Abstract
The lipid droplet (LD) is a cytoplasmic organelle, but it also exists in the nucleus under some conditions or in some cell types. New studies have revealed that nuclear LDs do not occur by haphazard entry of cytoplasmic LDs. Instead, they are generated by specific mechanisms that are increasingly understood. The inner nuclear membrane (INM) plays a critical role in nuclear LD formation in both mammalian hepatocytes and budding yeast, although in significantly different ways. Hepatocyte nuclear LDs derive from precursors of very low-density lipoprotein lacking apolipoprotein B-100, which form in the endoplasmic reticulum lumen and accumulate in intranuclear extensions of the perinuclear space called type I nucleoplasmic reticulum. In contrast, nuclear LDs in yeast are generated by triglyceride synthesized in the INM. Nuclear LDs in hepatocytes and budding yeast are both instrumental in the regulation of phospholipid synthesis; however, again they function in different ways. As the full functional importance is as yet unknown, the close relationship of nuclear LDs and the INM is an attractive target of research from both physiological and pathological perspectives.
Introduction
Lipid droplets (LDs) were long considered to be sedentary inclusions, like glycogen particles (Fawcett, 1981). Research over the past two decades has drastically changed this view, and LDs are currently considered to be cytoplasmic organelles that interact with other organelles in a regulated manner, with various physiological functions (Fujimoto and Parton, 2011; Gao and Goodman, 2015; Olzmann and Carvalho, 2019). LDs in the nucleus have not garnered much attention, mainly because they are few in number. Nuclear LDs might occur by chance entrapment of cytoplasmic LDs, but this presumption was challenged by two observations. The first was the frequent occurrence of nuclear LDs in some cell types, for example hepatocytes, which are not necessarily correlated with an abundance of cytoplasmic LDs. In contrast, adipose cells and steroidogenic cells (e.g., adrenocortical cells) contain abundant cytoplasmic LDs, but have few nuclear LDs (Ohsaki et al., 2016). Second, budding yeast deficient in Sei1 (Fld1), a homolog of mammalian seipin, contain a significant number of nuclear LDs (Cartwright et al., 2015; Wolinski et al., 2015). Seipin is a protein essential for normal LD formation (Walther and Farese, 2012; Olzmann and Carvalho, 2019) and its mutations cause severe lipodystrophy (Magre et al., 2001). Nuclear LDs in seipin-deficient yeast are likely a consequence of defects in LD biogenesis and might have pathological implications.
Two papers on hepatocyte nuclear LDs from our group (Ohsaki et al., 2016; Sołtysik et al., 2019) and one paper on budding yeast nuclear LDs from the Köhler lab (Romanauska and Köhler, 2018) showed that nuclear LD formation occurs independently from that of cytoplasmic LDs. These studies indicated that the inner nuclear membrane (INM) plays crucial roles in nuclear LD formation in both hepatocytes and budding yeast, although in drastically different ways. Yeast nuclear LDs appear to form in the INM, whereas most, if not all, hepatocyte nuclear LDs derive from LDs in the lumen of type I nucleoplasmic reticulum (NR), which is the inward extension of the perinuclear space (Figure 1(a); see the following text for detailed explanation of NR). Despite these differences, nuclear LDs in both hepatocytes and budding yeast are involved in the regulation of membrane lipid synthesis.

Nuclear LD formation in hepatocytes. (a) NR: Type I NR is made by intranuclear extension of the inner nuclear membrane (INM) alone, whereas type II NR is a protrusion of the cytoplasm covered by both the inner and the ONMs. (b) Under normal conditions, two VLDL precursors, ApoB-free lumenal LDs and lipidated ApoB, are generated in the ER lumen and form mature VLDL in the ER or in post-ER compartments. MTP is necessary for generation of both VLDL precursors, whereas ApoB is required only for lipidated ApoB. Under ER stress, ApoB decreases by degradation, whereas the MTP level is maintained and ApoB-free lumenal LDs thus increase. Lumenal LDs accumulate in the lumen of type I NR and eventually trespass into the nucleoplasm through defects in the INM. NR = nucleoplasmic reticulum; INM = inner nuclear membrane; ONM = outer nuclear membrane; LD = lipid droplet; ER = endoplasmic reticulum; VLDL = very low-density lipoprotein; MTP = microsomal triglyceride transfer protein; ApoB = apolipoprotein B.
