Abstract
Background:
Tuberculosis (TB), caused by Mycobacterium tuberculosis (Mtb), is an ancient disease that continues to pose a significant threat to global public health. Although the BCG vaccine, developed in the 1920s, remains the only approved TB vaccine, it has limited efficacy, particularly against pulmonary TB in adults. The ID93/GLA-SE vaccine, a recombinant subunit vaccine, shows promise by triggering immune solid responses and could be a key solution in combating TB, particularly in the face of rising drug-resistant strains and suboptimal current vaccines. It has the potential to address the unmet need for more effective interventions against drug-resistant TB, a growing global health issue that continues to challenge existing treatment options.
Objective:
To evaluate the immunogenicity and safety of ID93 + GLA-SE in BCG-vaccinated healthy adults.
Methods:
A comprehensive electronic search on PubMed (Medline), ScienceDirect, EMBASE, Scopus, and Cochrane Central database was conducted from inception till August 2024 for randomized controlled trials (RCTs) with a target population of BCG-vaccinated healthy adults. This review was conducted according to (PRISMA) criteria and registered with PROSPERO (CRD42024601450). This meta-analysis used Review Manager and forest plots for visual display. The outcomes were displayed as risk ratios (RR) with a 95% confidence interval.
Results:
The ID93 + GLA-SE vaccine showed strong immunogenicity, particularly in high doses, with robust IgG responses sustained up to day 421 in all studies, significantly higher than baseline, and seroconversion rates remained high through day 84. CD4 T-cell responses peaked after the third dose and remained elevated through day 421, whereas CD8 T-cell responses were minimal. Regarding adverse effects, the ID93 + GLA-SE vaccine significantly increases fatigue (RR 3.24, p = 0.005), myalgia (RR 5.82, p < 0.0001), and injection site pain (RR 4.12, p < 0.00001), compared to placebo, with consistent results across both high and low doses. However, there were no significant differences for upper respiratory tract infections, 0.83 (95% CI 0.38–1.84, p = 0.87) or 1.77 (95% CI 0.77–4.10, p = 0.18) headaches. Dose optimization remains crucial due to the higher side effect risks of increased doses.
Conclusion:
The ID93 + GLA-SE vaccine shows a solid safety profile and enhances immune responses, especially IgG and CD4+ T-cell activity, which is crucial for TB defense. Higher doses improve efficacy but increase side effects, highlighting the need for dose optimization. As a potential alternative to the BCG vaccine, especially in drug-resistant TB regions, further research should refine dosage and assess long-term safety.
Introduction
Tuberculosis, known as TB, is an infectious disease that has been known to man for thousands of years and is still ranked among the most dangerous illnesses in the world. 1 TB remains one of the leading causes of death from an infectious agent on a global level, despite the discovery of antibiotics and the development of vaccines. In the year 2022, about 1.3 million people died from this disease, and 167,000 persons were co-infected with HIV. In the same year, about 10.6 million people developed TB across all ages and regions of the world. 2 The disease disproportionately affects low- and middle-income countries, with two-thirds of new cases arising in just eight nations: India, China, Indonesia, Philippines, Pakistan, Nigeria, Bangladesh, and South Africa. 3 TB is an entirely preventable and curable illness, yet the disease remains a significant threat to the world’s health systems. 4 Tuberculosis is caused by Mycobacterium tuberculosis (Mtb), primarily by inhalation of aerosols containing the bacteria, and primarily affects the lungs but disseminates throughout the body. 1 TB affects millions of people all over the world and has high morbidity and mortality rates, which require significant preventive and treatment strategies. The standard of care for TB typically involves the use of multiple antibiotics, although increasing drug resistance makes the control of TB difficult. Developing an efficient vaccine is a global priority.
At present, the BCG vaccine developed in the 1920s is the only TB vaccine that has been approved for human use. BCG is an oral live attenuated vaccine obtained from Mycobacterium bovis, which is utilized to prevent severe forms of TB, such as TB meningitis and disseminated TB in children. However, the effects of this vaccine against pulmonary TB, regarded as the most frequent and infectious type, differ significantly in adult individuals. 5 A couple of long-term trials have shown that BCG confers about 52% protection against tuberculosis (TB), and the level of efficacy may vary significantly in different groups of people and geographical areas. These differences include genetic differences between the different BCG strains, differences in host immunity, and exposure to other mycobacteria.6,7
Moreover, protective immunity in BCG-vaccinated individuals decreases with age. 8 Many regions also face limited availability of the BCG vaccine. 9 The weaknesses of BCG have been the impetus toward looking for better vaccines against TB.
