Abstract
Drug interactions are key reasons for adverse drug reactions and attrition from market. Major infectious diseases causing morbidity/mortality in Ghana are malaria, tuberculosis, and HIV/AIDS. In this study, plant medicines commonly used to treat/manage these diseases in Ghana were investigated for their potential to modulate rat cytochrome P450 enzyme activities. Fluorescence and high-performance liquid chromatography–based assays were used to assess effects of antimalarial plant medicines, Fever (FEV), Mal-TF (MAL), and Kantinka terric (KT); anti-TB medicines, Chestico (CHES), CA + ST Pains + HWNT (TF), and Kantinka herbatic (KHB); and anti-HIV/AIDS medicines, Wabco (WAB), AD + T/AD (LIV) and Kantinka BA (KBA) on rat liver microsomal cytochrome P450 enzyme activities. Effects of medicines on rat biochemical and hematological parameters were also assessed. Generally, the medicines altered microsomal CYP1A1/1A2, CYP2B1/2B2, CYP2C9, and CYP2D6 activities. Only KBA elicited an increase (80%) in CYP1A1/1A2 activity. FEV, MAL, CHES, WAB, and LIV strongly inhibited the enzyme activity. All the medicines significantly inhibited CYP2C9 (24%-80%) activity. CYP2D6 activity increased after treatment with MAL, KBA, LIV, and TF. Also, MAL, WAB, LIV, KHB, and CHES increased CYP2B1/2B2 activity, while KT decrease the activity. Generally, the medicines altered liver function in the rats. Cholesterol levels declined after KBA treatment only. White and red blood cell counts, hemoglobin and hematocrit levels were significantly reduced in KT- and KBA-treated rats. Our results suggest that use of the medicines could have implications for drug interactions and safety, particularly if the medicines are administered over prolonged periods. Further investigations are imperative to establish clinical relevance of these results.
A major challenge in drug discovery is to predict pharmacokinetic drug interaction mediated by cytochrome P450 (CYP) enzyme induction and inhibition. 1 The CYP enzymes constitute one of the largest superfamilies of monooxygenase enzymes responsible for the metabolism of several drugs and foreign substances which are well studied. 2,3 The induction of CYP enzyme may result in enhanced metabolism of a co-administered drug, which is a substrate of a CYP isoenzyme and subsequently cause therapeutic failure or toxicity if metabolites are bioactive. Induction of CYP has been implicated in cases of clinically significant drug-drug interactions. 4 Inhibition of CYP may also results in accumulation and toxicity particularly if the drug metabolism depends mainly on the specific CYP. 5 Reports from a number of studies have suggested that drug interactions account for over 30% of harmful effects, thus several drugs have been withdrawn from the market due to drug-drug interactions. 3,5 Other studies have also shown that herbal products may cause CYP-mediated herb-drug interactions when co-administered with orthodox drugs. 6 –8 Herbal medicines such as St John’s wort, ginseng, and gingko, which are freely available over the counter, have been reported to cause serious interactions when co-administered with some prescribed drugs. 6 A survey conducted in the Ga East district of Ghana, revealed that about 50% of patients in the district co-use orthodox and herbal medicines (Appiah-Opong et al., unpublished data). Most of these people do not disclose their medicine use to their physicians. A similar trend has previously been observed in the United States. 9 Thus, limited information exists on the effects of herb-drug interaction. However, the potential for herb-drug interactions is higher than for drug-drug interactions since herbal medicines are usually mixtures of more than one active component. Adverse drug effects may occur as a result of such interactions. For example, St John’s wort, a popular antidepressant has been implicated in fatal interaction with cyclosporine, and there is evidence that the mechanism involves induction of CYP enzymes. 10 Given the common practice of consumption of herbal medicines with orthodox medicines, it is necessary to evaluate the modulation of CYP activity by plant medicines that are commonly used to treat or manage life-threatening infectious diseases such as malaria, HIV/AIDS, and tuberculosis (TB).
