Abstract
Sleep problems are widely prevalent and associated with various comorbidities including anxiety. Valerian (Valeriana officinalis L.) is a popular herbal medicine used as a sleep aid, however the outcomes of previous clinical studies are inconsistent. This study was conducted to update and re-evaluate the available data in order to understand the reason behind the inconsistent outcomes and to provide a broader view of the use of valerian for associated disorders. PubMed, ScienceDirect, and Cochrane Library were searched to retrieve publications relevant to the effectiveness of valerian as a treatment of sleep problems and associated disorders. A total of 60 studies (n=6,894) were included in this review, and meta-analyses were performed to evaluate the effectiveness to improve subjective sleep quality (10 studies, n=1,065) and to reduce anxiety (8 studies, n=535). Results suggested that inconsistent outcomes were possibly due to the variable quality of herbal extracts and that more reliable effects could be expected from the whole root/rhizome. In addition, therapeutic benefits could be optimized when it was combined with appropriate herbal partners. There were no severe adverse events associated with valerian intake in subjects aged between 7 and 80 years. In conclusion, valerian could be a safe and effective herb to promote sleep and prevent associated disorders. However, due to the presence of multiple active constituents and relatively unstable nature of some of the active constituents, it may be necessary to revise the quality control processes, including standardization methods and shelf life.
Introduction
Sleep Problems
Sleep plays a crucial role in maintaining brain functions and systemic physiology, and chronic sleep problems could have a significant impact on our health. 1,2 Insufficient sleep leads to reduced stress resilience, decreased quality of life, mood disorders, and cognitive, memory, and performance deficits. 3 It can also contribute to metabolic disorders including hypertension, dyslipidemia, cardiovascular disease, and type 2 diabetes mellitus. 2,4 In addition, it is associated with significantly increased dementia risk. 5,6 Problems with sleep are widely prevalent, 4 affecting 70 million (9-20%) of adults in the US and 45 million (7%) of adults in Europe. 7,8 Typical manifestation of sleep disorders include sleep deprivation or fragmentation and events that occur during sleep. 4 The major sleep disorders are insomnia, restless leg syndrome (RLS), 9 obstructive sleep apnea syndrome, and narcolepsy, 10,11 among which insomnia is the most common. 12 Sedative-hypnotic medications commonly prescribed for insomnia are γ-aminobutyric acid type A (GABAA) receptor agonists, antidepressants, and antihistamines. 13 However, the long term use of most sedative-hypnotic drugs is limited due to various side effects, such as cognitive and daytime performance impairments. 14 In recent years, herbal supplements, such as valerian (Valeriana officinalis L.), sour jujube seeds (Ziziphus jujuba Miller var. spinosa Hu ex H. F. Chou), and kava (Piper methysticum G.Forst.), have gained popularity as alternatives to prescription drugs to improve sleep quality without side effects. 15
Valerian for Sleep Problems and Associated Disorders
Recorded medicinal use of valerian dates back to the first century AD. In recent years, it is popular as a sedative and hypnotic. Historically, however, its metabolic stimulating features such as diuretic, carminative, and menstrual stimulant properties had been more valued. 16 It was in the middle ages that the use of valerian in treating nervous disorders and insomnia was recorded. 16,17 There are over 200 valerian species worldwide, including V. wallichii DC., V. edulis Nutt., and V. fauriei Briq., among which Valeriana officinalis L. is the most well known in Europe and North America as “valerian.” In the United States, valerian is regulated by the Food and Drug Administration (FDA) as a dietary supplement (https://ods.od.nih.gov/factsheets/Valerian-HealthProfessional/). According to the European Medicine Agency (EMA), the well-established uses of V. officinalis root include the relief of mild nervous tension as well as sleep disorders. For relief of nervous tension, recommended oral dosages are 400-600 mg dry hydroalcoholic extract or comminuted herbal substance (root) 0.3-3 g up to 3 times daily. 18 Valerian is considered relatively safe and well-tolerated, however EMA monograph notes gastrointestinal symptoms (e.g. nausea, abdominal cramps) as undesirable effects. Although hydroalcoholic extracts of valerian root in the recommended dosage improve sleep latency and quality, it is uncertain what constituents contribute to the efficacy. 18
The effectiveness of valerian as a sleep aid has been the major research focus, and several systematic reviews were conducted previously. A systematic review published in 2000, which analyzed 9 randomized clinical trials, found contradictory results and significant inconsistency in terms of patients, experimental design and methodology among the trials. 19 Another systematic review and meta-analysis, published in 2006, analyzed 16 studies and this study also found significant methodological problems. 20 Taibi et al. (2007) conducted a systematic review on 37 studies, of which 29 were controlled trials and 8 were open-label trials. They concluded that, although it is a safe herb, evidence did not support the clinical efficacy of valerian as a sleep aid for insomnia. 21 A meta-analysis of 18 randomized placebo-controlled trials, published in 2010, concluded that valerian’s effectiveness had not been demonstrated with quantitative or objective measures although valerian could improve subjective sleep quality. 22 So far, these inconsistent outcomes have not been fully explained. In addition, it is not clear if valerian is effective in treating other disorders associated with, and possibly contributing to, sleep problems. The aim of the current study is to up-date the available data, to evaluate the effectiveness of valerian as a treatment of sleep problems and associated disorders, and to discuss possible reasons behind the inconsistent research outcomes, by particularly focusing on the herbal preparations used in the studies.
Summary
In summary, this study will focus on: The herbal materials – extracts or whole root/rhizomes – that have been used in trials. Potential applications of valerian, including but looking beyond its use for sleep problems. Popular and effective herbal combinations.
