Abstract
Background.
Malaria is a major public health problem in developing countries. In Ethiopian, the seeds of Schinus molle are used for the management of malaria. Therefore, the current study aimed to evaluate in vivo antimalarial activity of hydro-alcoholic crude extract and solvent fractions of Schinus molle seeds in Plasmodium berghei infected mice.
Methods.
An 80 percent of methanolic crude extract and solvent fractions of Schinus molle seeds were tested for antimalarial activity at 100, 200 and 400 mg/kg doses. The parasitemia level, packed cell volume, body weight, survival of date and rectal temperature were used to evaluate the anti-malarial activity of the extracts. One-way ANOVA followed by post hoc Tukey’s HSD multiple comparison test was employed and the result was expressed in mean ± SEM (standard error of the mean).
Results.
The curative activity of the highest dose of crude extract and aqueous fraction of Schinus molle seeds was 69.86% and 73.82% (p < 0.001), respectively. In the prophylactic test, aqueous fraction had 72.39% (p < 0.001) suppression antimalarial activity. Among solvent fractions, only chloroform fraction was significantly attenuated packed cell volume reduction. The mice treated with crude extract and aqueous fraction had longer survival date than vehicle-treated mice (p < 0.001).
Conclusion.
The experiment finding showed that the crude extract and solvent fractions of Schinus molle seeds had significant curative and prophylaxis anti-plasmodial activity. This result revealed that the Schinus molle seeds extract has promising antimalarial activity against Plasmodium berghei. However, further confirmatory studies, isolation and characterization of the active constituents are recommended.
Background
Malaria is a blood-borne, protozoan infectious ailment caused by Plasmodium species. It is one of the most pressing public health issues in developing countries. 1 In Africa, particularly in sub-Saharan regions, it is one of the most serious medical issues with significant economic burden. 2 It is also the most prevalent infectious disease in Ethiopia (around 75%) 3 and is one of the leading causes of morbidity and mortality. Emergence and fast spread of multi-drug resistant Plasmodium parasites, development of insecticide resistance, and absence of effective vaccine are some of the challenges in the process of aversion and control of malaria. 4,5
In developing countries, most the time people use traditional medicines (TM) and occasionally it is the only source of medicine for the treatment of malaria. 6 In Ethiopia, the majority of the community uses herbal medicine preparations for the treatment of malaria. 7 However, scientific investigations on preparation of crude herbal medicines and the isolation of active ingredients is very minimal. 8 Schinus molle L (S.molle) is a medicinal plants which traditionally used for the treatment of various disease. It is commonly referred to as pink pepper or American pepper and belongs to the Anacardiaceae family which is native to subtropics of South America. 9
In Ethiopia, S.molle seeds are used to treat malaria by the community of Sasiga, Karsa Mojo and Mada Jalala, Western Ethiopia. It is taken orally after the seeds are powdered and dissolved in water. 10 –12 The plant is also used traditionally to treat of cough, tuberculosis, bronchitis, fever, 13 wound around the rectal area, 14 eye infection, allergy, hemorrhoids, respiratory infection, 15 jaundice, diarrhea, and tonsillitis. 16
Previous pharmacological reports indicate that S.molle fruit extract has antimicrobial, 13 immunomodulatory, antioxidant, 17 –19 anti-inflammatory, analgesic, 20 repellent, larvicidal, 21 antiviral, topical antiseptic as well as antifungal uses. 17 In addition, the S. mole seeds extract showed a good suppression antimalaria activity in 4-day suppression antimalarial test. However, other antimalarial activity test models were not. 22 In view of the above ethnobotanical and pharmacological studies, the current investigation aimed to evaluate in vivo curative and prophylaxis anti-plasmodial activity of the crude extract and solvent fractions of S. molle seeds.
Methods
Plant Material Collection and Preparation
The fresh seeds of S.molle were gathered from Maraki campus, University of Gondar, Ethiopia. The plant was identified and authenticated by Dr. Getinet Mersha (with voucher number AB015/2010) and deposited with biology department for future reference. The seeds of the plant were washed and air-dried in a shaded area at room temperature. Then the air-dried seeds were coarsely ground by using electric grinder. The powdered seeds were then placed in a plastic container at room temperature until extraction.
