physician-diagnosed ED and a concurrent comparison cohort of physician-diagnosed benign prostatic hyperplasia (BPH). Since accumulative epidemiological studies have established adequate evidence that there is an increased risk of prostate cancer in patients with clinical BPH, one shall be able to compare the incident prostate cancer in the ED cohort with the BPH cohort.
BPH is a prevalent men’s health problem worldwide. The hyperplasia is not a pre-malignant lesion in an enlarged prostate. However, for the past three decades, several studies have reported that BPH is associated with increased incidence of subsequent prostate cancer. In a massive population-based cohort study involving slightly more than 3 million Danish men followed for up to 27 years, clinical BPH was associated with a two- to threefold increased risk of prostate cancer development (Orsted, Bojesen, Nielsen, & Nordestgaard, 2011). In another Swedish nationwide population-based longitudinal study with 26 years follow-up of 86,626 men diagnosed with BPH, relative to the general population, patients with BPH harbor a 2% excess incidence of prostate cancer after 10 years of follow-up (Chokkalingam et al., 2003). A recently published meta-analysis demonstrated that BPH was associated with an increased incidence of prostate cancer (Dai, Fang, Ma, & Xianyu, 2016). The same study’s subgroup analysis by ethnicity indicates that the magnitude of the association between BPH and prostate cancer was somewhat stronger in Asians (RR, 6.09, 95% CI [2.96, 12.54]) than in Caucasians (RR, 1.54, 95% CI [1.19, 2.01]). Current research findings as reviewed above have identified that the association consistently observed in several large longitudinal studies is convincing. Thus, in the current study, it would be reasonable to expect an increased risk of prostate cancer in the BPH cohort. It would be worthwhile to compare the magnitude of prostate cancer risk between the ED cohort, the BPH cohort, and the general population cohort. Hopefully, the differences in the risk between them can better be appreciated.
Methods
Data Source
This study used a population-based retrospective cohort design, based on datasets derived from the Taiwan National Health Insurance Research Database (NHIRD). The National Health Insurance program of Taiwan started on March 1, 1995, and by 2007 more than 98% of Taiwan’s residents were enrolled in this single-payer system. The NHIRD is a large computerized database that includes registration files and original data on claim reimbursement. This database is accessible by qualified researchers in Taiwan for academic research purposes. The present study utilized the Longitudinal Health Insurance Dataset 2005, which consists of one million randomly selected enrollees, sampled from the Registry for Beneficiaries in the 2005. The authors have conducted several cohort studies using this dataset (Kok et al., 2016; Kok, Sung, Kao, Lin, & Tseng, 2015; Kok, Tsai, Su, & Lee, 2015). The current study was exempted from full review by the local Ethics Committee (certificate number KTGH-IRB 10525) and was performed according to the Declaration of Helsinki. Owing to the de-identified and anonymized nature of the database, informed consent was waived. This study was registered at ClinicalTrials.gov (NCT03009123).
Study Cohorts
The population for this research was constructed from the NHIRD, which consists of one million randomly sampled patients, from 2005 to 2013, including 489,855 male patients. The authors constructed an ED study cohort consisting of 3,593 men, older than age 40, with no preexisting prostate cancer, and two concurrent non-ED comparison groups. The first of these consisted of 14,372 men recruited from the general population (GENPOP group), with no preexisting prostate cancer, matched 1:4 with the ED group by age and index date. The controls were matched to the cases without replacement.The second comparison group was 3,593 men with BPH, and no preexisting prostate cancer, matched 1:1 with the ED group by age and index date of the ED case (Figure 1). Matching beyond 1:1 was not feasible due to the restriction from the pool of BPH patients.

Flowchart showing the construction of the erectile dysfunction (ED), general population (GENPOP), and benign prostatic hyperplasia (BPH) groups.