In Ghana, apart from orthodox medicines, there are several medicinal plants that are used as traditional therapies for management of these infectious diseases among others. These medicines are easily available over-the-counter, at herbal centers or on the local markets. It is necessary to assess the potential for these medicines to alter metabolism of orthodox medicines or other plant medicines used to treat other diseases. This information is useful for the prevention of adverse effects resulting from herb-drug or herb-herb interactions. In this study, we investigated the potential for modulation of CYP enzyme activity by plant medicines commonly used to treat or manage malaria, TB, and HIV/AIDS in Ghana. Effects of the medicines on hematological and biochemical parameters were also assessed.
Materials and Methods
Chemicals and Reagents
Ethoxyresorufin, pentoxyresorufin, diclofenac and dextromethorphan were purchased from Sigma-Aldrich (St Louis, MO, USA). All other chemicals and reagents used were of analytical grade and obtained from standard suppliers.
Ethical clearance for the study was obtained from the Institutional Review Board of the Noguchi Memorial Institute for Medical Research (NMIMR-IRB CPN 020/11-12), University of Ghana, Legon, Ghana.
Plant Medicines
Plant medicines used to treat or manage malaria, HIV/AIDS, and TB were kindly donated by herbal health facilities. Kantinka terric (KT), Kantinka BA (KBA), and Kantinka herbatic (KHB) were donated by Adom Herbal Clinic; Fever (FEV), Wabco (WAB), and Chestico (CHES) were from Amen Scientific Hospital; and Mal + TF (MAL), AD + T/AD (LIV), and CA + ST Pains + HWNT (TF) were from Top Herbal Clinic. Table 1 shows the constituents of the plant medicines used. The medicines were frozen at −80°C, lyophilized, and stored at −20°C until use.
Plants Medicines Used and Their Constituents.

a Sourced from Adom Herbal Center.
b Sourced from Amen Scientific Hospital.
c Sourced from Top Herbal Clinic.
Laboratory Animals, Pretreatment, and Preparation of Liver Microsomes
A total of 45 eight-week old male Sprague-Dawley rats (Hsd: SD strain) used for the study were obtained and maintained in the Animal Experimentation Department (Noguchi Memorial Institute for Medical Research, Legon, Ghana). The animals were housed in groups of 3 in stainless steel cages (34 cm × 47 cm × 18 cm) with soft wood shavings as beddings. They were fed with normal commercial pellet diet (AGRIMAT, Kumasi), given water ad libitum, and maintained under laboratory conditions (temperature 22°C ± 2°C, relative humidity 60%-70% and 12-hour light-dark cycle). All the animal procedures and techniques used in this study were approved by the Institutional Review Board of the Noguchi Memorial Institute for Medical Research. To evaluate the potential to regulate CYP expression, the herbal medicines (100 mg/kg body weight/day) were administered to the rats by oral gavage for 7 days. The negative control group was given the vehicle (water).
Twenty-four hours after the last treatment, the rats were euthanized in a chloroform chamber. Blood was drawn by cardiac puncture and livers immediately isolated, placed on ice and then stored at −80°C until use. Blood sample (1 mL) was collected from each animal into tubes with and without ethylenediaminetetraacetic acid (EDTA) for hematological and biochemical analysis. Subsequently, the livers from treated and untreated rats were excised and microsomal fractions were prepared by ultracentrifugation as previously described. 11 Protein concentration was determined by the method of Bradford, 12 with bovine serum albumin as the standard. The CYP activity was assessed using fluorescence and high-performance liquid chromatography (HPLC)–based assays.
Ethoxy- and Pentoxy-Resorufin O-Dealkylation
The effects of the medicines on ethoxyresorufin O-deethylase (EROD/CYP1A1/1A2) and pentoxyresorufin O-depentylase (PROD/ CYP2B1/2B2) in rats were determined as described. 11 Briefly, microsomal incubations consisted of the substrates, ethoxy- or pentoxy-resorufin (5 µM), microsomal protein (0.1 mg protein/mL) and 0.1 M phosphate buffer (pH 7.4). The reaction mixtures were preincubated for 5 minutes and reactions initiated by adding 100 µM NADPH (reduced form of nicotinamide adenine dinucleotide phosphate) and allowed to proceed for 20 minutes at 37°C. Reactions were terminated with 80% acetonitrile and 20% 0.5 M Tris solution. All experiments were performed in triplicate. Fluorescence measurements were done using a Tecan Infinite M200 Pro plate reader (λex = 530 nm and λem = 586 nm).
