Abstract
Proteins and lipids represent the two major constituents of biological membranes. Different organelles have different lipid compositions, which may be crucial for the execution and control of various organelle-specific functions. The interorganellar transport of lipids is dominated by mechanisms that are distinct from the vesicular mechanisms that underlie the interorganellar transport of proteins. Lipid transfer proteins (LTPs) efficiently and accurately mediate the trafficking of membrane lipids at the interfaces between different organelles. In this review, which focuses on sphingolipids, we describe the coordinated synthesis and transfer of lipids that occur at the endoplasmic reticulum (ER)-Golgi apparatus contact zones and discuss the impacts of lipid metabolism on membrane trafficking from the trans-Golgi network (TGN).
Abbreviations
ACBD3: acyl-coenzyme A binding domain containing 3; ARF1: ADP-ribosylation factor 1; CERT: ceramide transfer protein; CKI: casein kinase I; DAG: diacylglycerol; ER: endoplasmic reticulum; FAPP: four-phosphate adaptor protein; FFAT: two phenylalanines in an acidic tract; GlcCer: glucosylceramide; LacCer: lactosylceramide; LTP: lipid transfer protein; MOSPD2: motile sperm domain-containing protein 2; MSP: major sperm protein; Nir2: PYK2 N-terminal domain-interacting receptor 2; OSBP: oxysterol binding protein; ORP: OSBP-related protein; PH: pleckstrin homology; PI4K: phosphatidylinositol 4-kinase; PKC: protein kinase C; PKD: protein kinase D; PM: plasma membrane; PPM1L: protein phosphatase Mg2+/Mn2+-dependent 1 L; PtdIns: phosphatidylinositol; PtdIns(4)P: phosphatidylinositol 4-monophosphate; PtdSer: phosphatidylserine; SM: sphingomyelin; SRM: serine-repeat motif; TGN: trans-Golgi network; VAP: vesicle-associated membrane protein-associated protein
Lipids are major constituents of all cell membranes and are essential for the structure and function of membranes. Lipids not only serve as the hydrophobic matrix for cellular membranes but also play various roles, including acting as functional modulators of membrane proteins, transmembrane signaling molecules, and energy sources (van Meer et al., 2008; Dowhan et al., 2016).
Three classes of lipids comprise the major lipid constituents of biomembranes: glycerophospholipids, sphingolipids, and sterols. Glycerophospholipids feature a diacylglycerol (DAG) backbone and are widely present in prokaryotic and eukaryotic cells (Figure 1A) (Ridgway, 2016; Zhang and Rock, 2016). The most abundant glycerophospholipid in eukaryotes is phosphatidylcholine (PtdCho), which comprises up to 50% of the total phospholipid content (van Meer et al., 2008; Dowhan et al., 2016). Sphingolipids represent another class of lipids, featuring a sphingoid base backbone (Figure 1A). In contrast to glycerophospholipids, sphingolipids are ubiquitous in eukaryotes but are utilized by limited bacterial species among prokaryotes (Sohlenkamp and Geiger, 2016). The choline-containing sphingolipid sphingomyelin (SM) is predominant in vertebrates and comprises 5%–10% of total phospholipids (Dowhan et al., 2016). Fungi and plants do not utilize SM and instead employ sphingolipids with inositol moieties (Luttgeharm et al., 2016; Megyeri et al., 2016). Finally, sterols are a class of lipids with a steroid backbone and are found primarily in eukaryotes (Figure 1A). Cholesterol is an abundant sterol species found in mammalian cells (Ikonen, 2008; Luo et al., 2020), although it has a biased intracellular distribution. In the mitochondria, nuclear envelope, and endoplasmic reticulum (ER), cholesterol comprises less than 5% of the total lipid mass, whereas the proportions of cholesterol increase along the secretory pathway, with as much as 90% of total cellular cholesterol found in the plasma membrane (PM) (Lange et al., 1989; Lange, 1991).
