Abstract
Corticosteroids improve the complications of Covid-19 but may cause some side effects such as hyperglycemia. Royal jelly is one of the bee products that exert anti-inflammatory, insulin-like, and hypoglycemic activities. The present study was conducted to investigate the effect of royal jelly capsules on blood sugar and the clinical course of Covid-19 in the patients receiving corticosteroid therapy. In this clinical trial, 72 Covid-19 patients with positive reverse transcription polymerase chain reaction (RT-PCR) test and pulmonary involvement hospitalized in Shahrekord Hajar Hospital were enrolled and randomized into two groups: treatment (receiving corticosteroids and Royal Jelly 1000 mg capsules daily for 7 days) and placebo (given corticosteroids and placebo). Laboratory tests, blood sugar, and clinical courses were determined and compared. Data was analyzed using SPSS version 16. On day 7 after the onset of the intervention, the dosage and frequency of insulin, FBS level, and required corticosteroid showed a decrease in both groups but the inter-group difference was not significant (P > .05). As well, the Spo2 level indicated a non-significant increase and hospital stay length indicated a non-significant decrease in the intervention group (P > .05). Among the symptoms, only headache, cough, and dyspnea indicated an improvement in the intervention group (P < .05). Overall, the results indicated the short-term consumption of royal jelly could not significantly improve blood sugar and the clinical course of Covid-19; however, it could significantly improve headache, cough, and dyspnea in the patients.
Introduction
Covid-19 is known as a prevalent disease and one of the most prominent health issues worldwide. 1 As of November 4, 2021, Covid-19 affected approximately 247 million people worldwide, of whom 5 million died. Iran is one of the 10 most affected countries with over 6 million definite cases and around 120 000 deaths. 2 Although the majority of the cases are mild, in 20% of the patients, there are severe or critical symptoms that require hospitalization and intensive care. 3 Although Covid-19 mainly affects the respiratory system, many studies have found evidence of changes in hematologic, hepatic, and renal functions in hospitalized patients with Covid-19 infection, leading to the hypothesis that Covid-19 can cause damage to other organs and systems of the body through direct injury, drug toxicity or immune system-mediated response. 4 Early administration of steroids to Covid-19 patients has been reported to improve survival and reduce ICU length of stay. 5 However, a common side effect of corticosteroid therapy is hyperglycemia, which affects 20-50% of patients without a history of diabetes, 6 and hyperglycemia in patients with Covid-19 leads to worsened prognosis, and increases the risk of mechanical ventilation, shock, and multiple organ failure, which can negate the benefits of systemic corticosteroids. Therefore, blood sugar control is of special importance for these patients. 7
Royal jelly is one of the most nutritious bee products and its constituents include water, protein, simple sugars (monosaccharides), omega fatty acids, minerals and vitamins. 8 Studies have shown the antiviral, 9 anti-inflammatory, insulin-like 10 and blood sugar-modulating 11 properties of royal jelly. According to the above-cited findings and arguments, the present study was conducted to investigate the effect of royal jelly capsules on blood pressure, laboratory parameters and critical course of Covid-19 in patients receiving corticosteroid therapy.
Materials and Methods
The present study is a double-blind randomized clinical trial that was performed on patients with Covid-19 admitted to Hajar Hospital of Shahrekord in 2021. The sample size was calculated at 30 for each group based on similar studies and using the following formula. Finally, 36 people were enrolled to compensate for potential dropouts (1.96 = 1-α/2, 1-β = 1.28, S1 = 0.4, S2 = 0.7, D = 0.5).
12
Random allocation of participants to the control and intervention groups was performed using Random Allocation Software. Per the number of participants in the study, envelopes were prepared and the first envelope was labeled no. 1, the second envelope no. 2, etc, and in each envelope, the assignment of each patient was determined using the software. The assigned envelope was opened as soon as the participant entered, and according to the number in the envelope, the individual was assigned to one of the intervention or control groups (Figure 1).

Flowchart of the study.
The intervention group received corticosteroid therapy (methylprednisolone sodium succinate; 500 mg/8 ml diluents) and Tang Forte (Royal Jelly) 1000 mg Capsules daily for 7 days and the control group received corticosteroid therapy and placebo daily for 7 days.