Methods
In order to evaluate the therapeutic effects of valerian, systematic reviews and meta-analyses were conducted following PRISMA guidelines. 23 Searches A-C were conducted using PubMed (https://www.ncbi.nlm.nih.gov/pubmed), ScienceDirect (https://www.sciencedirect.com/search/advanced), and Cochrane Library (https://www.cochranelibrary.com/search).
Search Strategy
Search A:
Clinical studies on valerian, published between January 1980 and November 2019, were retrieved using search term “Valeriana” in PubMed database under article type: Clinical Trial.
Search B:
ScienceDirect advanced search was conducted using search term “Valeriana clinical trial.” Articles published between January 2000 and November 2019 were sought.
Search C:
To identify clinical trials using valerian, the Cochrane Library was searched (up to December 2019) using the search term ‘Valeriana officinalis’.
Study Selection
Studies were screened initially for relevance based on the title and abstract. Reviews, unrelated studies, and works without available full text were excluded. Studies evaluated the effectiveness of valerian alone or in combination on sleep or related health problems were considered relevant. The full texts of potential studies were assessed following the exclusion and listed below.
Exclusion criteria Articles published in any non-English language. Studies using unknown substance. Studies on non-human subjects.
The quality of randomized trials were assessed using Jadad scale. 24 In order to extensively collect currently available information, both controlled trials and observational studies were included for reviewing, irrespective of potential risk of bias (Figure 1).

PRISMA flowchart.
Meta-Analysis
The effectiveness of V. officinalis in promoting sleep and reducing anxiety were evaluated by meta-analyses. Due to the scarcity of data, all placebo-controlled studies reporting values that are convertible to effect sizes were included for statistical analysis, irrespective of trials’ quality (Jadad scale). The outcome variables were extracted from V. officinalis treatment group and placebo group by 1 author, which was checked by 2 authors. In order to evaluate the effectiveness as a sleep aid, data on subjective ‘sleep quality’ improvement by repeated administration (ranging between 5 days to 8 weeks) were included in the analysis. Due to the variable measures, both numerical scores and binary scores were converted to effect sizes for the analysis. 25 To evaluate the anxiolytic effect, a study reporting the overall emotional symptoms including anxiety and a study reporting binary outcomes, in addition to the studies reporting standard anxiety test scores, were included. When several measures were reported over time in the same study, the one with the longest follow-up period was selected. When more than 1 dose were tested, the result of the most effective dose was selected. Adjusted effect sizes (Hedges’ g) were calculated from summary measures such as means and standard deviations or confidence intervals, odds ratio (for binary outcomes), and sample sizes, using reported formula. 25– 27 Meta-analyses were performed using Meta-Essentials. 28 I2 statistic was used to estimate heterogeneity. Publication bias was evaluated using the funnel plots. 29
Results
Based on the inclusion and exclusion criteria described above, a total of 60 articles were selected for reviewing (Table 1 and 2). Both quantitative and qualitative studies with human subjects were included. Of 40 articles using valerian as a single herb, 36 studies were randomized controlled trials (RCT) 30–65 and 2 studies were observational. 66,67 Two studies addressed the potential induction of liver enzyme cytochrome P450 (CYP) isoforms. 68,69 In addition, 2 RCTs were conducted using valepotriates (80% didrovaltrate, 15% valtrate, and 5% acevaltrate) 70,71 (Table 1). Nineteen articles assessed the effectiveness of herbal combinations, of which 13 studies were RCT 65,72,73–80,81–83 and 6 were observational studies 84–89 (Table 2).
Valeriana spp. as a Single Herb.

* (1) data included in meta-analysis for sleep quality; (2) data included in meta-analysis for anxiety; (3) Jadad scale.
Abbreviations: ARCI-Addiction Research Center Inventory, CFF-Critical Flicker Fusion, CRT-Choice Reaction Time, DSST-Digit Symbol Substitution Test, DST-Digit Span Test, EEG-Electroencephalogram, HAM-A-Hamilton Anxiety Rating Scale, HAM-D-Hamilton Depression Rating Scale, ISI-Insomnia Severity Index, LARS-Line Analogue Rating Scales, LSEQ-Leeds Sleep Evaluation Questionnaire, MMSE-Mini-Mental State Examination, PANSS-Positive and Negative Syndrome Scale, PANSI-The Positive and Negative Suicide Ideation, PSQI-Pittsburgh Sleep Quality Index, SFST-Standardized Field Sobriety Test, SMT-Short term Memory Test, SST-Symbol Search Test, STDI-State Trait Depression Inventory, SVRT-Simple Visual Reaction Test, WASO-Wake Time After Sleep Onset.
Herbal Combinations.