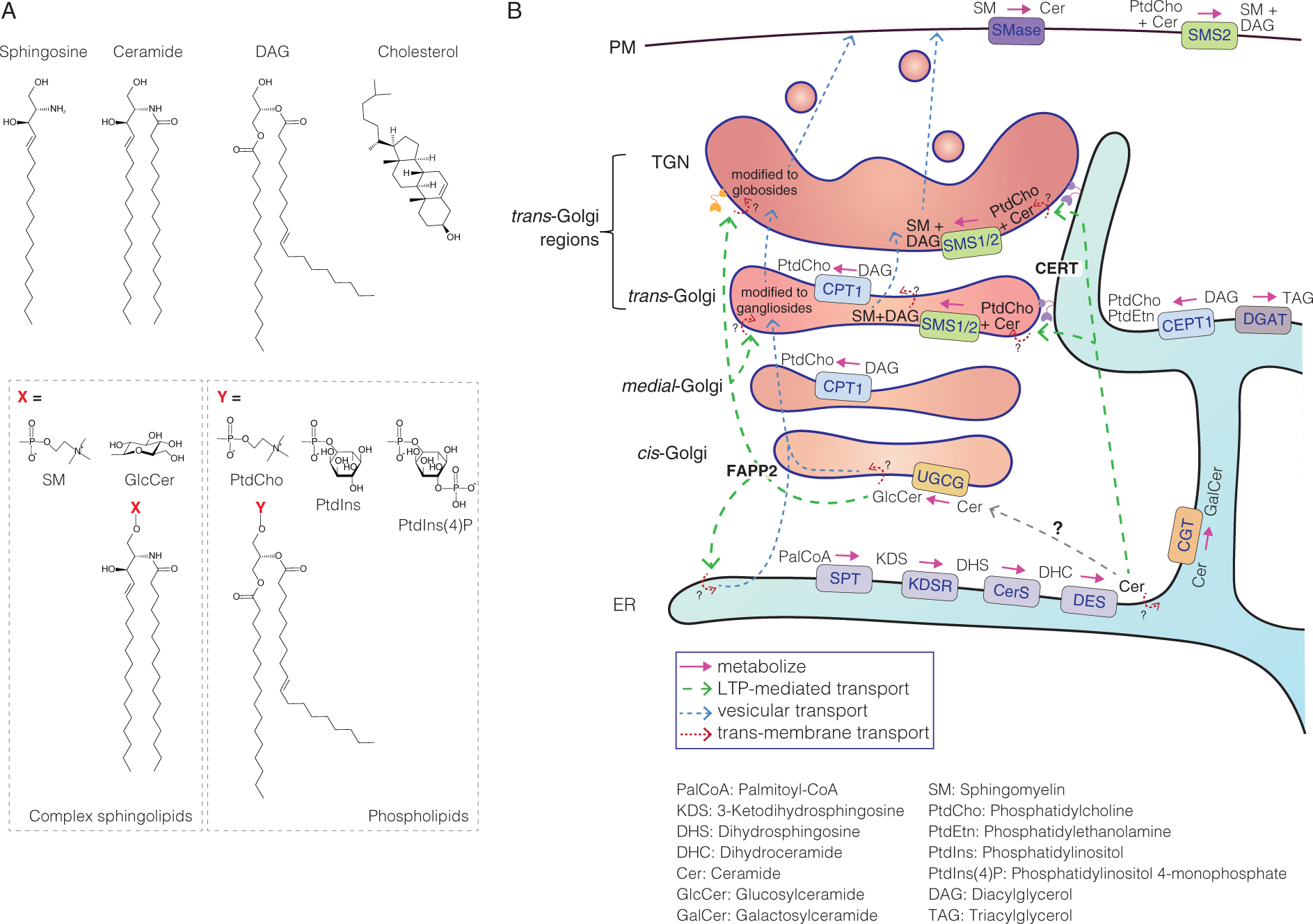
Structure and Metabolism of Mammalian Lipids.
The proportions of different lipid classes among total lipids (“lipid composition”) vary across tissue and cell types (White, 1973; Harayama and Riezman 2018). Moreover, the lipid compositions vary across cellular organelles (White, 1973; van Meer, 2008; Yang et al., 2018) (as above exemplified by cholesterol) and can even vary across nano-scale membrane domains of the same organelle (Lu and Fairn, 2018; Kusumi et al., 2020; Levental et al., 2020). Different leaflets of the lipid bilayers in a single organelle have been found to feature different lipid compositions; for example, choline-containing neutral phospholipids (i.e., PtdCho and SM) are enriched in the exoplasmic leaflet of the PM, whereas acidic phospholipids, such as phosphatidylserine (PtdSer) and phosphoinositides, are enriched in the endoplasmic leaflet (van Meer, 2008; Murate et al., 2015; Fujimoto and Parmryd, 2017). In particular, several organelles (such as the Golgi apparatus, endosomes, lysosomes, and PM) are characterized by specific phosphoinositide species, which are crucial for generating and/or maintaining these organelles (Choy et al., 2017; De Craene et al., 2017; Dickson and Hille, 2019).
The lipid composition of an organelle is determined not only by the metabolism of lipids present in each organelle but also by the transport of lipids among organelles. The interorganellar transport of lipids is dominated by mechanisms that are distinct from the vesicular mechanisms that regulate the secretory transport of proteins. Specifically, lipid-transfer proteins (LTPs) mediate the efficient and accurate transport of membrane lipids at local organelle contact zones (Chiapparino et al., 2016; Hanada, 2018). In addition, changes in the lipid compositions of biomembranes can often induce membrane deformation, which is required for certain cellular functions, including membrane trafficking. The fine-tuning of the local lipid composition, through the modulation of lipid metabolism, can provide a membrane with the potential to undergo tubulation, budding, and fusion.
This review contributes to the “ER-Golgi contact sites” review collection by focusing on the metabolism and lipid transport of sphingolipids. In particular, the ER-to-Golgi transfer of ceramide is mediated by a typical but non-redundant LTP, making this an excellent model for examining the molecular and atomic mechanisms that underlie the interorganellar trafficking of lipids at organelle contact zones (Hanada, 2014; 2018). Here, we will first describe the coordinated synthesis and transfer of lipids at ER-Golgi contact zones, in the context of sphingolipid metabolism. Next, we will illustrate the recycling cycle of lipid metabolism, coordinated by phosphatidylinositol 4-monophosphate [PtdIns(4)P] turnover, which is mediated by various LTPs, and then describe how lipid metabolism impacts membrane trafficking from the trans-Golgi network (TGN).