In subsequent sections, we will first look over the past literature on nuclear LDs and then discuss the mechanism of nuclear LD biogenesis in hepatocytes and budding yeast with a particular focus on their relationship with the INM including the type I NR membrane. The function of nuclear LDs, which is largely an unexplored territory at present, will also be discussed.
Nuclear LDs in Liver Cells
Early Studies
Presence of nuclear LDs, termed as nuclear inclusions, was initially reported in hepatoma cells by light microscopy using Sudan staining (Leduc and Wilson, 1959). Those LDs might be actually located in cytoplasmic invaginations deep into the nucleus, but electron microscopic (EM) studies that followed demonstrated that LDs were present within the nucleoplasm. The EM studies revealed LDs in four situations: (i) the nucleolus of rat liver cells during thioacetamide-induced carcinogenesis (Thoenes, 1964), (ii) LDs in the nucleoplasm and in the lumen of the endoplasmic reticulum (ER)/nuclear envelope/NR in rat liver treated with carcinogenic N, N′-dimethyl-4-aminoazobenzene (Karasaki, 1969, 1973), (iii) nuclear LDs in mouse liver treated with hepatotoxic polybrominated biphenyl (Martino et al., 1981), and (iv) nuclear LDs in mouse embryonic cells with lethal mutations (Hillman et al., 1970; Hillman and Hillman, 1975).
Study of nuclear LDs in liver cells has been expanded using sophisticated methods, including confocal laser-scanning microscopy and EM of serial sections, to include normal rat liver (Layerenza et al., 2013), human liver infected with hepatitis C (Uzbekov and Roingeard, 2013), mouse liver treated with perfluorooctanoic acid (a chemical structurally resembling fatty acids but showing hepatotoxicity and carcinogenicity; Wang et al., 2013), and in HepG2 cells treated with a cytotoxic clay (Maisanaba et al., 2013).
Mechanisms Underlying Nuclear LD Formation in Hepatocytes
The early studies suggested that nuclear LDs may occur in hepatocytes under unfavorable conditions, but the mechanism of nuclear LD formation and its functional significance were not known. Hepatocytes also contain many cytoplasmic LDs, reflecting their high lipogenic capacity. Active lipogenesis, however, does not necessarily lead to nuclear LD formation (Ohsaki et al., 2016), suggesting that hepatocytes may have some specific mechanism to form nuclear LDs.
Hepatocytes synthesize and secrete very low-density lipoprotein (VLDL; Kulinski et al., 2002; Hussain et al., 2012; Lehner et al., 2012). In the process of VLDL formation, two lipidic particles are synthesized in the ER lumen: lipidated apolipoprotein B-100 (ApoB) and ApoB-free lumenal LDs. They collaborate to generate mature VLDL in post-ER compartments (Figure 1(b)). The formation of lipidated ApoB requires ApoB and microsomal triglyceride transfer protein (MTP), which transfers lipids to the nascent ApoB polypeptide. In contrast, the formation of ApoB-free lumenal LDs requires MTP, but not ApoB. When cells are exposed to ER stress, ApoB decreases due to co- and posttranslational degradation, downregulating VLDL synthesis (Ginsberg and Fisher, 2009), whereas the MTP level is maintained (Qiu et al., 2005; Sołtysik et al., 2019). As a result, lipidated ApoB and VLDL decrease, whereas ApoB-free LDs increase in the ER lumen, which includes the perinuclear space. Eventually, large ApoB-free lumenal LDs accumulate in the type I NR lumen and then appear to enter the nucleoplasm through the NR membrane, which is disrupted, becoming nuclear LDs (Figure 1(b)). The formation of nuclear LDs concomitant with defective VLDL secretion is prominent in hepatocarcinoma cell lines and also occurs in mouse hepatocyte lacking lamina-associated polypeptide 1 (LAP1; Shin et al., 2019). MTP is crucial for the entire process, and thus MTP inhibition suppresses nuclear LD formation. The direct relationship between a lipoprotein precursor and nuclear LDs was unexpected, but it explains how nuclear LDs exist abundantly in hepatocytes (Lehner et al., 2012; Hussain, 2014).