The most successfully searched antigens under development are the ID93 vaccines. ID93 is a recombinant subunit vaccine derived from the 4 Mycobacterium tuberculosis proteins Rv1813, Rv2608, Rv3619, and Rv3620. 10 These antigens were chosen based on their immunogenicity and stage dependency in the TB infection cycle. Among these proteins, Rv3619 and Rv3620 are associated with the ESX-1 secretion system, an important virulence factor in M. tuberculosis but not present in M. bovis or the BCG strain. The remaining two antigens, Rv1813 and Rv2608, are shown to be expressed in both M. tuberculosis and M. bovis.11,12 These unique antigens are thought to activate the immune system’s humoral and cellular arms required for TB control. Indeed, ID93 is a combination of multiple antigenic peptides; nevertheless, it includes the TLR4 agonist GLA-SE (Glucopyranosyl et al.) in its formulation to boost immunogenicity. 13 This area is where adjuvants are essential since they help the antigens to enhance the immune response and improve the body's ability to defend against infections. Prior research indicates that GLA-SE boosts the generation of multifunctional CD4+ T cells capable of releasing cytokines such as IFN-γ, TNF, and IL-2. 10 These cytokines are essential for regulating immune responses to M. tuberculosis infection. The combination of ID93 with GLA-SE has been shown to provide strong protection against high-virulence M. tuberculosis strains in early experimental animal models.14–17 The immunological importance of ID93 ensures that it can compensate for the drawbacks of the BCG vaccine well. According to several computational predictions, one of its subunits, Rv2608, provides widespread epitope protection, especially for TB endemic populations, particularly in South Asia and Sub-Saharan Africa. 18 Such broad coverage ensures that the vaccine works in different population subgroups. Existing clinical studies on the ID93 vaccine conducted in the United States and South Africa suggest that it is safe for use across different population groups and effectively induces immunity, particularly in preventing both primary infection and the reactivation of latent TB. Overall, the vaccine was safe throughout these investigations, and the immune response was associated with high antibody formation and Th1-cytokine dominance. 10 Therefore, because of the favorable safety signals and the effective immune responses it can induce, ID93 could become the next-generation TB vaccine.
This meta-analysis aims to comprehensively review and synthesize all available published data on the safety and immune response of the ID93/GLA-SE vaccine. By systematically reviewing data from preclinical and clinical studies, this paper aims to give an overall rating of the viability of ID93 as a new TB vaccine. Since there is a worldwide need to come up with better and more efficient tools for addressing TB, especially with the current emergence of drug-resistant TB and suboptimal vaccines, ID93 could be seen as an ideal candidate for solving one of the world’s biggest health problems.
Methodology
This systematic review and meta-analysis was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) criteria and registered with PROSPERO (CRD42024601450) accessible at https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42024601450
Data sources and search strategy
This meta-analysis was conducted conclusively to the Preferred Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines. 19 A comprehensive electronic search of PubMed (Medline), ScienceDirect, EMBASE, Scopus, and Cochrane Central databases was conducted from inception to September 2024. The following search terms were used: “ID93,” “GLA-SE,” and “tuberculosis.” Two unbiased authors (ES and MK) searched without limitations or conflicts of interest. A search was conducted on “clinicaltrials.gov” for relevant published or unpublished clinical trials. Furthermore, we conducted a manual search of the reference lists of the included research, as well as related meta-analyses and review articles, to identify possibly relevant studies.
Study selection
The eligibility criteria were randomized controlled trials (RCTs) with a target population of BCG-vaccinated healthy adults. All studies had ID93 + GLA-SE tuberculosis vaccine compared with placebo (BCG-vaccinated individuals without ID93 + GLA-SE). While the case reports, review articles, expert opinions, comments, cross-sectionals, and editorials were excluded from the analysis.
Data extraction and assessment of study quality
Duplicate studies were removed from the list after exporting the retrieved articles to Endnote Reference Library Software. All reviewers then carefully assessed the remaining articles, and only those that met the previously stated eligibility conditions were included. All the articles were first screened based on the abstract and title. After that, the remaining articles were screened through the full text. A third reviewer (MSK) was consulted to resolve any disagreements regarding the results. An online Microsoft Excel spreadsheet was created using data from the completed RCTs for the baseline characteristics and outcomes. Baseline parameters are as follows: mean age (SD), dose (high dose and low dose), gender, baseline BMI, and ethnic origin (mixed, black, white). The following outcomes were included:
Safety outcomes
injection site pain
erythema
headache
myalgia, fatigue
upper respiratory tract infection.
Immunogenicity outcomes
IgG response
Geometric mean titer (GMT) values
seroconversion rates (SRR)
CD4 and CD8 T-cell responses
The modified Cochrane Collaboration risk of bias technique was utilized to check the reliability of RCTs. Quality assessment and risk of bias were independently assessed using the Cochrane Risk of Bias Tool 2 on all included RCTs by two investigators, and the results were matched. Any discrepancies were resolved by team consensus.
Meta-analysis
The meta-analysis used Review Manager (version 5.4. Copenhagen: Nordic Cochrane Centre, The Cochrane Collaboration, 2014). Forest plots were generated for the results’ visual display. The outcomes were displayed as risk ratios (RR) with a 95% confidence interval using the random effects model. A value of below 50% was deemed high for I2 (study heterogeneity).