Sphingolipid Metabolism at the ER-Golgi Contact Zone
De Novo Synthesis and Transport of Sphingolipids in Mammalian Cells
Sphingolipids, which are characterized by their unique sphingoid base moieties, represent 10% to 20% of all eukaryotic cellular lipids. Sphingosine is the major sphingoid base found in mammalian sphingolipids (Figure 1A), although various structural subclasses of sphingoid bases exist (Merrill, 2011; Manni et al., 2018). The N-acylated form of sphingosine is referred to as ceramide (for a wider definition, “ceramides” refer to all N-acylated forms of sphingoid bases, in a context-dependent manner) (Figure 1A). Ceramide is synthesized by a series of enzymatic reactions that occur in the ER (Figure 1B). First, the condensation between serine and palmitoyl-CoA generates 3-ketodihydrosphingosine, which is reduced to dihydrosphingosine. Dihydrosphingosine is N-acylated, to generate dihydroceramide, which is desaturated to produce ceramide. These reactions occur on the cytosolic side of the ER. Ceramide can then be converted into three different types of complex sphingolipids (Figure 1B). After being transported to the trans cisterna of the Golgi apparatus and the TGN (these regions are hereafter cumulatively referred to as trans-Golgi regions), ceramide is converted into SM. Ceramide that is transported to the cis-Golgi region is likely converted into glucosylceramide (GlcCer). GlcCer is then metabolized into more complex glycosphingolipids in the trans-Golgi regions. Ceramide is also converted into galactosylceramide (GalCer), on the luminal side of the ER, and GalCer can be sulfated to produce sulfatide, in several cell types. Intermembrane LTPs for ceramide [i.e., ceramide transfer protein (CERT)] and GlcCer [i.e., four-phosphate adaptor protein 2 (FAPP2)] have been identified.
Identification and Evolutionary Aspects of CERT
CERT was identified by a forward genetic approach. Among several Chinese hamster ovary (CHO) cell variants that are resistant to the SM-binding cytolysin lysenin, a variant, named LY-A, was found to be defective in the transport of ceramide from the ER to the Golgi apparatus, without causing any discernible deficiency in ER-to-Golgi protein transport Figure 1B; Hanada et al., 1998; Fukasawa et al., 1999). By performing a functional rescue screen, using a human cDNA expression library, a cDNA was identified whose expression was able to induce the phenotypic reversion of LY-A cells to that of wild-type cells. The protein product of this cDNA was named CERT (Hanada et al., 2003). The endogenous CERT gene in LY-A cells was demonstrated to contain a missense mutation, which disrupted the PtdIns(4)P-binding activity of CERT (see below) (Hanada et al., 2003). The importance of CERT for the de novo synthesis of SM was later confirmed in both model animals and human cultured cells by performing reverse genetic experiments (Rao et al., 2007; Wang et al., 2009; Yamaji and Hanada, 2014).
The Human Genome Organization (HUGO) Gene Nomenclature Committee has recently determined CERT1 to be the official symbol of the gene encoding CERT, although several alternative names have been used for this gene. CERT1 is unique, as no close isoform of CERT1 exists in the human genome. Human CERT1 produces at least two protein isoforms, encoded by different splicing transcripts. CERT (also known as GPBPΔ26/STARD11) consists of 598 amino acid residues is the most abundantly and ubiquitously expressed CERT1 isoform found in humans (Raya et al., 2000). The other isoform, CERTL (also known as GPBP), consists of 624 amino acid residues, due to an additional 26-amino acid fragment, just upstream of the steroidogenic acute regulatory protein-related lipid transfer (START) domain in CERT. CERTL/GPBP is expressed in several tissue types, including skeletal muscle and brain (Raya et al., 2000). CERTL and CERT have both been shown to mediate the ER-to-Golgi transport of ceramide in cells (Hanada et al., 2003). In contrast, CERTL/GPBP, but not CERT/GPBPΔ26, was reported to be secreted from cells, by a non-conventional mechanism (Revert et al., 2008). Other rare CERT isoforms may also exist. A previous study suggested the existence of another, longer isoform, consisting of 715 amino acid residues, that is translated from a non-canonical initiation ACG codon (Revert et al., 2008), and the current human genome assembly (Genome Reference Consortium Human Build 38 patch release 13; GRCh38.p13) predicts that a very long isoform, consisting of 752 amino acid residues, may be translated from the first ATG codon, found in the putative first exon of CERT1. However, whether these latter two isoforms are actually produced remains uncertain because their expressions have not yet been verified (Human BodyMap 2.0, 2011). Intracellular, endogenous CERT and CERTL, but not the putative longer isoforms, have been detected in HeLa cells by western blotting with anti-CERT antibodies (Yamaji and Hanada, 2014).