Tang Forte (Royal Jelly) 1000 mg contains 350 mg royal jelly extract, 2 mg of vitamin E and other compounds as color, beeswax, soy lecithin, soybean.
It should be noted that the study was double-blind and the principal investigator and patients, as well as the statistician knew about the group assignment of the participants only by code.
A data collection form was completed for each patient separately. For all participants, demographic data such as age, gender and Illness severity were recorded.
Liver function tests including aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphate (ALKP or ALP), total and direct bilirubin, kidney functions including blood urea nitrogen (BUN), creatinine, sodium, and potassium, hematological factors including hemoglobin, white blood cell (WBC), platelets count (PLT), and fasting blood sugar (FBS) were examined before enrollment on a daily basis and inflammatory factors including lactate dehydrogenase (LDH) and ferritin were determined weekly before enrollment and clinical symptoms including headache, dyspnea, cough, fever, chills, sore throat, abdominal pain, diarrhea and respiration, and severity of the disease were determined on days 1 and 7. 13
After data collection, the data were entered into SPSS software version 26 and analyzed using t-test, chi-square and repeated measures analysis of variance (ANOVA). The significance level (P-value) was considered <.05.
Results
In the present study, 72 patients with Covid-19 were hospitalized and divided into two groups of 36 patients, of which 6 patients were excluded from the study. Finally, statistical analysis was performed on data from 30 patients in the control group and 36 patients in the intervention group (Figure 1).
The mean age in the intervention and control groups was 43.97 ± 14.92 and 45.96 ± 13. 01 years, respectively. The gender distribution is presented in Table 1. The illness severity showed no difference between the two studied groups (P > .05). Based on the results, all patients in two groups had moderate disease severity. After the treatment, the severity of the disease was mild in all patients and they were discharged (Table 1).
Some Demographic Properties of Patients Participated in This Study.

According to Table 2 and t-test results, the studied groups in the first to seventh days were not significantly different and were matched in terms of dosage and frequency of required corticosteroids and insulin (P > .05). FBS on days 1-7 and oxygen saturation on days 1-6 were not significantly different between the two groups, but oxygen saturation on day 7 was significantly higher in the intervention group (P < .05).