Results
Study selection
The searches yielded 45 articles from PubMed, ScienceDirect, EMBASE, Scopus, and Cochrane Central databases. After filtering duplicates and excluding irrelevant articles based on title, six articles were examined for the availability of full texts and data related to the research objective. Following the assessment of complete texts, three articles were included for analysis.20–22 The study selection steps are presented in Figure 1.

PRISMA flowchart.
Characteristics of studies
All three included articles were randomized controlled trials done on BCG-vaccinated healthy adults. Detailed characteristics of the included studies and the results of each study are outlined in Table 1 (Baseline Characteristics). A study by Yu Hwa Choi et al. 22 was funded by Quratis Inc. and supported by a grant from the Korea Health Technology R&D Project through KHIDI, funded by the Ministry of Health and Welfare, Republic of Korea. A study by Adam Penn-Nicholson et al. 2018 20 was supported by co-funding from Aeras and the Paul G Allen Family Foundation. Additional grants were provided by the Bill & Melinda Gates Foundation and DFID (UK Government), with non-financial support from GSK and Sanofi Pasteur.
Baseline characteristics.
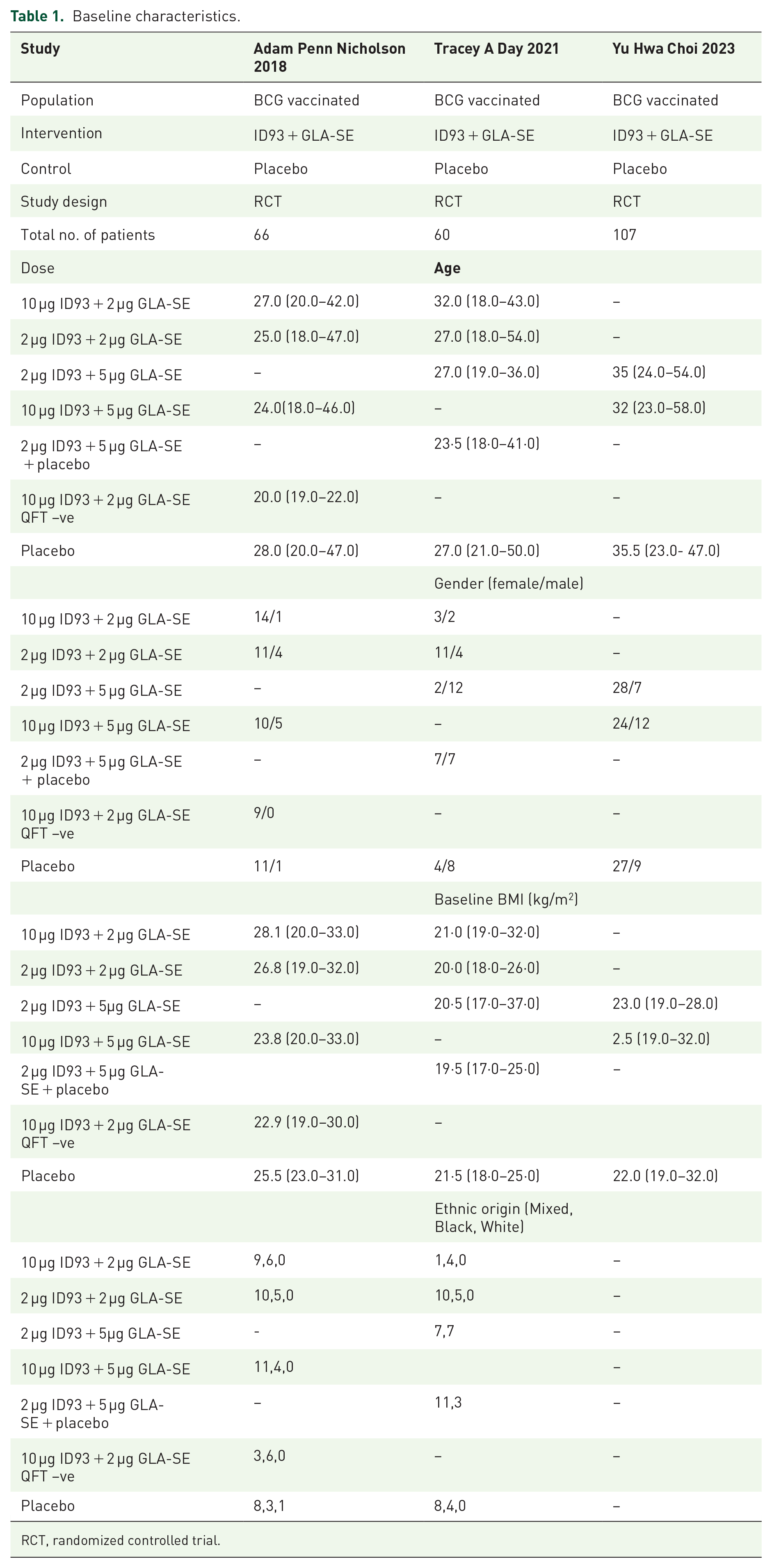
RCT, randomized controlled trial.