Chemicals and Equipment
The following chemicals and reagents were used: Giemsa stain (Science Lab, USA), trisodium citrate (Deluxe Scientific surgico, India), hydrochloric acid, potassium ferro cyanide, lead acetate, chloroform (Finkem Laboratory Reagent, India), absolute methanol (Okhla Industrial, India),isotonic saline (Addis Pharmaceuticals Factory, Ethiopia), ketamine (Rotexmedica, Germany), ethyl acetate (Research-Lab-Fine, India), acetic anhydride (Techno Pharm Chemical, India), and chloroquine phosphate (Addis Pharmaceuticals Factory, Ethiopia),Whatman filter paper No.1 (with pore size 11 μm) (Whatman, England), test tubes, beakers, funnels, measuring cylinders, glass rods, spatulas, oral gavages (oral feeding syringes), syringes (1 mL, 3 mL, and 5 mL) with needles, desiccators, digital analytical balance (Mettler Toledo, Switzerland), digital thermometer, isotonic saline, micro-hematocrit reader (Hawksley and Sons, England), microscope (CHK2-F-GS, Taiwan), rotary evaporator (Buchi type TRE121,Switzerland), centrifuge (Gelma Awhksley, England), and lyophilizer (Operan, Korea vacuum limited, Korea).
Extraction and Fractionation
A thousand gram (1kg) of dried coarsely powdered seeds of S.molle was macerated in hydro-methanol (80%). After 72 hours, the macerated blend first sifted by gauze and then, filtered by using filter paper no 1. The residue also re-macerated twice for 72 hours and filtered again. The filtered extract then placed in the deep freeze and allowed to dry by using lyophilizer. A portion of methanolic crude extract suspended in a separator funnel in distilled water. The suspension was then shaken by adding chloroform and the chloroform fraction was collected in flask beneath. Next to this, the aqueous portion was shaken with ethyl acetate to obtain the ethyl acetate fraction. The fractions residue (ethyl acetate and chloroform) were allowed to concentrate in rotary evaporator and the aqueous residue was lyophilized to obtain the final fractions. Finally, all fractions were kept in a refrigerator by using amber bottle until use.
Phytochemical Analysis of Extracts
Preliminary phytochemical screening test was performed to evaluate the presence of terpenes, alkaloid, steroidal compounds, phenolic compounds, tannin, triterpenoids, sesquiterpenoid, triterpenoid, saponin, and flavonoid using standard procedures. 23
Acute Toxicity Testing
Five female Swiss albino mice were randomly selected and fasted for 3 hours. Then, administered 2000 mg/kg dose of S.molle seeds for a single mouse through oral route. Similarly, food was withheld for 1hour after extract administration. Then, the mouse was observed for gross physiological changes and mortality for the next 24 hours. If death was not observed within 24 hours, then, the remaining 4 mice were dosed with the same dose and were observed any signs of toxicity for 14 days. 24
In Vivo Antimalarial Tests
Experimental Animals Grouping and Dosing
Six-eight weeks old, 24 to 35 gm body weight, either of sex Swiss albino mice were brought from Ethiopian Public Health Institution, Addis Ababa. The mice were kept in plastic cages with softwood shavings and presented in a 12/12 dim-to-light cycle with free access of food. Before starting experiment, the mice were acclimatized to lab conditions for 1 week. 25 According to Organization of Economic Co-operation and Development (OECD) guideline, if limit dose (2000mg/kg) not induced any sign of toxicity or mortality, we can use 1/10th of 2000mg/kg (200mg/kg) medium dose, 1/20th of 2000mg/kg (200/2 = 100 mg/kg) low dose and 1/5th of 2000mg/kg (200x2 = 400mg/kg) high dose of the study.
The mice were then randomly grouped into 5 groups (each group having 5 mice). The negative control group (Group I) were administered 0.5 ml/kg of distilled water as well as the tested groups (II, III, and IV) were received 100, 200, and 400 mg/kg of extract (both crude and solvent fraction) orally, respectively in all antimalarial testing models. In positive control group, the mice were received 25 mg/kg chloroquine phosphate. In all laboratory procedures, all mice were treated and cared in accordance with internationally recognized guideline. 26
Parasite and Parasite Inoculation
A chloroquine-sensitive Plasmodium berghei, obtained from Aklilu Lemma Institute of pathobiology, was used in this study. The donor mice with 30–37 percent of parasitemia levels were selected from previously infected mice by P.berghei. After confirming the required level of parasitemia, the mice were anesthetized with ketamine and cardiac punctured, then, the blood was collected via 0.5% trisodium citrate containing tube. Finally, the collected blood was diluted (by normal saline (0.9%)) and administered intraperitoneally for mice with dose of 0.2 ml. 27,28 The required volume of normal saline in the dilution of blood was calculating based on the percentage of parasitemia in blood of donor mice. 29
Curative Antimalarial Activity Test (Rane’s Test)
The remedial capability of the most active solvent fraction and the crude extract was done according to the techniques illustrated by Peters and Ryley. 30 On the 1st day (D0), selected mice were inoculated by standard inocula (1 × 107 infected red blood cells) intraperitoneally. After affirmation of parasitemia, on 3rd day (D2), the infected mice were randomly grouped into 5 groups and treated accordingly as described above. Then, the daily treatment was preceded for 5 consecutive days (up to D7). On day 7, thin blood film was prepared and stained by 10% of Giemsa. After this, their parasitemia level was determined using microscope.