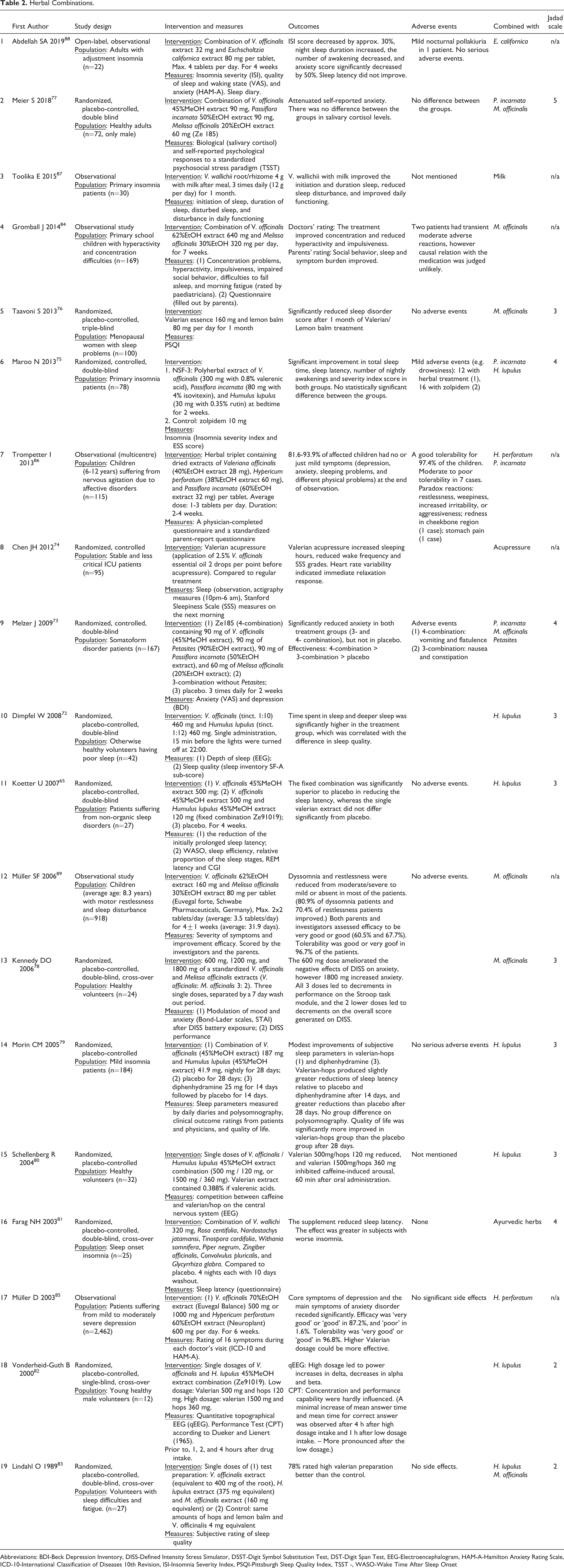
Abbreviations: BDI-Beck Depression Inventory, DISS-Defined Intensity Stress Simulator, DSST-Digit Symbol Substitution Test, DST-Digit Span Test, EEG-Electroencephalogram, HAM-A-Hamilton Anxiety Rating Scale, ICD-10-International Classification of Diseases 10th Revision, ISI-Insomnia Severity Index, PSQI-Pittsburgh Sleep Quality Index, TSST -, WASO-Wake Time After Sleep Onset
V. Officinalis as a Single Herb
Sleep problems
To evaluate the effectiveness of valerian for sleep problems, 40 articles using V. officinalis as a single herb were assessed. As a result, it was found that 23 studies addressed the effectiveness of valerian for sleep problems. Among them, 13 studies found valerian effective as a sleep aid. Three studies were performed with healthy subjects, 37,40,41 4 studies with insomnia patients, 36,42,52,66 and 1 study in RLS patients. 30 In addition, studies conducted with mental health patients, 67 cancer patients, 56 HIV-positive patients, 35 postmenopausal women, 57 and the elderly, 38 also found that valerian could improve sleep quality. 35,38,56,57 On the other hand, 10 studies found that valerian was not significantly effective compared to control 36,38,40,48,50,51,53,54,56,65 at least for the outcome measures used in those studies. Study outcomes were assessed using various measures, such as sleep questionnaires and diaries, such as Pittsburgh Sleep Quality Inventory (PSQI), 90 Insomnia Severity Index (ISI), 91 and Epworth Sleepiness Scale (ESS), to polysomnography. 92
Six studies measured immediate responses after single dose administration, 36,38,40 –42,50 while most studies evaluated the effectiveness of repeated doses. 30,35,54,56,57,65,67,36 –38,44,48,51 –53 The intervention periods ranged from 5 days to 8 weeks.
In order to extract possible associations between herbal preparations and study outcomes, the reports were classified into 4 groups (Table 1): 8 studies using hydroalcoholic extracts, 36,42,44,50,52–54,65 3 studies using aqueous extracts, 38,40,41 3 studies using extracts using unspecified solvents, 37,48,51 and 5 studies using herbal substance (the whole root / rhizome). 30,35,56,57,67 Among the 3 studies using aqueous extracts, 2 studies with single dose found improved subjective sleep quality and latency in healthy volunteers 40,41 (RCTs [Jadad scale 2 and 5]), while there was no significant difference in polysomnography 40 (RCTs [Jadad scale 2]). Schulz et al. found that repeated administration of V. officinalis aqueous extract (for 1 week) increased deep sleep (slow-wave sleep: SWS) in elderly subjects, although single dose of the same preparation did not have any significant impact on polysomnography 38 (RCT [Jadad scale 2]). As a single dose, V. officinalis hydroalcoholic extracts did not improve sleep quality 36,50 (RCT, Jadad scale 2-3), however improved REM sleep was observed in insomnia patients 42 (RCT [Jadad scale 2]). Repeated administration of hydroalcoholic extracts also led to inconsistent outcomes: sleep quality was improved in 4 studies using extract 600 mg (2 to 6 weeks) 36,37,44,52 (RCTs [Jadad scale 2-5]), whereas 3 studies using extract 300-600 mg (5 days to 4 weeks) found no improvement 53,54,65 (RCTs [Jadad scale 3-5]). Donath et al. found improved sleep latency and deep sleep after 2 weeks, while no improvement was observed after a single dose 36 (RCTs [Jadad scale 2]), which suggests that repeated administration is needed to exert observable effects. There were 3 studies using extracts with unspecified procedures. 48,51,66 The outcomes from those studies were also inconsistent: negative outcomes for 2 studies 48,51 (RCTs [Jadad scale 5]) and positive for an observational study. 66 On the other hand, all 5 studies using the dried root/rhizome showed that the interventions led to improved sleep at least in 1 subgroup 30,35,56,57,67 (RCTs [Jadad 3-5] and 1 observational study), suggesting that maximum efficacy could be expected from the root / rhizome before extraction.