Although oxysterol-binding protein (OSBP)-related family proteins have been identified in a range of organisms, from single-cell eukaryotes to humans, no functional homolog of CERT has been identified in single-cell eukaryotes, thus far. Phylogenetic analysis suggested that CERT may have co-evolved with SM (Hanada, 2014). Although the yeast species Saccharomyces cerevisiae does not contain SM or CERT, ceramide (more precisely phytoceramide) is transported from the ER to the Golgi apparatus in yeast, where it is converted into inositolphosphoceramide, a complex sphingolipid found in yeast cells (Dickson, 2008; Funato et al., 2020). In yeast cells, vesicular transport(s), which is regulated by OSBP homolog (Osh) proteins, has been suggested to represent the major pathway for the transfer of ceramide between the ER and the Golgi apparatus, under normal culture conditions (Kajiwara et al., 2014). In addition, Nvj2, which contains an SMP domain as a lipid transfer domain, may also be involved in the ER to medial-Golgi ceramide transfer process under ER stress conditions (Liu et al., 2017).
Structure of CERT and Its Interacting Partners
CERT contains several functional domains and motifs (Figure 2A): an N-terminal pleckstrin homology (PH) domain, which preferentially binds PtdIns(4)P; a serine-repeat motif (SRM); a FFAT (two phenylalanines in an acidic tract) motif, which binds vesicle-associated membrane protein-associated protein (VAP); and a C-terminal START domain, which effectively encloses and transfers ceramide (Hanada et al., 2003; Kawano et al., 2006; Kumagai et al., 2007). Notably, several other proteins that act at the ER-Golgi interface (i.e., OSBP, FAPP1, and FAPP2) also contain Golgi-targeting PH domains with demonstrated PtdIns(4)P-binding activity (Figure 2A) (Wong et al., 2019). Although the PH domains found in OSBP and FAPPs bind and require both PtdIns(4)P and the small GTPase ADP-ribosylation factor 1 (ARF1) to be properly recruited to the trans-Golgi regions, the CERT PH domain does not bind nor require ARF1 (Levine and Munro, 2002; Godi et al., 2004). Instead, biochemical and structural analyses have identified a weak affinity for the CERT PH domain towards acidic lipids, such as PtdSer (Sugiki et al., 2012), suggesting its suitability for interactions with the anionic surface of the Golgi membrane.
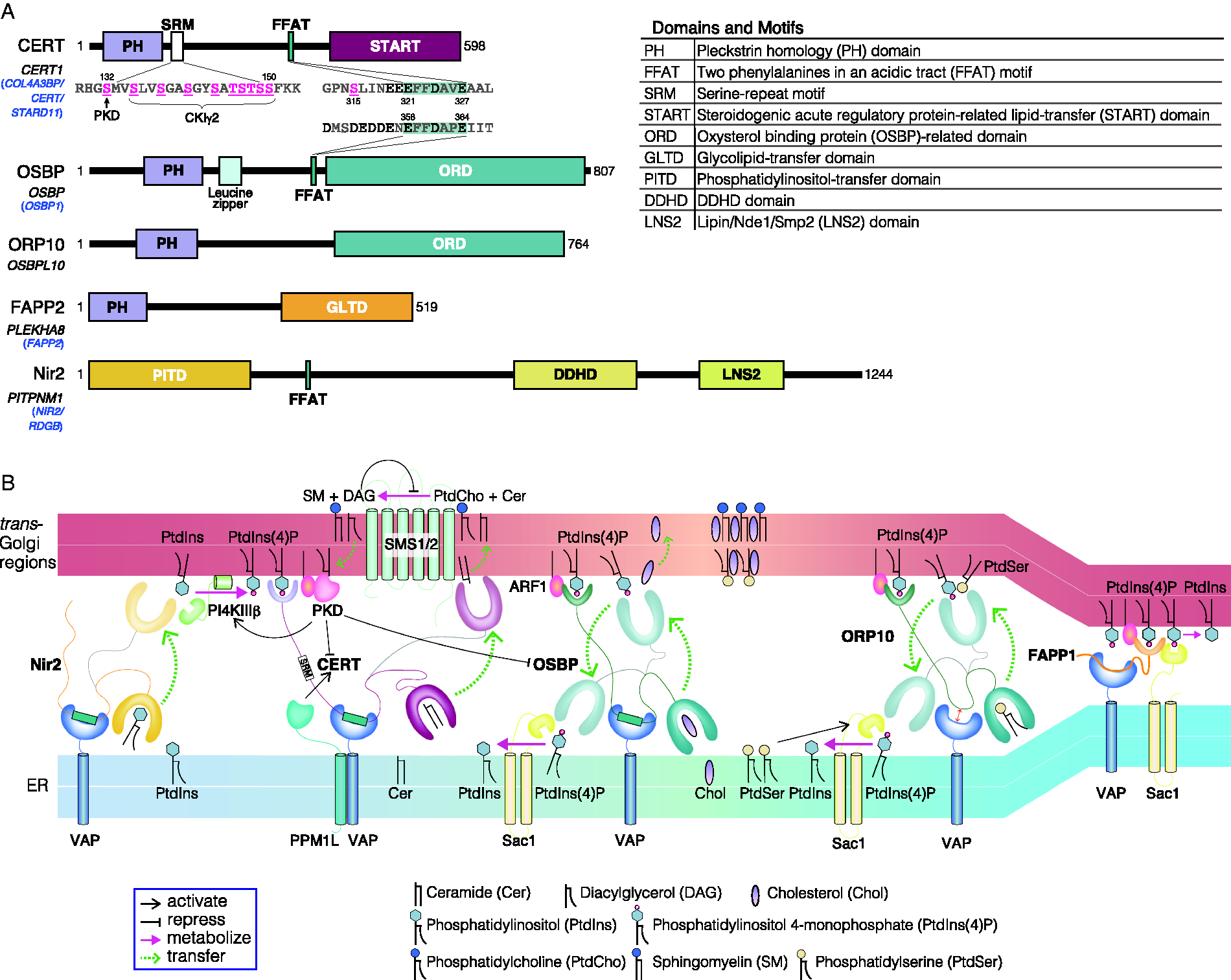
LTPs and Their Functional Coordination at ER-Golgi Contact Zones.