Diclofenac Hydroxylation
To determine modulation of CYP2C9 activity by the medicines, the enzyme activity was assessed by measuring the formation of 4-hydroxydiclofenac from diclofenac in the treated microsomal fractions. 13 Reaction mixtures consisted of the substrate, diclofenac (6 µM), microsomal protein (0.1 mg protein/mL) and 0.1 M phosphate buffer (pH 7.4). Preincubations were done for 5 minutes and the reactions were carried out in the presence of 100 µM NADPH, for 10 minutes at 37°C. Reactions were terminated by adding methanol. Duplicate experiments were performed. Product formed was measured using an isocratic HPLC (Shimadzu, Japan) method. A C18 column was used at a carrier flow rate of 0.6 mL/min. The mobile phase consisted of 60% (v/v) 20 mM potassium phosphate (pH 7.4), 22.5% (v/v) methanol, and 17.5% (v/v) acetonitrile. Chromatographic peaks were monitored at the wavelength of 280 nm.
Dextromethorphan O-Demethylation
The effects of the plant medicines on rat liver microsomal CYP2D6 levels in each of the 3 treated groups of rats were evaluated by measuring the formation of dextrorphan from dextromethorphan, as described. 13 The reaction mixture comprising rat liver microsomal fraction (0.1 mg protein/mL), 4.5 µM dextromethorphan, and 0.1 M potassium phosphate buffer was preincubated for 5 minutes before 100 µM NADPH was added and the reaction was allowed to proceed for 45 minutes. Reactions were terminated by adding 60 mM zinc sulfate. Duplicate experiments were performed. The mobile phase consisted of 24% (v/v) acetonitrile and 0.1% (v/v) triethylamine adjusted to pH 3 with perchloric acid. The carrier flow rate was 0.6 mL/min. Metabolite formation was monitored using an isocratic HPLC procedure with a C18 column fluorescence detection (λex = 280 nm and λem = 310 nm).
Biochemical and Hematological Assays
To determine the impact of medicines on biochemical parameters, the blood samples removed from the treated and control animals were centrifuged at 1500 rpm for 10 minutes. The sera obtained were used for determination of levels of alanine aminotransferase (ALT), aspartate amino transferase (AST), albumin (ALB), direct (conjugated) bilirubin (DBIL), total bilirubin (TBIL), γ-glutamyltransferase (GGT), cholesterol (CHOL), trigycerides (TG), creatinine (CRE), and glucose (GLU). Measurements were done using a Selectra E Clinical Chemistry Analyzer (Dieren, the Netherlands) with commercial kits (ELITech Clinical Systems, France). Hematological parameters, including white blood cell (WBC) and red blood cell (RBC) counts, hemoglobin (HB), and hematocrit (HCT) levels, mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH) level, and mean corpuscular hemoglobin concentration (MCHC) were also measured on blood samples delivered into EDTA tubes, using an automated hematology analyzer (KX-2IN, Sysmex Corporation, Japan).
Data Analysis
Percent induction of CYP activity by drugs and plant medicines was calculated from the ratio of the activity of treated to control samples. Student’s t test was used for comparing of differences between means to determine significant levels. A P value <.05 was considered statistically significant.
Results
CYP Assays
Figure 1 shows that plant medicines KT, KBA, and TF did not significantly alter CYP1A1/1A2 activity, but KHB caused an 80% increase in activity of the enzyme, while the remaining medicines strongly inhibited the enzyme activity in the range 54% to 85.8%. The strongest inhibitory activity (85.8%) was caused by WAB.

Increase in EROD (CYP1A1/1A2) activity by drugs and plant medicines used to treat/manage malaria, tuberculosis, and HIV/AIDS in Ghana. Assay conditions are described in section “Ethoxy- and Pentoxy-Resorufin O-Dealkylation.” The charts represent mean and standard deviations of triplicate experiments. The asterisk (*) indicates statistically significant difference (P < .05) from untreated control experiments as determined by the Student’s t test (refer to section Plant Medicines for names of medicines).