Involvement of INM in Nuclear LD Formation in Hepatocyte
Disruption of normal VLDL synthesis does not fully account for nuclear LD formation in hepatocytes under ER stress. Another important factor is the development of the type I NR. The type I NR is a thin sheet-like or tubular structure made by intranuclear extensions of the perinuclear space, also known as the nuclear envelope, covered by the INM. This is different from type II NR, which refers to nuclear invaginations of the cytoplasm that include both the outer nuclear membrane (ONM) and the INM (Malhas et al., 2011; Figure 1(a)). The NR has been observed in various cell types and may increase in several pathological conditions, including cancer (Drozdz and Vaux, 2017). Several functions have been proposed for the NR, but it remains unclear if type I NR has any function distinct from type II NR. For example, one putative NR function is to regulate Ca2+ concentration deep inside the nucleus, which may affect gene transcription, but this function can be exerted by either type I or type II NR. In contrast, the mechanisms proposed for NR formation, such as pushing-in from the cytoplasm or pulling-in from the nucleoplasm, are mostly applicable only to type II NR (Malhas et al., 2011). Type I NR formation must be accompanied by separation of the INM and the ONM, which are normally bridged by the linker of nucleoskeleton and cytoskeleton (LINC) complex. Consistently, knockdown of SUN1, an INM transmembrane protein forming the LINC complex, induces type I NR increase (Ohsaki et al., 2016).
NR formation may be correlated with de novo phospholipid synthesis. Overexpression of CTP:phosphocholine cytidylyltransferase α (CCTα), the rate-limiting enzyme of the Kennedy pathway for phosphatidylcholine (PC) synthesis, causes NR elongation (Lagace and Ridgway, 2005). Moreover, de novo synthesized phospholipids were shown to be incorporated preferentially to NR and contribute to NR growth (Drozdz et al., 2017). These studies did not necessarily distinguish between type I and type II NR, but in cells deficient in proteolytic maturation of lamin A, which accumulate prelamin A, prominent elongation of both type I and type II NR was demonstrated to occur in a CCTα-dependent manner (Goulbourne et al., 2011). These results indicated that increased PC synthesis by CCTα activation promotes NR development.
Expression of the catalytic-dead mutant of CCTα can also induce NR development, probably through deforming the INM by insertion of the amphipathic domain (Lagace and Ridgway, 2005; Gehrig and Ridgway, 2011). It is not known, however, to what extent this nonenzymatic mechanism contributes to NR formation in cells (see later for a further discussion).
In hepatocytes under ER stress, type I NR increases in both number and length (Sołtysik et al., 2019). This change occurs even when MTP is inhibited, indicating that it is not directly related to VLDL synthesis. Because ER stress in nonhepatocyte cell lines does not induce type I NR, it appears that type I NR is regulated by a cell-type-specific mechanism. The close relationship between nuclear LDs and NR is most evident during mitosis of hepatocytes (Sołtysik et al., 2019). When a hepatocyte with many nuclear LDs divides, the daughter cells initially have no nuclear LDs but gradually regain them thereafter. In contrast, NR is already present immediately after mitosis. The new nuclear LDs appear to emerge from those NR membranes in an MTP-dependent manner.
