Abstract
Cervical cancer is the fourth most common cancer among women worldwide, with 660,000 new cases and 350,000 deaths reported in 2022. This disease mainly affects women in low- and lower-middle-income countries (LMICs), where 90% of cases occur. This study explored how LMICs are responding to the World Health Organization’s (WHO) call to eliminate cervical cancer. It looked at cervical cancer rates, the use of human papillomavirus (HPV) vaccines, and suggested strategies to fight the disease. The study analyzed the rates of cervical cancer and deaths in LMICs, the effectiveness of HPV vaccination programs, and how well screening efforts are working. To compare data across countries, it used age-standardized incidence rates (ASIRs) and age-standardized mortality rates (ASMRs) per 100,000 women per year. In 2022, the ASIR was 25.4% in lower income countries and 16.9% in lower-middle-income countries. The ASMR was also higher in lower income countries (18.5%) compared to lower-middle-income countries (10.3%). HPV vaccine coverage remains low in many LMICs, with significant differences between the rates of first and final doses completed. By 2050, the number of new cervical cancer cases is expected to rise. Although the WHO aims to eliminate cervical cancer by achieving 90% HPV vaccination by age 15, 70% screening coverage, and 90% access to treatment, many challenges remain in LMICs. These include weak healthcare systems, poor infrastructure, and a lack of health education. Overcoming these challenges through better vaccine distribution, improved screening programs, and stronger healthcare systems is essential to eliminate cervical cancer.
Plain language summary
Cervical cancer is the fourth most common cancer in women worldwide, with 660,000 new cases and 350,000 deaths in 2022, mostly in low- and middle-income countries (LMICs). This review highlights the high disease burden in LMICs, where cervical cancer rates and deaths are significantly higher than in wealthier nations. HPV vaccination coverage remains low, and screening efforts are limited due to weak healthcare systems and poor infrastructure. By 2050, new cases are expected to rise. Achieving the WHO’s goal to eliminate cervical cancer requires stronger healthcare systems, improved vaccine distribution, and effective screening programs.
Background
Cervical cancer is a form of cancer that develops in the cervix, the lower portion of the uterus that attaches to the vagina. It is predominantly caused by persistent infection with high-risk strains of human papillomavirus (HPV). 1 With an estimated 660,000 new cases and 350,000 deaths in 2022, cervical cancer ranks as the fourth most frequent cancer in women worldwide. 2 The primary etiology of cervical cancer in females is the human papillomavirus. Oral contraceptives, diminished immunity, tobacco use, alcoholism, early sexual initiation, and multiple sexual partners constitute supplementary risk factors. Genetic predisposition significantly influences the etiology of cervical malignancies. The majority of women in low- and lower-middle-income countries (LMICs) do not know anything about cervical cancer, so when it does occur, it comes as a complete shock. 3 HPV testing for cervical screening is usually followed by histological evaluation, often augmented by radiological imaging. 4 Persistent infection with high-risk HPV strains is the primary cause of cervical cancer, accounting for nearly all cases.5,6 Cervical cancer is the fourth most prevalent malignancy among women and is the leading cause of female fatalities in LMICs. 7 In LMICs, cervical carcinoma persistently represents a significant public health concern owing to the inadequate accessibility of preventive measures, screening protocols, and therapeutic interventions. The economic burden of cervical cancer is significant, with treatment costs that can range from a few hundred dollars to as much as 70,968 international US dollars in certain instances. 8 Additionally, the clinical presentation of cervical cancer in these regions frequently occurs at advanced stages, which complicates treatment outcomes. 9 The most common type of cervical cancer, squamous cell carcinoma, is typically detected at an advanced stage. 9 A need for earlier detection strategies is indicated by the fact that the mean age of diagnosis is higher than the global average. 9 Cervical cancer screening coverage is substantially influenced by socioeconomic disparities, as many women are unable to access essential services. 10 Vaccination against HPV is essential; however, the availability and utilization of this vaccine differ significantly between higher income countries and LMICs.6–11 Early detection and management have been improved by the transition from traditional cytology to more sensitive HPV-based assays in screening methods. 11 A multidisciplinary approach is essential for effective management, as treatment modalities include chemotherapy, radiation therapy, and surgery. 5 Surgical options, such as hysterectomy, can result in 5-year overall survival rates exceeding 98% for early-stage cervical cancer. 5 The purpose of this study is to evaluate how LMICs are reacting to the WHO’s call to end cervical cancer by (1) analyzing the actual human impact of cervical cancer by calculating the number of women who are afflicted and lose their lives to the disease in various LMICs, (2) investigating how successfully HPV vaccination programs are being implemented in these countries, (3) analyzing the various strategies and solutions that LMICs are trying to put in place to eliminate cervical cancer. This includes looking at practical challenges like vaccine availability, storage issues, and whether girls and women can actually access these life-saving vaccines and examining innovative approaches that work within the constraints of limited healthcare resources. The goal is to speed up the process of eliminating cervical cancer by identifying what is working and what is not, particularly in places where healthcare systems face significant challenges.
Methodology
This narrative review aimed to analyze trends in cervical cancer, evaluate the use of HPV vaccines, and examine screening methods in LMICs. To enable comparisons between countries, data were standardized utilizing age-standardized incidence rates (ASIRs) and age-standardized mortality rates (ASMRs) per 100,000 women per year. Specific ASIR and ASMR figures for the year 2022 were obtained from the Global Cancer Observatory (GCO) 12 database, maintained by the International Agency for Research on Cancer (IARC). The ASIR and ASMR measurements were obtained from the “Cancer Today” data visualization tool of the GCO database. To extract data regarding only LMICs and cervical cancer, applicable filters were chosen. 12 Countries were classified as low- or lower-middle-income according to the World Bank’s Income Classification for the fiscal year 2025. 13 Information regarding HPV vaccination rates for both the initial and final doses among females aged 9–14, as well as for the overall adult female population in the year 2023, was collected from WHO immunization data sources. 14 Projections for new cervical cancer cases through to 2050 were also evaluated for both low- and lower-middle-income nations using data sourced from the GCO database, applying the “Cancer Tomorrow” data visualization tool. 12
This narrative review compiled information from a variety of sources. The criteria for selecting literature were based on several essential principles. First, literature was selected for its direct relevance to the main themes of the review: trends in cervical cancer, the use of HPV vaccines, and the methodologies and challenges of screening, particularly concerning LMICs. Second, priority was given to data and publications from reputable international health organizations, including the World Health Organization (WHO), the International Agency for Research on Cancer (IARC), and the GCO, especially for statistics related to incidence, mortality, and vaccination coverage; peer-reviewed studies and official reports from these organizations were crucial. Third, to maintain an “updated review,” greater emphasis was placed on recent publications (generally from the last 5–10 years, barring landmark studies) and influential research providing thorough insights. Fourth, the selection aimed to offer a comprehensive overview of the current scenario, addressing pathogenesis, prevention, treatment, ongoing challenges, and future prospects. Database searches in PubMed and Google Scholar, utilizing general keywords like “cervical cancer,” “Human Papillomavirus,” “HPV vaccination,” “screening,” and “LMICs,” complemented information from these primary organizational sources. Since this is a narrative review, a systematic search protocol such as Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) was not implemented, a fact also acknowledged in section Limitation.