Effects of the plant medicines on rat liver microsomal CYP2B1/2B2 activity is shown in Figure 2. Five of the medicines significantly increased the enzyme activity in the range 20.8% to 60.2%. Among the 3 antimalarial plant medicines, MAL treatment resulted in an increase in PROD activity (23.8%) while KT caused a reduction in the activity (22.7%). The FEV treatment did not significantly modulate the enzyme activity. Two anti-HIV medicines, WAB and LIV caused increase in enzyme activity of 20.8% and 27.5%, respectively, whereas KBA did not alter the enzyme activity significantly. Similarly, 2 anti-TB medicines, KHB and CHES, caused increases in PROD activity (47.1% and 60.2%, respectively) while TF did not significantly alter the enzyme activity.
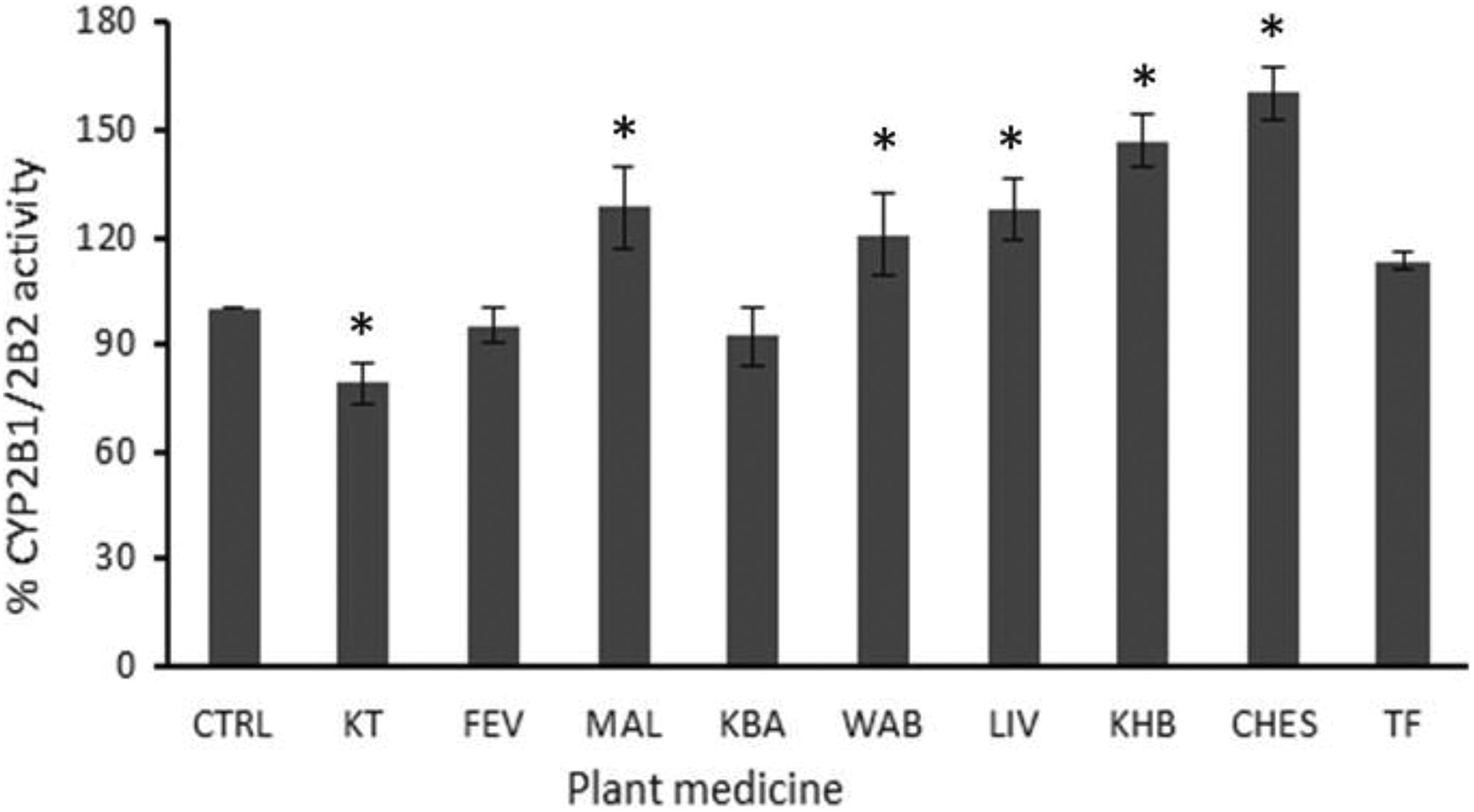
Increase in PROD (CYP2B1/2B2) activity by drugs and plant medicines used to treat/manage malaria, tuberculosis, and HIV/AIDS in Ghana. Assay conditions are described in section “Ethoxy- and Pentoxy-Resorufin O-Dealkylation.” The charts represent mean and standard deviations of triplicate experiments. The asterisk (*) indicates statistically significant difference (P < .05) from untreated control experiments as determined by the Student’s t test.