It has been suggested that valerian may be useful to improve subjective sleep quality 22 and that repeated administration is required to obtain significant effects. 36 In order to evaluate if repeated administrations could consistently improve sleep quality, a meta-analysis was carried out (Figure 2). Studies using repeated valerian administrations were assessed to extract numerical values for the meta-analysis, and 10 randomized placebo-controlled trials reporting quantifiable results 30,34–36,51–54,56,57 were included in the analysis. The combined effect size was 0.36 (95% CI: -0.08 to 0.81) (Figure 2A). The funnel plot indicated missing data (Figure 2B) and high heterogeneity was observed (I2=85.33%). As described above, differences in herbal preparation could potentially contribute to the heterogenous outcomes. In fact, subgroup analysis for the whole root 30,35,56,57 (RCTs [Jadad 3-5]) and the extract 34,36,51–54 (RCTs [Jadad 2-5]) revealed that the combined effect size for the whole root was considerably higher 0.83 (95% CI: 0.03 to 1.62), compared to the extract (0.10; 95% CI: -0.02 to 0.22) (Figure 3), further supporting the use of the whole root rather than extract.

Sleep quality improvement by repeated administration of V. officinalis. Ten studies were included in meta-analysis. Positive values indicate enhanced sleep quality. (A) Forest plot for sleep quality improvement. Effect sizes (Hedges’ g) for 10 studies (blue circles) and combined effect size (green circle) are shown. Black horizontal bars indicate 95% CI. (B) Funnel plot. Included data points are presented as filled blue circles and imputed data are shown as open orange circles.

Subgroup analysis for V. officinalis whole root and extract as a sleep aid. Subgroup analysis was performed with 4 studies (1 ∼ 4) using the whole root/rhizome and 6 studies (5 ∼ 10) using extracts. Effect sizes for each group are shown as red circles. Black horizontal bars indicate 95% CI.
Anxiety
Anxiety was assessed by Hamilton Anxiety Rating Scale (HAM-A): a clinician-rated scale that considers psychological and somatic factors, 93,94 State-Trait Anxiety Inventory (STAI): a patient-rated scale that measures state and trait anxiety, 95,96 mental stress test using the color/word interference task, 97 or other questionnaires. Seven studies examined valerian’s anxiolytic potential, and positive outcomes were observed in 6 studies. 33,35,43,60,63,71 Valerian standardized extract 600 mg per day for 1 week reduced psychological and physiological stress reactivity under stress in healthy adult subjects 43 (RCT [Jadad scale 1]). Average 81.3 mg per day of valepotriates for 4 weeks reduced the score of HAM-A in generalized anxiety disorder patients 71 (RCT [Jadad scale 5]), suggesting that valepotriates are the active anxiolytic constituents. Valerian root / rhizome 530 mg per day for 4 weeks reduced anxiety in HIV-positive patients receiving efavirenz 35 (RCT [Jadad scale 3]), suggesting valerian could prevent neuropsychiatric adverse effects caused by the antiretroviral medication. Valerian extract 100 mg 33 or root / rhizome 100 mg, 60 single dose 60 min before the initiation of surgical operation, reduced anxiety in anxious patients undergoing dental operation 33,60 (RCTs [Jadad scale 5]). In addition, valerian extract 1,260 mg per day for 7 days for 3 menstrual cycles effectively reduced premenstrual anxiety 63 (RCT [Jadad scale 5]). However, an internet-based study with volunteers with non-clinical anxiety and insomnia found no significant difference in STAI compared to placebo 51 (RCT [Jadad scale 5]). Figure 4 shows the result of meta-analysis for valerian’s anxiolytic effects based on 7 randomized placebo-controlled trials that reported quantitative outcomes 34,35,47,49,51,60,63 (Figure 4A). The funnel plot indicated significant publication bias, missing data in the area of negative outcome, and high heterogeneity (I2=93.13%) (Figure 4B). Subgroup analysis revealed high variability for valerian extracts 34,47,49,51,63 (RCTs [Jadad scale 3-5]), whereas positive outcomes were observed for the whole root 35,60 (RCTs [Jadad scale 3 and 5]). However, due to the scarcity of data (2 studies for the whole root), it is difficult to draw a conclusion.

V. officinalis for anxiety. Eight studies were included in meta-analysis. Positive values indicate either reduced anxiety or enhanced calmness. (A) Forest plot for anxiety reduction. Effect sizes (Hedges’ g) for each study (blue circles) and combined effect size (green circle) are shown. (B) Funnel plot. Included data points are presented as filled blue circles. (C) Subgroup analysis for V. officinalis whole root (1 and 2) and extracts (3 ∼ 8).
Two studies investigated the effects of V. officinalis extracts on anxiety-associated brain activities. 31,34 Single dose of 300 mg extract reduced intracortical facilitation, possibly via GABAA enhancement 31 (RCT [Jadad scale 4]), and hydroalcoholic extract 300 mg per day for 4 weeks increased frontal brain region alpha coherence, which was correlated with anxiolysis 34 (RCT [Jadad scale 5]). These findings suggest that V. officinalis could induce immediate as well as long-lasting effects on anxiety-associated brain activities.
Other therapeutic effects
In addition to insomnia and anxiety, several other therapeutic benefits were suggested (Figure 5). Valerian aqueous extract 750 mg per day for 8 weeks was effective in reducing the symptoms of obsessive-compulsive disorder (OCD) 55 (RCT [Jadad scale 2]). Valerian root / rhizome 530 mg per day for 1 month improved cognition in haemodialysis patients 32 (RCT [Jadad scale 2]), and valerian root / rhizome 1,060 mg per day for 8 weeks prevented cognitive dysfunction after coronary bypass surgery 61 (RCT [Jadad scale 5]). Valerian root / rhizome 675 mg or extract 1,060 mg per day for 8 weeks reduced severity and frequency of hot flashes in menopausal and postmenopausal women 59,64 (RCTs [Jadad scale 4]). In addition, valerian root / rhizome 765 mg per day for 3 days for 2 cycles reduced pain severity and syncope in young females with dysmenorrhea 58 (RCT [Jadad scale 5]), and valerian extract 1,260 mg for 7 days for 3 cycles reduced premenstrual symptoms 63 , supporting the traditional use of valerian as a menstrual stimulant. 16

Valerian’s active constituents and potential therapeutic benefits.
