Abstract
Pioneering work in the 1990s started to address an interesting question. How is the main cellular energy source, adenosine triphosphate (ATP), imported into the mammalian endoplasmic reticulum (ER)? Despite its high-energy demand, large volume, and structural as well as functional complexity, the ER harbors no intricate system for ATP synthesis or regeneration. Although the original biochemical reconstitution approaches established hallmarks of the ATP transport into the ER including nucleotide selectivity, affinity, and antiport mode, the more recent live-cell imaging methods employing sensitive, localized molecular probes identified the elusive ATP/adenosine diphosphate (ADP) exchanger. According to its selectivity and localization, the identified SLC35B1 protein was rebranded AXER. Here, we discuss the identification and regulation of AXER plus the cytosolic partners (AMP-activated protein kinase, AMPK) and subcellular structures (mitochondrial–ER contact sites, MERCs) acting in concert with it to orchestrate energy homeostasis of the mammalian ER. Furthermore, we combine the two seemingly controversial regulatory mechanisms (lowER and CaATiER) in a unifying hypothesis.
Cells are notorious rebels. Designed as open systems that exchange matter and energy with their surroundings cells do not adhere to the second law of thermodynamics. Instead, cells decrease their internal entropy by generating disorder in the environment and disturbing the equilibrium. To do so, their method of choice is called metabolism (evolving from the Greek word metabolismos meaning change or overthrow). To generate and maintain complexity, cells couple energy liberating catabolic processes with energy demanding anabolic reactions and synthesize biomass. Thus, cellular metabolism maintains internal order by creating external disorder (Demirel and Sandler, 2002).
One of the most crucial metabolites that connect anabolic and catabolic reactions is adenosine triphosphate, ATP. This compound harnesses and releasably stores free energy from catabolic oxidation processes in its high-energy phosphoanhydride bonds. Although other metabolites such as adenosine diphosphate (ADP), phosphoenolpyruvate, carbamoyl-phosphate, 2,3-bisphospho-glycerate, or phosphocreatine also carry a high-energy phosphate moiety, none is used as versatile as ATP. ATP drives many fundamental cellular reactions including nucleotide and protein synthesis, cycling of ions such as Na+, K+, H+, or Ca2+ across various membranes, metabolic activity by kinases, or the motility of organelles and muscle cells. In addition, ATP can serve as purinergic signaling molecule for cell–cell communication (Bonora et al., 2012; Cavaliere et al., 2015). Despite this cornucopia of processes depending on ATP, sources of ATP synthesis are limited to substrate level phosphorylation by cytosolic glycolysis as well as the mitochondrial tricarboxylic acid (TCA) cycle (actually, one guanine triphosphate (GTP) is generated as well as three NADH/H+ and one FADH2 per cycle, i.e., Acetyl-CoA) and oxidative phosphorylation involving the ATP synthase (Bonora et al., 2012; Hahn et al., 2016; Schorr and van der Laan, 2018). Hence, in eukaryotic cells, ATP-consuming and ATP-generating processes are not always colocalized and can occur in separate organelles. For example, secretory proteins are transported in an unfolded manner across the endoplasmic reticulum (ER) membrane and can be folded with the help of ATP-consuming and ATP-dependent chaperones of the ER lumen. However, to the best of our knowledge, neither autonomous de novo ATP synthesis nor ADP phosphorylation occurs in the ER. Similarly, phosphorylation of secretory and resident proteins by kinase Fam20C in the ER as well as the nonautonomous Golgi apparatus relies on exogenous ATP (Cui et al., 2015; Pollak et al., 2018; Zhang et al., 2018; Yu et al., 2020).
Recent data from others and us provide evidence for a previously misclassified membrane protein (SLC35B1) that transports ATP into the ER as energy source for the folding process mediated by ER luminal chaperones (Klein et al., 2018; Yong et al., 2019). This transporter of the ER membrane, which exchanges ATPin and ADPout, was termed AXER and provided insights into questions about maintenance and resupply of ER energy homeostasis. Details of the AXER-mediated ATP import into the ER together with associated aspects of cellular energy homeostasis and regulatory mechanisms are the main focus of this review.
Summary of Proteins Involved in the ER Energy Homeostasis.

Note. Protein names are listed alphabetically together with their most likely location (cytosol [C], ER membrane [ERM], ER lumen [ERL]), function, abundance in HeLa cells (taken from Hein et al., 2015) as well as ATP- and Ca2+-dependence. NEF = nucleotide exchange factor.