Risk of bias assessment
The quality of individual studies’ analysis was conducted according to the study design using the Cochrane Risk of Bias Tool 2, 23 where this checklist was used to analyze the risk of bias in the studies. The results of the risk of bias assessment are presented in Figure 2.

Risk of bias assessment.
Safety evaluation
ID93 + GLA-SE versus placebo compared to fatigue
Figure 3 shows a forest plot comparing the impact of ID93 + GLA-SE versus placebo on fatigue across two dosage subgroups of ID93 + GLA-SE: high dose (10 μg) and low dose (2 μg). For high doses, the pooled analysis from three studies shows a significant effect favoring ID93 + GLA-SE, with a risk ratio of 4.97 (95% CI 1.69–14.60), with a significant effect (p = 0.004). This indicates that participants receiving ID93 + GLA-SE were nearly five times more likely to experience fatigue than placebo. However, the risk ratio for low dose is 1.83 (95% CI 0.53–6.34), with no significant effect (p = 0.34). Overall, when combining both fatigue categories, the treatment significantly increases fatigue, with a risk ratio of 3.24 (95% CI 1.44–7.31, p = 0.005), driven primarily by the reduction in dose. There is no evidence of heterogeneity across the studies (I² = 0%), indicating consistent results.

Fatigue forest plot.
Comparison of ID93 + GLA-SE versus placebo concerning myalgia/muscle pain
Figure 4 shows a forest plot comparing the effect of ID93 + GLA-SE versus placebo on myalgia/muscle pain across two dosage subgroups of ID93 + GLA-SE: high dose (10 μg) and low dose (2 μg). For high doses, the pooled analysis from the studies shows a significant benefit of ID93 + GLA-SE with a risk ratio of 6.02 (95% CI 1.86–19.48) with significant effect (p = 0.003), indicating a marked gain in myalgia compared to placebo. Similarly, the risk ratio for low doses is 5.63 (95% CI 1.74–18.25), also showing a significant effect (p = 0.004). Overall, the combined risk ratio for both categories is 5.82 (95% CI 2.54–13.37, p < 0.0001), demonstrating that ID93 + GLA-SE significantly increases myalgia symptoms compared to placebo. No significant heterogeneity was found between the studies (I² = 0%), showing consistent results across studies.

Myalgia forest plot.
ID93 + GLA-SE versus placebo compared to upper respiratory tract infections
Figure 5 shows a forest plot comparing the effect of ID93 + GLA-SE versus placebo on upper respiratory tract infections (URTIs), across two dosage subgroups of ID93 + GLA-SE: high dose (10 μg) and low dose (2 μg). In the dose group, the pooled analysis of three studies gives no significant effect of ID93 + GLA-SE, with a risk ratio of 0.91 (95% CI 0.23–3.64, p = 0.90), showing no gain in URTI incidence as compared to placebo. Similarly, the risk ratio for low dose is 0.79 (95% CI 0.30–2.10, p = 0.64), also showing no significant effect. Overall, the combined risk ratio for the high-dose and low-dose categories is 0.83 (95% CI 0.38–1.84, p = 0.87), indicating that ID93 + GLA-SE does not significantly increase the risk of URTIs compared to placebo. There is no practical heterogeneity between the studies (I² = 0%), which means results are consistent across studies.