Inclusion and Exclusion Criteria
The inclusion criteria for the ED cohort were that subjects should be male, older than age 40, with no preexisting prostate cancer, diagnosed with ED (302.72 and 607.84), according to the International Classification of Diseases, Ninth Edition, with Clinical Modification (ICD-9-CM) between 2005 and 2013, and with medical claims at least three times a year. There are two specific ICD-9-CM codes for ED diagnosed by a physician or urologist: 607.84 for organic ED and 302.72 for psychogenic ED. Claims-based ED research is valid, according to numerous studies published in Taiwan (Chao, Chen, Wang, Li, & Kao, 2015; Chen et al., 2015; C. Y. Chou, Yang, Chou, Hu, & Huang, 2016; P. S. Chou et al., 2015; Hsu, Lin, & Kao, 2015; Huang, Lin, Chan, Loh el, & Lan, 2013; Kao et al., 2016). The day of the first ED claim was considered the index date. Patients who were younger than 40 or older than 99, had a history of preexisting (before 2006) ED, outpatient diagnosis fewer than three times in a year, or preexisting prostate cancer prior to ED diagnosis were excluded.
The inclusion criteria for the BPH cohort were physician-diagnosed BPH by the specific ICD-9-CM code of 600 traceable at least three times at the outpatient or one time claim under the inpatient discharge diagnoses. Exclusion criteria were younger than 40 or older than 99, preexisting prostate cancer, and medical visits fewer than two times a year. For the GENPOP cohort, same age criteria, adequate medical visits criteria for follow-up, and having no preexisting prostate cancer at accrual applied. BPH coexistence was allowed for the individuals of the GENPOP cohort.
Confounding Factors
The confounding medical comorbidities included in the study were DM, dyslipidemia, hypertension, stroke, coronary artery disease (CAD), peripheral artery disease (PAD), chronic obstructive pulmonary disease (COPD), chronic kidney disease, depression, anxiety, smoking-related diagnoses, and alcohol use disorder, each of which has its own corresponding ICD-9-CM code/codes (Kok et al., 2016; Kok, Sung, et al., 2015; Kok, Tsai, et al., 2015). These medical comorbidities were examined at baseline up to the end of the first follow-up year.
Outcome Measure
The primary outcome of this longitudinal study was incident prostate cancer, defined by at least three times outpatient or one-time inpatient physician-diagnosis using the specific ICD-9-CM code of 185 for prostate cancer.
Follow-up
All patients were followed up until the incidence of prostate cancer, death (censored), or the end of 2013. Person-time was calculated (Figure 1). Incident prostate cancer, detected during the first year of follow-up, was excluded from overall risk estimation to reduce protopathic (reverse causation) bias.
Statistical Analyses
Distributions of age groups, insurance premiums, geographic regions, urbanization levels, medical comorbidities, and medication between the ED and non-ED groups were displayed. The medical comorbidities to be analyzed in this study were those abovementioned confounding factors. Prostate cancer incidence rate (IR) using person-year approaches in the three cohorts were reported so that IR ratio can be readily calculated if needed to compare the estimated risk of developing prostate cancer between the ED and non-ED groups. Cox proportional hazards regression analysis was used to calculate the hazard ratio (HR) and its 95% CI for the ED cohort, compared to the two non-ED cohorts. The statistical analyses were conducted using the SAS statistical package (SAS System for Windows, version 9.4; SAS Institute, Cary, NC, USA). IRs and crude HRs for developing prostate cancer were calculated, stratified by age, comorbidities, insurance premium, geographic location, and urbanization level. In addition, a Cox model was constructed to show the aHR for developing prostate cancer development after controlling for age, insurance premium, geographic region, urbanization level, smoking-related diagnosis, alcohol use disorder, and testosterone use. All comorbidities were mutually adjusted.
Results
During the 11,449 person-year follow-up for the ED group, 24 incident prostate cancers developed. During the 44,486 person-year GENPOP group follow-up, there were 33 incidents of prostate cancer. The BPH group had 11,221 person-years of follow-up, during which there were 25 incident prostate cancers. Regarding the IR per 1,000 person-years, GENPOP group had an IR of 0.74; whereas when compared to the BPH group whose IR was 2.23, the ED group had a comparable IR of 2.1 (Table 2). The risk for subsequent prostate cancer was increased 2.63-fold in the ED group (aHR, 2.63; 95% CI [1.51, 4.59]; p < .0001) after controlling for age, insurance premium, geographic region, residential urbanization level, smoking-related diagnosis, alcohol use disorder, and the presence of major medical comorbidities. When compared to patients with BPH, the aHR for prostate cancer development for patients in the ED group did not rise to the level of statistical significance, with aHR of 0.83, 95% CI [0.46, 1.48]. Cumulative prostate cancer incidence curves for the ED group and the two concurrent non-ED comparison groups are reported in Figure 2. A log-rank test identified a statistically significant difference between the ED and GENPOP groups (p < .0001), but not between the ED and the BPH groups (Figure 2).