Figure 3 shows that all the plant medicines strongly inhibited the CYP2C9 activity in the rat liver by 23.9% to 80.7%. The strongest inhibitor was KBA, which showed 80.7% inhibitory effect. The effects of the plant medicines on CYP2D6 are shown in Figure 4. Five out of the nine medicines did not alter CYP2D6 activity. Significant increases in CYP2D6 activity were found in MAL-, KBA-, LIV-, and TF-treated rats.

Increase in CYP2C9 activity by drugs and plant medicines used to treat/manage malaria, tuberculosis, and HIV/AIDS in Ghana. Assay conditions are described in section “Diclofenac Hydroxylation.” The charts represent mean and standard deviations of duplicate experiments. The asterisk (*) indicates statistically significant difference (P < .05) from untreated control experiments as determined by the Student’s t test.

Increase in CYP2D6 activity by drugs and plant medicines used to treat/manage malaria, tuberculosis, and HIV/AIDS in Ghana. Assay conditions are described in section Dextromethorphan O-Demethylation.” The charts represent mean and standard deviations of duplicate experiments. The asterisk (*) indicates statistically significant difference (P < .05) from untreated control experiments as determined by the Student’s t test.
Biochemical and Hematological Parameters
Results on the biochemical and hematological parameters in the rats treated with the antimalarial, anti-HIV, and anti-TB medicines are shown in Tables 2, 3, and 4, respectively. The results obtained on the biochemical indices show significant increase of 80.7% in serum ALT level in rats treated with the antimalarial plant medicine KT, while CHES caused a decrease in the enzyme level by 23.7%. The other plant medicines did not cause any significant changes in ALT levels of the treated rats (Tables 2 -4). Similarly, significant changes in serum AST levels were only observed of KT-, MAL-, LIV-, and KBA-treated rats (20.3%-28.1%). In contrast, serum ALB levels were not significant altered in any of the rats treated with the medicines. Furthermore, increases in DBIL levels were found in rats treated with KBA (110%) and KHB (180%), but no significant changes occurred in other plant medicine-treated rats (Tables 2 -4). Levels of serum TBIL were also elevated in KBA (130%), LIV (92.3%), CHES (76.9%), and TF (69.2%)-treated rats. Serum GGT levels declined in FEV, MAL, WAB and CHES-treated rats (68%-76%) while increased levels were found in LIV (112%) and TF (68%) treated rats. There were no significant changes in the GGT levels in KBA- and KHB-treated rats.
Biochemical and Hematological Parameters of Rats on Antimalarial Medicines.a

Abbreviations: KT, Kantinka terric; FEV, Fever; MAL, Malaria + TS; ALT, alanine aminotransferase; AST, aspartate aminotransferase; ALB, albumin; DBIL, direct (conjugated) bilirubin; TBIL, total bilirubin; GGT, γ-glutamyltransferase; CHOL, cholesterol; TG, triglycerides; CRE, creatinine; GLU, glucose; WBC, white blood cells; RBC, red blood cells; HB, hemoglobin; HCT, hematocrit; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration.
a Values are means ± SD of at least 2 experiments.
b Statistically significant difference (P < .05) compared with control experiment (untreated).