The NR in hepatocytes appears to be related to two different stages of nuclear LD formation. The first is at the stage when ApoB-free lumenal LDs grow in the type I NR lumen and eventually escape into the nucleoplasm through defects in the NR membrane (Sołtysik et al., 2019). The NR membrane is generally associated with lamins (Gehrig et al., 2008), but curiously, the type I NR membrane around large ApoB-free lumenal LDs is devoid of lamins (Sołtysik et al., 2019). This deficiency is likely to make the membrane mechanically weak, so that expansion of ApoB-free lumenal LDs by MTP-mediated lipid accretion may physically cause the disruption. Here, how a local lamin deficiency arises is not known, but promyelocytic leukemia (PML) protein-II (PML-II), which is crucial for nuclear LD formation in hepatocytes, binds linearly along the INM when overexpressed and induces local lamin depletion (Jul-Larsen et al., 2010; Ohsaki et al., 2016). The PML-II domain necessary for binding to INM is enriched with positive charges, suggesting that the interaction of PML-II and acidic residues in the INM might be involved.
The NR–nuclear LD connection in hepatocytes also exists when nuclear LDs grow. This growth is not sensitive to MTP inhibition, but is suppressed by triacsin C, an inhibitor of acyl-CoA synthase (Sołtysik et al., 2019). Diacylglycerol acyltransferase 2 (DGAT2), which distributes in the NR and nuclear LDs (Ohsaki et al., 2016), is likely to be involved in this phase of nuclear LD growth. DGAT2 is thought to take a hairpin-like configuration and move from the ER to cytoplasmic LDs via a membrane bridge (Wilfling et al., 2013). The distribution of DGAT2 suggests that nuclear LDs are connected to the NR membrane at a certain stage.
Nuclear LDs in Other Mammalian Cell Types
Small intestinal epithelial cells harbor LDs in the ER lumen and the perinuclear space in apolipoprotein B deficiency (Hamilton et al., 1998) and after oil feeding (Palay, 1960). These observations indicate that small intestinal epithelial cells, which secrete chylomicrons, may form nuclear LDs by a mechanism similar to those in hepatocytes.
Other types of cells that are not thought to secrete lipoproteins also contain nuclear LDs. Stromal cells in the human sclera in vivo and U2OS, an osteosarcoma cell line, may belong to this category (Ohsaki et al., 2016; Petrea et al., 2018). Still others, such as HeLa, can also harbor nuclear LDs, albeit much fewer in number. The mechanism of nuclear LD formation in these cells is not known, but the presence of lipid-synthesizing enzymes in the INM (Agarwal et al., 2011; Prasad et al., 2011; Tsai et al., 2016) suggests that nuclear LDs in mammalian cells may be generated in the INM as shown in yeast (Romanauska and Köhler, 2018; see later).
Nuclear LDs in Budding Yeast
Nuclear LDs in Seipin-Deficient Yeast
Budding yeast (Saccharomyces cerevisiae) does not appear to have nuclear LDs under normal culture conditions, but yeast lacking either the yeast seipin Sei1 (Fld1) or its partner Ldb16 exhibit LDs in the nucleus, along with small, clustered LDs and a few supersized LDs in the cytoplasm (Cartwright et al., 2015; Wolinski et al., 2015). Deficiency of seipin homologs in Arabidopsis thaliana also induces nuclear LD formation (Taurino et al., 2018). Seipin exists as undecamer (human; Yan et al., 2018) or dodecamer (Drosophila melanogaster; Sui et al., 2018) in the ER membrane; seipin of budding yeast was also suggested to adopt a similar configuration (Binns et al., 2010). Although the molecular function of seipin remains an open question, it is located at the ER–LD junction (Szymanski et al., 2007; Fei et al., 2008; Figure 2(A)) and is thought to be involved in the initial process of LD formation by properly partitioning neutral lipids and proteins between the ER and LDs and also in the subsequent stage of LD maturation (Grippa et al., 2015; Salo et al., 2016; Wang et al., 2016; Salo et al., 2019). One notable property of the seipin molecule is its strong affinity to acidic phospholipids, especially phosphatidic acid (PA; Yan et al., 2018). A number of studies found that seipin deficiency induces an increase of PA in the total cells (Fei et al., 2011b; Sim et al., 2012; Jiang et al., 2014; Wolinski et al., 2015; Pagac et al., 2016) and in the microsome (Fei et al., 2011b; Pagac et al., 2016), suggesting that seipin may play a critical role in PA metabolism.