Prophylactic Antimalarial Test (Repository Test)
Assessment of the repository ability of extract was conducted according to the techniques described by Fidock et al. 27 The dosing of mice was started on the 1st day and continued up to 4th day (D3). On day D4, each mouse was infected with standard inocula intraperitoneally. After 72 hours of inoculation (D7), a thin blood smear was made and then parasitemia level was determined.
Parasitemia Level Measurement
Thin smears were prepared by using 76 × 26 mm slides from each mouse tail. Then, fixed and stained by using absolute alcohol and 10% Giemsa blue for 15 minutes respectively. After staining, the slides were allowed to gently wash using demineralized water and air dry. Finally, the air-dried slides were observed under an oil immersion microscope with 100 times of magnificent nosepiece. After counting 6 fields from each slide, the percentage of parasitemia level was calculated [25] according to the following equation [20].
Packed Cell Volume Measurement
The effectiveness of tested extract to anticipate hemolysis due to invading parasitemia is determine by measuring Packed cell volume (PCV). The blood from each mouse was collected by using heparinized capillary tubes and filled up to 3/4th of total volume. Then, the filled capillary tubes were sealed with dry sealing clay and allowed to centrifuge in micro-hematocrit centrifuge with a speed of 12,000 revolutions per minute. After 5 minutes, the sealed tubes were taken off from micro-hematocrit centrifuge and measured the percentage of PCV using a calibrated micro-hematocrit reader. 31
Determination of Mean Survival Time
Similar to other parameters, mean survival time (MST) is also important to evaluate the effectiveness of plant extracts against malarial infection. For an extract to be considered as an active antimalarial agent, mice in the tested groups should have greater mean survival time than the vehicle-treated group. In all the above models, each mouse was observed from parasite inoculation (D0) to 30 days (D29) for determining survival time.
The mortality of each group of mice was recorded and mean MST of each group was calculated using the following mathematical equation. 28
Monitoring of Body Weight and Temperature Changes
Measuring the changes of rectal temperature and body weight are important to access the efficacy of medicinal plant extracts against malarial infection. 27 In the case of curative test, the measurement of rectal temperature and body weight of each mouse was taken before parasitemia inoculation (on D0) and after inoculation at day 7. In the prophylactic test, the measurement (body weight and rectal temperature) was taken at day 1 (before starting treatment) and at day 7 (after treatment).
Anesthesia and Euthanasia
The study was done for 30 consecutive days. After completed the experiment, mice were Euthanatized according to the guideline. 32
Data Analysis
The raw data was analyzed using SPSS version 21.0 software. All data were presented in the form of mean ± SEM (standard error of the mean). One-way ANOVA followed by Tukey’s HSD multiple comparison tests were used to analyze and compare the mean difference between values of the measured parameters. A p-value of less than 0.05 was considered statistically significant.
Results
Extraction Yield
The total percentage of yield extract was 17.2% and had a deep red color. The yields of the dried solvent fractions were 11.6gm (29.1%) chloroform, 4.8 gm (12%) ethyl acetate, and 23.6 gm (57.5%) aqueous.
Acute Oral Toxicity Test
The result of acute oral toxicity test showed that there was no visible sign of gross physical, behavioral changes or resulting death at 2 thousand mg/kg/dose of S.Molle within the first 24 hours and the following 14 days. This indicated that LD50 of S.molle seeds extract is greater than 2000 mg/kg.
In Vivo Antimalarial Activity Test
Antimalarial of Crude and Solvent Fractions in Rane’s Test
Crude extract of S.Molle seeds decreased parasitemia level by 38.42%, 55.10%, and 69.86% for SM100, SM200 and, SM400, respectively compared to NC mice. Similar to the crude extract, the result from all solvent fractions (except chloroform and Ethyl acetate 100 mg/kg) showed that the parasitemia level was reduced significantly (p < 0.001) as compared to vehicle-treated mice. Maximum inhibition (72.39%) of parasitemia levels was attained by 400 milligram per body weight (in KG) dose of the aqueous fraction. The parasitemia inhibition seen with CQ25 was significantly (p < 0.001) higher than the crude extract and aqueous fraction (Table 1).