Cumulative incidence of prostate cancer in the erectile dysfunction (ED), general population (GENPOP), and benign prostatic hyperplasia (BPH) groups, compared with a log-rank test. A statistically significant difference exists between the ED and GENPOP groups (p < .0001), but not between the ED and the BPH groups.
Baseline demographic characteristics and comorbidities for the ED and comparison groups are presented in Table 1. The mean age of the patients in the ED group and the non-ED group were similar, at approximately 57 years. However, insurance premiums, geographic regions, urbanization level, smoking-related diagnoses, alcohol use disorders, and comorbidities (including DM, dyslipidemia, hypertension, stroke, CAD, PAD, COPD, chronic kidney disease, depression, and anxiety) differed significantly between the two groups (Table 1). In this cohort, neither patients with ED, nor comparison subjects, took any phosphodiesterase type 5 (PDE5) inhibitor.
Demographic Characteristics and Comorbidities at Baseline of the Erectile Dysfunction and the Double Concurrent, Age-, and Index Date-Matched, Comparison Groups.
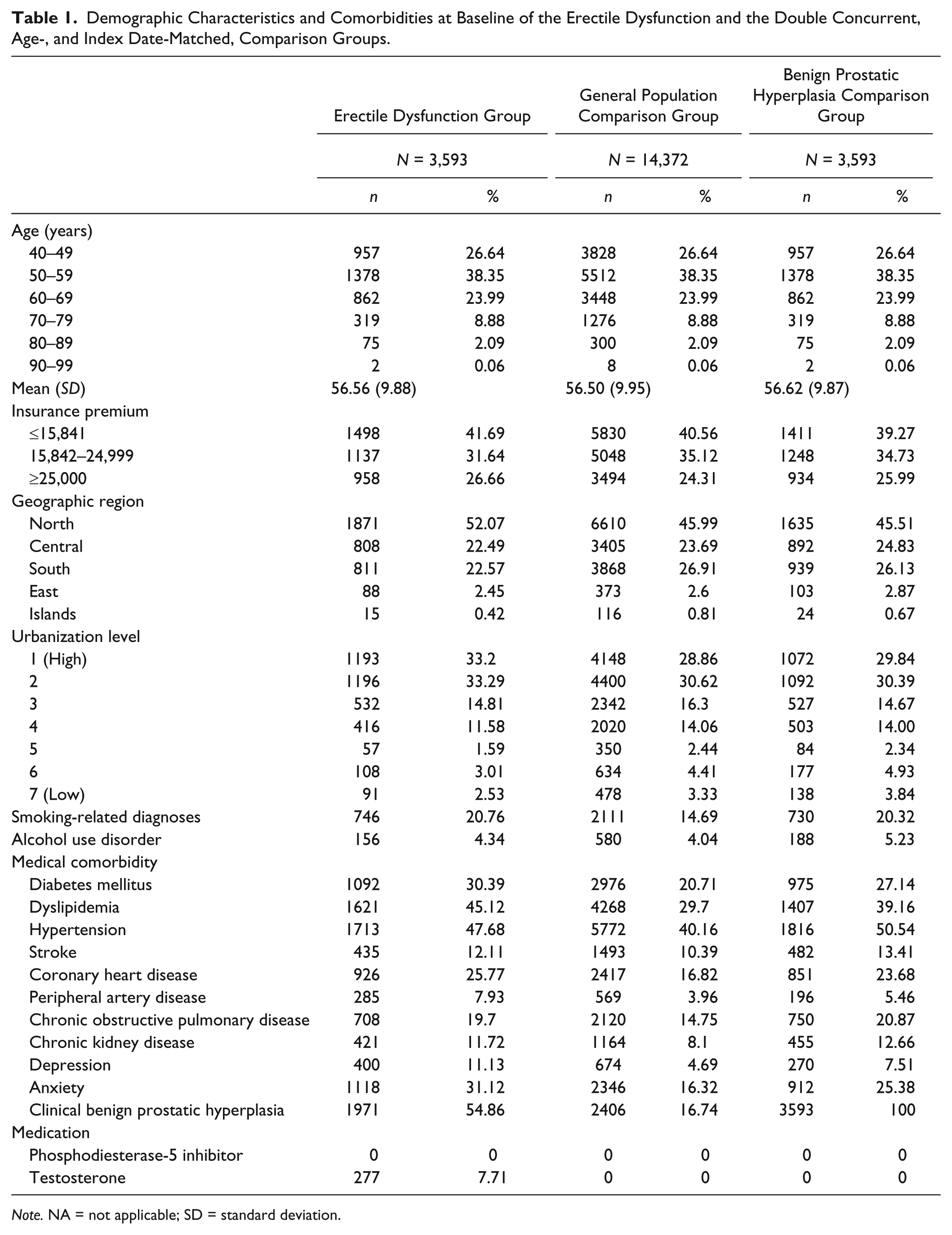
Note. NA = not applicable; SD = standard deviation.