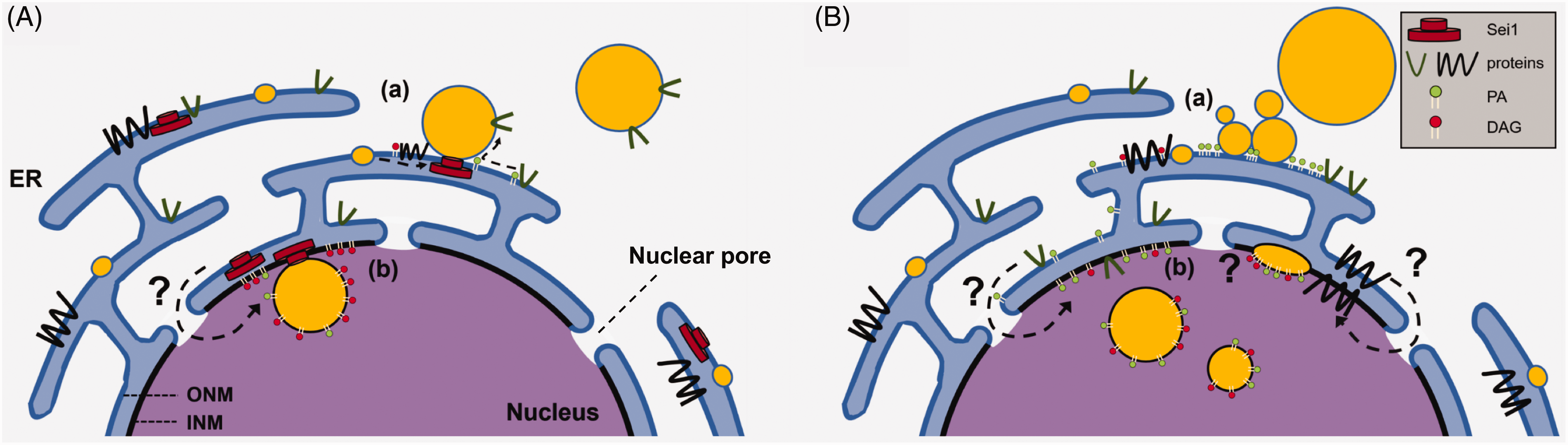
Nuclear LD formation in budding yeast. (A) Wild-type yeast. (a) Seipin (Sei1) exists as oligomers at the ER–LD junction and aids in the orderly transport of proteins and lipids from the ER to budding LDs. Sei1 plays a critical role in the regulation of PA in the ER/ONM, probably by binding PA and also by affecting PA-synthesizing enzyme activity. (b) Sei1 may also localize to the INM and support the budding of nuclear LDs, but how the Sei1 oligomer reaches the INM remains unclear. The INM has its own pool of PA and DAG, and the downregulation of phospholipid synthesis channels more PA to the synthesis of DAG and triacylglycerol (TAG) that facilitates nuclear LD formation. (B) Sei1-deficient yeast. (a) Cytoplasmic LDs form in irregular sizes. PA accumulates at the ER–LD junction. Free PA in the ER/ONM may increase through the absence of Sei1 binding and the aberrant activation of PA synthesis. (b) Nuclear LDs exist more frequently than in wild-type yeast, but the nuclear LDs and the INM might not form a normal bridge. The increase of nuclear LD formation might be caused by an increase of PA in the INM. INM = inner nuclear membrane; ONM = outer nuclear membrane; ER = endoplasmic reticulum; PA = phosphatidic acid; DAG = diacylglycerol.
It is not clear why seipin deficiency induces nuclear LD formation in budding yeast. If seipin exists only in the ER/ONM and not in the INM (see otherwise later), it is reasonable to assume that nuclear LDs occur because the normal seipin functionality in the ER/ONM is lost.