Various LTPs contain FFAT or FFAT-like motifs, which enable anchoring to the ER through interactions with ER-resident VAP proteins. VAP proteins contain a major sperm protein (MSP) domain and a C-terminal transmembrane domain and are widely conserved in eukaryotes. The MSP domain of VAPs can bind numerous non-ER proteins that contain FFAT or FFAT-like motifs, mediating communications between the ER and various organelles (Murphy and Levine, 2016). The human genome encodes two VAP isoforms, VAP-A and VAP-B, both of which are ubiquitously expressed in various tissues. Another ER-anchored protein, motile sperm domain-containing protein 2 (MOSPD2), also contains an MSP domain and was identified as a novel FFAT-binding protein (Di Mattia et al., 2018). MOSPD2 has also been shown to interact with CERT in an MSP domain- and FFAT motif-dependent manner. MOSPD2 is characterized by the presence of a CRAL-TRIO domain, which is a lipid transfer domain, although the ligand(s) and physiological functions of CRAL-TRIO domain in MOSPD2 remain unsolved.
Post-Translational Regulation of CERT
The function of CERT is regulated by phosphorylation in at least two different contexts (Kumagai and Hanada, 2019). The hyperphosphorylation of serine and threonine residues in the SRM downregulates CERT function. The inhibition of SM de novo synthesis and decreased cholesterol or SM contents in the PM can affect the phosphorylation status of the CERT SRM (Kumagai et al., 2007). This finding may indicate that the cross-regulation of cholesterol and SM levels is crucial for sustaining the biophysical properties of the PM, although the underlying mechanisms of this relationship are unclear. Serine 132 (S132), the most N-terminal serine in the SRM, is phosphorylated by protein kinase D (PKD), followed by the sequential phosphorylation of downstream serine and threonine residues by casein kinase I γ2 (CKIγ2) (Fugmann et al., 2007; Kumagai et al., 2007; Tomishige et al., 2009). The hyperphosphorylation of the SRM can result in the simultaneous downregulation of the activities of both the PH and START domains of CERT. This is presumably due to a conformational change in CERT that is associated with an interaction between the PH and START domains, which likely mutually masks these two domains (Kumagai et al., 2007; Prashek et al., 2017). Solution-based NMR analysis has demonstrated that the hyperphosphorylated SRM interacts with the PH domain, which partially compromises the PtdIns(4)P-binding activity of the PH domain (Sugiki et al., 2018).
The protein phosphatase Mg2+/Mn2+-dependent 1 L (PPM1L, also known as protein phosphatase 2Cε) is involved in the dephosphorylation of the CERT SRM (Saito et al., 2008). PPM1L is an ER-resident protein that interacts with VAP-A (Saito et al., 2008) and the Golgi-resident protein ACBD3 (acyl-coenzyme A binding domain containing 3) (Shinoda et al., 2012). ACBD3 also binds and recruits phosphatidylinositol 4-kinase III β (PI4KIIIβ) to the TGN (Shinoda et al., 2012), where PI4KIIIβ synthesizes PtdIns(4)P (Sasaki et al., 2012). When recruited to the ER by binding VAP, CERT may become dephosphorylated by PPM1L, which may cause a conformational change that facilitates an interaction with PtdIns(4)P, generated by PI4KIIIβ.
Another phosphorylation site that has been identified in CERT is serine 315 (S315), which is just upstream of the FFAT motif (Olsen et al., 2006). The phosphorylation of S315 increases the affinity of CERT for VAP, which enhances the ER-to-Golgi trafficking of ceramide (Kumagai et al., 2014). The phosphorylation of S315 extends the acidic stretch at the CERT FFAT motif, which likely enhances the affinity of CERT for VAP. Interestingly, the amino acid sequence upstream of the FFAT motif in OSBP is extensively acidic and is likely to have a higher affinity for VAP than CERT that has not been phosphorylated at S315 [because an interaction of VAP with OSBP but not with CERT was reported in an interactome study (Huttlin et al., 2017)] (Figure 2A). The specific kinase(s) and phosphatase(s) that are responsible for the phosphorylation status of CERT S315 remain unidentified.
CERT can oligomerize into a homo-trimer or even higher oligomers (Revert et al., 2018), although whether oligomerization affects CERT function remains unclear. The oligomerization of CERT may strengthen the association between CERT and target organelle membranes, via organelle-binding modules (i.e., the PH domain and the Golgi apparatus or the FFAT motif and the ER), which may act to stabilize a CERT-mediated ceramide transfer zone at the ER-Golgi interface. OSBP dimerization is dependent on the region located between the PH domain and the FFAT motif of the protein (Ridgway et al., 1992; Jamecna et al., 2019). An in vitro analysis has suggested that OSBP dimers dissociate more slowly from PtdIns(4)P-containing liposomes than OSBP monomers (Jamecna et al., 2019).
