Abstract
Background:
Extrapulmonary tuberculosis (EPTB) poses a public health challenge, particularly among individuals with human immunodeficiency virus (HIV). However, many EPTB cases arise in those without HIV, leaving the underlying factors unclear.
Objectives:
This study aims to characterize patients with pulmonary tuberculosis (PTB) and EPTB, exploring the differences in associated factors for each type in patients with and without HIV.
Design:
An analytical observational study was conducted on a cohort of tuberculosis (TB) patients diagnosed between 2014 and 2021 in a referral hospital in Bogotá, Colombia.
Methods:
Patients were categorized into PTB or EPTB based on the site of infection. Data on demographic and clinical variables were collected, comparing the two groups. A multivariate logistic regression model was created to identify factors associated with EPTB compared to PTB.
Results:
The study encompassed 533 patients: 310 with PTB and 223 with EPTB, of which only 65 (14.7%) were HIV positive. PTB patients exhibited higher rates of active smoking, smoking cessation, diabetes mellitus (DM), and other pulmonary diseases. The logistic regression identified HIV infection as the only factor associated with EPTB (OR 2.36 (1.54–3.61),
Conclusion:
EPTB is primarily associated with HIV infection, while smoking, DM, COPD, other pulmonary diseases, and immunosuppressant use are associated with PTB. Variability in associated factors for specific EPTB forms underlines the need for tailored research into the predisposing factors for EPTB, particularly in HIV-negative patients.
Introduction
Tuberculosis (TB) constitutes a significant public health issue and since 2023 has once again become the leading cause of death by a single infectious agent, replacing COVID-19. 1 In 2023, an estimated 8.2 million new infections (84% extrapulmonary tuberculosis (EPTB) and 16% pulmonary tuberculosis (PTB)) and 1.25 million deaths were reported worldwide, according to data from the World Health Organization (WHO). Only 6.1% of incident cases and 161,000 deaths were in patients with human immunodeficiency virus (HIV). 1 Colombia is a high TB prevalence country, with 14,060 new cases reported in 2021 and an incidence rate of 25.9 per 100,000 inhabitants, as per the National Institute of Health (INS) data. PTB is the most common form of presentation, accounting for 84.8% of cases in the country.2,3 Until 2019, a decreasing trend in TB incidence had been observed; however, in 2020 and 2021, there was a resurgence, possibly due to reduced preventive measures and timely diagnosis programs during the COVID-19 pandemic, 1 which began to recover in 2022.
The progression and dissemination of TB infection depend on the host's immune response. In most cases, it is expected that proper containment of the infection will be achieved through the action of Th1 CD4+ lymphocytes. These lymphocytes produce interferon-gamma, which activates local macrophages and promotes the formation of granulomas, preventing the proliferation of the bacillus. In patients with impaired cellular immunity, the mechanisms that promote containment are insufficient, allowing for its dissemination. In this manner, extrapulmonary forms of tuberculosis develop, with three primary modes of dissemination: by contiguity, by hematogenous route, or through the spread of respiratory secretions to the digestive tract. 4 Infection with HIV has been recognized as the primary risk factor for extrapulmonary dissemination due to its known alteration of cellular immunity through the reduction of CD4+ T lymphocytes. 5 However, EPTB commonly presents in patients without HIV, and there is a lack of clarity regarding the factors that predispose to its occurrence in this population.
Most studies on risk factors for EPTB are in HIV-positive patients. Some descriptive studies in patients without this coinfection have identified associated factors such as female sex, social marginalization, stay in prisons, and rural housing6 –13; however, these were conducted in very selected contexts and populations, with variable and sometimes discordant results. In Colombia, descriptive studies report the frequency of presentation of EPTB14 –17 and, to our knowledge, only one has evaluated possible associated factors, finding a higher frequency of EPTB with higher developmental indices, contrary to the findings of previous studies that associated EPTB with underdevelopment. 18
Thus, it is possible to state that the information available about the characteristics of HIV-negative patients who develop EPTB is limited and there is a lack of clarity about the factors associated with its occurrence. It has been suggested that these patients may have other conditions that lead to immunosuppression, such as the use of immunomodulators, active neoplasms, or chronic diseases such as type 2 diabetes mellitus (DM) or chronic kidney disease (CKD), but studies have not consistently shown this.7,8,12 Knowing the specific risk factors for EPTB is important because it can facilitate clinical suspicion, timely diagnosis, and the design of specific screening or follow-up programs. This study aims to evaluate the clinical characteristics and possible differences between patients with and without HIV and between patients presenting with PTB and EPTB to propose hypotheses about the factors associated with its occurrence.