We speculate that an increase of free PA in the INM might be involved: namely, that a significant portion of PA may be sequestered by the seipin oligomer in the ER/ONM normally (Yan et al., 2018), but when seipin is absent, that PA may become freely diffusible and an increased amount can reach the INM. PA in the INM can be converted to diacylglycerol (DAG) by Pah1, a PA phosphatase (Santos-Rosa et al., 2005). Dga1 and Lro1, which are DAG-acyl transferases, may be present in the INM and synthesize TAG to generate nuclear LDs (Figure 2(B)).
The increase of free PA in seipin-deficient cells may also arise because seipin has an inhibitory effect on glycerol-3-phosphate acyltransferase (GPAT; Pagac et al., 2016). Seipin in human cells was also suggested to bind 1-acylglycerol-3-phosphate-O-acyltransferase 2 (AGPAT2) and lipin-1 (Talukder et al., 2015), but the interaction of Sei1 with the yeast homolog of these enzymes has not been detected (Pagac et al., 2016).
Nuclear LD Formation in the INM
The INM of budding yeast appears to have an innate ability to produce lipid esters. In wild-type yeast, fluorescence biosensors detected a low level of PA and abundant DAG in the INM (Figure 2(B); Romanauska and Köhler, 2018). In the absence of Cds1 and Ino4, nuclear LDs formed with intense PA signals on their surface (Romanauska and Köhler, 2018). Cds1 is an enzyme that converts PA to CDP-DAG, which is a precursor for all glycerophospholipids in budding yeast. Ino4 is part of the Ino2-Ino4 complex that upregulates transcription of Cds1 and other genes encoding phospholipid synthetic enzymes, by binding a UASINO sequence in their promoters (Henry et al., 2012). The result suggests that PA increases as a result of decrease in the conversion of PA to CDP-DAG, and thus more PA is channeled to the synthesis of DAG and triacylglycerol (TAG) in the INM. Consistently, Cds1 was shown to localize in the INM (Romanauska and Köhler, 2018).
Notably, co-depletion of Sei1 and Ino4 caused abnormal LD–membrane adherence in both the INM and the ER/ONM. Furthermore, bimolecular fluorescence complementation with Nup60, a nucleoplasmic basket protein in the nuclear pore complex, suggested that Sei1 may also exist in the INM (Romanauska and Köhler, 2018). This result led authors to speculate that Sei1 may be involved in the membrane bridge formation between the INM and nuclear LDs, as it probably does between the ER and cytoplasmic LDs (Salo et al., 2016; Wang et al., 2016; Figure 2(B)). This suggests that the INM might be able to generate nuclear LDs by a mechanism similar to the cytoplasmic LD formation in the ER/ONM. Nuclear LDs form only rarely, however, even when budding yeast is cultured with oleic acid (Romanauska and Köhler, 2018). Moreover, TAG generated in the INM predominantly gives rise to cytoplasmic LDs (Barbosa et al., 2019). These results suggest that nuclear LD formation in the yeast INM is not an efficient process.
With regards to the aforementioned scenario, it is intriguing how Sei1 reaches the INM. The seipin oligomer is estimated to be 22 nm in diameter and 7 nm in height (human; Yan et al., 2018) and 15 nm in diameter and 8 nm in height (Drosophila melanogaster; Sui et al., 2018), the size of which might limit diffusion through the nuclear pore. TorsinA, which also forms a large oligomer (Demircioglu et al., 2019), is thought to reach the INM by undergoing disassembly and reassembly, which is assisted by two cofactors, luminal domain-like LAP1 (LULL1) in the ER and LAP1 in the INM (Goodchild et al., 2015). It is not known whether Sei1 utilizes a similar mechanism. In addition, although the abnormal LD–membrane adherence in Sei1-deficient cells is consistent with Sei1’s putative function in membrane bridge formation (Salo et al., 2016; Wang et al., 2016), it was not observed in previous studies on budding yeast deficient in Sei1 alone (Szymanski et al., 2007; Fei et al., 2008, 2011a; Wolinski et al., 2011; Cartwright et al., 2015; Wolinski et al., 2015). This discrepancy suggests that the abnormal LD–membrane adherence may occur only when Ino4 is also deficient, causing general perturbation of membrane lipids (Romanauska and Köhler, 2018).