FAPP2 Mediated GlcCer Transport
GlcCer synthase (also known as UDP-glucose ceramide glucosyltransferase) is encoded by UGCG in the human genome and localizes to the cis-Golgi region, with its catalytic site oriented to the cytosolic side (Halter et al., 2007). The synthesis of GlcCer is independent of CERT (Hanada et al., 2003; Yamaji and Hanada, 2015), although how ceramide synthesized in the ER is delivered to the cis-Golgi region remains unclear. After synthesis, GlcCer is delivered to more distal Golgi region(s), where it can be converted into lactosylceramide (LacCer). The intra-Golgi transport of GlcCer is mediated by FAPP2, a member of the glycolipid-transfer protein family (Figure 1B) (D’Angelo et al., 2007; 2013). An alternative model has also been proposed, in which GlcCer is transported to the ER, in a FAPP2-dependent manner, and then GlcCer is delivered from the ER to the Golgi apparatus, in a vesicle-dependent manner (Halter et al., 2007).
FAPP2 contains a PtdIns(4)P-preferential PH domain, which interacts with PtdIns(4)P and ARF1 (Godi et al., 2004). ARF1 has been suggested to stimulate the association between FAPP2 and the cis-Golgi region, allowing FAPP2 to capture its lipid ligand GlcCer (D’Angelo et al., 2013). After enclosing GlcCer in the lipid-binding cavity, FAPP2 is thought to undergo a conformational change, which may enhance its affinity for PtdIns(4)P and promote the subsequent translocation of FAPP2 to the PtdIns(4)P-abundant TGN. ACBD3 has been suggested to directly interact with FAPP2 to enhance site-specific GlcCer transport (Liao et al., 2019).
The catalytic glycosyltransferases sites required for the synthesis of LacCer and more complex glycosphingolipids are oriented towards the lumen of the trans-Golgi regions. Thus, GlcCer must transfer across the organelle membrane to be converted into LacCer, although the entity that mediates this transmembrane movement of GlcCer has not yet been identified. A type of ABC transporters can catalyze the transmembrane transport of short-chain GlcCer, such as C6-NBD-GlcCer (van Meer et al., 2006), but not natural GlcCer species (Halter et al., 2007). A type of P4-ATPases has also been demonstrated to mediate the transmembrane transport of GlcCer at the PM (Roland et al., 2019), but the exoplasmic side-to-cytosolic side transport of GlcCer by P4-ATPase occurs in the topologically opposite direction necessary for GlcCer conversion into LacCer in the lumen of the Golgi apparatus.
Impacts of Lipid Metabolism at the ER-Golgi Contact Zone on Membrane Trafficking
Concerted Regulation of Lipid Metabolism at the ER-Golgi Contact Zone
DAG, which is formed simultaneously during SM synthesis, can affect lipid metabolism at the ER-Golgi contact zone, both directly and indirectly (Figure 2B). For instance, DAG directly inhibits SM synthase activity (Hanada et al., 1991), whereas DAG (in concert with ARF1) indirectly affects the activities of multiple protein factors that are involved in lipid metabolism, through the recruitment of PKD to the Golgi apparatus (Baron and Malhotra, 2002; Pusapati et al., 2010). The recruitment of PKD to the Golgi triggers a recycling cycle of lipid metabolism, through the comprehensive regulation of LTPs, which is coordinated with PtdIns(4)P turnover, as described below.
PKD phosphorylates PI4KIIIβ to activate it (Figure 2B) (Hausser et al., 2005), and the activated PI4KIIIβ generates PtdIns(4)P, which, in turn, promotes the translocation of PtdIns(4)P effectors (e.g., CERT and OSBP) to the trans-Golgi regions. ARF1, which plays a key role in the recruitment of transport vesicle-forming factors to the Golgi apparatus (Donaldson and Jackson, 2011; Tan and Gleeson, 2019), is also involved in the recruitment of PI4KIIIβ to the Golgi apparatus (Godi et al., 1999). A clathrin adaptor GGA2 (Golgi-localized, gamma-adaptin ear homology, ARF-binding), which requires the GTP-bound form of ARF1 and PtdIns(4)P for TGN localization, has been suggested to bind and recruit PI4KIIIβ (Daboussi et al., 2017).
OSBP and some of its related proteins can recognize not only sterol but also PtdIns(4)P, catalyzing the sterol/PtdIns(4)P exchange between the ER and Golgi (Figure 2B), although these sterol-transfer proteins are capable of mediating the intermembrane transfer of sterol without the counterflow partner PtdIns(4)P (de Saint-Jean et al., 2011; Mesmin et al., 2013). Upon arrival at the ER, through the OSBP-mediated exchange reaction, PtdIns(4)P is degraded into PtdIns, by the PtdIns(4)P phosphatase Sac1 (Wakana et al., 2015; Zewe et al., 2018), and PtdIns is then transported from the ER to the trans-Golgi regions, presumably by the PtdIns-transfer protein Nir2 (PYK2 N-terminal domain-interacting receptor 2) along with the concentration gradient of PtdIns (Peretti et al., 2008).