Biochemical and Hematological Parameters of Rats on Anti-HIV Medicines.a

Abbreviations: KBA, Kantinka BA; WAB, Wabco; LIV, AD + T/AD; ALT, alanine aminotransferase; AST, aspartate aminotransferase; ALB, albumin; DBIL, direct (conjugated) bilirubin; TBIL, total bilirubin; GGT, γ-glutamyltransferase; CHOL, cholesterol; TG, triglycerides; CRE, creatinine; GLU, glucose; WBC, white blood cells; RBC, red blood cells; HB, hemoglobin; HCT, hematocrit; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration.
a Values are means ± SD of at least 2 experiments.
b Statistically significant difference (P < .05) compared with control experiment (untreated).
Biochemical and Hematological Parameters of Rats on Antituberculosis Medicines.a

Abbreviations: KHB, Kantinka herbatic; CHES, Chestico; TF, CA + ST Pains + HWNT; ALT, alanine aminotransferase; AST, aspartate aminotransferase; ALB, albumin; DBIL, direct (conjugated) bilirubin; TBIL, total bilirubin; GGT, γ-glutamyltransferase; CHOL, cholesterol; TG, triglycerides; CRE, creatinine; GLU, glucose; WBC, white blood cells; RBC, red blood cells; HB, hemoglobin; HCT, hematocrit; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration.
a Values are means ± SD of at least 2 experiments.
b Statistically significant difference (P < .05) compared with control experiment (untreated).
No significant changes occurred in serum CHOL levels of rats treated with plant medicines, with the exception of KBA-treated rats in which a decrease of 44.4% was observed (Tables 2 -4). Increase in serum TG level of about 40% to 50% were observed in KT-, KBA-, and KHB-treated rats whilst reduced level was observed in those treated with CHES (30%). Similarly, serum CRE levels were elevated in KT (40.2%) and KHB (32.5%) treated rats, but no significant changes were found in the levels of rats treated with the other plant medicines.
Significant increase in serum GLU level (38%) was observed in KT-treated rats, whereas decrease in levels by about 30% to 40% was found in WAB-, KHB-, and TF-treated rats.
Results on hematological parameters tested showed significant decrease in counts of WBC in KBA (88%), KT (72.8%) and KHB (34%) treated rats, whereas an increase of 20% was found in rats treated with FEV (Tables 2 -4). No significant changes were observed in rats treated with the remaining medicines.
Red blood cell counts, hemoglobin and hematocrit levels of rats treated with KT and KBA declined significantly while the other plant medicines did not cause any significant changes to these parameters in the rats (Tables 2 -4). The MCV, MCH, and MCHC of the treated rats were not significantly altered except with the latter, which was significantly increased (23.3%) after treatment with KBA.
Discussion
Studies on effects of plant medicines on important drug metabolizing enzymes such as CYP are important for early detection of potential clinically relevant pharmacokinetic herb-drug interactions. These interactions pose a risk of toxicity or therapeutic failure and constitute a major cause of attrition during drug development or their removal from the market. Herb-drug interactions are even more critical because plant medicines comprise a mixture of several bioactive components therefore the potential for herb-drug interactions is higher than drug-drug interactions since usually there are less targets involved in the latter.