The formation of nuclear LDs in budding yeast does not appear to be accompanied by NR formation, whether it is induced by the Sei1/Ino4 deficiency or by suppression of phospholipid synthesis (Romanauska and Köhler, 2018). This is different not only from nuclear LD formation in hepatocytes (Ohsaki et al., 2016) but also from that in seipin-deficient plant cells (Taurino et al., 2018), which are not likely to synthesize lipoprotein. The role played by type I NR in nuclear LD formation remains to be studied.
Function of Nuclear LDs
Regulation of Phospholipid Synthesis
Nuclear LDs in hepatocytes recruit CCTα, the major CCT isoform, thereby activating PC synthesis (Ohsaki et al., 2016; Sołtysik et al., 2019). ApoB-free lumenal LDs contain abundant phosphatidylethanolamine, a cone-shaped phospholipid (Wang et al., 2007), so its direct descendant, nuclear LDs, are likely to be the same. The propensity of cone-shaped phospholipids to form phospholipid packing defects may facilitate the binding of CCTα to nuclear LDs through an amphipathic helix (Prevost et al., 2018).
Inactive CCTα is present in the soluble phase and, upon binding to membranes, becomes activated through a conformational change (Cornell and Ridgway, 2015). The INM and the cytoplasmic LD have been reported as locations of CCTα recruitment and activation (Watkins and Kent, 1992; Lagace and Ridgway, 2005; Krahmer et al., 2011; Aitchison et al., 2015; Haider et al., 2018). In hepatocytes, nuclear LDs appear to be a major platform of CCTα activation, because significant changes in PC synthesis were caused by an increase and a decrease of CCTα recruitment to nuclear LDs by manipulating the expression level of perilipin-3 (Sołtysik et al., 2019). Here, perilipin-3 competes with CCTα in nuclear LD binding and appears to function as a regulator of CCTα (Figure 3(a)). The increase of nuclear LDs in hepatocytes under ER stress is thought to be a feedback mechanism to upregulate PC synthesis (Sołtysik et al., 2019), leading to mitigation of the stress through expansion of the ER lumenal capacity (Schuck et al., 2009).

Nuclear LD and regulation of phospholipid synthesis. (a) Hepatocytes. CCTα and perilipin-3 bind to nuclear LDs in a competitive manner. An increase in perilipin-3 expression decreases CCTα bound to nuclear LDs and downregulates PC synthesis, whereas a decrease in perilipin-3 exerts the opposite effect. (b) Budding yeast. Sequestration of Opi1 to nuclear LDs, cytoplasmic LDs, and the ER/ONM allows the Ino2-Ino4 activator complex to bind UASINO, causing transcriptional activation of genes related to phospholipid synthesis. Nuclear LDs might have a different significance than the other Opi1-binding structures because they are located close to the gene transcription site. PC = phosphatidylcholine; CCTα = CTP:phosphocholine cytidylyltransferase α.
Nuclear LDs in budding yeast are also involved in the regulation of lipid synthesis by Opi1 sequestration. Opi1 is the repressor of the Ino2-Ino4 activator complex (Henry et al., 2012). When the PA level in the ER is relatively high (e.g., yeast in exponential growth in the absence of added inositol), Opi1 is retained in the ER through interaction with both PA and Scs2, which leads to active transcription of UASINO-containing genes (Loewen et al., 2004). Opi1 sequestration to nuclear LDs likely works similarly, increasing phospholipid synthesis in response to lipid overload (Figure 3(b)), but in this respect the more abundant cytoplasmic LDs, may be more important (Romanauska and Köhler, 2018). Proximity to the location of gene transcription is speculated to impart additional significance to nuclear LDs. Selective abrogation of nuclear LD formation without affecting cytoplasmic LDs may be needed to clarify this point.