The impact of cervical cancer on LMICs
The year 2022 witnessed an anticipated 662,301 cases of cervical cancer, along with 348,874 mortalities, documented on a global scale; the age-standardized incidence rate was recorded at 14.1 cases per 100,000 women-years, while the mortality rate was noted at 7.1 deaths per 100,000 women-years. 15
Among the spectrum of gynecological malignancies, cervical cancer constitutes the predominant etiological factor for cancer-related death in LMICs. Infection with oncogenic human papillomavirus strains is a prominent cause of invasive cervical cancer. Since HPV is a virus transmitted through sexual contact, certain factors such as early marriage, having multiple sexual partners, and engaging in unprotected sexual practices elevate the likelihood of the incidence of cervical cancer. 16
According to the World Bank, for the 2025 fiscal year, if a country’s GNI per capita (calculated using the World Bank Atlas method) is $1145 or less in 2023, then it is defined as a low-income country, whereas for a lower-middle-income country, the GNI per capita is between $1146 and $4515. 13
Within different lower-middle-income countries, ASIRs show that the newly discovered cases of cervical cancer vary greatly, from as high as 95.9/100,000 in Eswatini (95.9), to over 50/100,000 in Tanzania (64.8), Lesotho (60.5), Comoros (52), to ASIR even less than 6/100,000 in Tunisia (5.3), Egypt (2.8), Iran (2.5) (Table 1). 15 A comprehensive graphical representation of the age-standardized cervical cancer incidence and mortality rates per 100,000 women-years in low-income economies in 2022, arranged in decreasing order of incidence, is provided in Figure 1. 12
The impact of cervical cancer on economies with low incomes in 2022.
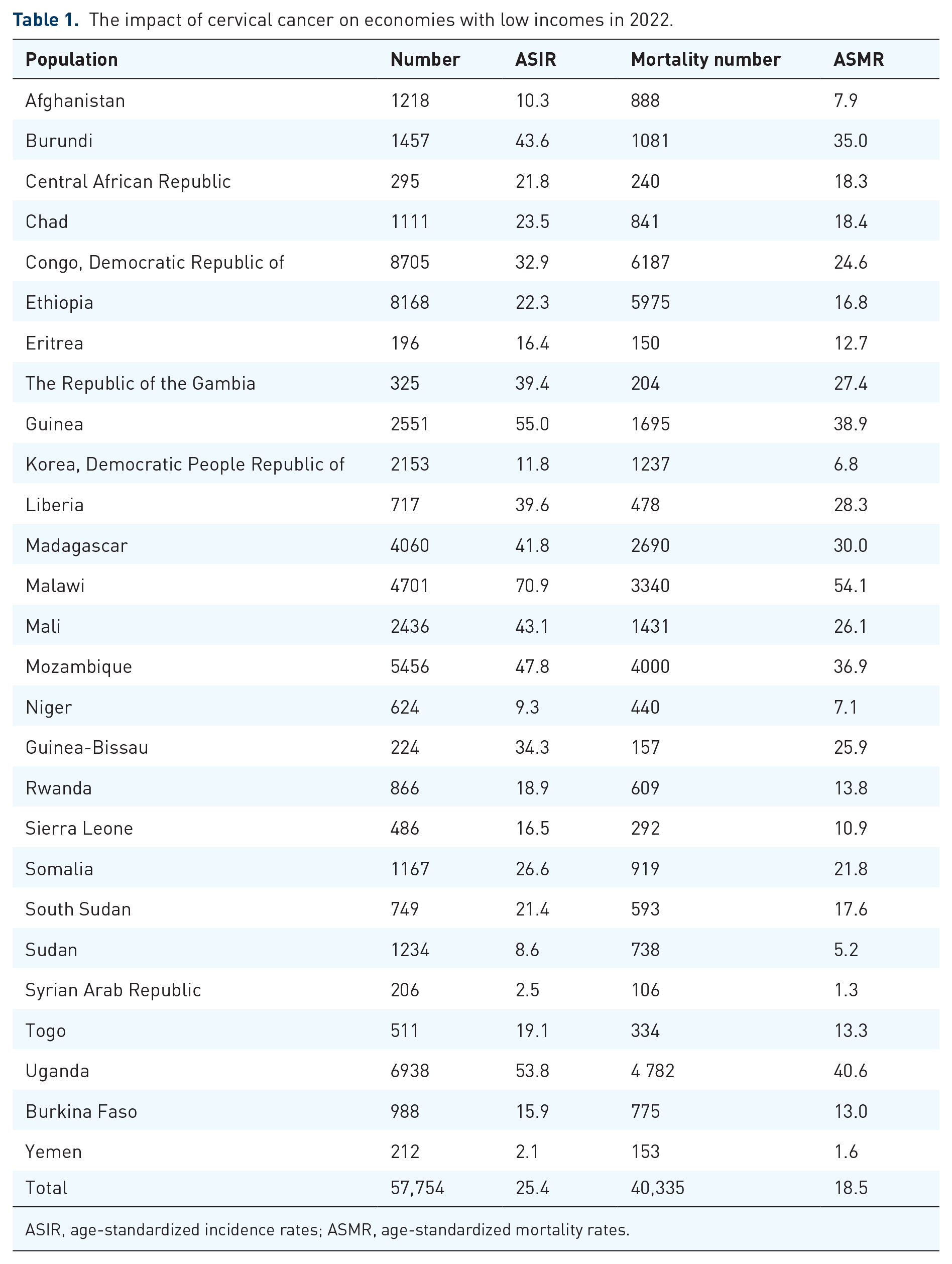
ASIR, age-standardized incidence rates; ASMR, age-standardized mortality rates.