Other Valerian Species
Three studies investigated the effectiveness of other Valeriana spp. as a sleep aid: 2 studies on V. edulis 42,46 and 1 study on V. wallichii. 70 V. edulis 20 mg/kg for 2 weeks reduced sleep latencies and improved sleep quality in children with intellectual deficit and primary sleep problems 46 (RCT [Jadad scale 5]), and V. edulis hydroalcoholic extract 450 mg single dose increased REM sleep and reduced the number of awaking in insomnia patients 42 (RCT [Jadad scale 5]). A concoction of valepotriates from V. wallichii 300 mg per day for 15 days reduced Wake Time After Sleep Onset (WASO) and improved sleep quality in insomnia patients 70 (RCT [Jadad scale 3]). Of note, chemical characteristics of the 3 species (V. officinalis, V. edulis, and V. wallichi) significantly differ; valerenic acids (valerenic acid, acetoxyvalerenic acid, hydroxyvalerenic acid) are specific to V. officinalis, whereas valepotriates (valtrate and isovaltrate) are common among the 3 species, 98 suggesting that valepotriates at least in part contribute to valerian’s sleep promoting activity. Due to the scarcity of data, it was not possible to perform meta-analysis for V. edulis and V. wallichi.
Valerian in Herbal Combinations
In addition to the effectiveness of valerian alone, 19 studies evaluated the effectiveness of valerian in combination with other herbs or other agents, among which 16 studies used V. officinalis, 65,72,82–86,88,89,73–80 2 studies used V. wallichii, 81,87 and 1 study used an unknown valerian species. 76 One study treated menopausal women with valerian essence in combination with lemon balm and observed sleep improvement, however the species and extraction methods were unidentified. 76
Among 16 reports using herbal combinations with V. officinals, 8 studies examined the effects on sleep quality and/or latency and all those interventions led to positive outcomes. 65,72,75,79,83,86,88,89 Six studies evaluated anxiolytic potential, and all those studies found positive outcomes at least for one of the measures at one of the tested dosages 73,77,78,85,86,88 (RCTs [Jadad scale 3-5] and 3 observational studies). However, a study using the combination of V. officinals, Passiflora incarnata (syn. P. edulis), and Melissa officinalis extracts (single dose) found that the intervention attenuated anxiety under stress, although it did not alter the level of salivary cortisol, a physiological parameter of stress reactions 77 (RCT [Jadad scale 5]). In another study, V. officinalis and M. officinalis combination (extract weight ratio 3:2) at 1,800 mg (single dose) increased anxiety while the same formulation at 600 mg reduced anxiety 78 (RCT [Jadad scale 3]). Both of these negative outcomes were observed for single doses in healthy subjects. On the other hand, the other 4 studies, conducted with subjects with adjustment insomnia, 88 somatoform disorder patients 73 (RCT [Jadad scale 4]), depression patients 85 (observational), and children with affective disorders 86 (observational), found that the herbal combinations with V. officinalis were effective in reducing anxiety. Of note, when depression patients were treated with V. officinalis (500 or 1,000 mg extract) and Hypericum perforatum (600 mg extract) for 6 weeks, higher valerian dose (1,000 mg) reduced anxiety more effectively 85 (observational). In addition, a study found that a combination of V. officinalis and M. officinalis extracts was effective in reducing the symptoms in children with hyperactivity and concentration difficulties 84 (observational). In a study with Intensive Care Unit (ICU) patients, V. officinalis essential oil combined with acupressure effectively induced relaxation and improved sleep 74 (observational). V. wallichii with 8 ayurvedic herbs improved sleep in sleep onset insomnia patients 81 (RCT [Jadad scale 4]), and V. wallichii with milk improved sleep in primary insomnia patients 87 (observational).
With respect to the potential herbal partners of V. officinalis, Humulus lupulus was the most frequently used in the clinical trials. All 7 reports that examined the effects on sleep or associated parameters observed sleep improvement 65,72,79,83 (RCTs [Jadad scale 2-3]), EEG power alteration toward sleep 82 (RCT [Jadad scale 2]), and reduced arousal 80 (RCT [Jadad scale 3]) were observed. M. officinalis used in 5 trials, 73,77,78,84,89 was the second most frequently combined herb, followed by P. incarnata used in 4 trials. 73,75,77,86 The 2 herbs were used in combination with V. officinalis for sleep problems and anxiety, resulting in positive outcomes. 73,75,77,78,84,86,89 Two studies were conducted on the combination of V. officinalis and H. perforatum with depression patients 85 (observational) and children with affective disorders 86 (observational), and the intervention reduced anxiety, 85,86 improved sleep, 86 and reduced depression symptoms. 85 Two studies used M. officinalis and P. incarnata 73,77 (RCTs [Jadad scale 4-5]), and 1 study used H. lupulus and P. incarnata 75 (RCT [Jadad scale 4]) in combination with V. officinalis. These 4 frequently used herbal partners and the study outcomes are summarized in Figure 6. It was not possible to perform meta-analysis for herbal combinations due to the highly variable outcome measures.

V. officinalis and herbal partners. Frequently used herbal partners and potential uses supported by evidence are shown with number of studies and total number of subjects (n).