The ER and Its Energy Requirements
The ER is a large, single-copy organelle in nucleated cells. It spans as continuous reticular network all the way from the nuclear envelope close to the periphery, which is confined by the plasma membrane. Smooth tubules and rough sheets is a catch phrase often associated with the ER and refers to both different morphological structures of the mesh-like continuum and the binding of ribosomes (Palade, 1956; Porter and Palade, 1957; Shibata et al., 2006; Friedman and Voeltz, 2011). This dynamic, shape-shifting behavior of the ER is discussed best in the light of a second ER relevant catch phrase coined by Westrate et al.’s (2015) saying, form follows function. Smooth tubules, which are devoid of associated ribosomes, take part in the synthesis and elimination of many lipophilic molecules. Enzymes of the smooth ER are involved in lipid and steroid synthesis as well as phase 1 and 2 reactions of the biotransformation process for apolar endo- and xenobiotics (Cribb et al., 2005; Lehman-McKeeman and Ruepp, 2018). However, binding of ribosomes to the ER membrane transforms smooth tubules into rough sheets, thereby, promoting protein transport across the ER membrane as well as the downstream maturation steps such as protein folding, modification, oligomerization, quality control, and degradation (Puhka et al., 2007; Barlowe, 2010; Shibata et al., 2010). Common for both the smooth and the rough ER is that their continuous lumen serves as Ca2+ reservoir within mammalian cells, that, under resting conditions, exceeds the cytosolic Ca2+ level by multiple orders of magnitude (Clapham, 2007; Feske, 2007; Sammels et al., 2010).
A detailed review of the key players of ER-associated processes and their energy requirements (cf. Table 1) was recently provided by Depaoli et al. (2019). In short, most ATP-consuming reactions associated with the ER occur on the cytosolic leaflet of the ER membrane. These include for example the ER Ca2+ ATPase (SERCA), the antigen peptide transporter 1 (TAP) with an ATP binding cassette, membrane-embedded kinases (IRE1 and PERK) of the unfolded protein response (UPR), or members of the retro-translocation process during ER-associated degradation (ERAD) such as HRD1 and the associated ATPase (Cdc48). In addition, the GTP-driven protein synthesis by ribosomes attached to the ER translocon occurs as energy-demanding process at the cytosolic leaflet of the ER membrane. On the other side, that is, within the ER lumen, ATP is required mainly for chaperone mediated processes. The chaperones in question include the ATP-consuming immunoglobulin heavy chain binding protein (BiP) and the single-pass type I membrane protein calnexin (CNX) plus the soluble protein calreticulin (CRT) both of which use ATP as a cofactor. The former two, BiP and palmitoylated CNX, can also be found in association with the translocon complex and support the protein transport of incoming precursor proteins (Dudek et al., 2009; Lakkaraju et al., 2012; Zimmermann, 2016). In addition, BiP was also identified as (a) master regulator of the UPR interacting with the signal transducers IRE1, PERK, and ATF6 (Walter and Ron, 2011; Plate and Wiseman, 2017) as well as (b) regulator of the ER Ca2+ homeostasis regulating both the passive Ca2+ efflux via the Sec61 complex directly (Schäuble et al., 2012; Schorr et al., 2015) and the Ca2+ import via SERCA indirectly, that is, in cooperation with ERdj5 (Ushioda et al., 2016). Thus, BiP represents the proverbial Swiss Army knife of the ER lumen supporting processes related to Ca2+ and protein homeostasis (Figure 1A), with the latter including protein translocation, folding, surveillance and degradation (Dudek et al., 2009; Melnyk et al., 2015). BiP’s versatility and central role in the ER will be discussed next.

Functions and Key Players of ER Homeostasis Influenced by BiP. A: The octagon represents the ER with BiP as the main Hsp70-type luminal chaperone influencing a plethora of relevant functions. As major ATP-consuming protein of the ER, BiP is linked to nucleotide homeostasis via the ATP/ADP exchanger AXER (12 o’ clock position). BiP also regulates, either as direct interaction partner or as regulating factor of additional proteins not shown in the cartoon, Ca2+ homeostasis of the ER. This includes Ca2+ influx via SERCA, Ca2+ efflux through IP3 receptor (IP3R) and its stabilizing chaperone Sigma1 receptor (Σ1R), or inter-organelle communication via MERCs, the mitochondrial-ER contacts (1–5 o’ clock position). In addition, BiP is a main regulator of processes contributing to proteostasis that include protein surveillance by the UPR sensors IRE1, PERK, and ATF6 (I/P/A), protein degradation during ERAD including one of the putative retro-translocons such as HRD1 in conjunction with the Cdc48 ATPase (not shown), protein stabilization through folding and refolding as well as protein transport via the Sec61 complex (6–11 o’ clock position). B: Multifunctionality of BiP is guaranteed by the so-called Hsp70 cycle. As shown on the right, BiP consist of an NBD and an SBD carrying a lid. The lid provides structural flexibility and variable substrate binding affinity of BiP. In the ATP-bound state the SBD is in the open conformation allowing rapid binding and release, that is, low affinity for a protein substrate. Context-specific interaction of BiP with one of the eight known Hsp40 co-chaperones (ERj1–ERj8) catalyzes ATP hydrolysis and BiP is converted into the ADP-bound form showing a reduced binding and release kinetic enabling a high substrate affinity. Subsequent substrate release is mediated by a NEF such as Grp170 or Sil1 and allows BiP to enter a new cycle. ER = endoplasmic reticulum; ATP = adenosine triphosphate; ADP = adenosine diphosphate; SBD = substrate-binding domain; NBD = nucleotide-binding domain.