The proportional hazards assumption was assessed in SAS by the graph of the log[-log(survival)] versus the log of survival time graph, which identified that the assumption was not violated. IRs, crude HRs, and aHRs of developing prostate cancer in the ED group, versus the comparison groups, are presented in Table 2. Patients with ED, between 50 and 64 years old, had a 3.4-fold, statistically significant increased risk (aHR, 3.43, 95% CI [1.48, 7.94], p < .01) of developing prostate cancer. Patients with ED younger than 50 or older than 64 years old did not demonstrate statistically significant increased risk (Table 2). Individuals who resided in the most urbanized cities, the top tier of the seven urbanization levels, had up to a 3.2-fold increase in the future risk of prostate cancer (aHR, 3.19, 95% CI [1.35, 7.58], p < .01). Subjects with ED who paid the lowest insurance premiums had a statistically significant threefold increase in the future risk of prostate cancer with an aHR of 3.12 (95% CI [1.51, 6.48]). Except for dyslipidemia which was associated with a 2.5-fold increased risk of prostate cancer, no other medical comorbidities including DM, hypertension, stroke, CAD, PAD, COPD, chronic kidney disease, and depression or anxiety affected cancer risk (Table 2).
Incidence Rates and Adjusted Hazard Ratios for Prostate Cancer Development Comparing Erectile Dysfunction (ED) Group With Two Concurrent Comparison Groups, Stratified by Age, 10 Selected Comorbidities, Insurance Premium, Geographic Location, and Urbanization Level.

Note. To reduce protopathic bias, we excluded the incident cases of prostate cancer in the first year follow-up from risk calculation.
ED = erectile dysfunction; GENPOP = general population comparison group; BHP = benign prostatic hyperplasia comparison group; CI = confidence interval; Dx = diagnosis; HR = hazard ratio; IR = incidence rate per 1000 PY; NA = not applicable; PY = person-year.
p < .05, **p < .01, ***p < .001, ****p < .0001.
The results were different when the second comparison group was men with BPH. ED had a neutral effect on prostate cancer development when comparison was made in patients with ED to patients with BPH, with an aHR of 0.83 (95% CI [0.46, 1.48]); the risk was even reduced in patients with DM (aHR, 0.24, 95% CI [0.06, 0.99], p < .05) or hypertension (aHR, 0.4, 95% CI [0.16, 0.98], p < .05); Table 2). Other than these two variables, when comparing the ED and BPD groups, no factors significantly increased or decreased risk for future development of prostate cancer.