Methods
An analytical observational single cohort study was conducted and reported according to the STROBE statement. 19 All patients over 18 years of age, notified to the “Secretaría Distrital de Salud” in Bogotá Colombia by the San Ignacio University Hospital (HUSI) between 2014 and 2021 with a diagnosis of TB, were included. The study was approved by the ethics and research committee of HUSI and the Javeriana University.
Freeman’s formula was used to calculate the sample size. 20 Patients included had TB confirmed by smear microscopy, culture, molecular methods, or histology. Patients with a diagnosis of latent TB, empirical diagnosis, no data on HIV coinfection, and those without complete medical history data were excluded.
A standardized format was used to systematically collect information from electronic medical records stored in the Integrated Hospital Administration System (SAHI). The RedCap platform was utilized for this purpose.21,22 Demographic variables, comorbidities, medications, and hospital care data—including TB diagnostic method, HIV infection status, follow-up duration, and mortality were collected for each patient.
After collecting the information, patients were classified based on the site of involvement in PTB and EPTB. Using the WHO guidelines 23 as a reference, PTB was defined as involving the tracheobronchial tree or lung parenchyma. EPTB was defined as a disease outside the tracheobronchial tree. Patients with both pulmonary and extrapulmonary involvement were classified as PTB.
Alcohol consumption was considered if it was reported as such in the medical history at admission. Data on weight, height, and body mass index (BMI) recorded in the medical records were obtained. Malnutrition was identified if this diagnosis appeared in the clinical nutrition assessment or if the BMI was less than 18. Lung disease was considered as the presence of chronic obstructive pulmonary disease (COPD), asthma, idiopathic pulmonary fibrosis, other interstitial diseases, bronchiectasis, cystic fibrosis, silicosis, and sequelae of previous TB or anthracosis. Autoimmune diseases were classified as systemic lupus erythematosus (SLE), rheumatoid arthritis, Sjögren's syndrome, dermatomyositis, mixed connective tissue disease, spondyloarthropathies, multiple sclerosis, vasculitis, and others. Significant steroid consumption was considered the equivalent of 10 mg of prednisolone per day for 1 month before diagnosis. The diagnosis of cancer was confirmed when active disease was present.
Descriptive statistics were utilized to present the sociodemographic data of the population, along with clinical and paraclinical variables. Mean and standard deviation or median and interquartile range were used for continuous variables, depending on the data distribution. A Shapiro-Wilk test was used to assess the assumption of normality. For categorical variables, absolute numbers and percentages were reported. The difference between groups (PTB and EPTB) was assessed according to the nature of the variables using student’s
Subgroup analysis was conducted for both the HIV-positive and HIV-negative populations. To assess the factors associated with presenting with EPTB, a multivariate logistic regression model was performed, including clinically relevant variables and those with an initial
Results
A total of 802 patients were notified to the “Secretaría de Salud” between January 2014 and December 2021. Of these, we excluded 41 patients under 18 years of age, 21 patients in whom HIV testing was not performed, 76 patients with latent TB, 40 patients without complete data in the clinical history, 29 patients with empirical diagnosis, and 62 patients in whom TB diagnosis was then ruled out (initially reported for empirical management or had final isolation of a non-tuberculous mycobacterium). In the end, 533 patients were included for analysis.