BiP, a Pivotal ATP-Consuming Chaperone of the ER
To fulfill its essential role in protein biogenesis, the ER of all nucleated cells in all kingdoms of life contains a Hsp70-type molecular chaperone, HSPA5, also termed BiP or glucose-regulated protein 78 (Grp78; Haas and Wabl, 1983; Hendershot et al., 1988). Typically, BiP is present in the ER lumen in millimolar concentration and depends on both a high Ca2+ content (800 µM) and the hydrolysis of ATP (Lievremont et al., 1997; Hamman et al., 1998; Bertolotti et al., 2000; Tyedmers et al., 2003; Zahedi et al., 2009; Schäuble et al., 2012; Hein et al., 2015). As expected from a Hsp70, a flexible linker region connects the amino-terminal nucleotide-binding domain (NBD) and the carboxy-terminal substrate-binding domain (SBD), which can reversibly bind to hydrophobic peptides within polypeptides (Flynn et al., 1991; Kumar et al., 2011). In this way, binding and release of a substrate peptide are coupled to an ATPase cycle, which supports folding or conformational changes of BiP’s polypeptide substrates (Smock et al., 2010; Marcinowski et al., 2011). Typically, there are several hydrophobic peptides within polypeptides that may simultaneously or sequentially be bound by BiP. For a productive ATPase cycle, BiP is in the center of a proteinaceous tripartite system (Figure 1B), which additionally involves a J-domain carrying Hsp40-type co-chaperone (termed ERj or ERdj) and a nucleotide exchange factor (NEF). Binding of BiP to one of the multiple ERjs stimulates its ATPase activity and triggers an increase in substrate affinity. The reverse reaction is mediated by binding of BiP to an NEF (such as Sil1/BAP and Grp170/HYOU1), which triggers the exchange of ADP for ATP and converts BiP back to the state with low substrate affinity (Melnyk et al., 2015). Thus, each reversible interaction of BiP with substrate peptides is linked to the hydrolysis of one ATP to ADP and one inorganic phosphate, which in sum amounts to quite a lot of ATPs being hydrolyzed given the millimolar concentration of BiP and its perpetual activity. The currently known set of eight ERjs and two NEFs in human cells mediates substrate specificity of the ATPase cycle of BiP (Figure 1B). In this way, BiP’s functions relate (a) to the ER import of precursor polypeptides, folding/assembly of newly imported polypeptides, export into the cytosol plus degradation by the cytosolic proteasome of misfolded polypeptides as well as (b) to the regulation of folded proteins such as the ER membrane-resident sensors of the UPR (i.e., IRE1, PERK, and ATF6) or the α-subunit of the polypeptide-conducting Sec61-channel (Hennessy et al., 2000, 2005; Zhao and Ackerman, 2006; Dudek et al., 2009; Walter and Ron, 2011; Wang and Kaufman, 2012).
In the context of the latter, BiP was shown to support protein transport into the ER in two different ways. First, BiP together with the ER membrane-resident Sec63/ERj2 acts as an allosteric effector of the Sec61 complex for channel opening and concomitant insertion of the precursor polypeptide into the channel by binding to the luminal loop 7 of Sec61α (Haßdenteufel et al., 2018; Schorr et al., 2020). Next, BiP can act as a molecular ratchet on certain incoming and unfolded precursor polypeptides in transit through the Sec61-channel (Tyedmers et al., 2003). In addition, BiP is required for efficient closing of the Sec61 channel to prevent excessive Ca2+ leakage from the ER, again by binding to the luminal loop 7 of Sec61α (Schäuble et al., 2012). Therefore, siRNA-mediated depletion of BiP from HeLa cells was found to stimulate Ca2+ efflux from the ER via the Sec61-channel, as do protein misfolding in the ER or inhibition of SERCA by Thapsigargin (Lang et al., 2011; Schäuble et al., 2012).
Additional studies suggested that the different activities of BiP at the Sec61 complex are linked to different co-chaperones. Depletion of the integral membrane protein Sec63/ERj2 caused a substrate-specific defect in protein import (Lang et al., 2012; Schorr et al., 2015). In contrast, depletion of the ER luminal proteins ERj3 and ERj6, caused a Ca2+ specific phenotype, which resulted from inefficient sealing of the Sec61 channel by BiP (Schorr et al., 2015). In sum, functionality of the abundant ER luminal chaperone BiP is linked to two metabolites, ATP and Ca2+. Therefore, BiP occupies a central position in ER homeostasis by integrating proteostasis, Ca2+ balance, and energy homeostasis of the ER (Lang et al., 2017).
Protein-Mediated ATP Import Into the ER
As stated earlier, BiP is present in the ER lumen in millimolar concentration and depends on ATP hydrolysis as well as a high Ca2+ concentration for its various functions. Moreover, ATP hydrolysis by BiP results in ADP and, therefore, necessitates ADP and phosphate removal from the ER. To fulfill the energy demand of BiP and other abundant ER chaperones as well as the ER resident kinase Fam20C (Yu et al., 2020) both ATP import and ADP export should be coupled by an efficient exchanger.