Nir2 affects DAG level of the Golgi apparatus by regulation of DAG consumption through the CDP-choline pathway during PtdCho synthesis (Litvak et al., 2005). At the Golgi membrane, PtdIns is phosphorylated by PI4KIIIβ, at the expense of ATP. The functionally interconnected system, consisting of OSBP, Sac1, PI4KIIIβ, and Nir2, at the ER-Golgi contact zone can drive the transfer of cholesterol against its concentration gradient, from the ER to the Golgi apparatus, at the expense of ATP. The CERT-dependent movement of ceramide occurs from the ER to the Golgi apparatus, along a ceramide concentration gradient, because ceramide synthesized in the ER is converted to SM upon its arrival at the trans-Golgi regions. The ER-resident protein Sac1 is also capable of dephosphorylating the PtdIns(4)P embedded in the Golgi membrane at the ER-Golgi contact zone. FAPP1 at the trans-Golgi region senses increased PtdIns(4)P and forms a complex with Sac1 and VAP, both of which are integral ER membrane proteins, which renders the catalytic site of Sac1 accessible to Golgi-embedded PtdIns(4)P (Figure 2B) (Venditti et al., 2019). Sac1 is predicted to feature a linker of up to 7-nm long (Gatta and Levine, 2017), and FAPP1-mediated contact is estimated to be narrower than OSBP-mediated contact, allowing Sac1 to act between the opposing organelles at the contact zone. Although whether OSBP and FAPP1 mediate PtdIns(4)P turnover at different zones of the trans-Golgi regions remains unclear, the reduced affinity of the FAPP1 PH domain for PtdIns(4)P, compared with that of OSBP (Levine and Munro, 2002), implies that FAPP1 is recruited to a limited trans-Golgi region, with a particularly high concentration of PtdIns(4)P.
A reduction in the Golgi PtdIns(4)P level impairs the recruitment of CERT to the trans-Golgi regions, and, as a consequence, the ER-to-Golgi transport of ceramide and the de novo synthesis of SM become downregulated. The PKD-mediated phosphorylation of CERT also inhibits ceramide transfer, causing a structural change, as described above (Kumagai et al., 2007). In contrast, PKD phosphorylation inhibits the trans-Golgi-localization of OSBP (Nhek et al., 2010). Notably, the PKD-dependent phosphorylation of OSBP does not affect the cholesterol-binding/transfer abilities or the PtdIns(4)P-binding abilities of OSBP (Nhek et al., 2010; Goto et al., 2018), and PKD-independent phosphorylation can modulate the function of OSBP, in response to alterations in cellular cholesterol and SM levels (Goto et al., 2012). The PKD-mediated phosphorylation of CERT and OSBP is likely involved in the coordinated feedback regulation mechanism of the SM synthesis pathway.
The functions of CERT and SM synthase appear to be finely controlled in cells, through several feedback pathways. After being transported to the trans-Golgi regions by active CERT, ceramide is converted to SM, producing the by-product DAG, which acts as a feedback inhibitor of SM synthase and an activator of PKD. As described above, activated PKD triggers the PI4KIIIβ-dependent production of PtdIns(4)P in the trans-Golgi regions, which upregulates the function of CERT. In parallel, activated PKD downregulates the function of CERT through CERT S132 phosphorylation, which represses DAG production and SM synthesis. Meanwhile, the cholesterol/PtdIns(4)P exchange mediated by OSBP and its related proteins also affects the Golgi PtdIns(4)P levels, which, in turn, affects the recruitment of CERT to the trans-Golgi regions. In contrast, because SM is prone to form domains with cholesterol in phospholipid membranes (Radhakrishnan et al., 2000; Engberg et al., 2016), the synthesis of SM in the Golgi apparatus may also facilitate the Golgi-oriented movement of cholesterol. Collectively, given that the functions of these enzymes and transport proteins are either positively or negatively modulated by their metabolites or lipid ligands, both directly and indirectly, at the ER-Golgi contact zone (Figure 2B), the overall system may serve as a rheostat, to maintain the levels of various lipids (i.e., SM, DAG, PtdIns(4)P, and cholesterol) within appropriate ranges in the ER and Golgi apparatus.
Golgi PtdIns(4)P Pools Are Modulated by Multiple LTPs at the ER-TGN Contact Zone
As described above, OSBP is capable of bridging ER and Golgi membranes, utilizing its dual membrane-targeting domains, and modulates the Golgi PtdIns(4)P pool. In addition to OSBP, a recent study identified the OSBP-related proteins (ORPs) ORP10 and ORP9 (in combination with OSBP) as stabilizers of the ER-Golgi contact zone (Venditti et al., 2019).
ORP10 transfers PtdSer from the ER, where PtdSer is de novo synthesized, to the trans-Golgi regions, in place of transferring cholesterol (Figure 2B) (Venditti et al., 2019). In addition to the tethering function of ORP10, which binds both the ER and the trans-Golgi regions, the PtdSer-transfer activity of ORP10 also stabilizes the ER-Golgi contact zone. Altered PtdSer synthesis affects PtdIns(4)P levels, at both the PM and the Golgi (Sohn et al., 2016). A mutation in PtdSer synthase (encoded by PTDSS1) renders the synthase constitutively active (Kuge et al., 1998), and the overproduction of PtdSer enhances Sac1 activity, through an unknown mechanism, and triggers a reduction in PtdIns(4)P levels at both the PM and the Golgi. This effect implies the presence of a PtdSer-dependent mechanism that coordinates PtdIns(4)P levels between the PM and the Golgi.