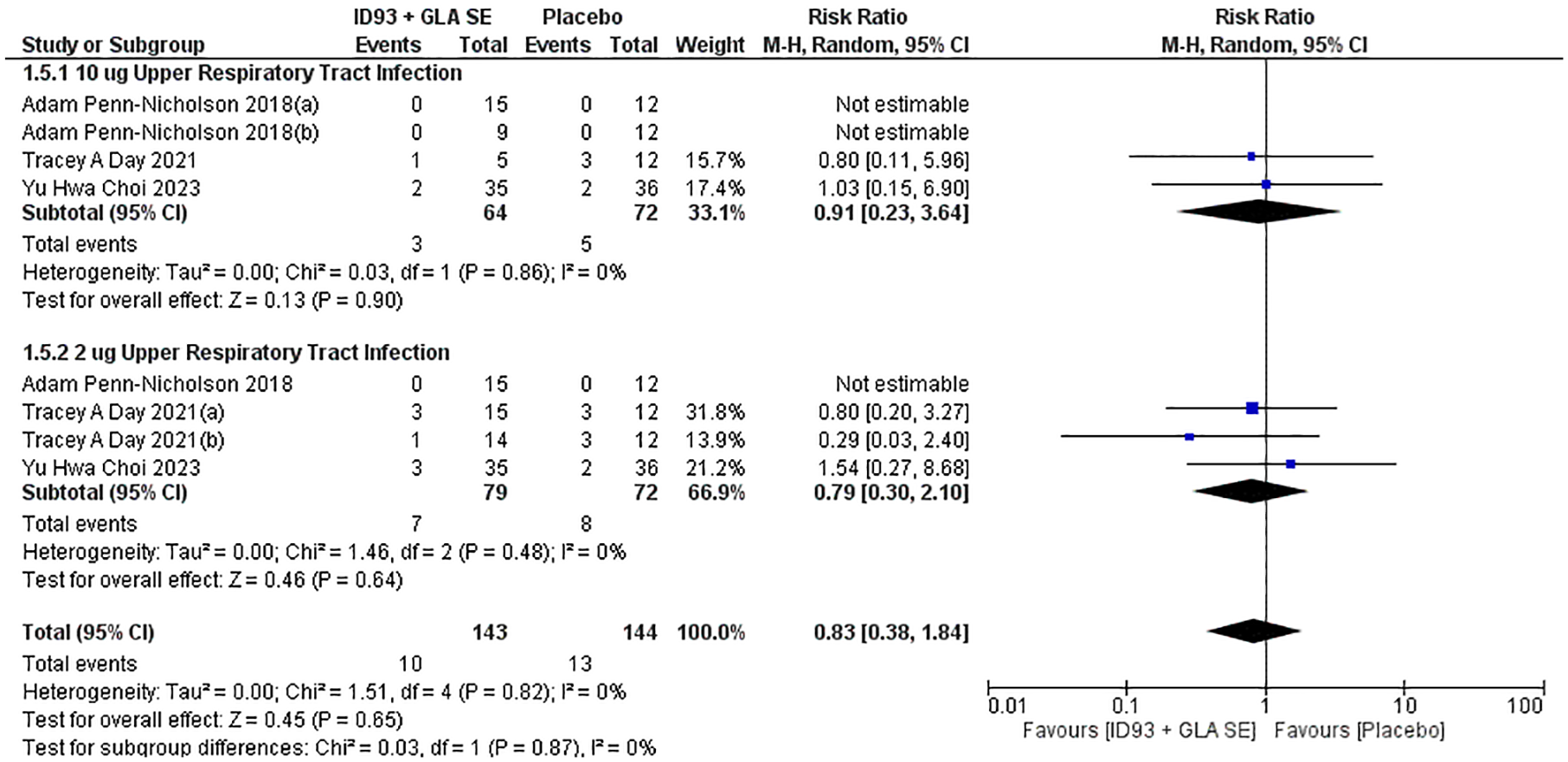
Upper respiratory tract infection forest plot.
ID93 + GLA-SE versus placebo compared to injection site pain
Figure 6 shows a forest plot comparing the effect of ID93 + GLA-SE versus placebo on injection site pain across two dosage subgroups of ID93 + GLA-SE: high dose (10 μg) and low dose (2 μg). In high dose, the pooled analysis of three studies shows a significant effect of ID93 + GLA-SE, with a risk ratio of 5.75 (95% CI 2.97–11.15, p < 0.00001) and consistent results across studies. Similarly, the risk ratio for low doses is 3.07 (95% CI 1.73–5.46, p = 0.0001). Combined analysis of both subgroups showed the overall RR is 4.12 (95% CI 2.68–6.32), indicating that across all doses, participants receiving ID93 + GLA-SE are about four times more likely to experience injection site pain compared to placebo. The analysis gives low overall heterogeneity (I² = 4%), and the results are highly significant (p < 0.00001), demonstrating a strong association between the treatment and increased pain.

Injection site pain forest plot.
ID93 + GLA-SE versus placebo compared to headache
Figure 7 shows a forest plot comparing the effect of ID93 + GLA-SE versus placebo on headache occurrence between the ID93 + GLA-SE group and a placebo group, with two dosage subgroups of ID93 + GLA-SE: high dose (10 μg) and low dose (2 μg). For high doses, the pooled analysis from three studies shows no significant effect of ID93 + GLA-SE, with a risk ratio of 2.88 (95% CI 1.13–7.29, p = 0.03), indicating this effect is not statistically significant. However, in low doses, the RR is 0.81 (95% CI 0.16–4.08, p = 0.79), suggesting no significant difference in headache risk between the groups. When combining both subgroups, the overall RR is 1.77 (95% CI 0.77–4.10), indicating a non-significant increase in headache risk for the ID93 + GLA-SE group compared to placebo, and the overall effect is not statistically significant (p = 0.18).

Headache forest plot.
Secondary outcomes
Other secondary outcomes, such as decreased ALT, decreased blood pressure, and erythema, were reported by either two or a single study out of our three RCTS and, hence, were not reported in our analysis.
Publication bias
Since there are fewer RCTS (3), we could not make funnel plots and perform Egger’s regression test for assessing publication bias.