Although the antimalarial, anti-TB, and anti-HIV plant medicines used in the study are commonly used in Ghana, there have not yet been any reports on their effects on drug metabolizing enzymes. In view of this, we assessed these plant medicines, commonly used to treat malaria and TB and to manage HIV/AIDS in Ghana for their potential to induce CYP-mediated drug interactions. Some conventional drugs were used as controls for comparison. Several studies have shown that some plant medicines and their derivatives have the potential to induce or inhibit CYP enzymes. 14 –17 The significant increase in rat liver CYP1A1/1A2 activity caused by KHB (64.2%) could have implications for drug interactions, resulting in therapeutic failure, if the medicine is co-administered with drugs metabolized by the enzyme. 18 A previous study has shown that Mangifera indica, which is a component of KHB inhibits CYP1A2 in human hepatocytes; however, the converse was found in the present study. 19 This could be due to the fact that the other 2 components of the products increase the enzyme activity strongly, neutralizing the inhibitory effect of M indica. On the other hand, the strong inhibitory activity on the CYP1A1/1A2 exhibited by FEV, MAL, LIV, WAB, and CHES could have implications for adverse effects for drugs taken concurrently with these plant medicines if the drugs that are substrates of CYP1A1/1A2 and have narrow therapeutic window. 20 The strong inhibitory activity shown by products could be due to combined or individual effects of the constituents of the plant products shown on Table 1. For instance, the inhibitory effect of FEV was possibly caused by Parinari robusta since Morinda lucida, the only other component in the product has been reported to have no inhibitory effect on human recombinant CYP1A2 activity. 15
Among the medicines tested, MAL, LIV, WAB, CHES, and KHB increased rat liver CYP2B1/2B2 activity to different extents. The enhanced enzyme activity due to the medicines suggest their potential to cause drug interactions. 4 Apart from possible therapeutic failure, significant increases in enzyme activities could also result in toxicity if increased levels of electrophilic or bioactivated metabolite(s) are generated. 21 The remaining medicines caused weak inhibition of the enzyme activity, which are not likely to have implications for drug interactions. Azadirachta indica present in MAL has been shown inhibit CYP activity in mice, thus, the observed increase in enzyme activity may be due to Alstonia boonei and Vernonia amydalina, which are the other components present in the product. 22
In this study, all the medicines significantly inhibited CYP2C9 activity, the strongest being KBA (80.7%). This suggests that the medicines have strong potential to cause drug interaction if taken with drugs metabolized by the CYP2C9 such as S-warfarin, an anticoagulant used to block clotting avoid its migration to other parts of the body. 23 It is worth noting that CYP2C9 is responsible for metabolism of about 12.8% of the drugs on the market, 24 therefore inhibition of this enzymes could cause therapeutic failure. On the other hand, CYP2D6 activity was significantly elevated by MAL, KBA, LIV, and TF, while the other medicines did not have significant effect on the activity of the enzyme. These results suggest that the 4 medicines have the potential to cause CYP-mediated drug interaction when co-administered with drugs metabolized by CYP2D6, including Primaquine, which is the only antimalarial drug available to clinicians for the treatment of relapsing forms of malaria. 25 So far, CYP2D6 has been known to be a noninducible enzyme. 2 Therefore, it may be interesting to explore the factors accounting for the increase in the enzyme activity among the others. CYP2D6 accounts for metabolism of about 25% of the drugs on the market and it is highly polymorphic, an attribute that results in poor, intermediate, and ultra-rapid metabolism of medicines. 2 Thus, modulation of CYP enzyme activity by these plant medicines could have implications for metabolism of several drugs on the market.
The biochemical and hematological analysis on blood samples of the plant medicine–treated rats were performed to assess toxicity of the medicines. Parameters used to assess the integrity of the liver after treatment were serum ALT, AST, albumin, direct bilirubin, total bilirubin, and GGT. Each of the plant medicines caused significant alteration in one or more of the parameters for liver function investigated, except serum albumin level, which was not significantly altered by any of the medicines. In Ghana, use of plant medicines have generally been considered as safe because they originate from nature and most of them have also been used for several decades. However, lately there have been suggestions that plant medicines can contribute to organ damage, particularly to liver and kidney damage, due to their popularity and the scarcity of safety studies on them. Therefore, it is imperative that studies are performed on all the plant medicines on the market to ensure their safety for use by humans. Drug-induced liver injury is a major challenge to physicians and a leading cause of death from acute liver failure in the United States. 26,27 Elevated bilirubin levels were measured after treatment with the medicines KHB, KBA, LIV, CHES, and TF. Elevated bilirubin levels have implications not only for liver damage but also cardiovascular damage. 28 The enzyme GGT is critical for glutathione homeostasis, 29 thus, alteration in levels of this enzyme caused by 6 (FEV, MAL, WAB, LIV, CHES, and TF) out of the 9 medicines tested suggests their potential to cause oxidative stress as well as liver damage, particularly if these plant medicines are consumed over a prolonged period, considering that in this study the plant medicines were administered over a period of 7 days.