The Effect of Crude and Aqueous Fraction of S. molle Seeds on Parasitemia Level and Survival Time of Infected Mice in the Rane’s Test.
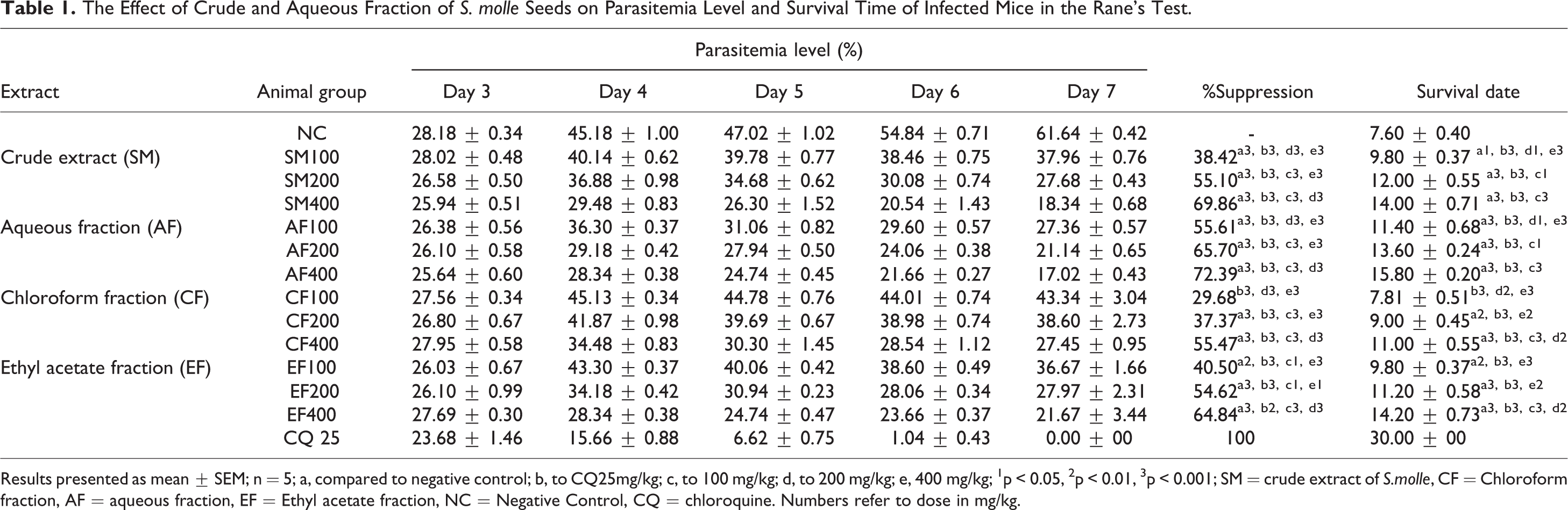
Results presented as mean ± SEM; n = 5; a, compared to negative control; b, to CQ25mg/kg; c, to 100 mg/kg; d, to 200 mg/kg; e, 400 mg/kg; 1p < 0.05, 2p < 0.01, 3p < 0.001; SM = crude extract of S.molle, CF = Chloroform fraction, AF = aqueous fraction, EF = Ethyl acetate fraction, NC = Negative Control, CQ = chloroquine. Numbers refer to dose in mg/kg.
Mean survival time analysis revealed that mice received all doses of crude (except SM100 (p < 0.05) and aqueous fraction lived longer (with significant value p < 0.001) than the mice in NC. The mice treated with the largest dose of aqueous fraction and crude extract lived 15.80 ± 0.20 days and14.00 ± 0.68 days, respectively).The survival date of mice treated by a middle dose of ethyl acetate and larger dose of both ethyl acetate and chloroform fractions was significantly prolonged (p < 0.001) than mice in NC group (Table 1).
The mean comparison analysis indicated that all doses (SM100, SM200, and SM400) of crude extract attenuated body weight reduction significantly (p < 0.05, <0.001 and <0.001 respectively) compared to NC group. A maximum prevention of weight loss related to the elevation of the parasite level was shown at largest dose of crude extract. There was no detectable difference in preventing PCV reduction associated with parasitemia levels between all doses of crude extracts and NC groups. Regarding rectal temperature, the crude extract of S.molle seeds attenuated significantly (p < 0.05 for SM200, p < 0.001 for SM200 and SM400) rapid loss of temperature compared to the NC. (Table 2).