Immunogenicity evaluation
IgG response
In Table 2, Adam Penn-Nicholson’s 2018 study 20 shows that a robust IgG response was observed, particularly after the third dose. The increase was significant in the 10 µg ID93 + 5 µg GLA-SE group, with sustained elevated IgG levels beyond day 294. The IgG responses remained higher than pre-vaccination levels at later stages, especially in the higher-dose groups (10 µg ID93 + 5 µg GLA-SE and 10 µg ID93 + 2 µg GLA-SE). In Tracey A. Day’s 2021 study, 21 significant IgG responses were seen after the second dose, with higher doses (10 µg ID93 + 5 µg GLA-SE) yielding a peak response. IgG levels remained elevated up to Day 224. The 2 µg ID93 + 2 µg GLA-SE group also had a notable response, though of a lesser magnitude than higher doses. Yu Hwa Choi’s 2023 study 22 recorded peak IgG responses by day 84, particularly in the higher dose (10 µg ID93 + 5 µg GLA-SE) cohort, and these responses were decreased up to day 421. IgG levels dropped slightly but remained significantly higher than baseline across all dose groups, while the placebo group showed no notable rise in IgG throughout the time period.
Humoral and cell-mediated response to different doses of ID93 + GLA-SE and placebo.
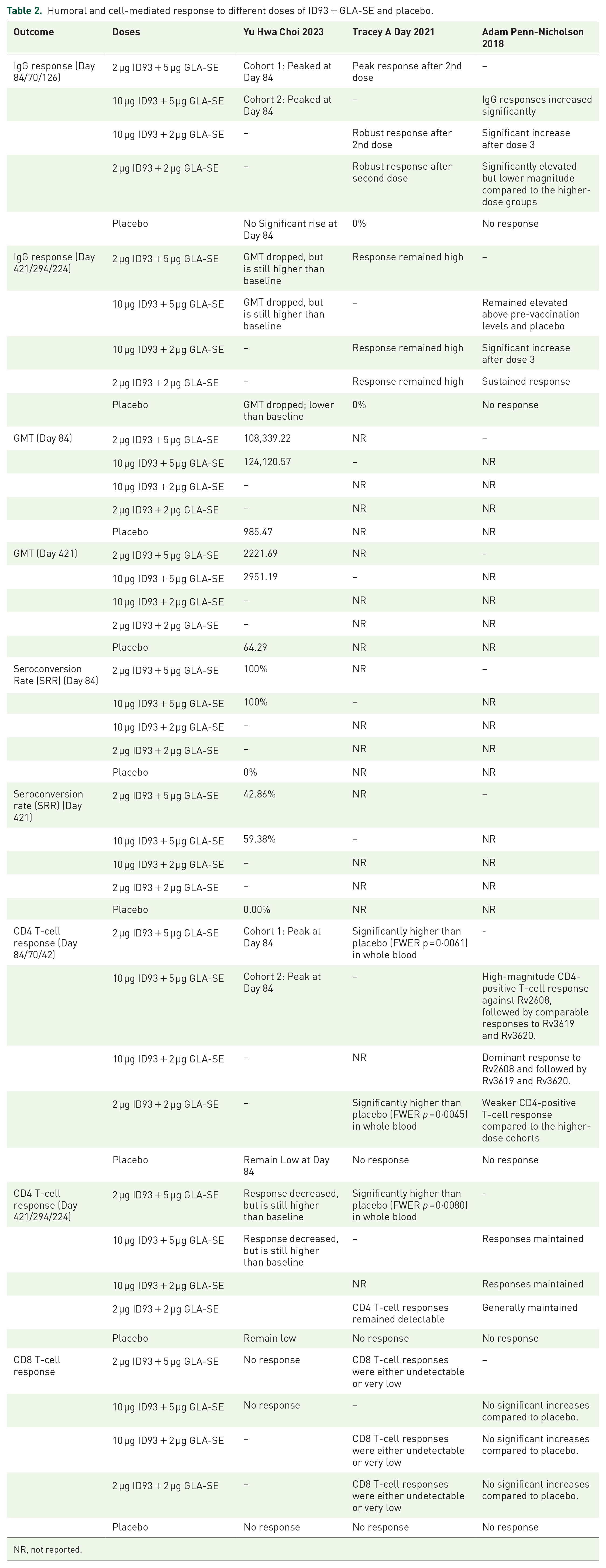
NR, not reported.
GMT values
Yu Hwa Choi’s 2023 study 22 analyzed high GMT values in the vaccinated cohorts at day 84, with the 10 µg ID93 + 5 µg GLA-SE group showing the highest values. By day 421, GMT values had dropped but were still much higher than baseline, particularly in the (10 µg and 2 µg groups). The placebo group’s GMT values remained low throughout the trial, again showing no significant immune response compared to the vaccinated groups. However, Adam Penn-Nicholson’s 2018 20 and Tracey A. Day’s 2021 21 studies did not report any GMT values. All these findings are mentioned in Table 2.