Generally, rat serum total cholesterol level was not significantly altered on treatment with the plant medicines, suggesting that the synthetic function of the liver was not compromised by those plant medicines. 30 KBA however caused a reduction in cholesterol level. Apart from its potential adverse effect on the liver, since this medicine is used to manage HIV/AIDS, its chronic intake could result in lowering of cholesterol levels in the patients. Significant decrease in cholesterol level could have implications for the normal function of several parts of the human body due to the requirement of cholesterol for several functions, including brain and nerve function and synthesis of hormones and cell membranes. 30 Increased levels of triglycerides were observed after treatment of the rats with the medicines, KT, KHB, and KBA while a decreased level was found in CHES. The results suggest that these medicines have potential to alter lipid profile. High levels of triglycerides and cholesterol indicate high risk of atherosclerosis or coronary artery disease. 31 –33 Effect of the plant medicines on kidney function was assessed by measuring rat serum creatinine levels after treatment. Generally, levels of serum creatinine were not significantly changed after the treatment, except in KT- and KHB-treated rats where elevated levels were found. Elevated creatinine level indicates damage to the kidneys. 34 Thus, our results suggest that ingestion of KT and KHB adversely affected the kidneys of the rats. However, unlike KHB, an anti-TB agent which is more likely to be used for a prolonged period, KT is used in treating malaria with a shorter term of treatment, therefore the risk may be limited. To determine the effect of the medicines on glucose metabolism, random serum glucose levels were measured after treatment. Increase in the serum glucose level was found in KT-treated rats, whereas glucose levels were decreased in WAB-, KHB-, and TF-treated rats. The results suggest the potential of WAB, KHB, and TF to cause hypoglycemia, while KT could cause hyperglycemia when ingested.
The hematological parameters were assessed in this study to determine the effect of the medicines on blood and blood-forming tissues. Generally, most of the medicines tested did not significantly alter the hematological parameters tested, except KT and KBA, which reduced levels of WBC, RBC, HB, and HCT. The MCHC was rather enhanced after treatment with KBA. Treatment with FEV increased the WBC counts while treatment with KHB resulted in a decrease in the count. White blood cells play a major role in cellular immunity by forming antibodies in response to antigens or foreign substances in the body. Thus, increase in WBC count caused by FEV suggests the potential of the plant medicine to boost the immune system. 35 Significant lowering of WBC counts found after treatment with the medicines could be due to their toxic effects. This also suggests their potential to affect the immune system adversely by reducing the ability to decrease inflammation, fight off invading infections and diseases, 35,36 especially if taken over a prolonged period. Decreases in the hematological parameters by KT and KBA also suggest that intake of these medicines may result in anemia. 36
Conclusion
Our study has shown that some common plant medicines used to treat/manage malaria, TB, and HIV/AIDS in Ghana, have the potential to cause CYP-mediated drug interactions. Since the anti-TB and anti-HIV medicines are usually taken over prolonged periods, caution must be taken when the medicines are co-administered with other drugs that are metabolized by CYP enzymes that the plant medicines modulate. Changes in biochemical and hematological parameters of the rats caused by some of the plant medicines suggest that chronic use of those plant medicines could result in adverse effects. It is recommended that future studies explore the clinical relevance of the potential herb-drug interactions and adverse biochemical and hematological parameters reported for some of the medicines in this study.
Footnotes
Authors’ Note
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgments
We thank Adom Herbal Clinic, Top Herbal Clinic, and Amen Scientific Hospital for providing the plant medicines used in work. We also appreciate the technical support of Mr F. Attigah, Department of Clinical Pathology and Messrs M. Quartey and D. Appiah both of Department of Animal Experimentation, Noguchi Memorial Institute for Medical Research.
Author Contributions
RAO conceived the study, participated in animal study, data analysis, and drafted the manuscript. IT participated in animal study, in vitro assays, and data analysis. EO-A, AA, and PA participated in animal study and in vitro assays. EA-D conducted the biochemical and hematological assays. SA participated in the animal study and drafting of the manuscript. BA participated in the animal study. AKN participated in the study design and manuscript writing. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by Third World Academy of Sciences (TWAS) Re-entry grant, Trieste, Italy (10-179RG/CHE/AF/AC_G–UNESCO FR 3240245989) and Noguchi Memorial Institute for Medical Research, Legon, Ghana.
Ethical Approval
Ethical clearance for the study was obtained from the Institutional Review Board of the Noguchi Memorial Institute for Medical Research (NMIMR-IRB CPN 020/11-12), University of Ghana, Legon, Ghana.