The Effect of Solvent Fractions of S. molle Seeds on Rectal Temperature, Body Weight and Packed Cell Volume of Infected Mice in Rane’s Test.
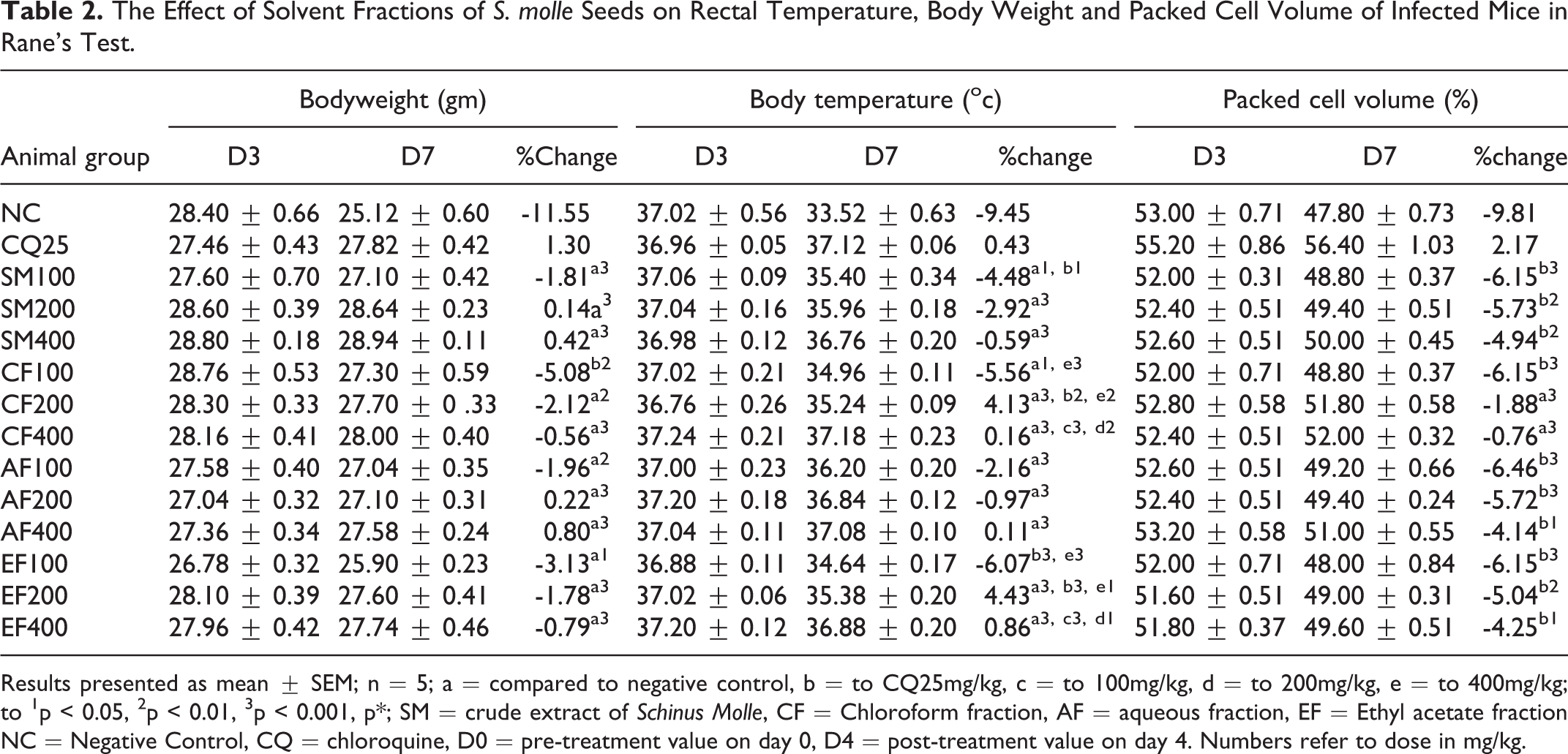
Results presented as mean ± SEM; n = 5; a = compared to negative control, b = to CQ25mg/kg, c = to 100mg/kg, d = to 200mg/kg, e = to 400mg/kg; to 1p < 0.05, 2p < 0.01, 3p < 0.001, p*; SM = crude extract of Schinus Molle, CF = Chloroform fraction, AF = aqueous fraction, EF = Ethyl acetate fraction NC = Negative Control, CQ = chloroquine, D0 = pre-treatment value on day 0, D4 = post-treatment value on day 4. Numbers refer to dose in mg/kg.