Safety
There were no serious adverse events reported in the studies included in this review (60 studies, total n=6,894). Paradoxical stimulation, 16,99 such as agitation and restlessness, was experienced only in minority. 30,78,86 In older women with insomnia, valerian extract increased WASO, 53 suggesting it could have stimulatory effects. However, a study found that the same formulation at a higher dose increased anxiety while a lower dose was anxiolytic, 78 suggesting that paradoxical stimulation could be avoided by cautious dosing. Six studies investigated possible adverse effects on cognitive performance after V. officinalis extract intake (single dose) in healthy adults 37,39,47,49,62 and elderly volunteers. 45 V. officinalis standardized extracts at 100-1,600 mg did not impair cognitive or psychomotor performance 37,39,45,47,49,62 and were proven safer compared to triazolam, temazepam (benzodiazepines), and diphenhydramine (antihistamine). 45,47 Valerian did not cause any adverse events in postmenopausal women with insomnia, 57 older women with insomnia, 53 children with intellectual deficits and primary sleep problems, 46 and psychophysiological insomnia patients. 36 Mild adverse events were reported in RLS patients (vivid dreams and fatigue), 30 arthritis patients with sleep disturbance (dizziness and sleepiness), 54 sleep-disturbed subjects (drowsiness), 50 insomnia patients (gastrointestinal symptoms), 42,70 and outpatients with stress-induced insomnia (vivid dreams, drowsiness, heavy dream and depression), 66 however there was no clear association with the treatments. Potential herb-drug interaction was addressed by examining the effects on cytochrome p450 (CYP) expression levels, and no significant impact was detected at least for CYP1A2, CYP2D6, CYP2E1 and CYP3A4/5. 69,68 As a whole, valerian is safe in all ages.
Discussion
Concurring sleep problems and anxiety are frequently observed and their onset and course are interrelated. 100,101 In addition, these disorders are associated with a number of comorbid conditions including depression, 101– 103 dementia, 5,6 OCD, 104 and hot flashes 105– 107 (Figure 6). Our results demonstrated that sleep promotion and anxiolytic effects were the major therapeutic benefits expected of valerian (V. officinalis), and this herb could be also useful in treating OCD, cognitive dysfunction, menopausal hot flashes, as well as menstrual problems. However, the study outcomes considerably differed particularly when herbal extracts were used. We address this issue by discussing potential mechanisms underlying the therapeutic actions of valerian.
Key Constituents and Mechanisms of Actions
Although the use of valerian extracts as a sedative and sleep aid dates back to the 18th century and a number of constituents have been identified in the last 120 years, 108 the active constituents and underlying mechanisms for the reported activities remain obscure. 109 The identified constituents include iridoids known as valepotriates (valtrate, isovaltrate, didrovaltrate and acevaltrate), 110 essential oil constituents including monoterpenes (e.g. borneol, bornyl acetate), sesquiterpenes (e.g. valerenal, valerenic acid), and carboxylic compounds (valeric / isovaleric acid), 111 lignans, 112 flavonoids, 108,113 and low levels of γ-aminobutyric acid (GABA). 114 Valeriana edulis (Mexican valerian) Valeriana wallichii (Indian valerian) contain higher levels of iridoids, while valerenic acid and acetoxyvalerenic acid are unique to V. officinalis. 98
Valerian and GABAergic Signaling
GABA is an inhibitory neurotransmitter within the central nervous system and is a key target of pharmacotherapies in the treatment of anxiety and sleep disorders. 115,116 GABA is present in the central nervous system (CNS), where it plays a role in the maintenance of balance between excitatory and inhibitory neurotransmissions. GABA acts via 3 subclasses of receptors termed GABAA, GABAB, and GABAC, each of which has distinct characteristics. GABAA and GABAC receptors are ligand-gated ion channels, while GABAB members are G-protein coupled receptors. GABAA receptors are heteropentameric transmembrane protein complexes made up of α1-6, β1-3, γ1-3, δ, ε, θ, π subunits, and have numerous allosteric binding sites. 116 Although it is not very likely that GABA in valerian, taken orally, directly exerts any therapeutic action, valerenic acid and valerenol can allosterically modulate GABAA receptors to enhance the response of GABAA to GABA in vitro, 117– 119 and both valerenic acid and valerenol exerted anxiolytic activity in a GABAA β3 subunit-dependent manner in mice. 117 On the other hand, valerenic acid derivatives (e.g. acetoxy valerenic acid and hydroxy valerenic acid) bind the identical sites without inducing allosteric modulation, suggesting that these constituents can potentially compete with valerenic acid. 120 In fact, in an anxiety model in rodents, a valerian extract containing valerenic acid at a high level and acetoxy valerenic acid at a low level exhibited higher anxiolytic potency compared to an extract with acetoxy valerenic at a high level, indicating that standardization with respect to the total amount of valerenic acids (both valerenic acid and acetoxy valerenic acid) is inappropriate. 120 In addition, an aqueous extract of valerian induced GABA release in rat brain synaptosomes in vitro, 121 suggesting the presence of hydrophilic constituents potentially promotes GABAergic signaling. Expected mechanisms of actions are summarized in Figure 5. Of note, the expression patterns of GABA receptors in humans are sex- and age-dependent, and GABAA β3 subunit levels in the superior temporal gyrus were lower in older women than old men, 122 suggesting that valerian’s actions via GABAergic signaling could depend on the sex and age.