Comparison of SpO2, FBS, Dosage and Frequency of Regular Insulin and Corticosteroids Required Between Studied Groups.

* Significant difference (P < .05), t-test
The studied groups in the first to seventh days were not significantly different in ALKP, BillT, creatinine, WBC, hemoglobin and platelets (P > .05). BillD on days 5 and 6 and BUN on days 2, 5 and 7 were significantly lower in the intervention group than in the control group (P < .05) but on other days, no difference was observed between the two groups. AST on days 1-3 and ALT on day 1 was significantly higher in the intervention group than in the control group (P < .05) but there was no difference between the two groups on other days (Table 3, Figure 2).

Changes in oxygen saturation (a), FBS (b), AST (c), BUN (d), ALKP (e), ALT (f), WBC (g), Cr (h), PLT (i), HB (j) in studied groups (Code 1 = intervention; Code 2= Control).
Comparison of Hepatic, Renal and Hematological Function
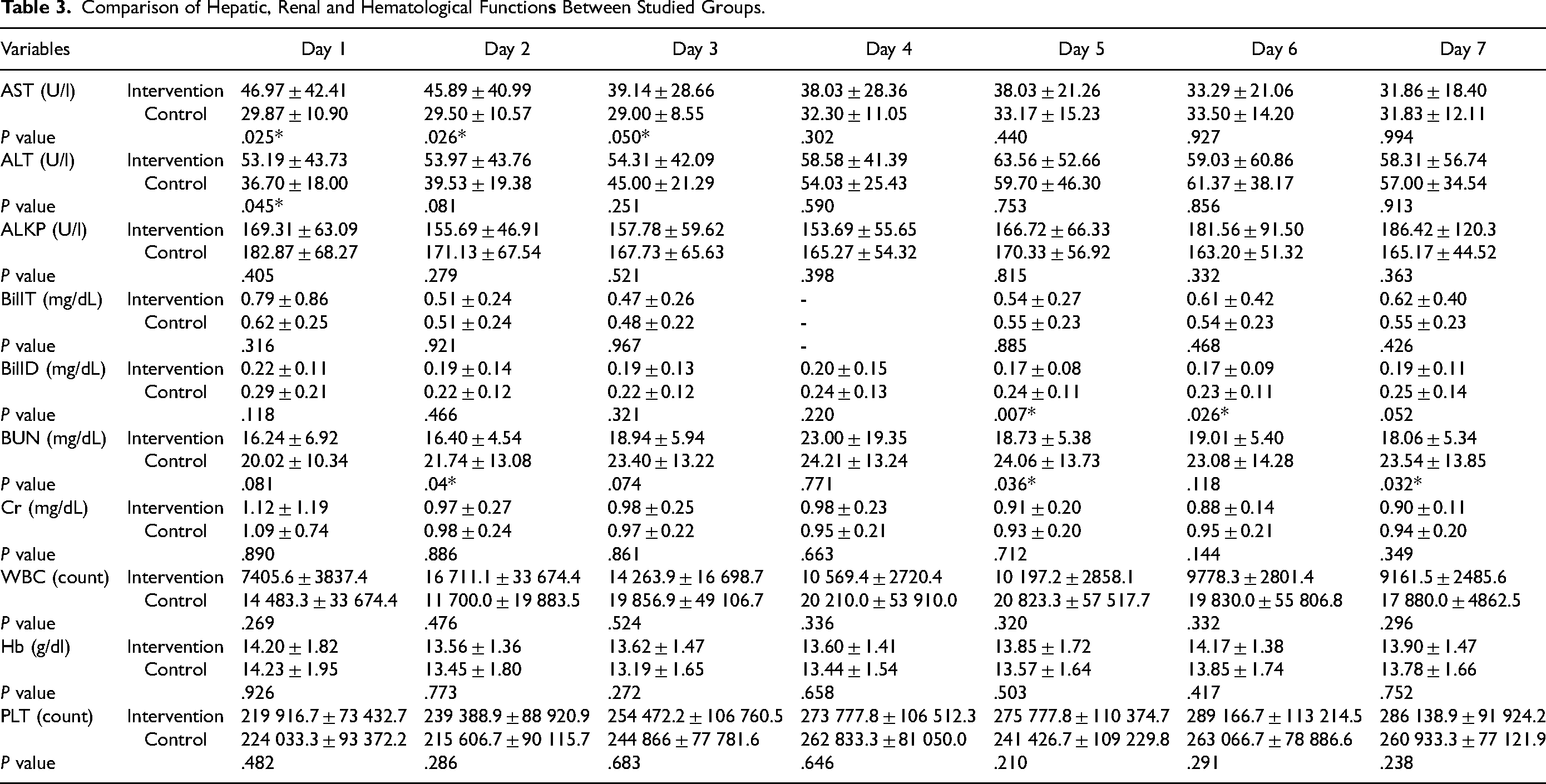
Repeated measures ANOVA showed that the studied variables significantly changed during the period (P > .001) but the two groups were not significantly different in changes in SpO2 (P = .289), FBS (P = .464), AST (P = .131), ALKP (P = .436), BUN (P = .073), PLT (P = .386), creatinine (P = .880), WBC (P = .387) and Hb (P = .559) (Table 3, Figure 2).
According to our results, the frequency distribution of disease severity, fever, chills, sore throat, abdominal pain, diarrhea and breathing before the intervention and on day 7 were not significantly different between the study groups (P > .05, Table 4).
Comparison of Disease Severity and Symptoms During the Study in Studied Groups.

Cough and dyspnea were significantly higher on the seventh day in the intervention group (P < .05) but did not differ between the two groups on the first day. Headache on days 1 and 7 was significantly different between the two groups and were higher in the intervention group (P < .05).
According to the results (Table 5), length of hospital stay was lower in the intervention group than in the control group (P > .05). As well, ferritin and LDH on days 1 and 7 were not significantly different between the two groups (P > .05).
Comparison of Ferritin, LDH and Length of Hospital Stay Between Studied Groups.