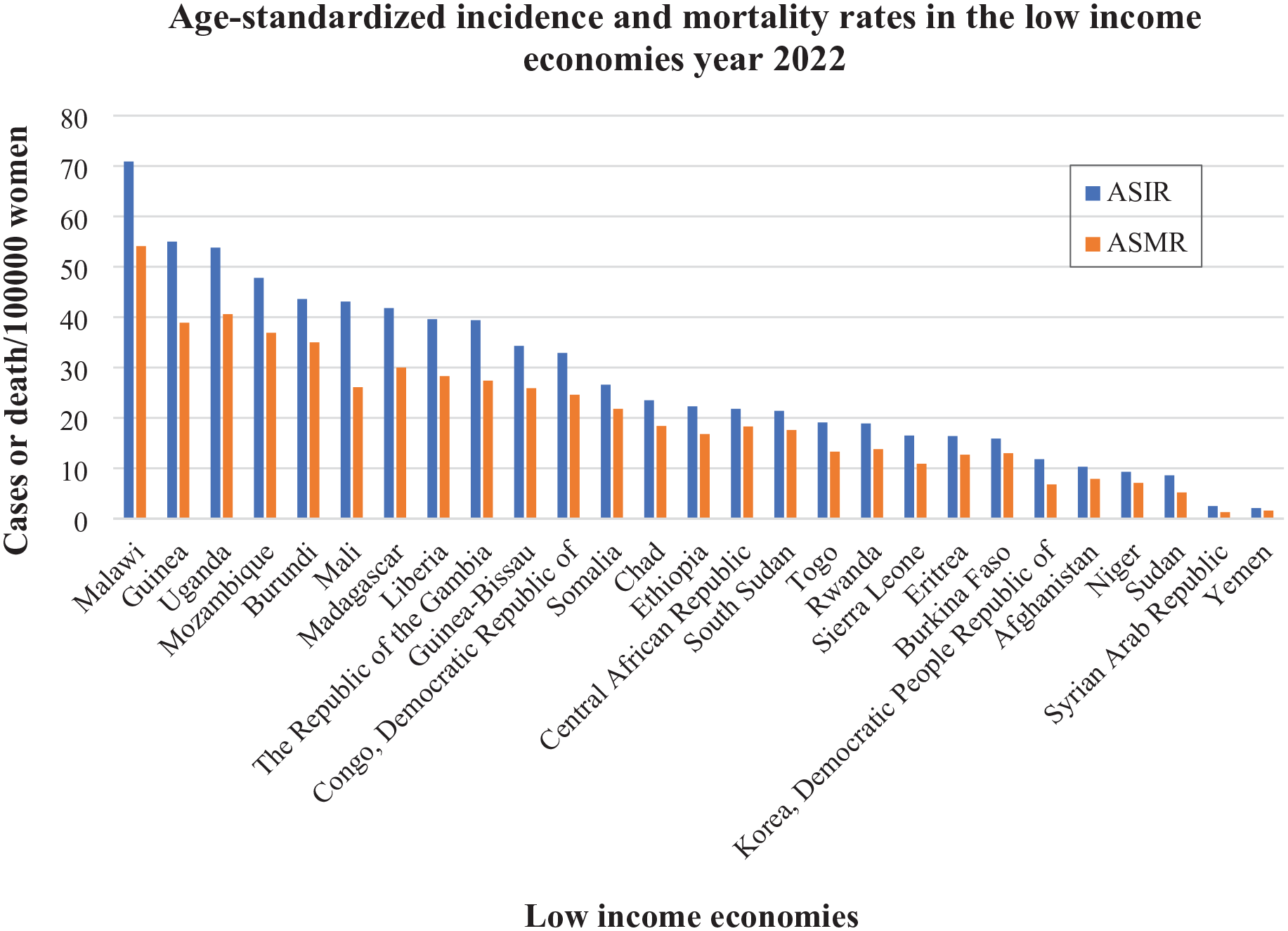
Globally, age-standardized rates of cervical cancer incidence and mortality cases per 100,000 women-years in low-income economies in 2022, displayed in decreasing order of incidence (as per descending order of ASIR). 12
When comparing the ASMRs of developing countries with low-income economies, the death rates exhibit significant variation, ranging from over 30/100,000 in Malawi (54.1), Uganda (40.6), Guinea (38.9), Mozambique (36.9), Burundi (35) to less than 7/100,000 in Korea Democratic People’s Republic (6.8), Sudan (5.2), Yemen (1.6), and Syrian Arab Republic having the lowest ASMR of 1.3/100,000 women-years (Table 2). 15 A thorough graphical visualization of the age-standardized cervical cancer incidence and mortality rates per 100,000 women-years in lower-middle-income economies in 2022, arranged in decreasing order of incidence, is provided in Figure 2. 12
The impact of cervical cancer in economies with lower-middle incomes in 2022.

ASIR, age-standardized incidence rates; ASMR, age-standardized mortality rates.

Worldwide age-standardized cervical cancer incidence and mortality rates per 100,000 women-years in lower-middle-income countries in 2022, arranged in decreasing incidence order (as per descending order of ASIR). 12
Cervical cancer: Identification, analysis, and elimination
Screening
LMICs have the greatest incidence and fatality rates of cervical cancer. 2 It remains the primary cause of cancer death among women in LMICs, where 90% of cases occur, despite the effectiveness of cervical cancer control programs in high-income nations, where widespread, high-quality screening has been in place for decades. 17 With nearly 660,000 new cases in 2022, cervical cancer is the fourth most common cancer in women globally. About 94% of the 350,000 cervical cancer deaths that year occurred in LMICs. Southeast Asia, Central America, and sub-Saharan Africa have the greatest incidence and fatality rates of cervical cancer. Inequalities in access to immunization, screening, and treatment facilities, risk factors like HIV incidence, and social and economic determinants like sex, gender bias, and poverty are all linked to regional variations in the burden of cervical cancer. It is six times more likely among women with HIV than in the general population, and HIV is considered to be responsible for 5% of all cervical cancer cases. 18 Since cervical cancer disproportionately affects younger women, 20% of children who lose their mother to the disease do so as a result of cervical cancer. 19
Pathogenesis
The most typical way that HPV infection is spread is through sexual contact, while it can also spread through nonsexual encounters. Women are disproportionately impacted, particularly those who have several sexual partners. Perinatal transmission of other viruses or microorganisms can also result in HPVs being passed from mother to child. There are three primary areas of the cervical mucosa: the ectocervix, transformation zone, and endocervix. Through microabrasions or microwounds, HPVs penetrate the epithelium and attack basal cells. They serve as stem cells and target actively dividing basal-layer cells. HPV penetrates target cells through endocytosis after cellular receptors attach to the HPV L1 capsid protein. Within 24 h, the virus penetrates the nucleus through post-endocytic trafficking, and the tubulin-mediated route transports the viral DNA there. 20 There are currently over 450 different genotypes of HPVs (a stable double-stranded DNA virus with a small genome size ranging from 42 to 55 nm) based on the nucleotide sequence relatedness of the encoded major capsid protein L1 into alpha, beta, gamma, mu, and nu genera. In addition to two late genes, structural major L1 and structural minor L2, and a noncoding region that contains all the regulatory elements for transcription and the origin of viral replication, known as the long control region (LGR) or the upstream regulatory region (URR), these genotypes include the long control region (LCR), which regulates genome replication and transcription of six early genes, namely E1, E2, E4, E5, E6, and E7, as well as a noncoding region that contains all the regulatory elements for transcription and the origin of viral replication. 60% of cases are resolved in a year, and 90% of cases are resolved in 2 years. The host immune response clears most HPV infections in a varied period of time, ranging from a few months to 2 years. Unfortunately, the host cannot detect the viral protein and is unable to establish a strong cell-mediated immunity or a sufficient innate immune response after the initial HPV infection has been eradicated (escaping immune detection). 21 Despite becoming less prevalent in high-income countries, CC continues to be the primary cause of cancer-related fatalities among women in LMICs, according to the World Health Organization (WHO). Age-standardized mortality rates are lower in Western Europe, North America, Australia, New Zealand, and Western Asia than in South, East, and West Africa, as well as Melanesia. 22 The suggested worldwide goals are 90% vaccination coverage for girls under the age of 15, 70% HPV testing coverage for women between the ages of 35 and 45, and 90% treatment coverage, including palliative care. 22
Integrating early diagnosis and prevention through HPV vaccination
Early detection of cervical cancer is best achieved through routine screening. Cervical cancer can be detected using the HPV and Pap tests. These tests can be performed alone or in tandem, sometimes known as a co-test. It has been shown that regular screening can prevent cervical cancer and save lives. The odds of successfully treating cancer and precancers are significantly increased by early identification. Avoiding delays in diagnosis can also be achieved by being aware of any cervical cancer symptoms. 23 This awareness is a key to curing cervical cancer.