Valerian and Serotonergic Signaling
Serotonin (5-HT) plays essential roles in the regulation of sleep and mood, and serotonergic system is a promising therapeutic target for psychiatric disorders including anxiety, depression, and sleep disorders. 123– 126 Both valerian extract and valerenic acid exhibit partial agonist activity at 5-HT receptor 5A (5-HT5A) in vitro, 127 suggesting the involvement of serotonergic system in the actions of valerian. There are 7 families of its receptors (5-HT1-7). Although the functional significance of 5-HT5A remains obscure, 5-HT5A is present at high levels in the regions that regulate circadian rhythms (the suprachiasmatic nucleus, the intergeniculate leaflet, 128,129 the median raphe nuclei, and dorsal raphe nucleus) and colocalized with serotonin, suggesting the auto-receptor role of 5-HT5A in circadian regulation. 128 In addition, 5-HT5A is expressed in the brain areas such as the cerebral cortex, hippocampus, amygdala and hypothalamus, 129 suggesting that it is potentially involved in stress reactivity and resilience. Indeed, a study demonstrated that 5-HT5A antagonists could have different sedative, anxiolytic, and anti-depressant properties 130 (Figure 6). These data suggest that valerian, as a 5-HT5A partial agonist, may act differently depending on what circumstances the subjects were placed and this could possibly explain the inconsistent outcomes among the trials.
Valerian and Adenosine Signaling
Adenosine receptors play important roles in mood and anxiety disorders. 131 Adenosine A1 receptor activation reduces anxiety-like behavior in mice 132 and positive allosteric modulators of adenosine A1 receptor could be potential anxiolytic therapy. 133,134 In addition, adenosine helps maintain brain homeostasis by preventing over excitation and regulating the sleep-wake rhythm. 135– 137 Sleep stages are divided into 1 stage of rapid eye movement (REM) sleep and 4 stages of non-rapid eye movement (NREM) sleep (Stages 1-4) that are characterized by increasing sleep depth. 4,138 The deeper stages (Stages 3 and 4) are believed to be the most restorative and collectively called SWS. 4,138 Adenosine acting through A1 receptors facilitates sleep and increases slow wave activity (SWA) in SWS, whereas A1 receptor antagonism suppresses SWA. 139 Importantly, 2 studies found that V. officinalis could increase SWS, 36,38 possibly contributing to the improved subjective sleep quality while objective sleep latency remained unchanged. 36 These observations suggest that valerian may promote the sleep quality by inducing deep sleep via adenosine A1 receptor signaling. In fact, a polar extract of valerian exhibited partial agonistic activity at A1 receptors 140 and valerian contains hydrophilic constituents that interact with adenosine signaling, such as lignans of a olivil derivatives that is a partial agonist of adenosine A1 receptors. 112,141 Thus, valerian’s hydrophilic constituents could exert the anxiolytic and sleep-inducing effects via adenosine signaling. Importantly, while the hydrophilic extract was agonistic, isovaltrate in the hydrophobic extract acted as an inverse agonist at adenosine A1 receptor. 140 These findings indicate the hydrophilic and hydrophobic components of valerian act in opposite directions at adenosine receptors, 140 supporting the traditional use of water-based extract as a sleep aid, 16,140 and possibly explaining the contradictory findings regarding the efficacy of valerian hydroalcoholic extracts.
Valepotriates
Valepotriates are unstable thermolabile compounds that rapidly decompose. The characteristic odor of dried valerian is due to isovaleric acid released from decomposing valepotriates. 142 Although the mechanistic roles of valepotriates in the therapeutic effects are yet to be clarified, pre-clinical evidence suggests that valepotriates exert anxiolytic 143 and antidepressant-like effects, 144,145 possibly via the dopaminergic and noradrenergic neurotransmission but not via the serotonergic system. 145 The monoaminergic neurotransmitters (dopamine, noradrenaline, and 5-HT) play overlapping roles in the etiology of anxiety and depression, and medications targeting these systems are proved to be effective in treating both anxiety and depression. 146 Moreover, those monoamines play a role in sleep-wake regulation. 147 Thus, the presence of valepotriates is likely to be essential in treating these disorders. In addition, valtrate reduced the serum glucocorticoid level in a rat anxiety model, 148 suggesting hypothalamus-pituitary-adrenal axis modulatory potential of valepotriates. Considering the significant association between neuroendocrine-immune axis imbalance and stress vulnerability and resilience, 149 the potential impact on the hypothalamus-pituitary-adrenal axis is particularly important. Stress can disturb the sleep, and stress reactivity is implicated in vulnerability to insomnia and other stress-related disorders such as anxiety and depression. 8,150 Furthermore, glucocorticoid imbalance could lead to amyloid formation and tau accumulation, hallmarks of Alzheimer’s disease, 151 and elevated glucocorticoid levels are associated with neurodegenerative disorders. 152 Thus, valepotriates may prevent stress-induced neuropsychiatric disorders including anxiety and depression, as well as cognitive dysfunction, by restoring endocrine-immune balance. Expected mechanisms of action are summarized in Figure 6.
Quality and Shelf Life
As described above, multiple constituents, including valerenic acids, lignan, and valepotriates, differentially contribute to the therapeutic effectiveness of valerian. However, most of the commercially available valerian extracts are standardized to the levels of valerenic acids, while the contents of valepotriates are often unknown. Importantly, Bos et al. compared freshly prepared tincture (70% EtOH extract) and the same extract stored at room temperature for 2 months, and found that the levels of valepotriates (valtrate and isovaltrate) after 2 months were less than 5% of the original, whereas the levels of valerenic acids (valerenic acid and acetoxyvalerenic acid) were not affected. 98 This finding suggests that the shelf life of valerian extracts may be significantly shorter if the levels of valepotriates are considered. It is possible that the negative outcomes observed in the trials using valerian extracts, rather than the raw materials, could have been caused by the loss of valepotriates in those extracts.
In the United States, valerian-containing products are widely available as dietary supplements, in various forms under various trade names. Examples are Valerian (NOW FOODS, IL, US; Nature’s Way, WI, USA), Standardized Valerian (root extract standardized to 0.8% valerenic acids; Nature’s Way), and Amantilla (hydroalcoholic extract; NutraMedix, FL, USA), to name a few. In Europe, the Committee on Herbal Medicine Products (HMPC) concluded that valerian-containing medicinal products have to be assessed by the responsible national authorities. Available products include Baldrian (85% ethanol extract; Kneipp, Würzburg, Germany), and Baldrianwurzel (70% ethanol extract; Hofapotheke St. Afra, Augsburg, Germany). Herbal combinations are also available (e.g. Valeirana-Melissa extract [standardized to 0.8% valerenic acids; Bonusan, Rotterdam, Netherlands]). Of note, the recommended storage temperatures for most of these products are at or below room temperature (around 25°C).