Before AXER
Originally, classical biochemical approaches were employed in the putative identification and characterization of ATP carriers or ATP/ADP exchangers of the ER membrane. ATP import was reconstituted into proteoliposomes, harboring the total set of mammalian (rat liver, dog pancreas) or yeast ER membrane proteins, and this set of membrane proteins was subjected to purification of the transport activity by various standard methods. In none of these systems, however, did the biochemical approach identify the elusive carrier or exchanger (Clairmont et al., 1992; Mayinger and Meyer, 1993; Guillen and Hirschberg, 1995; Mayinger et al., 1995; Kim et al., 1996; Kochendörfer et al., 1999; Shin et al., 2000). However, the approach established certain hallmarks for the transport activity: protein-mediated (i.e., saturable and temperature/pronase/inhibitor [4,4'-diisothiocyano-2,2'-stilbenedisulfonic acid (DIDS), N-ethymaleimide (NEM)] sensitive), nucleotide selectivity (i.e., specificity for ATP and ADP), high nucleotide affinity (i.e., low micromolar KM), antiport transport mode (i.e., requirement for nucleotide preloading of the proteoliposomes), and Mg2+ independence (i.e., insensitivity to EDTA). More recently, ER membrane-resident ATP/ADP antiporters have been identified with the help of genetic approaches in the plant Arabidopsis thaliana (ER-ANT1) and in the alga Phaeodactylum tricornutum (PtNTT5) (Leroch et al., 2008; Chu et al., 2017). These proteins, however, do apparently not represent the major carriers for chemical energy in the ER of these green organisms and do apparently not have mammalian and yeast orthologs. Thus, until very recently ubiquitous proteins catalyzing the ATP uptake and the concomitant ADP release at the ER membrane had remained unknown at the molecular level. Yet, recent work established a set of hallmarks for cell type-specific regulation of ATP homeostasis in the ER of HeLa and INS-1 cells (Vishnu et al., 2014). Accordingly, Ca2+-efflux from the ER into the cytosol is coupled to both increased ADP phosphorylation in the cytosol (dominant in HeLa cells) plus mitochondria (dominant in INS-1 cells) as well as increased ATP uptake into the ER by an at the time unknown ATP carrier in the ER membrane, which itself is stimulated by Ca2+-efflux from the ER into the cytosol. Furthermore and as discussed later, the cytosolic Ca2+- and AMP-activated protein kinase (AMPK), the master regulator of energy metabolism with a link to the UPR (Preston and Hendershot, 2013; Boß et al., 2016), is essential for increased ADP phosphorylation at least in HeLa cells (Vishnu et al., 2014).
Since AXER
Screening databases for solute carriers (SLCs) that are located in the ER membrane and share the expression pattern in human tissues together with BiP drew attention to SLC35B1 (Hediger et al., 2013; Schlessinger et al., 2013). Previously, SLC35B1 was annotated as a member of the nucleotide-sugar transporter family (Nakanishi et al., 2001; Reyes et al., 2006; Dejima et al., 2009), hence its synonym UDP-galactose transporter-related protein 1 (UGTrel1). Depletion of the corresponding orthologs in Saccharomyces cerevisiae (HUT1), Schizosaccharomyces pombe (HUT1), and Caenorhabditis elegans (HUT-1) caused aberrant protein folding in the ER and loss of ER homeostasis as well as impaired larval development in case of the worm (Nakanishi et al., 2001; Dejima et al., 2009). Subsequent heterologous expression of SLC35B1 in Escherichia coli revealed that SLC35B1 is highly specific in transporting ATP and ADP, not nucleotide sugars, and acts in antiport mode (Klein et al., 2018). Furthermore, the heterologous expression product was reconstituted into proteoliposomes and found to show all four established characteristics of nucleotide selectivity (i.e., specificity for ATP and ADP), high nucleotide affinity (i.e., low micromolar KM), antiport transport mode (i.e., requirement for nucleotide preloading of the proteoliposomes), and Mg2+ independence (i.e., insensitivity to EDTA). Moreover, siRNA-mediated depletion of SLC35B1 from HeLa cells in combination with both ratiometric live cell imaging in real-time of cytosolic Ca2+ with Fura-2 and of ER ATP with the genetically encoded ATP sensor ERAT 4.01 was found to reduce ER ATP levels and, as a result, BiP activity. Consistent with the previously established hallmarks of ER energy homeostasis, artificially – that is, Thapsigargin – induced Ca2+ release from the ER was found to trigger SLC35B1-mediated ATP uptake into the ER. In sum, the data suggested that SLC35B1 facilitates ATP import into the ER and concomitant ADP export from the ER in human cells. Therefore, SLC35B1 was termed AXER, short for ATP/ADP exchanger in the ER membrane (Klein et al., 2018). These findings, which were already confirmed for additional cell lines such as CHO cells and INS-1 cells (Yong et al., 2019), may explain why the aforementioned AXER orthologs in yeast (HUT1) and C. elegans (HUT-1) have been found to play a more general role in ER homeostasis as would be expected for a nucleotide sugar transporter (Nakanishi et al., 2001; Dejima et al., 2009). Whether or not AXER is the only ATP carrier or ATP/ADP exchanger present in the mammalian ER membrane remains to be seen. In retrospect, the very low cellular concentration of AXER (18 nM; Sec61-channel, 300 nM; BiP, 8,235 nM in HeLa cells; Hein et al., 2015) paired with its high affinity for ATP may very well explain why the original biochemical approaches failed to identify the protein despite observing metabolite transport.