Discussion
This population-based longitudinal study investigated whether men with ED have an increased risk for subsequent prostate cancer. Although the biological rationale of a link between the two is weak, a formal study at a population level is still worth undertaking to prove or refute the association. This study’s results revealed that men ≥ 40 years old, diagnosed with ED, do have a significantly 2.6-fold increased risk of developing prostate cancer, compared to the general population of men. As the authors have presented the argument for BPH as a risk factor for the development of prostate cancer earlier in the article, the cumulative incidence function reveals that both groups, ED and BPH, had a similar increased risk of prostate cancer. The cumulative incidence curves are not separable, and their difference is not statistically significant (Figure 2). This article is the first cohort study to examine potential temporal relationships between the diagnosis of ED and subsequent development of prostate cancer using double concurrent comparison cohorts (groups). The current results lead us to conclude that like BPH, ED may associate with increased risk of prostate cancer.
Since this study’s data demonstrate that the risk of subsequent prostate cancer in patients with ED may not be different from that in patients with BPH, it would be interesting now to compare the magnitude of risk in terms of the aHR between the current study ED cohort and the published BPH cohort. Take the massive Danish study for comparison (Orsted et al., 2011): the multivariate-aHRs for prostate cancer incidence were 2.22 (95% CI [2.13, 2.31]) in men hospitalized and 3.26 (3.03, 3.50) in men operated for clinical BPH versus general population controls. Given that this study’s ED cohort has an adjusted HR of 2.63 (95% CI [1.51, 4.59]), it is no wonder that there was no statistical difference when comparing ED to BPH on the risk of prostate cancer incidence.
The strength of this research is its study design that used two concurrent comparison groups, to match the exposure group by age and index date. Doing so, the authors can then be able to simultaneously compare and appreciate the difference in the magnitude of risk with both a general population and a subcohort whom an increased risk has been established from the previous research findings. There was no drop-out or cross-over of participants between the study groups. To avoid protopathic bias (reverse causation), the incident cases of prostate cancer detected during the first year of follow-up, in either group, were excluded from precise risk estimation using the Cox model.
A previous study has reported that Caucasian men with ED had significantly lower education and lower annual income than men without ED. This indicates that socioeconomic variables may play an important role in ED (Walz et al., 2008), and is consistent with the results of this study. This article showed that insurance premiums, geographic region, and urbanization level differed significantly between patients in the ED and comparison groups (Table 1). Sexual dysfunction in men is associated with a variety of common medical problems that require multidisciplinary management (Montorsi et al., 2010). Men with medical comorbidities can have worse erectile function than those without comorbidities (Lewinshtein et al., 2006). This article’s results demonstrated that the following major medical comorbidities are statistically more prevalent in patients with ED than in patients without ED, accrued from the general population: DM (30% vs. 21%), dyslipidemia (45% vs. 30%), hypertension (48% vs. 40%), stroke (12% vs. 10%), CAD (26% vs. 17%), PAD (8% vs. 4%), COPD (20% vs. 15%), chronic kidney disease (12% vs. 8%), depression (11% vs. 5%), and anxiety (31% vs. 16%; Table 1). Testosterone deficiency is a concern as it is associated with several medical conditions, including diabetes, hypertension, and obesity. However, the relationship is not certain to be causal, although a strong relationship exists between testosterone deficiency and metabolic syndrome (Traish, Miner, Morgentaler, & Zitzmann, 2011).
There were some potential limitations to this work. The study design was a retrospective, claims-based design, based on the NHIRD database, which does not contain biological data such as testosterone levels or concentrations of prostate-specific antigens. Also, elements of the subjects’ lifestyle history, such as having a sedentary lifestyle, physical activities, abdominal obesity, and fruit and vegetable consumption were not available in the database. The lack of such information could potentially bias the effect estimate. Late manifestation, and therefore late diagnosis of prostate cancer (or misdiagnosis), may have occurred. This could have produced a misclassification bias.
Conclusions
This cohort study, with up to 8 years of follow-up, demonstrated that an increased risk of prostate cancer was observed in patients ≥ 40 years old, with ED, and interestingly the magnitude of the risk may be somewhat similar to that of the BPH patients when patients with ED were compared to patients with BPH. This concurrent, double comparison, group longitudinal study revealed a positive association between ED and subsequent prostate cancer development.
Footnotes
Acknowledgements
The authors are grateful to the National Health Insurance Administration, Ministry of Health and Welfare, Taiwan, and the National Health Research Institute, Taiwan, for kindly providing access to the research data for this study. The interpretation and conclusions contained herein do not represent those of the institutions above.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