The clinical and demographic characteristics are summarized in Table 1. Of the 533 patients included 310 were PTB patients and 223 were EPTB patients. The median age was higher in patients with PTB compared to those with EPTB (60 years interquartile range (IQR) 37–71 vs. 48 years IQR 33–66,
Clinical and demographic characteristics of patients with pulmonary and extrapulmonary tuberculosis.

COPD, chronic obstructive pulmonary disease; EPTB, extrapulmonary tuberculosis; PTB, pulmonary tuberculosis.
Most of the patients were affiliated with the contributory system, with a slightly higher proportion affiliated to the subsidized system in the PTB group (13.23% vs. 8.07%,
Regarding comorbidities, DM and pulmonary diseases were more frequent in patients with PTB (see Table 1), while HIV was more frequent in patients with EPTB (14.83% vs. 29.15%
The most frequently reported autoimmune diseases were SLE and rheumatoid arthritis (RA). The frequency of immunosuppressive drug use was low in both groups (17.09% in PTB and 13% in EPTB). No differences were found in the use of steroids or biologic therapy between the two groups, with a slightly higher percentage of patients on other immunosuppressants in the PTB group (8.71% vs. 4.48%,
Regarding the diagnosis and treatment received (see Table 2), in the PTB group, diagnosis by smear microscopy or culture was more frequent, while in patients with EPTB, the most frequent form of diagnosis was histology. Four patients with pleural TB were diagnosed with adenosine deaminase (ADA). Both groups showed a similar frequency of diagnosis with molecular testing. The length of hospital stay was longer in patients with EPTB (10 days IQR 4–21 vs. 18 days IQR 10–30,
Characteristics of the diagnosis and treatment of the tuberculosis episode.
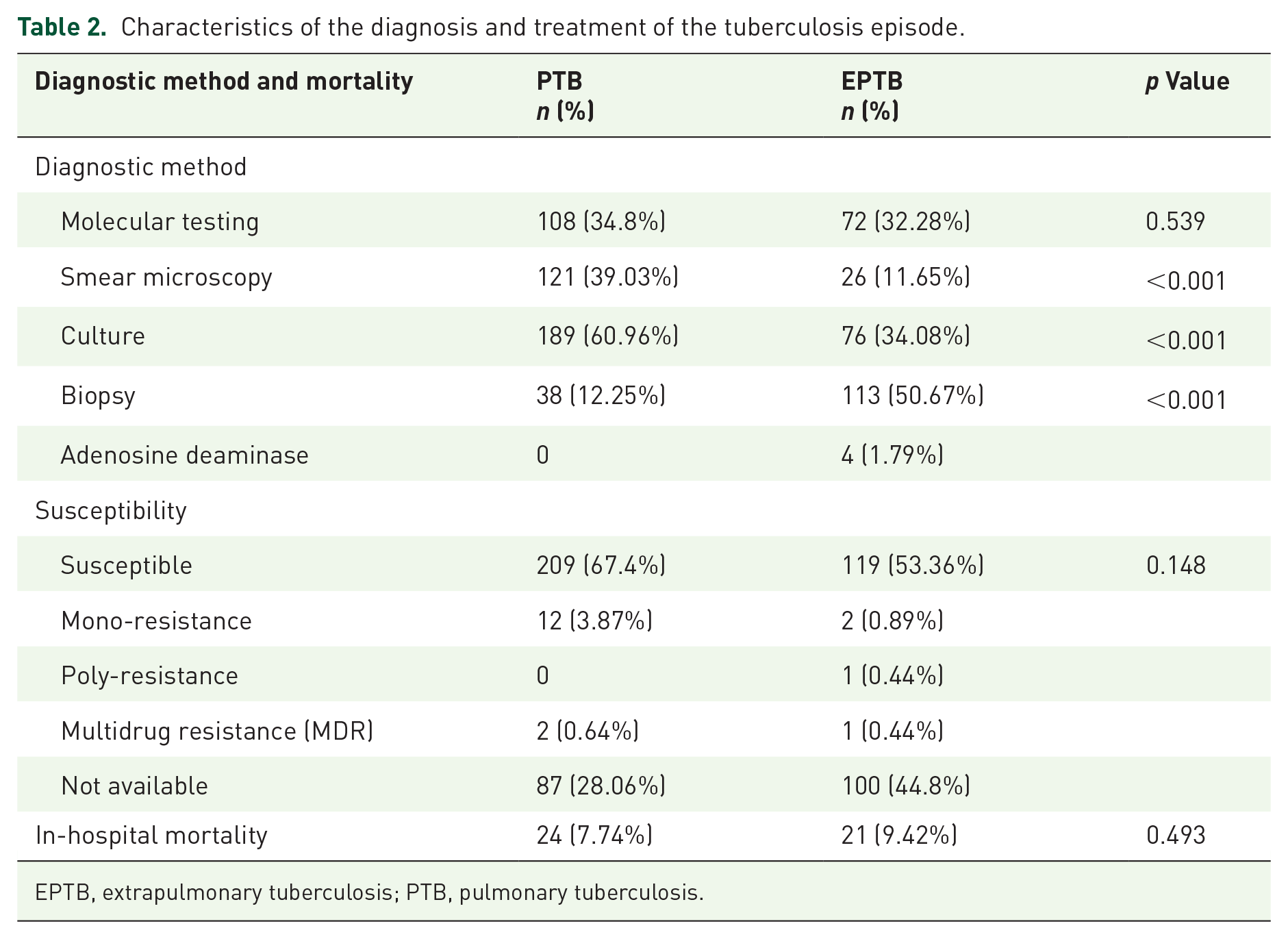
EPTB, extrapulmonary tuberculosis; PTB, pulmonary tuberculosis.
Susceptibility profile data were only found in 71.9% of the patients with PTB and 55.1% of the patients with EPTB. Of these, the most were susceptible strains, with a low frequency of multidrug-resistant (MDR) TB in both groups. There was no significant difference in mortality.