Regulatory Circuits and Signaling Pathways for Energy Homeostasis of the ER
Up to now, two seemingly controversial concepts have emerged for the regulation of ATP homeostasis in the mammalian ER (Figure 2). Based on experiments in HeLa cells, AXER was suggested to be part of a regulatory circuit and a Ca2+-dependent signaling pathway, termed lowER (Figure 2A), which was proposed to act in vicinity of the Sec61 complex at the ER membrane, thereby, guaranteeing sufficient ATP supply to the ER (Klein et al., 2018). The initial experimental data to characterize this putative signal transduction pathway suggested that a high ATP/ADP ratio in the ER lumen allows BiP to prevent Ca2+ leakage from the ER via the Sec61 channel and that a low ATP/ADP ratio, for example, because of increased protein import or folding or misfolding, triggers dissociation of BiP from the Sec61-channel and allows Ca2+ leakage from the ER (Schäuble et al., 2012). In the cytosol, Ca2+ could bind to calmodulin (CaM; Erdmann et al., 2011) and activate AMPK via CAMKK2 and finally the glycolytic enzyme 6-phospho-fructo-2-kinase (PF2K) and, in noncancer cells, Ca2+ could activate Ca2+-dependent dehydrogenases of the TCA cycle. Activated PF2K causes increased ADP phosphorylation by glycolysis, leading to ATP import into the ER via AXER. AXER itself may also be activated by Ca2+ efflux from the ER either directly or indirectly (Vishnu et al., 2014; Klein et al., 2018). We note that mammalian AXER comprises a putative cytosolic IQ motif in the loop between transmembrane domains 2 and 3 which suggests that AXER may very well be directly regulated by Ca2+-CaM. Subsequent normalization of the ER ATP/ADP ratio allows BiP to prevent the Sec61-mediated Ca2+ leakage and results in inactivation of lowER. SERCA, which pumps Ca2+ back into the ER lumen, balances the passive Ca2+ efflux and protein phosphatase 2 (PP2) dephosphorylates AMPK. Activated AMPK was previously shown to lead to reduced cap-dependent translation and therefore ties lowER to the UPR (Preston and Hendershot, 2013). Thus, lowER can be expected to represent the first line of defense of a cell against emerging ER stress.

Hallmarks of the Two Signaling Cascades LowER and CaATiER Regulating ER Energy Homeostasis. A: The ER low energy response (lowER) is activated by an increased need for protein folding. Misfolded proteins require BiP to dissociate from the Sec61 channel causing localized Ca2+ leakage from the ER. In the cytosol, Ca2+ triggers a downstream signaling process near the ER membrane causing immediate activation of the ATP importer AXER for ATP transport into the ER as well as the kinase AMPK for ATP generation by glycolysis. B: The Ca2+-antagonized transport into the ER (CaATiER) describes the preferential mitochondrial supply of ATP to the ER in response to a high protein misfolding load. In this model, Ca2+ release from the ER was partially rerouted into the mitochondria to boost the TCA cycle as well as blocking the AXER mediated ATP import into the ER after an extended period of time. For a more detailed description of both models, see text. Of note, in both models, AXER seems to represent the crucial solute carrier allowing ATP transport into the ER. ER = endoplasmic reticulum; ATP = adenosine triphosphate; ADP = adenosine diphosphate; TCA = tricarboxylic acid.
Based on experiments in HeLa, INS-1, and CHO cells, AXER was proposed to be part of another regulatory mechanism, termed Ca2+-antagonized transport into the ER or CaATiER (Figure 2B), which was supposed to guarantee sufficient ATP supply to the ER (Yong et al., 2019). The initial experimental data to characterize this putative signaling pathway demonstrated that mitochondria supply ATP to the ER and a SERCA-dependent Ca2+ gradient across the ER membrane is necessary for ATP transport into the ER via AXER. The following scenario was suggested: “Under physiological conditions, increases in cytosolic Ca2+ inhibit ATP import into the ER lumen to limit ER ATP consumption.” Furthermore, “ER protein mis-folding increases ATP uptake from mitochondria into the ER” (Yong et al., 2019). In the CaATiER model, the short-term increase in ER ATP levels upon SERCA inhibition was disregarded, because it was followed by a long-term decrease over the following ten minutes.