Discussion
In this study that was aimed to investigate the effect of royal jelly capsules on blood sugar and clinical course of Covid-19 in the patients under corticosteroid therapy, the results indicated that the short-term use of royal jelly could not significantly improve blood sugar and clinical course of Covid-19; however, it could significantly improve the cough and dyspnea. The effectiveness of royal jelly on blood sugar parameters has been investigated in a number of studies. For example, in the study of Pourmoradian et al (2014) on type 2 diabetic women, the daily consumption of 1000 mg of royal jelly for 8 weeks significantly reduced HbAC1 and FBS levels and significantly increased insulin concentration. 14 However, in the study of Shidfar et al on diabetic men and women, daily consumption of 1000 mg of royal jelly for 8 weeks did not have a significant effect on insulin, glucose and HOMA-IR. 15 In the study of Mobasseri et al (2015) on diabetic patients, consumption of 1000 mg royal jelly did not cause significant changes in FBS and insulin at 0, 60 and 120 min after consumption, indicating that royal jelly had no significant immediate effects on glycemic factors in patients with type 2 diabetes. 16 In the present study, in contrast to the study of Pourmoradian et al, but in line with the studies of Shidfar et al and Mobasseri et al, royal jelly did not have a significant effect on glycemic parameters. Inconsistent findings may be due to demographic differences (eg, gender), duration of treatment, etc In this regard, Omer et al (2019), in a systematic review of eighteen articles, reported that despite the positive effect of royal jelly on blood sugar regulation, the quality of evidence indicates that royal jelly reduces blood sugar activity in the long term and its effect is slight in the short term. 11 This confirms the present study's results with respect to the ineffectiveness of short-term use of royal jelly on blood glucose parameters. In addition, a meta-analysis by Mahboubi et al (2019) on 205 participants for FBS and 130 participants for HbA1c showed that royal jelly reduced FBS consumption by 0.95 mg/dl and HbA1c by 0.32, yet not statistically significantly. Therefore, it was argued that royal jelly supplement does not have a beneficial effect on glycemic indices. However, due to methodological constraints and confounding factors such as differing diets and populations, it was recommended that well-designed and controlled studies be conducted to examine the main confounding factors potentially involved. 17 In the systematic study of Maleki et al (2019), it was reported that out of twelve articles eligible for inclusion in analysis, only seven studies showed a significant reduction in FBS following the consumption of royal jelly. 10 Based on the above-mentioned and the findings of the present study, it seems that consumption of royal jelly for the short term does not have a significant effect on FBS and insulin consumption in Covid-19 patients; however, further studies are recommended due to the small sample size of the present study.
The results also showed that in the first to seventh days, ALKP and BillT were not significantly different between the two groups; however, BillD on days 5 and 7 was significantly lower in the intervention group than in the control group, but no difference was observed on other days. AST on days 1-3 and ALT on day 1 were significantly higher in the intervention group than in the control group, but on other days no difference between the two groups was noticed. Despite the higher mean AST and ALT levels in the intervention group, AST levels in the intervention group decreased by 15.11 units per liter but in the control group increased by 1.96 units per liter during the study period; ALT increased by 4.4 unit/l in the intervention group and by 20.3 unit/l in the control group, demonstrating the effectiveness of royal jelly for preventing ALT increase and AST decrease. However, the repeated measures ANOVA results did not show a significant difference in any of the above parameters in the two groups. Animal studies have shown the effect of royal jelly on the reduction of liver enzymes in rats with streptozotocin- and carbon tetrachloride-induced hepatotoxicity.18,19 In addition, in a clinical trial by Sargazi et al (2020) on addicts, it was observed that consumption of royal jelly for 8 weeks significantly reduced liver enzymes AST and ALKP compared to the control group. 20 Another study by Basirat Dehkordi et al (2019) on multiple sclerosis patients showed that taking royal jelly for 6 weeks significantly reduced ALT and ALKP compared to the control group. 21 In the present study, in line with the above-cited studies, royal jelly prevented the increase of ALT and the decrease of AST, although the changes in analysis of variance were not significant, which is probably due to the short treatment period, small sample size and lack of matching of groups in terms of AST and ALT prior to the intervention; therefore, further studies with higher doses and larger sample size are recommended.
The results of the present study also showed that royal jelly had no significant effect on renal and hematological factors, although the mean BUN levels on days 2, 5 and 7 were significantly lower in the intervention group than in the control group. However, the repeated measures ANOVA did not show a significant difference in any of the renal and hematological parameters between the two groups. Animal studies have shown the protective effects of royal jelly against kidney damage and toxicity induced by diabetes, 22 cisplatin, 23 ischemia-blood transfusion 24 and doxorubicin. 25 However, human studies evidence on its effectiveness is limited and contradictory. In a study of swimmers, royal jelly was found to be ineffective during a 30-day exercise program and increased BUN and creatinine levels due to its high amino acid content. 26 However, a study on cancer patients treated with cisplatin and receiving royal jelly three times before and during chemotherapy showed no significant effect on creatinine and BUN, 27 which is consistent with our study. However, the difference in the results could be due to certain variables such as demographic differences and treatment period.