Seroconversion rate
Choi’s 22 study reported that seroconversion remained high (100%) through day 84 and dropped moderately by day 421, with the 10 µg group having a 59.38% SRR. 2 µg ID93 + 5 µg GLA-SE dose also sustained a reasonable level of seroconversion (42.86%). In comparison, the placebo group had 0% SRR throughout. However, Adam Penn-Nicholson 2018 20 and Tracey A. Day 2021 21 studies did not report any SRR values as shown in Table 2.
CD4 T-cell response
In Adam Penn-Nicholson 2018, 20 CD4-positive T-cell responses peaked after the third dose, especially in the 10 µg ID93 + 5 µg GLA-SE group. The response was dominant against antigens Rv2608, Rv3619, and Rv3620, with sustained activity through day 421. In Tracey A. Day 2021, 21 these responses remained significantly higher than the placebo group, which had no meaningful CD4 T-cell activity. Yu Hwa Choi’s 2023 22 study also recorded a peak CD4 T-cell response at day 84, decreasing responses through day 421. Though the CD4 response decreased over time, it remained higher than baseline and was still significantly elevated compared to the placebo group. CD4 response in the placebo group remained low throughout the period. Table 2 illustrates these findings.
CD8 T-cell response
In Table 2, Adam Penn-Nicholson 2018 20 found that there was little to no CD8-positive T-cell response in any of the cohorts and no discernible change compared to the placebo group. According to Tracey A. Day 2021, 21 comparable results were seen, with CD8-positive T-cell responses being either negligible or undetectable in all vaccination groups compared to the placebo. Yu Hwa Choi 22 confirmed the lack of CD8 T-cell responses. Across all dose groups, CD8 T-cell activity remained very low or undetectable, indicating that CD4-positive responses were more commonly elicited by the vaccine than CD8 responses.
Discussion
Our meta-analysis evaluated the safety and immunogenicity of the ID93 + GLA-SE vaccine against tuberculosis, a novel tuberculosis vaccine developed to reduce the global tuberculosis burden. TB remains among the top 10 causes of death around the world, with around 10 million new cases and 1.5 million deaths per year, particularly affecting low- and middle-income countries (LMICs). 24 The newly emerging strain of drug-resistant TB, including multidrug-resistant (MDR) and extensively drug-resistant (XDR) TB, has further complicated treatment strategies and management, necessitating the development and use of more effective vaccines. 25 Our study is about the safety and immunogenicity produced by the ID93 + GLA-SE vaccine, its comparison with existing interventions, and the literature.
ID93 + GLA-SE was found in our study to have a greatly favorable safety profile, local and systemic AEs are seen to an extent manageable in tandem with the vaccine. Injection site pain, myalgia, and fatigue were the common side effects in the current study; this is expected from adjuvant vaccines that enhance the immune reactions.26–28 Still, compared to the placebo, there was no hike in serious adverse effects, which proves that the vaccine is safe in the short run. Surprisingly, our analysis found that URTIs are infrequent in this vaccine group, even though vaccines with some strong adjuvants, such as GLA-SE, may pose a threat of respiratory infections through overstimulation of the immune system with subsequent inflammation and increased vulnerability to infections. 29 However, we did not observe a significant increase in URTIs, indicating that systemic immunization with the ID93 GLA-SE vaccine does not elicit excessive inflammation that may be dangerous to patients with poor health, hence making the vaccine safer for patients at higher risk for infections. These results differ from a previous study on adjuvant vaccines, like influenza or pertussis, where respiratory infections were reported as the expected adverse event.30,31 On the other hand, the lack of such infections may be attributed to the specificity of ID93 + GLA-SE in targeting TB antigens and not a general immune stimulation, coupled with the fact that the immune response it elicits appears more targeted and well-controlled.
Regarding immunogenicity, ID93 + GLA-SE displayed a solid and sustainable immune response characterized by the ability to induce high titers of IgG antibodies and CD4 + T cells with multifunctional potential. Such T cells generate the pivotal cytokines interferon-gamma (IFN-γ), tumor necrosis factor (TNF), and interleukin-2 (IL-2), of which IFN-γ is perhaps the single most central figure in controlling Mycobacterium tuberculosis infections. 32 The activated T cells, also termed multifunctional T cells, are considered key players in immune activation because they secrete many cytokines at once and directly positively affect the intracellular bacteria’s lysis and maintain long-lasting immunity. 33 The immunogenicity of ID93 + GLA-SE is due to its antigens and adjuvant content. ID93 is constituted of four MTB antigens—Rv2608, Rv3619, Rv3620, and Rv1813, created to trigger immunological responses against various Tb phases, starting from the initial colonization up to the latent phase of infection. 10 Using multiple antigens increases the chances of producing broad immune activity that could help to inhibit ongoing tuberculosis and the recurrence of the latent form. 18 The adjuvant in this vaccine, GLA-SE, is a synthetic Toll-like receptor 4 (TLR4) agonist to augment the activation of antigen-presenting cells and enhance cytokine production in the innate immune system. It tends to enhance the immunogenicity of other supplemented vaccines by increasing the antigen presentation and the T-helper 1 (Th1) immune response, believed to play a role in controlling intracellular pathogens like Mtb. 34 The integrated form of ID93 and GLA-SE is designed to prime the immune system to respond vigorously from the first meeting with Mtb. Potentially providing superior protective actions to the current Bacillus Calmette-Guérin (BCG) vaccine, with variable effectiveness. 35
This study also showed that high- and low-dose formulations of ID93 + GLA-SE have a comparable impact. Elevated immune responses were observed in the high-dose group, which needs to be attributed to the antibody titer and T-cell stimulation index compared to the low-dose group. This is similar to other vaccines with dose-response relations, where different vaccine concentrations show significantly different immunogenicity.36,37 Nonetheless, the high-dose group also experienced higher toxicity, injection site reactions, and fatigue, suggesting that the high-dose safety profile needs to be closely monitored. On the other hand, the low-dose group presented with fewer AEs while presenting a slightly lower figure for immunogenicity results. Altogether, it emphasizes the necessity of dose optimization in the context of the current development of vaccines because even very low dosages may provide sufficient immunogenicity when associated with the enhanced safety margin. Ideally, future research will be required to continue fine-tuning the delicate balance between the safety and efficacy of this treatment protocol.