These findings raise, from our point of view, the interesting possibility that there may actually be two phases associated with Ca2+-coupled ER ATP homeostasis. A first phase that corresponds to lowER and a second one that was termed CaATiER. Such a biphasic regulatory scenario would be consistent with the observations that in HeLa cells the ATP for ER uptake is initially supplied mainly by anaerobic glycolysis and subsequently by oxidative phosphorylation and that there are, for example, cell-type specific variations, possibly reflecting the different proportions of substrate level- and oxidative-phosphorylation of ADP in different cell types and under different metabolic conditions. Further support for this scenario comes from the facts that various proteins of mitochondrial–ER contact sites (MERCs) were found to be co-immunoprecipitated together with AXER from HeLa cell detergent extracts including BiP, Calnexin, IP3R1, IP3R3, and SERCA2b (Klein et al., 2018). The question is what’s the point of having these two phases? In the experiments, which gave rise to the CaATiER model, the authors also studied the effect of protein misfolding in the ER and observed that it stimulates Ca2+ transfer from the ER to mitochondria and ER ATP homeostasis becomes more dependent on oxidative phosphorylation. Therefore, we propose that lowER may describe the ad hoc regulation of ER homeostasis under physiological conditions, that is, whenever the ATP to ADP ratio drops in the ER, to prevent problems of protein misfolding and that CaATiER may best describe pathophysiological conditions such as protein misfolding, where the demand for ATP becomes particularly high.
Tying Together lowER, CaATiER, and MERC
The biphasic AXER response to increasing ER stress aligns well with the previous observation of increased ER–mitochondrial coupling during early phases of ER stress to promote oxidative phosphorylation (Bravo et al., 2011). As its low cellular abundance might have prevented this so far, AXER itself has not been described as classic constituent of the MERC or the synonymous biochemically purified fraction called MAM (mitochondria-associated ER membrane) (Giacomello and Pellegrini, 2016). Yet, AXER associates with various members of MERCs as well as of the translocon complex and mitochondrial ATP synthase (Klein et al., 2018). The dynamic nature of the MERC structure including variable membrane distance, protein composition, subcellular localization, metabolic coupling, and metabolite flux could contribute to the different phases of Ca2+ dependent (cf. lowER) and Ca2+ inhibited (cf. CaATiER) ATP transport into the ER lumen by AXER (Myhill et al., 2008; Bui et al., 2010; Cerqua et al., 2010; Naon and Scorrano, 2014; Raturi et al., 2016). Actually, an analogous biphasic response to allosteric effectors such as Ca2+ and ATP is also seen for other key players of the ER-mitochondria crosstalk. While a low Ca2+ (or ATP) concentration activates the prominent MERC member IP3R, a high Ca2+ (or ATP) concentration triggers its inactivation. Similarly, the mitochondrial Ca2+ uniporter (MCU) shows a biphasic response to increasing Ca2+ levels. However, in the case of MCU the Ca2+ effect is time-dependent, that is, of a kinetic nature. While short-term Ca2+ elevations activate MCU, extended Ca2+ elevations inactive it (Mak et al., 2001; Moreau et al., 2006; Rizzuto et al., 2009). Hence, the seemingly controversial Ca2+ dependencies of AXER could indeed represent its cellular mechanism of regulation and prevent excessive ATP drainage into the ER in order to avoid spreading of ATP shortage, UPR and stress to other organelles. Regarding cellular homeostasis, a localized and confined imbalance (such as ER stress) might be easier to salvage than a spreading pan-organellar disturbance.
The hypothetical model combining AXER, lowER, CaATiER, and dynamic MERCs is shown in Figure 3. Under resting conditions, AXER transports cytosolic ATP into the ER and the MERCs are at a relaxed distance. A deviation from homeostasis by a minor protein misfolding load would cause BiP to segregate from the Sec61 complex, Sigma-1 receptor (a MERC constituent), and the UPR sensors. While the latter (UPR sensors) would trigger a slow translational/genetic program, the former two (Sec61 complex and Sigma-1 receptor) would initiate a fast and immediate ER Ca2+ release. Ca2+ efflux from the Sec61 complex and diffusion near the ER membrane could activate AXER and, as a first line of defense, cytosolic ATP import into the ER (Vishnu et al., 2014; Klein et al., 2018). Further Ca2+ efflux via IP3R (stabilized by BiP-free Sigma-1 receptor) across the MERCs into mitochondria could activate the TCA cycle to promote mitochondrial respiration and shorten the MERCs into a primed distance (Hayashi and Su, 2007; Bravo et al., 2011). In case this immediate danger response fails to reestablish protein and energy homeostasis CaATiER takes over. MERCs contract further to a tight distance allowing preferential transport of mitochondrial ATP via AXER into the ER (Yong et al., 2019). With MERCs in the tight distance mode, mitochondria would also act as Ca2+ sink and suppress Ca2+ dependence of AXER (Figure 3B). Finally, Yong et al. (2019) managed to demonstrate recovery of ER ATP levels and, thereby, reversibility of the homeostasis <=> lowER <=> CaATiER axis upon removal of the applied ER stressor (Figure 3C). Surprisingly, after washout of the reversible SERCA inhibitor, the refilling of ER Ca2+ and ER ATP was temporally offset from each other with the recovery of ER ATP levels being, for a so far unknown reason, much delayed.