All types and doses of the fraction (except low dose of chloroform) anticipated body weight loss significantly (p < 0.001) as compared to NC mice. With regard to rectal temperature, all solvent fractions (except chloroform fraction at 100 mg/kg) attenuated the rapid loss of rectal temperature significantly (p < 0.001) compared to NC group. Among fractions, only middle (200mg/kg) and larger (400mg/kg) doses of chloroform fraction could prevent reduction of PCV compared to the NC group (Table 2).
Effect of Crude Extract and Solvent Fractions on Prophylactic Test
Similarly, the fraction which had the highest anti-plasmodium activity (aqueous) in curative test was further assessed for its effectiveness in prophylactic test. In prophylactic text, the aqueous fraction was reduced the parasitemia level significantly (p < 0.001) compared to NC mice. It showed that dose-dependent reduction of parasitemia level by 54.84%, 69.76%and 79.78% for AF100, AF200, and AF400, respectively as compared to NC mice. The survival date analysis showed that all doses of the aqueous fraction was prolonged the survival date significantly as compared to NC mice (Table 3).
The Prophylactic Effects of Aqueous Fraction of S. Molle Seeds in Malaria Infected Mice.

Results presented as mean ± SEM; n = 5; a, compared to negative control; b, to CQ25mg/kg; c, to 100 mg/kg; d, to 200 mg/kg; e, 400 mg/kg; 1p < 0.05, 2p < 0.01, 3p < 0.001; AF = aqueous fraction, NC = Negative Control, CQ = chloroquine. Numbers refer to dose in mg/kg.
As shown in Table 4, the aqueous fraction ablated body-weight reduction significantly by p < 0.05, p < 0.01 and p < 0.001 at dose of AF100, AF200, and AF400, respectively as compared to the NC. The aqueous fraction of S.molle seeds also attenuated rapid reduction of rectal temperature significantly (p < 0.001) as compared to NC. Similar to the curative test, all doses of aqueous fraction was not prevented PCV reduction in prophylaxis test compared to NC (Table 4).
The Effect of Aqueous Fraction of S. molle Seeds on Temperature, Weight and Packed Cell Volume of Infected Mice in Prophylactic Tests.
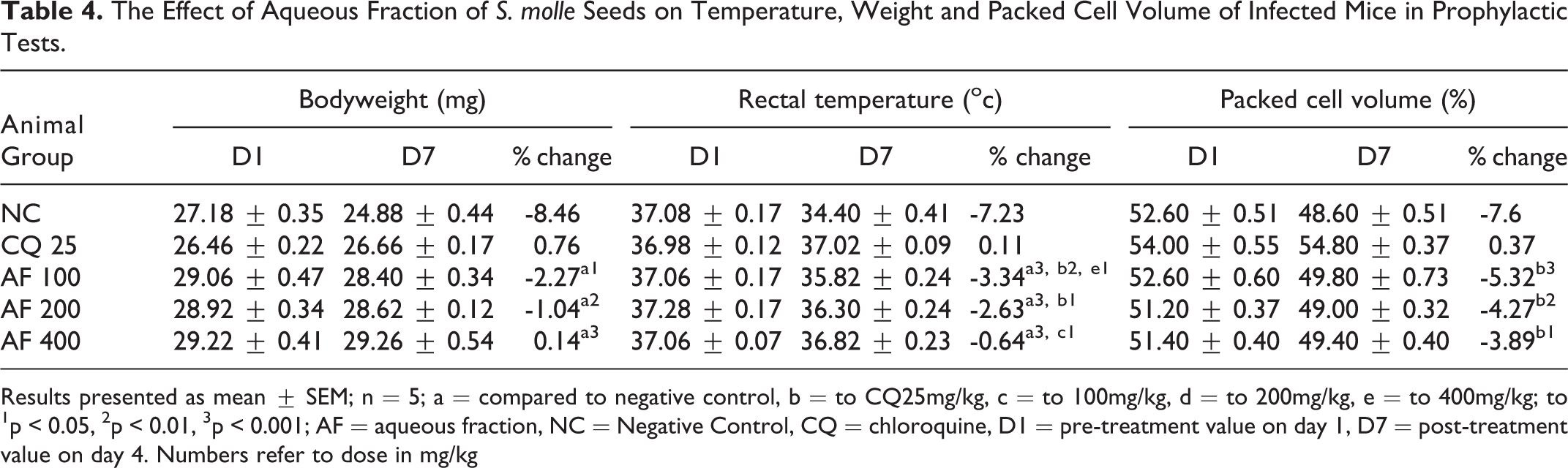
Results presented as mean ± SEM; n = 5; a = compared to negative control, b = to CQ25mg/kg, c = to 100mg/kg, d = to 200mg/kg, e = to 400mg/kg; to 1p < 0.05, 2p < 0.01, 3p < 0.001; AF = aqueous fraction, NC = Negative Control, CQ = chloroquine, D1 = pre-treatment value on day 1, D7 = post-treatment value on day 4. Numbers refer to dose in mg/kg
Phytochemical Screening
The test result revealed that the extract of S.molle seeds have saponins, alkaloids, phenolic compounds, flavonoids, cardiac glycosides, tannins, terpenoids, triterpenoid, and anthraquinones. However, diterpenoid and steroids were absent from c extract (Table 5).