Since more than 80% of occurrences of cervical cancer occur in LMICs, the timetable for eliminating the illness there is longer. 24 Lack of equipment, logistical and financial difficulties, high rates of loss to follow-up, and inadequate funding for specialist training are some of the obstacles that successful screening programs in LMICs must overcome. 25 Though organized screening programs at the population level have decreased cervical cancer incidence by 50% to 80%. 26 The majority of malignancies occur in people who are new to care or are rarely screened in settings with strong screening systems.27,28 For instance, the screening results from the Demography and Health Survey from 2010 to 2019. Out of 16 nations, only three (Colombia, Cameroon, 2018; Benin) received information about the results of the most recent screening test and subsequent visits for women who had abnormal results. Information regarding treatment for individuals with positive or abnormal test results was only available in Colombia. 7.9% of women in Benin, 4.6% in Colombia, and 1.6% in Cameroon had abnormal or positive test results among those who had cervical cancer screenings. A follow-up visit was performed by 100.0% of individuals with positive or abnormal results in Benin, compared to 72.6% in Cameroon and 49.2% in Colombia. In Colombia, 33.6% of patients who sought follow-up care due to abnormal or positive screening results were treated in some way. 10
If a woman experiences atypical bleeding, excessive vaginal discharge, chronic pain, exhaustion, weight loss, vaginal discomfort, or leg edema, she should consult a doctor. After a diagnosis is confirmed by clinical assessments and testing, patients are referred for treatment options such as palliative care, chemotherapy, radiotherapy, and surgery. With features like a multidisciplinary team, treatment decisions that follow national guidelines, and holistic psychological, spiritual, physical, and palliative care, management pathways for invasive cancer care provide timely referral and support. As cervical screening increases in LMICs, more invasive cervical cancer cases will be detected, especially in previously unstudied groups. Thus, strategies for cancer management and referral must be established in addition to prevention. 2 Early identification of precancers and cervical cancer is advised by the American Cancer Society. A primary HPV test should be performed every 5 years for anyone between the ages of 25 and 65. Screening should begin at age 25. If primary testing is not accessible, a Pap test or co-test may be used. If there is a history of major precancer, testing should continue for at least 25 years. DES exposure or immune system suppression may necessitate more frequent screening for high-risk people. 29
Improving information availability and public awareness is essential for HPV prevention and control. Cervical cancer and HPV infection can be avoided by vaccination between the ages of 9 and 14. Beginning around age 30, screening can identify and cure cervical illness. Six HPV vaccines are currently available worldwide as of 2023, with girls ages 9–14 receiving priority. In order to lower the prevalence of HPV, boys should also receive vaccinations. Voluntary male circumcision, condom use, and quitting smoking are further preventative strategies. 30
The HPV vaccine prevents the majority of cases of genital warts and cervical cancer. The vaccine gives the body a safe way to raise immune system awareness of certain HPV strains. This suggests that a person’s body will be able to eliminate specific viral strains more easily if they are contracted later. The Food and Drug Administration (FDA) of the United States has authorized the Gardasil nine vaccine. People 9 years of age and up can take it. It is possible to administer this vaccine concurrently with other vaccinations. Routine HPV vaccination is advised by the Centers for Disease Control and Prevention (CDC) at the age of 11 or 12. Getting vaccinated before engaging in sexual activity is the best course of action.
People under the age of 15 can receive two doses, six to twelve. People under the age of 15 can receive two doses spaced six to twelve months apart. For people who start the vaccination series later, between the ages of 15 and 26, three doses of the vaccine are advised. These immunizations last for 6 months.
In contrast, during pregnancy, the HPV vaccine is not administered. If a person has severe, life-threatening allergies or has an allergic reaction following their initial HPV vaccination, they should not get the HPV vaccine. Additionally, those who are moderately or seriously ill should postpone getting a HPV vaccination until they are well. 31
The World Health Organization (WHO) has also just approved the use of Cecolin, a fourth HPV vaccine agent, in a single-dose regimen. Based on new research, this decision satisfies WHO’s 2022 recommendations for the alternative, off-label use of HPV vaccines in single-dose schedules. As part of the WHO’s global effort to eliminate cervical cancer, 90% of girls should have received all recommended immunizations by the age of 15. 32
Continuing struggles in tackling cervical carcinoma in low- and lower-middle income countries
If 90% of girls are vaccinated by the age of 15, 70% of women between the ages of 35 and 45 are screened, and 90% of cervical cancer patients in LMICs receive treatment, cervical cancer can be eradicated. Today, cervical cancer can be prevented with the HPV vaccine and screening, and it can be effectively treated with chemotherapy, radiation therapy, and surgery. Still, more than 500,000 new cases were reported globally. 33 Every year, half a million women worldwide are affected by cervical cancer, and half of them pass away. Countries with poor and moderate incomes have a disproportionately high prevalence of the condition. 34 Due to a lack of infrastructure, qualified medical services, health education, and logistics, women in LMICs frequently do not have access to preventative vaccinations and screening. To guarantee that everyone has access to HPV vaccination, these problems must be resolved. In certain nations, screening services must be set up, and qualified experts must be on hand. Cervical cancer prevention requires a multifaceted fight that includes introducing new technology, updating health education, and increasing public awareness. To overcome these obstacles, cooperation between governments, academics, the commercial sector, and civil society is crucial. Successful HPV testing deployment depends on laboratory preparedness, but it also requires a quality culture with consistent training, strong monitoring, and quality assurance procedures. 35 In LMICs, self-sampling testing, single-dose HPV vaccine, and new technology are effective ways to prevent cervical cancer. For secondary prevention and management, cooperation is required. 36 Experts point to risk factors, community-based social entrepreneurship, and socioeconomic inequality as important problems in LMICs, especially when it comes to rural–urban discrepancies. 37 Age, history of alcohol use, marital status, reproductive disorders, educational attainment, and live births are risk factors for HPV infection. In remote locations, women over 30 should have access to cervical cancer screening. 38 Expanding HPV self-sampling social entrepreneurship projects may be made easier with further study on the relationship between worker performance and empowerment. 39
Numerous real-world studies have shown the safety and efficacy of the HPV vaccination program in preventing and treating HPV infection and related disorders since June 2006, when the vaccines were initially authorized in the United States. However, there are a number of obstacles, such as the high cost of vaccines, their inaccessibility, and the unsuitable conditions for storage or shipment. Furthermore, in the majority of LMICs, the general public is unaware of national immunization programs and diseases linked to HPV. The fact that HPV vaccines do not offer protection against all forms of HPV is one of the primary obstacles to their widespread use. 40