Other Possible Functions
The close association of nuclear LDs with the PML nuclear body (PML-NB; Ohsaki et al., 2016) suggests a tantalizing possibility that nuclear LDs may modify PML-NB functions (Bernardi and Pandolfi, 2007; Lallemand-Breitenbach and de The, 2018). PML-NBs are thought to be made by ring-like shells of PML protein oligomers, and they recruit many partner proteins to the center of the ring mainly through SUMO–SUMO interactions (Sahin et al., 2014). Interestingly, nuclear LDs are often located right in the middle of PML-NBs (Ohsaki et al., 2016) and may thus obliterate the space for partner proteins. It is also intriguing that PML-NBs have been suggested to be membrane-less organelles generated through phase separating condensation (Banani et al., 2017). This means that a lipid droplet (a nuclear LD) exists in the middle of a liquid droplet (PML-NB); this arrangement may influence the structural property of PML-NBs.
Functionally, PML potently activates fatty acid oxidation through the peroxisome proliferator-activated receptor (PPAR) signaling pathway (Carracedo et al., 2012; Ito et al., 2012). Nuclear LDs may play a role in fine-tuning this PML-NB function by regulating the fatty acid concentration in its vicinity. In a similar vein, the cholesterol synthesis intermediate 14-demethyl-14-dehydrolanosterol (also called follicular fluid meiosis-activating sterol, FF-MAS), which is a ligand for liver X receptor α, was shown to be stored as an ester in nuclear LDs (Gatticchi et al., 2017). Nuclear LDs may also be involved in proteasomal protein degradation in PML-NBs, in a similar manner as cytoplasmic LDs (Ohsaki et al., 2006; Welte and Gould, 2017). It is currently unknown whether nuclear LDs in mammalian cells interact directly with genomic DNA or DNA-binding proteins, as reported in bacteria (Zhang and Liu, 2017) and Drosophila melanogaster (Li et al., 2012).
Another possible significance of the nuclear LD and PML-NB association is related to virus infection. On one hand, PML-NBs play a major role in intrinsic and innate immune defense against virus infection (Scherer and Stamminger, 2016). On the other hand, cytoplasmic LDs are exploited by viruses as a site of replication, a lipid source, and other purposes (Cheung et al., 2010; Heaton and Randall, 2010; Meyers et al., 2016; Viktorova et al., 2018; Laufman et al., 2019). In this respect, it is of note that several viruses appear to target and affect nuclear LDs. Human adenovirus type 5 dissolves PML-NB using its E4 Orf3 protein, which directly interacts with the PML-II domain that is crucial for nuclear LD formation (Leppard et al., 2009). Hepatitis C virus core protein, which distributes to LDs when expressed alone, binds PML-NBs and inactivates their apoptosis-inducing function (Herzer et al., 2005). Expression of VP8, a tegument protein of bovine herpesvirus 1, targets PML-NBs and increases nuclear LDs (Zhang et al., 2015, 2019). These results suggest that nuclear LDs may be utilized by viruses to evade host defense mechanisms.
Concluding Remarks
In this article, we discussed nuclear LDs and their close relationship with INM, considering both hepatocytes and budding yeast in parallel. Little is known about not only nuclear LDs but also type I NR, which is difficult to study by conventional methods. Moreover, the wide heterogeneity of the nuclear membrane proteome in different tissues (Korfali et al., 2012) and the difference between budding yeast and mammalian cells, including phospholipid distribution (Tsuji et al., 2019), preclude the extrapolation of results between cells/species. Nevertheless, just as split GFP technology made it possible to identify proteins in the INM (Smoyer et al., 2016), the development of new methodology should solve the mysteries of nuclear LDs. More surprises are expected to come in this understudied realm.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Grants-in-Aid for Scientific Research from the Japan Society of the Promotion of Science (KAKENHI) to Y. O. (18K06829) and T. F. (15H05902 and 18H04023).