The results of the present study also revealed that the studied groups did not differ significantly in WBC, ferritin and ldhy on days 1-7. The repeated measures ANOVA results did not show a significant difference in the mean values of WBC, ferritin and LDH between the intervention and control groups. A number of studies have examined the anti-inflammatory effects of jelly royal. A study by Aslan et al (2015) showed that royal jelly significantly reduced the levels of the pro-inflammatory cytokines tumor necrosis factor alpha (TNF-α), Interleukin (IL)-1β and IL-18 in the blood and kidney tissue samples of ethylene glycol-receiving mice. 28 In one animal study, royal jelly reduced peripheral blood leukocytes and pneumonia in asthmatic mice. 29 In addition, a clinical trial in obese individuals showed that royal jelly consumption for 8 weeks significantly reduced the inflammatory marker C-reactive protein and significantly increased the anti-inflammatory marker adiponectin compared to the placebo. 30 In contrast to the above-cited studies, royal jelly did not produce a significant effect on inflammatory factors, which is probably due to demographic differences, short treatment period and small sample size, and further studies are thus recommended to examine this finding.
Our results also showed that oxygen saturation on days 1-6 was not significantly different between the two groups, but the variable on day 7 was significantly higher in the intervention group. However, the repeated measures ANOVA results did not show a significant difference in mean oxygen saturation between the two groups. The results of the present study also showed that the severity of the disease and clinical symptoms such as fever, chills, sore throat, abdominal pain, diarrhea and breathing before the intervention and on day 7 were not significantly different between the studied groups. However, cough and dyspnea on the seventh day were significantly lower in the intervention group than in the control group. Although headache on days 1 and 7 in the control group was significantly higher and the groups were not matched before the intervention, its reduction was higher in the intervention group (41.7%) than in the control group (16.6%). Besides this, our results showed that the studied groups had a non-significant difference in length of hospital stay so that the length was non-significantly higher in the intervention group than in the control group, which indicates the ineffectiveness of royal jelly for reducing the severity of the disease and length of hospital stay.
Although some studies on animal models and some in vitro studies have shown the antiviral effect of royal jelly,31,32 its effectiveness on improving patient outcomes in viral diseases has not yet been investigated. In the present study, for the first time, this effect was evaluated in patients with Covid-19 and no significant improvement of patient outcomes was observed, although this could be due to the low dose of royal jelly, short treatment period and/or small sample size. Further studies are recommended to reach more definitive observations.
One of the limitations of our study was lack of matching of groups in terms of ALT and AST and day 1 headache, which could not be matched, but we compared the trend of changes between the two groups during the period using repeated measures ANOVA, yielding no significant difference in AST and ALT between the two groups during the period.
Conclusion
Our results showed that administration of royal jelly for seven days had no significant effect on blood sugar, dosage and frequency of insulin and corticosteroid, liver and kidney function indices, inflammatory markers, hemoglobin, platelets, disease severity, and most clinical symptoms. However, it could significantly improve some symptoms such as cough and dyspnea after treatment. Future studies can address the use of the royal jelly during the treatment of Covid-19 for the long term to potentially improve most of the symptoms due to this disease.
Footnotes
Acknowledgements
The authors would like to thank the Clinical Research Development Unit, Hajar Hospital, Shahrekord University of Medical Sciences, Shahrekord, Iran for their support, cooperation and assistance throughout the period of study.
Author Contributions
ZH, MAS, and AS designed the study. HP and NS collected the data. AA performed data analysis and interpretation. All authors drafted the manuscript and approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The protocol of this study was reviewed and approved by the Ethics Committee of Shahrekord University of Medical Sciences (IR.SKUMS.REC.1400.063). IRCT registration number for this study is IRCT20210616051596N1.
Funding
The study was funded by Deputy of Research and Technology, Shahrekord University of Medical Sciences, Shahrekord, Iran (Grant number: 5842)