Despite the considerable outreach to a significant cohort of eligible females by numerous LMICs, additional efforts are necessary to eliminate misinformation, complete the vaccination regimens, ascertain the target demographic, monitor the efficacy of the program, and ensure its long-term viability. Overall, certain problems remain unresolved and require additional research. 41
Efficient strategies for the prevention and management of HPV-related diseases, including regular screenings, health regulations, ongoing patient care, and initiatives for public education and awareness, are lacking in LMICs. 42 Therefore, failing to fully comprehend eligibility requirements may result in a missed immunization opportunity or an unintended vaccine. For instance, in the first year of the HPV vaccine’s introduction, Zimbabwe faced difficulties in identifying eligible individuals and was beset by a number of issues, such as a lack of social mobilization, inadequate instruments and training, and poor staff transport. According to healthcare professionals, the absence of a qualified team to implement immunization, insufficient financing to give vaccines, and inadequate staff and supply transportation were the top three challenges. 43 Furthermore, there have been claims of an increase in death rates among individuals who have received the HPV vaccine, even though no vaccine-associated deaths have been recorded to date. 44 Also, implementing HPV vaccination programs is complicated by unique economic circumstances, parental mistrust, infrastructure problems, and improper identification of eligible females. Vaccinating girls with HIV and conducting an accurate census of out-of-school girls are further hurdles. Rumors, staff or community resistance, or formal consent processes can all be used to limit school vaccinations. Effective social mobilization and high HPV vaccine uptake have been shown in LMICs in spite of these obstacles. 45 Four low-income economies, fourteen lower-middle-income economies, and six upper-middle-income economies comprised the twenty-four countries from which a meta-analysis conducted between 2008 and 2020. 46 HPV vaccination uptake in LMICs from 2006 to 2020 was 61.69%. In high-uptake countries, the female target population was 77.93%, consisting of 33.32% in the secondary target population and 87.98% in the primary target population. The target population for females was 4.72% in low-uptake countries. The number of girls vaccinated increased from 1.4 million in 2006 to 2014 to a projected 3.3 million in 2020. Only 1% of the 47 million women who were given the entire course, nevertheless, came from LMICs. 47 As of March 2022, 117 nations, representing roughly one-third of the worldwide target population and 60% of WHO member states, had incorporated the HPV vaccination into their regular national immunization regimens.
A considerable number of women, particularly in nations with poor vaccination rates, are thought to be unprotected and live in areas without an adequate cervical cancer screening program, according to the most recent assessment. 48 Seventy-five percent of low-income countries, seventy percent of lower-middle-income countries did not incorporate the HPV vaccine into their national immunization programs in 2021. Bhutan, Bolivia, Brazil, Cambodia, Cameroon, India, Kenya, Nepal, Peru, Rwanda, Uganda, and Vietnam are among the countries that have chosen to use school-based delivery models.49–51
The five “I” framework: Guiding principles for implementation
The five “I” framework for HPV vaccines, which is based on the Consolidated Framework for Implementation Research (CFIR), lists the most important factors that affect how many people get vaccinated against HPV. This framework focuses on how different factors can work together to help or hinder vaccination attempts, especially in communities with a lot of diversity. The parts that follow go into more detail about each of the five domains. 52
An overview of the system: Recognizing the issue at hand is the starting point for any solution. Finding the primary causes and potential outcomes is the subject of the second stage. The third step is to think of creative solutions that are completely fresh to the problem. Implementing the strategy or intervention plan is the subsequent phase. Reviewing the outcomes and evaluating success is the last one. 53
The “intervention” is the new method or practice that is being used, which in this case is the HPV vaccine. It is very important that vaccines like Cervarix® and Gardasil® work because they protect almost 100% of people from certain kinds of HPV that cause cervical cancer. 54
The inner setting domain encompasses the attributes of the organizations or entities administering the immunization. Elements such as resource availability, organizational culture, and implementation readiness significantly influence vaccine uptake. 55 Strategies to improve the inner setting include routinely identifying sources of vaccines at no or low cost and providing continuing education for providers. This education emphasizes the significance of vaccination and effective communication tactics, which are essential for enhancing vaccination rates. 55 ’ 56
The outer setting encompasses the broader community context, including the socioeconomic and cultural environment where the target population resides. Raising community awareness and providing information regarding HPV and its vaccines are essential for surmounting obstacles and enhancing acceptability.55,57 The COVID-19 pandemic has developed a distinctive environment that has influenced the outer setting by requiring the establishment of new partnerships and approaches. To accommodate the changing needs of safety net healthcare systems, these adjustments were necessary for HPV vaccination programs. 56
Healthcare workers and staff who are in charge of giving the vaccine are included in the step that involves people. What they know, how they feel, and what they believe about the vaccine can have a big effect on how many people get it. To make sure that the vaccine is communicated and given correctly, healthcare providers must be trained and given help.55,58 This stage entails identifying all individuals and groups with a vested interest in the subject at hand. This may encompass the individuals, healthcare providers, policymakers, and community organizations involved in HPV vaccination. 58
Challenges associated with the uptake and distribution of the HPV vaccine in LMICs are attributed to the financial implications of the vaccine and its delivery, insufficient prioritization, the complexities involved in administering vaccines to adolescents, and a deficiency in vaccine supply that has been unable to match the swift increase in global demand. Implementation of the five “I” framework endorses innovative strategies aimed at enhancing operationalization of the vaccination program at the national level especially in LMICs. 59
Financing two doses of a comparatively costly vaccination while carrying out monitoring and follow-up to guarantee adequate coverage of the second dose is challenging for LMICs. 60 Certain existing studies suggest that a single-dose administration of the HPV vaccine may offer potentially effective protection against HPV infection in healthy young individuals. 61 Thus, the single-dose HPV vaccination strategy may simplify the prime challenges regarding vaccine cost and distribution in LMICs.
In regions with limited resources, establishing efficient methods for vaccination is hindered due to the limited availability of reliable data. 62 Consequently, keeping track of increasingly precise data in both educational institutions and public settings for small-scale planning and monitoring, along with determining eligible groups, has become a sheer necessity. 63 Information systems and data optimization to align with eligibility criteria can be significantly enhanced by investing in staff training in healthcare and educational institutions. Zambia utilizes online digital recording technology to identify and observe vaccinated girls, categorized by age and method of administration in order to determine vaccination coverage rates. 53 Likewise, other LMICs can also come up with cost-effective ways by incorporating technology with conventional methods.