The results are in harmony with the relative reference literature concerning adjuvant subunit vaccines, embracing the possibility of enhancing immune responses against many other pathogens, such as pertussis and influenza.30,31 The conclusion of the present study shifts in the same direction as the ability of adjuvants such as GLA-SE, which can be helpful in further vaccination against Tb. Being a global killer, TB affects mainly LMICs, and developing TB vaccines is essential. Current prevention strategies against TB include the BCG vaccine, which provides poor protection against adult pulmonary TB and has variability in effectiveness in different populations. 5 Management approaches have also been made difficult by increased MDR and XDR TB cases since common antibiotics have a reduced potency against these strains. 38 The administration of ID93 with GLA-SE is a relatively novel therapeutic angle. It is proposed to enhance TB prevention through boosted immune responses and protection against all states of TB infection.
Strengths and limitations
Another benefit of our work is that it systematically builds on the most recently available pooled data from several RCTs to show that ID93 + GLA-SE is safe and immunogenic. This analysis also shows that, assuming protection in BCG-vaccinated populations, the vaccine can offer protection where BCG is already implemented. The three primary studies employed for this analysis are mainly randomized controlled trials (RCTs) of healthy, BCG-vaccinated individuals conducted in various settings, such as South Africa and South Korea. The results are more robust in external validity as the mentioned populations are those most at risk of developing TB and where vaccine intervention would be targeted. 39 The included studies also reported outcomes of extended follow-up, as TB is a chronic disease, and lasting immunity is crucial for any vaccine candidate to be considered effective. 40
Nevertheless, there are several limitations to our study: First, there was a lack of high-risk populations, including people with HIV, children, and those with latent TB. This limits the external validity of the findings to these groups because they are likely to have differential reactions to the vaccine. Moreover, the studies employed a variety of assays and guidelines for detecting immune responses, which added variability to the immunogenicity data, thereby complicating direct comparison. In addition, there was no uniformity and measurable data in all the studies for critical immunogenicity endpoints, making it possible to do more complex subgroup comparisons. These limitations have pointed out that more extensive studies with a lengthier follow-up period, better-defined and standardized assay, and a more heterogeneous population sample are required to optimally assess the utility of ID93 + GLA-SE as a new generation TB vaccine.
Conclusion
The study identified the ID93 + GLA-SE vaccines as generally safe with a few AEs, injection site pain, and fatigue, which are easily tolerable. Robust immune responses, particularly boosted IgG levels and CD4+ T-cell activity, have been seen with ID93 + GLA-SE, which are crucial for tuberculosis prevention. Considering the high and low doses, higher doses showed improved efficacy but also increased the incidence of adverse effects, demonstrating the importance of dose optimization. It is found to be a potential adjunct to the BCG vaccine, especially in areas where drug-resistant TB is increasing. Priority should be given to refining the dosage in further studies, assessing long-term safety and efficacy, and investigating its combination with other TB vaccines or therapies to strengthen global TB control efforts.
Supplemental Material
sj-docx-1-tav-10.1177_25151355251344473 – Supplemental material for Evaluating the immunogenicity and safety of ID93 + GLA-SE in BCG-vaccinated healthy adults: a systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-docx-1-tav-10.1177_25151355251344473 for Evaluating the immunogenicity and safety of ID93 + GLA-SE in BCG-vaccinated healthy adults: a systematic review and meta-analysis of randomized controlled trials by Erum Siddiqui, Mohammad Saad Khan, Maliha Khalid, Harshika Khaim Chandani, Umaimah Naeem, Muhammad Mohsin Khan and Syed Owais Wasti in Therapeutic Advances in Vaccines and Immunotherapy
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