Phytochemical Constituents of Crude Extract and Solvent Fractions of Seeds of S. Molle.

Key: (-) = absent; (+) = present
Discussion
In this study, in vivo antimalarial test model was used which is important to evaluate the possible pro-drug effect and involvement of the immune system in killing parasites. 6 In this study, the antimalarial activity screening was done by using chloroquine-sensitive P.berghei which used for the identification of several antimalarial medications like mefloquine, chloroquine, halofantrine, and artemisinin derivatives. Determination of the percentage inhibition of parasitemia is a gold standard parameter in all anti-plasmodial test models. 28 According to previous study, the study plant had promising antimalarial activity in Peter’s antimalarial activity test model. 22 Therefore, the current study aimed to evaluate the curative and prophylaxis antimalarial activity of Schinus molle seeds.
In the Rane’s test, blood smears were prepared and the parasitemia level was determined each day to assess the remedial capacity of tested extract. In the current investigation, the crude extracts of seeds of S.molle have reduced the parasitemia level by 69.86%. The extract to be considered as active ought to decrease the parasitemia level at least by 30%. 33,34 Therefore, the extract of S.molle seeds is considered as active and endowed the potential antimalarial activity. This strong curative antimalarial activity may be related to secondary metabolites that are present in its seeds. The seeds contained alkaloids, flavonoids, phenolic compounds, and terpenoids which might be contributed to this antimalarial activity. Among those secondary metabolites, alkaloids have critical importance and strong antimalarial effects. 35,36 In addition to this, S.molle had immunomodulatory, antioxidant, 18 anti-inflammatory, and analgesic effects 37 that might contribute to its antimalarial activity. In addition, S.molle seeds extract showed a dose-dependent curative effect that could be attributed to the concentration of bioactive principles in doses delivered to experimental animals. This type of dose-dependent antimalarial activity was observed with other medicinal plants. 38 Aqueous fraction was possessed the highest blood curative activity than ethyl acetate and chloroform fraction. A similar finding was reported the aqueous extraction had better chemo-suppressive activity than the other nonpolar solvent (chloroform). 39 This might be explained by the possible localization of the most active components in aqueous fraction than ethyl acetate and chloroform fraction of S.molle seeds.
Then again, the comparison between crude and the aqueous fraction, the aqueous fraction produced better chemo-suppressive activity than the crude extract. This finding was in line with the crude extract of S.guineense 40 and the aqueous fraction of Croton macrostachys extract. 25 The result sections showed that all doses of S.molle seeds extracts began to decrease parasitemia levels after the second dose of extract. This might be suggesting that the plant extract has delayed onset of action. The extracts had the highest anti-plasmodial activity in curative model further investigated its activity in the prophylaxis antimalarial test model. In our study, aqueous fraction had the highest antimalarial activity in curative tests compare to crude and other solvent fractions. Similar to the curative test, the aqueous fraction also had dose-dependent high chemo-prophylactic activity. In the repository test, the aqueous fraction showed that a relatively lower reduction percentage of parasitemia levels than in curative test. This might be related to rapid liver metabolism and clearance of the active ingredient.