The five “I” framework gives us a complete picture of how to apply the HPV vaccine, but it is important to remember the problems and restrictions that come with each domain. For example, the long-term effectiveness of vaccines and the need for ongoing education and awareness campaigns are both very important problems that need constant study and attention. 57
Contribution of HPV vaccination to cervical cancer prevention in low- and lower-middle-income regions
LMICs must prioritize HPV vaccination because cervical cancer is still a big problem there. By increasing vaccine availability, we can diminish the effects of cervical cancer and enhance public health in these regions by greatly lowering the rates of morbidity and mortality associated with cervical cancer. Following the launch of HPV vaccines in 2006, countries have progressively included the vaccine in their national immunization schedules. The WHO first recommended HPV vaccines in 2009, employing a three-dose regimen for girls aged 9–14 years. 2 As of 2023, six HPV vaccinations are available worldwide. All provide protection against the high-risk HPV types 16 and 18, which are responsible for the majority of cervical malignancies, and have demonstrated safety and efficacy in preventing HPV infection and cervical cancer. 2 Recent evidence indicates that a single-dose regimen may offer comparable protection to two or three administrations, with sustained antibody levels maintained for up to 16 years. 64 Prophylactic vaccines effectively prevent new infections and reinfections, but do not eliminate existing infections. 65 Therapeutic HPV vaccines are being developed to elicit immunity against existing infections and lesions, perhaps averting cancer progression. DNA-based vaccines are specifically promising because they can stimulate both cellular and humoral immune responses, providing enduring immunity. 52
The HPV vaccination is crucial for preventing cervical cancer, yet uptake among girls varies significantly across regions. Here, the graphs illustrate HPV vaccine coverage among females under 15 years and the whole female population, classified by country.
Figures 3 and 4 highlight the HPV vaccine coverage for females under 15 years across various countries, distinguishing between the first and last doses.

Projections for 2023 regarding the HPV vaccine uptake among women aged 9–14 in low- and lower-middle-income countries, with at least one dose received, are listed in alphabetical order by country, based on the information sourced from the WHO Immunization Data portal. 14

Projections for 2023 regarding the HPV vaccine uptake among women aged 9–14 in low- and lower-middle-income countries, with full recommended dose received, are listed in alphabetical order by country, based on the information sourced from the WHO Immunization Data portal. 14
The initial vaccination uptake is high in many countries (e.g., Mexico, Sri Lanka, and Palau, at 99% for the first dose), but it drops significantly for the last dose. Palau, for example, falls from 99% to 79%. Some countries, like Mexico and Sri Lanka, maintain exceptionally high coverage for both doses, suggesting strong vaccination completion rates. Countries such as Indonesia and Jamaica show extremely low coverage for both doses (1% and 5% for the first dose in Indonesia and Jamaica, respectively), with minimal improvement for the last dose. Certain countries experience steep declines between the doses. For instance, Kenya drops from 29% to 17%, and the Bahamas from 15% to 6%. While some countries maintain moderate coverage without a significant decline. For example, Ecuador drops only slightly from 90% to 86% (Tables 3 and 4) (Figures 3 and 4). 14
HPV vaccination under 15 years (first dose) year 2023. 14
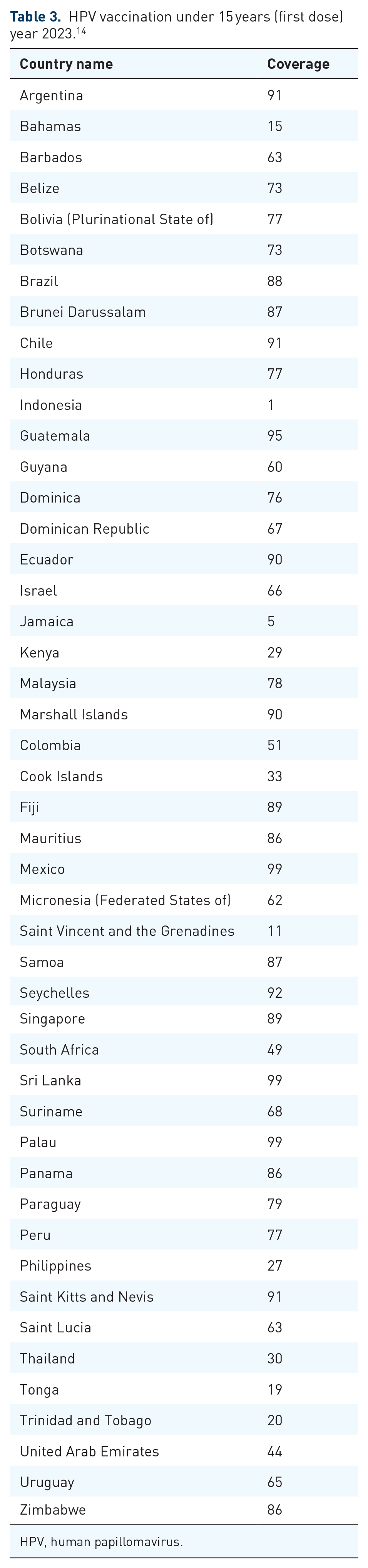
HPV, human papillomavirus.
HPV vaccination under 15 years (last dose) year 2023. 14 .
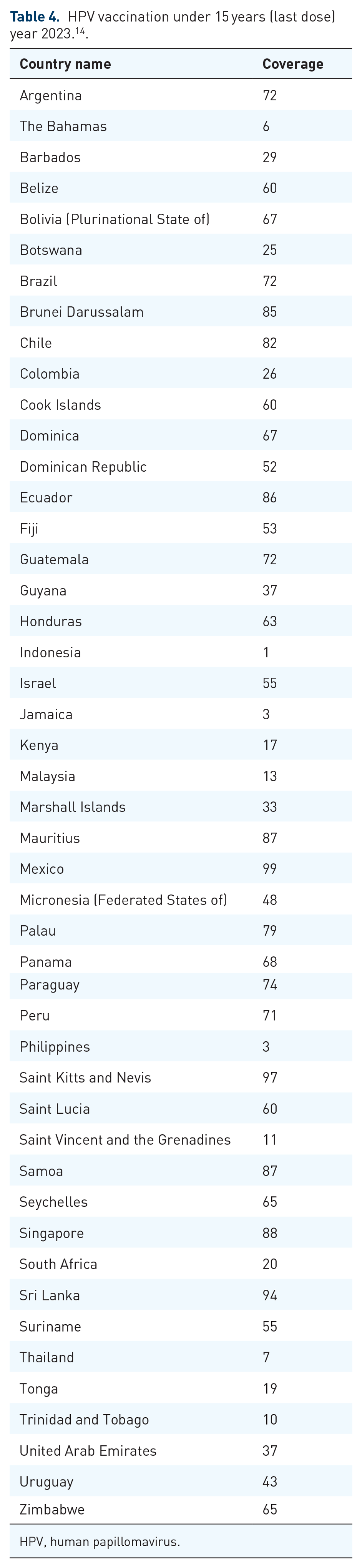
HPV, human papillomavirus.