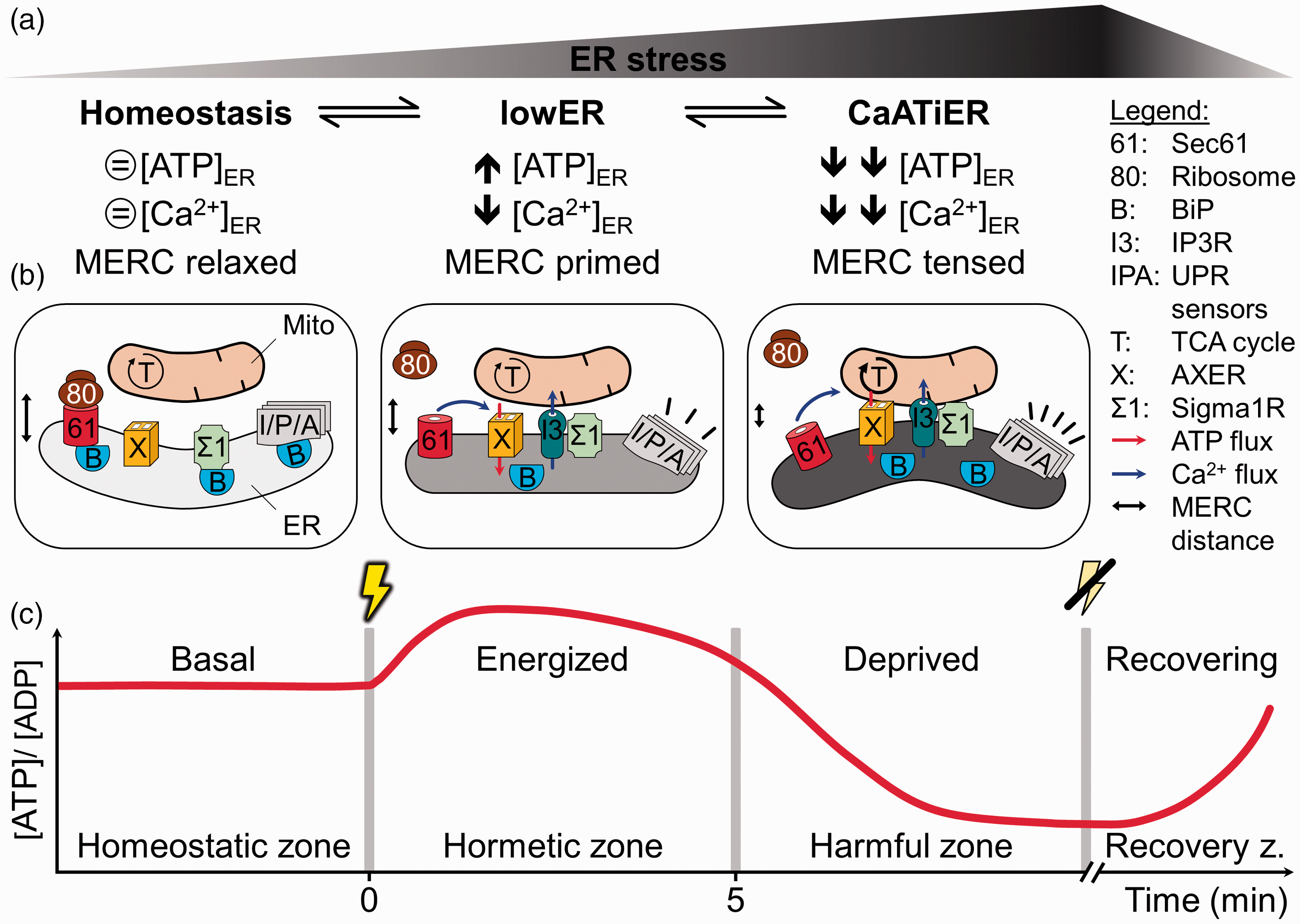
The ER Holo-Care Model as an Attempt to Unify the Controversial LowER and CaATiER Observations. A: With an increasing level of ER stress, often equivalent to an increasing amount of misfolded proteins, cells respond in a successive manner away from homeostasis to lowER to CaATiER. Each phase or response zone is illustrated by distinctive fluctuations in ATP and Ca2+ levels as well as (putative) alterations in MERC distance. B: Proteins and complexes thereof, metabolite fluxes as well as subcellular structures that mediate the transitions from homeostasis to lowER to CaATiER are indicated. Although the changes in metabolite flux (ATP and Ca2+) occur on a quick scale (i.e., allegro = fast, quickly, and bright), the MERC contraction is of moderate speed (i.e., andante = at a walking pace), and the full activation of the UPR response emerges with rather slow tempo (i.e., adagio = slowly with great expression). C: The diagram shows a trace of the ER [ATP]/[ADP] ratio over time in response to a protein folding stressor (yellow thunderbolt). In a first line of defense the ER becomes energized by an increased [ATP]/[ADP] ratio via lowER to prevent propagation of the protein folding issue. Unresolved, a further increase of misfolded proteins would cause vehement energy deprivation and eventually even shut down of the ATP import by CaATiER. Effective clearance of ER stress (strike-through thunderbolt) commences the [ATP]/[ADP] ratio to recover. In adherence to the principle of hormesis, the zones were coined homeostatic, hormetic (lowER), and harmful zone (CaATiER) followed by the recovery phase. The four phases balancing ER energy requirements were collectively summarized as holo-Care principle. A more detailed description of the model is also given in the text. ER = endoplasmic reticulum; ATP = adenosine triphosphate; ADP = adenosine diphosphate; MERC = mitochondrial–ER contact site; UPR = unfolded protein response.