Although OSBP and ORP9 are cholesterol-transfer proteins that act at ER-Golgi contact zones, their functions and regulatory systems appear to be different. OSBP is involved in the regulation of SM synthesis, whereas ORP9 has been suggested to transfer cholesterol from the TGN to the ER (Ngo and Ridgway, 2009). OSBP recruitment to the Golgi apparatus is affected by PI4KIIIβ and cholesterol, whereas ORP9 recruitment is not (Ngo and Ridgway, 2009). Although OSBP and ORP9 have analogous activities (i.e., the extraction/transfer of sterols and PtdIns(4)P, and ER-TGN membrane tethering activities, through interactions with PtdIns(4)P and VAPs), these two LTPs may interact with different PtdIns(4)P pools or other lipid effectors, contributing to the formation of contacts in different sub-regions of the trans-Golgi regions (Ngo and Ridgway, 2009; Liu and Ridgway, 2014).
Lipid Metabolism Affects Cargo Sorting
Mature and active proteins that reach the TGN must be promptly and correctly exported to their sites of action (De Matteis and Luini, 2011; Di Martino et al., 2019). PKD and DAG, both of which modulate PtdIns(4)P production, are crucial for the formation of membrane vesicles and cargo sorting at the TGN (Roth, 2004; Capasso et al., 2017; Wallroth and Haucke, 2018; von Blume and Hausser, 2019). Membrane trafficking is also affected by the metabolism of other lipids. For instance, the level of DAG in the Golgi apparatus is affected by the synthesis of SM (which generates an equimolar amount of DAG as a by-product) in the trans-Golgi regions. Here, we summarize the sorting mechanisms in which the above-mentioned regulators of lipid synthesis and/or transfer are involved.
Cargoes from the TGN are sorted to at least three major destinations: apical, basolateral, and lysosomal membranes. PKD regulates basolateral sorting (Yeaman et al., 2004). The recruitment and activation of PKD at the TGN are mediated by a signaling cascade, which includes the protein G β-γ dimer, phospholipase C (PLC), and protein kinase C (PKC). A G-protein-coupled receptor has been shown to mediate PKD activation via the PLC/PKC signaling pathway (Xu et al., 2015). The inactivation of PKC results in the retention of cargo at tubules that elongate from the TGN membrane, and PKD activation is essential for the normal fission of basolateral trafficking vesicles from the TGN (Liljedahl et al., 2001; Yeaman et al., 2004; Bossard et al., 2007).
As earlier mentioned, PI4KIIIβ is an effector of PKD. The ER-TGN contact zone dynamics, which are modulated by PtdIns(4)P metabolism, couple the LTP-mediated trafficking of lipids with the sorting of certain cargos at the TGN. For example, the disruption of ER-Golgi contact zones by VAP depletion decreases TGN DAG levels and impairs the formation of a subtype of TGN-to-PM secretory vesicles (Wakana et al., 2015). Moreover, the FAPP1-Sac1-VAP-dependent consumption of PtdIns(4)P at the TGN downregulates the transport of ApoB100 secretion but does not affect the secretion of albumin and α1-antitrypsin (Venditti et al., 2019), suggesting that a specific set of cargoes is regulated by PKD signaling.
Perspectives
Organelle contacts have recently gathered increasing attention not only in basic cell biological research but also in terms of medical research, including human diseases that are associated with organelle contacts (Dumoux and Hayward, 2016; Strating and van Kuppeveld, 2017; Peretti et al., 2019; Prinz et al., 2020). The accurate spatiotemporal regulation of lipid-transfer at organelle contact zones is vitally important. The artificial anchoring of CERT to the mitochondria induced Bax-dependent apoptosis (Jain et al., 2017), and specific amino acid substitutions in the SRM, which are expected to render CERT constitutively active, have been associated with intellectual disability in humans (Fitzgerald et al., 2015; Lelieveld et al., 2017).
Lipid metabolism at the ER-Golgi contact zone, associated with LTP-mediated lipid transport, impacts membrane traffic, revealing its crucial role in cellular fitness. However, several issues remain unresolved, including the physiological significance of CERT oligomerization, and several factors crucial for the organization of the ER-Golgi contact zone have not yet been identified. Furthermore, whether “ER-Golgi contact sites” can be sub-classified into diverse functional zones remains an important open question that should be addressed in the future. The clarification of the functions of specific interorganellar dynamic contact regions should further deepen our understanding of the cell systems, which can contribute to further applications, such as drug discovery.
Footnotes
Acknowledgments
The authors would like to thank Drs. Keigo Kumagai and Shota Sakai (Department of Biochemistry and Cell Biology, National Institute of Infectious Diseases) for their helpful discussions on lipid and organelle biology.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclose the receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the MEXT KAKENHI (No. JP17H06417) to K. H. and AMED-CREST (No. JP20gm0910005j0006) to K. H.