Figures 5 and 6 depict HPV vaccine coverage among the overall female population across various countries, distinguishing between the first and last doses. Effective vaccination programs are demonstrated by Uganda, Tanzania, and Burkina Faso, which exhibit excellent first-dose coverage (97%–99%) and sustain high levels for the last dosage. While first-dose coverage is high in Bhutan, Cambodia, and Rwanda (87%–91%), last-dose completion rates are much lower. Poor retention following the first dose indicates obstacles to finishing the immunization series in nations like Lesotho (44% to 25%) and Eritrea (27% first dose to 14% last dose). Countries with low vaccination uptake or limited accessibility include Bangladesh (21% first dose) and Mauritania (18% first dose, further falling). Solomon Islands, Sao Tome and Principe, and Tuvalu maintain comparatively constant numbers between doses, indicating effective follow-up procedures. It can be observed that a notable gap exists between the first and last doses in many countries, highlighting potential challenges in maintaining vaccine completion. LMICs generally show lower completion rates compared to higher income nations. Some countries exhibit exceptionally high initial coverage but face drop-offs for the last dose (Tables 5 and 6) (Figures 5 and 6). 14

Projections for 2023 regarding the HPV vaccine uptake among adult women in low- and lower-middle-income nations who are receiving only the initial dose based on the information sourced from the WHO Immunization Data portal. 14
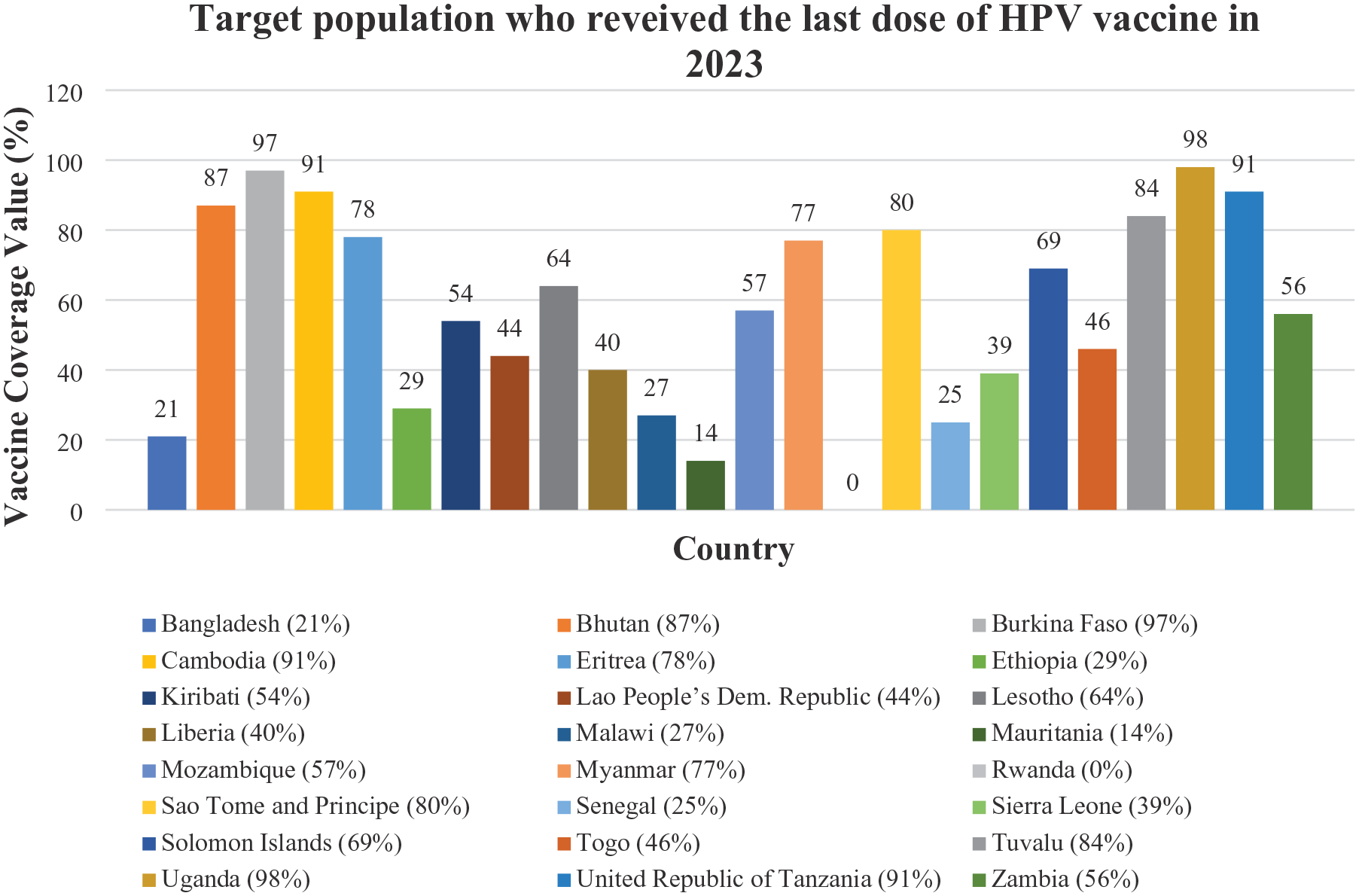
Projections for 2023 regarding the HPV vaccine uptake among adult women who complete the full recommended dosage in low- and lower-middle-income nations, based on the information sourced from the WHO Immunization Data portal. 14
HPV vaccination in adult women (first dose) year 2023. 14

HPV, human papillomavirus.
HPV vaccination in adult women (last dose) year 2023. 14

HPV, human papillomavirus.