Eventually, this delay between Ca2+ and ATP refilling could also indicate that the ATP import into the ER is decoupled from the Ca2+ status and the putative involvement of other regulators should be considered. In this case, another regulatory strategy of biochemical cascades based on feedback inhibition or feedforward stimulation by pathway intrinsic metabolites, including intermediates or end-products, could be feasible. Shin et al. (2000) observed using the classical approach of reconstitution that ATP transport into proteoliposomes is trans-stimulated by ADP. Thus, ER ATP consumption by protein folding, chaperoning or early prevention of misfolding would increase the ADP level in the ER and temporarily trans-stimulate AXER-mediated ATP import equivalent to the lowER signaling concept. As before, if this immediate danger response fails to reestablish ER homeostasis CaATiER kicks in. Alternatively, MERCs are also known to be enriched in cholesterol as well as enzymes for sterol and phospholipid synthesis (Vance, 1991; Hayashi et al., 2009; Flis and Daum, 2013; Vance, 2014) Interestingly, initial efforts in yeast by Mayinger et al. identified the Sac1 protein as crucial regulator of ER ATP import (Mayinger and Meyer, 1993; Mayinger et al., 1995; Kochendörfer et al., 1999). With Sac1 functioning in Golgi phospholipid metabolism, the data point at the unknown role of membrane lipids in regulating ER ATP import.
Taken together, the two controversially discussed regulatory mechanisms (Figure 2) of the recently identified ER ATP exchanger AXER are probably two successional phases of the same signaling response (Figure 3). In fact, the biphasic shape of the ER ATP levels during the biphasic defense response mounted by the ER resembles the hormetic principle (Mattson, 2008; Bhakta-Guha and Efferth, 2015; Calabrese, 2015). Following an initial disruption of homeostasis, hormesis can be defined as the transient, beneficial compensatory mechanism to a low dose of stress which will vanish upon longer or more vigorous exposure of the same stress, then causing damage. The underlying dose–response curve of such a hormetic response resembles an inverted U-shape. Within this also biphasic progress, the favorable compensatory adaption to the low dose of stressor is called the hormetic zone. Thus, progression from ER balance to lowER to CaATiER and the corresponding inverted U-shape of the ER ATP level during unresolved ER stress represents the classic transitions of hormesis from the homeostatic to the hormetic to the harmful zone (Figure 3C).
In an attempt to combine the transitional phases of the ER energy homeostasis from balance to imbalance (lowER and CaATiER) and recovery thereof, we propose to embed the terms in a more unifying acronym called ER holo-Care (homeostasis, lowER, CaATiER, recovery).
Open Questions
The identification of AXER opens up an exciting avenue of research addressing the energy homeostasis of one the cell’s largest organelles. However, many open questions remain on multiple layers. On an organismal level, we will need to find answers about AXER orthologs and its functional conservation in other species. Work with the AXER homolog HUT-1 in C. elegans has shown that it is essential only during larval development, but not in the adult worm (Dejima et al., 2009). On the cellular level, more detailed studies will be needed to define the exact subcellular localization of AXER and its participation in the MERC structure. In addition, AXER’s role as well as importance in different cell types should further our understanding of the ER ATP import. This line of research will likely be complemented by studies under different environmental conditions or stresses. Finally, a major focus on AXER should also happen at the molecular level. With its, at least currently, unique function among the ER membrane proteins, it should represent an interesting candidate for structural determination by cryo-EM or X-ray crystallography. Aside from a more detailed investigation of the regulatory mechanism described earlier, the regulation of AXER by posttranslational modifications, other allosteric effectors including protein–protein interactions as well as the role of the different AXER isoforms warrants an exciting time ahead of us. Maybe the identification of AXER will also pave the way for the identification of more ATP transporters in other secretory organelles lacking autonomous ATP generating systems. Even for the ER, there may be the need for an additional ATP transporter, one which does not work as an exchanger but imports nucleotide when the organelle expands during development or stress.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: S. L. and R. Z. are grateful for their continuous funding by the Deutsche Forschungsgemeinschaft (SFB 894 and IRTG 1830).