An effective anti-plasmodial active agent derived from medicinal plant extract has relied on anticipated body weight reduction, rectal temperature reduction, and anemia. 27 All doses and types of extraction (crude and solvents) of S.molle seeds ablated body weight reduction significantly. The finding is supported by study done on crude and solvent fraction of A. schimperi 38 and C.macrostachyus. 41 The highest dose (400 mg/kg) of crude extract and aqueous fraction of S.Molle seeds showed that a significant body weight gain. Other studies done on plant extract like seeds of B.nigra 42 and leaves of B.rotundifolia 43 showed that similar finding with current study. This may be related to the nutrient content, appetite-stimulating activity or possession of other pharmacologic activities of S.molle seeds like immunomodulatory, 18 analgesic, 37 and antipyretics activity. 13
In malarial infected mice decreasing internal body temperature is a common future which associated with the rapid decline of metabolism rate. 27 Ideally, active compounds should have to prevent rapid dropping of the body temperature. In this investigation, a significant dose-dependent attenuation of the rectal temperature reduction effect was seen in all tested models. This finding is strengthened by other medicinal plants’ effects on rectal temperature such as leaves of B.rotundifolia. 43 This might be associated with the quick reduction of parasitemia levels by the study plant extracts. Among solvent fraction, the aqueous fraction had the highest preventive activity against rectal temperature reduction than other extracts. This might be due to the possible localization of the responsible active ingredient for this effect into the aqueous fraction. In the curative test, active constituents ought to attenuate the rapid dropping of body temperature from baseline and return to normal temperature range. 27 The rectal temperature was increased in mice treated with all doses of aqueous fraction and crude extract (except SM100). This may be due to the extracts had a high level of reduction of parasitemia levels as well as appetite-stimulating activity. 34
As indicated by Lamikanra et al, the PCV parameter is estimated to assess the viability of the extracts in anticipating hemolysis because of the spread of the parasite in RBCs. 44 The pathogenesis of malaria causes severe anemia due to hemolysis of RBCs, distraction normal RBCs, and erythropoietin inhibition by reticuloendothelial cells action or multiplication of parasites. 45 In addition, the malaria parasites cause certain cells to produce free radical like reactive oxygen species thereby resulting in hemoglobin degradation. 46 Along these lines, anti-plasmodium agents extracted from plants has relied upon anticipation of the decrement of PCV. In this study, chloroform fraction was the only fraction which prevent the reduction of PCV significantly. This finding corroborates with other medicinal plants chloroform fraction such as C.macrostachys leaves 41 and D.Angustifolia. 47 Failure of other extracts to prevent reduction of PCV might be associated with presence of saponin which have a strong hemolysis effect. 48
The effectiveness of the anti-plasmodial activity of medicinal plant extract was also measured by the MST in P. berghei infected mice. 49 In our study, both solvent fractions and crude extract prolonged MST significantly but much shorter than standard drug. This finding corroborates with a study done on N.sativa. 50 This may be related to the shorter duration of action due to the rapid elimination of plant extracts from the body. Moreover, the aqueous fraction prolonged MST more than crude and other solvent fractions in all antimalarial activity test models. The extract had MST longer than 12 days and is considered as an active antimalarial agent 51 which further confirmed the potential anti-plasmodium activity. According to Deharo et al 49 classification, the crude extract of S.molle seeds has a good antimalarial effect while the aqueous fraction has very good antimalarial.
Conclusion
This investigation revealed that S.molle seeds extracts showed significant antiplasmodial activity in both prophylaxis and curative antimalarial tests. The aqueous fraction demonstrated the highest antimalarial activity. The extracts also attenuated body weight loss and the rapid dropping of temperature. However, except chloroform fraction, none of the extracts prevented the reduction of PCV significantly. Moreover, both crude and solvent fractions prolonged the mean survival time of mice compared to the negative control in all antimalarial activity test models. Generally this finding may partly justify the traditional claim of plant to treat malarial infection. However, further evaluation is needed to isolate, identify, and characterize the active ingredients responsible for the observed antimalarial activity of the plants as well as chronic toxicity evaluation of the studied plant.
Footnotes
Authors’ Note
ABM contributed to designing the study, performed the experiment and write up of the final research, and manuscript preparation and finalization. MG and DAG participated in data analysis, data interpretation and supervision of the study. EMB contributed to experimenting, write up of the final research and manuscript preparation. All authors read and approved the final manuscript. In laboratory procedures, all mice were handled and cared according to the internationally accepted guidelines. Ethical clearance was requested and obtained with approval number SoP/45/10 from the department of Pharmacology animal Ethics Committee, College of Medicine and Health Sciences, University of Gondar.
Acknowledgments
We thank the University of Gondar for the material support to conduct this study. Our gratitude also goes to Ethiopian Health and Nutrition Research Institute for supplying mice.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