Future projections for HPV vaccination and new cases in LMICs
Potential scientific and manufacturing advancements are emerging that could significantly impact future HPV vaccination efforts. A critical aspect is the growing body of evidence regarding the sufficiency of a single dose of the HPV vaccine in securing long-term immunity. Analyses of extant opportunistic data have indicated the potential benefits of this vaccination approach and have catalyzed the commencement of clinical trials aimed at a more rigorous assessment. Should the findings validate the effectiveness of a one-dose regimen, the implications could be monumental—diminishing the costs associated with vaccine procurement and administration, streamlining logistical operations, and facilitating the provision of gender-neutral services and broader age eligibility. 41
In regions with low- and lower-middle-income economies, access to preventive measures is greatly limited, leading to delays in identifying dysplastic lesions and cervical cancer until they have progressed to advanced stages. The restricted options and methods for treatment further increase mortality rates. Moreover, in resource-poor countries, the growing incidence of HIV, along with reduced funding for antiretroviral treatments, worsens the progression of HPV. A crucial consideration in the future implications of cervical cancer research is the pursuit of global health equity. Efforts to enhance vaccine distribution, develop cost-effective screening methods, and utilize artificial intelligence for diagnostic purposes should align with a commitment to reducing health disparities and improving outcomes for all women, regardless of their geographic location. 66
Projected incidences of new cases are anticipated to markedly escalate in the majority of LMICs between the years 2022 and 2050. The demographic spectrum encompasses a broad range from infancy to individuals aged 85 and above, specifically among the female population. In the context of low-income nations, the Democratic Republic of the Congo, Ethiopia, Uganda, Mozambique, and Malawi are predicted to exhibit the highest incidence rates by 2050, with estimates of 23,098, 22,692, 19,705, 13,608, and 12,688 cases, respectively. Additionally, other nations such as Mali, Somalia, Sudan, Chad, Niger, Togo, Sierra Leone, and Yemen are projected to experience new cases ranging from 6,000 to as low as 400, with Eritrea representing the country with the least estimated incidence (Figure 7). 12

Projections for the HPV vaccine uptake among adult women in low-income nations from 2022 to 2050, based on the information obtained from IARC GCO database. 12
In lower-middle-income nations, projections indicate a significant escalation in the incidence of cervical cancer by the year 2050. Countries such as India, Indonesia, Tanzania, Nigeria, Bangladesh, and the Philippines are anticipated to report the highest incidence rates, with expected new cases numbering 229,056, 50,669, 33,488, 33,138, 17,305, and 15,472, respectively. Additionally, in regions including Angola, Senegal, Nepal, Algeria, Haiti, Lesotho, Comoros, Solomon Islands, Cape Verde, and Belize, the incidence is projected to persistently increase, with estimates varying from approximately 8,000 to as few as 77 cases, wherein Samba is expected to record the lowest future incidence of only 19 cases. Across all aforementioned nations, the emergence of new cases is forecasted to rise, with markedly higher estimates for 2050 in comparison to the year 2022 (Figure 8). 12

Projected HPV vaccine coverage rates for adult women in lower-middle-income nations from 2022 through 2050, based on the information obtained from IARC GCO database. 12
Cervical cancer prevention and management are primarily supported by two key approaches: Pap screening for the early identification of cancerous changes and the implementation of vaccinations to protect against human papillomavirus (HPV), which helps to prevent the progression of cervical cancer. 45
Despite the fact that a large number of LMICs have achieved considerable coverage among eligible female populations, additional efforts are imperative to dispel misinformation, ensure the completion of vaccination series, assess the target demographic, evaluate program efficacy, and guarantee the sustainability of initiatives. Overall, the resolution of certain challenges remains ambiguous and warrants further scholarly exploration. LMICs demonstrate deficiencies in the effective prevention and treatment of HPV-related pathologies, including but not limited to regular screening protocols, comprehensive public health strategies, ongoing patient care, and initiatives aimed at enhancing public education and awareness. Consequently, a limited comprehension of eligibility parameters may result in missed opportunities for vaccination or the misallocation of vaccination efforts. 45
WHO initiatives: Toward the eradication of cervical cancer
Getting rid of cervical cancer is a global health concern. The WHO’s Global Strategy aims to get rid of cervical cancer by focusing on mass screening and vaccination against HPV subtypes that can cause cancer. The goal of this project is to get rid of cervical cancer completely within the next 100 years. It is the first noncommunicable disease to be the target of such an effort. 67 Vaccination strategies include HPV vaccination. For example, by 2030, the WHO wants 90% of girls to have received all recommended vaccinations by the age of 15. 68 And the other is gender-neutral vaccination, which involves expanding vaccination to include boys, which can speed up the process of eradication and improve herd immunity. 69 Screening programs encompass Efficient screening, including HPV testing, which is very essential. The objective is for 70% of women to have screening by the age of 35 and again by 45. 68 The conventional screening technique, the Pap smear, is widely recognized in high-income nations, but in LMICs, this method is constrained by insufficient financial resources. 70 The VIA (visual inspection of acetic acid) screening method is extensively utilized in LMICs due to its cost-effectiveness and minimal resource requirements. 70 Primary HPV DNA testing every 5 years has been proven to be the most successful and cost-efficient screening strategy, resulting in a considerable reduction in cervical cancer mortality. 71 Mobile health (mHealth) projects have demonstrated potential in enhancing awareness and engagement in cervical cancer screening. Mobile health has equipped women with early warning signs and indications, along with enhanced awareness of various risk factors, HPV vaccination, and treatment alternatives.
Additionally, gaining knowledge about health can encourage people to embrace recommended, research-supported behavior changes, like screening, which can lead to a reduction in cancer-related illness and death.72,73 An essential component of efficient therapy is enhancing human capacity through the education of gynecologic oncologists and the division of labor. Integrating surgical systems and improving access to quality treatment requires a multipronged strategy. The capacity-building process has made use of two main strategies: collaborative practice and education for gynecologic oncologists. 74 While tremendous progress has been made, problems still exist, such as the need for long-term infrastructure and regulations to support these projects. Addressing these impediments is critical to reaching the aim of cervical cancer elimination in LMICs.
Limitation
It is important to emphasize that this research was carried out as a narrative review. Rather than using a systematic literature search protocol or following specific guidelines like PRISMA, we selected our cited literature and data sources, which may have introduced selection bias based on the authors’ existing knowledge and viewpoints. While this narrative format provides a general overview of the subject, it may constrain the thoroughness of the review, as pertinent studies or data points could have been unintentionally left out. As a result, the findings should be viewed with the awareness that they reflect a synthesis from a nonsystematic literature survey, which could influence the overall dependability and replicability in contrast to a formal systematic review.
Our analysis is heavily based on information reported by international organizations such as the WHO, IARC, and GCO. The reliability of ASIR, ASMR, and vaccination coverage statistics can vary significantly among LMICs due to inconsistencies in data collection and reporting. There may also be limited data available for certain regions or subpopulations within countries.
Additionally, a notable challenge faced during this study was the inconsistency in income classification across countries. In particular, discrepancies were noted between the categorizations from the GCO, the WHO immunization portal, and the World Bank’s official income classifications for the applicable fiscal year. For example, some countries were designated as “low-income” in one source while being classified as “lower-middle-income” in another. Although attempts were made to follow a specific classification system (e.g., the World Bank FY25 criteria as described in the methods), aligning data from sources that may utilize varying or outdated classification versions poses a limitation. This inconsistency may result in the misclassification of certain countries within the analyses provided (such as in tables that categorize countries by income status) and could impact the accuracy of the aggregate statistics reported for each income category.
Conclusion
The manuscript emphasizes the significant global impact of cervical cancer in terms of disease burden, along with the limited awareness and access to preventive measures for cervical cancer in LMICs. By doing this, it becomes clear that if there is to be any progress, more has to be done to improve vaccine distribution and to make screening and treatment measures more accessible to marginalized populations. The intention is to harmonize gaps in health and increase clinical outcomes among women, no matter the district. The age-standardized incidence rates of cervical cancer per 100,000 women-years in 2022 are 25.4% for lower income countries and 16.9% for lower-middle-income countries. Significant disparities exist between the initial and final dose completion rates in numerous LMICs, resulting in suboptimal coverage of the HPV vaccine. The estimated number of new cases is expected to increase by the year 2050. The manuscript signifies the need for early detection and routine screening of cervical cancer and its consequent spread and need to spread the accessibility of HPV testing suitable care in LMIC. This could help speed diagnosis and improve outcomes with treatment. Further research is needed to substantiate the efficacy of a one-dose protocol of HPV vaccination. If effective, and it has been proven so, it would drastically cut costs and simplify the logistics of vaccination. Our future prevention and treatment efforts with regard to cervical cancer should focus on addressing health inequities. As a result, it calls for gender-equal access to screening and vaccination services everywhere.