Limitation
The current study has reviewed the effectiveness of valerian without specifying target populations. This raises a limitation as the etiology underlying sleep problems and associated health problems could be diverse depending on age, sex, and general health conditions. Consequently, there could be potential differences in the outcomes depending on the target populations, however such possibilities were not addressed in this review. In addition, heterogeneity in the outcome measures limited the number of data sources for meta-analysis, which in turn limits the generalizability of the results. Further research is needed to assess the effectiveness for specific populations and outcomes.
Conclusion
According to the 1905 edition of King’s American dispensatory, 99 ‘valerian excites the cerebro-spinal system.’ and ‘in medicinal doses it acts as a stimulant-tonic,…’, which suggests that valerian is not a simple hypnotic or anxiolytic agent. In addition, the text continues that ‘The extract of valerian is worthless, but the fluid extract has been found to possess all the medicinal virtues of the root’, 99 suggesting that extraction methods could play a crucial role in preserving the therapeutic potency. Valerian has diverse chemical properties. As summarized in Table 3, the differences in polarity can lead to variability in the quality of valerian extracts depending on the extraction solvents. Our study has found that the hydroalcoholic extracts used in the clinical trials are variable with respect to the solvents (Table 4), and their phytochemical properties are not characterized in most cases. Furthermore, there is no information regarding the storage conditions such as temperature and storage period. Considering the low stability of some of the important active constituents, 98,142 it is unknown to what extent the active constituents had been preserved at the end of those trials. The absence of such information limits the discussion as to why some extracts were ineffective while others exhibited effectiveness in those clinical trials. Due to the multiple active constituents and complex mechanisms of actions (Figure 5), it is imperative to use well characterized plant materials and extracts in the future clinical trials, in order for the evaluation of valerian’s true effectiveness to be possible.
Polarity Indices of Major Constituents.

Polarity index values were obtained from PubChem database (https://pubchem.ncbi.nlm.nih.gov).
XLogP3-AA (computed by XLogP3 3.0) indicates hydrophobicity, whereas Topological Polar Surface Area (computed by Cactvs 3.4.6.11) and Hydrogen Bond Donor / Acceptor (computed by Cactvs 3.4.6.11) indicate hydrophilicity.
Solvents for Valerian Extract Preparation.

This study demonstrated that valerian could be a safe and useful herb alone and also in combination in treating sleep problems, anxiety, and associated comorbidities. One possibility for exploration that arises is whether valerian is particularly useful in treating insomnia where there are higher levels of anxiety. In fact, the observation that V. edulis reduced sleep problems most effectively in children with hyperactivity 46 may suggest that valerian is more useful in treating insomnia associated with particular psychiatric conditions such as anxiety. The safety has been proven in a wide range from childhood 46,84,86 to old age. 38,45,53 Sleep problems are associated with a number of comorbidities including anxiety, depression, and dementia in both adulthood 123,153 and childhood. 154 As we age, the quantity and quality of sleep decrease, 155,156 and insomnia and daytime drowsiness are frequently reported in the older population. 157 Thus, valerian may help improve the quality of life in all ages by improving sleep quality, thereby preventing a number of psychiatric and cognitive dysfunctions. However, due to the presence of multiple active constituents and the unstable nature of some components, it may be useful to revise the standardization methods and shelf life of the extracts. Repeated treatments with the whole root / rhizome consistently promoted sleep quality at 450-1410 mg per day for 4-8 weeks, whereas valerian extracts 300-600 mg per day for 5 days-4 weeks resulted in inconsistent outcomes. Evidence is poor with respect to the effectiveness of single dose intervention as a sleep aid. Developing effective standardization methods would be desirable. Meanwhile, the usage of whole herbal substances (root / rhizome), rather than extracts, may be the way to obtain optimal efficacy for the time being.
Lastly, due to the subjective nature of sleep problems and associated mood disorders, there remains a difficulty in determining the best measure for the efficacy of interventions. The ultimate goal of any therapies, whether it be traditional, conventional or integrative, beyond disease ‘cure’ or modulation, is to improve the quality of life for patients. While objective measures in a laboratory setting provide valuable information and mechanistic understanding, it may be useful to consider additional and more important factors that need to be addressed specifically in each individual. Such measures and tailor-made approaches would necessarily involve sufficient interaction and communication between patients and practitioners. Developing effective trial strategies to address this issue is a future challenge.
Supplemental Material
Supplemental Material, PRISMA_checklist_200107 - Valerian Root in Treating Sleep Problems and Associated Disorders—A Systematic Review and Meta-Analysis
Supplemental Material, PRISMA_checklist_200107 for Valerian Root in Treating Sleep Problems and Associated Disorders—A Systematic Review and Meta-Analysis by Noriko Shinjyo, Guy Waddell and Julia Green in Journal of Evidence-Based Integrative Medicine
Footnotes
Authors Note
All research done by the authors.
Acknowledgments
We thank the collaborative environment at University of Westminster, Heartwood Education, and Chiba University, which enabled this study.
Author Contributions
NS wrote this systematic review with input and contributions of GW and JG. NS, GW and JG planned the search in databases and defined criteria for the selection of articles. NS carried out the search, screen the materials and proposed a selection. All authors contributed to the evaluation of the studies. All authors contributed to the interpretation of data and discussion. All authors reviewed the whole manuscript, read and approved the submitted version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