The distribution by the sites of involvement in patients with PTB is shown in Figure 1. Pleural, lymph node, and miliary were the most frequent sites. Figure 2 shows the comparison of the sites of involvement in HIV-positive and HIV-negative patients. Most forms of EPTB were more frequent in patients without HIV, except for miliary and lymph node TB, which were more frequent in patients with this coinfection.

Sites of involvement in patients with extrapulmonary tuberculosis.

Distribution of extrapulmonary tuberculosis involvement in HIV-positive and HIV-negative patients.
In the subgroup analysis, in HIV patients, there were no significant differences in age or sex between PTB and EPTB patients, with a median age of 37.5 and 36 years, respectively. A higher frequency of anemia of any grade (50% vs. 56.9%) was found in patients with EPTB, with a greater proportion within the range of mild-to-moderate anemia. The frequency of comorbidities other than HIV was very low.
The characteristics of the HIV-negative patients are outlined in Table 3. The age range was similar in patients with PTB and EPTB, with male predominance in both groups. A higher frequency of smoking cessation and active smoking was observed in patients with PTB. Among comorbidities, there was a higher prevalence of DM, CKD, and other pulmonary diseases in patients with PTB. Cancer was also more frequent in PTB patients, but this was not statistically significant. There were no differences in other comorbidities or the use of immunosuppressive medications.
Clinical and demographic characteristics of PTB and EPTB patients in HIV-negative patients.

COPD, chronic obstructive pulmonary disease; EPTB, extrapulmonary tuberculosis; PTB, pulmonary tuberculosis.
Table 4 shows the results of the logistic regression model. Univariate analysis found an association between HIV infection and extrapulmonary presentation of TB (OR 2.36; 95% CI 1.54–3.61). Smoking cessation (OR 0.6; 95% CI 0.39–0.92), DM (OR 0.44; 95% CI 0.23–0.85), COPD (OR 0.17; 95% CI 0.07–0.36) and the group of other pulmonary diseases (OR 0.22; 95% CI 00.06–0.79) were identified as having a lower association with EPTB than PTB. In the multivariate analysis, the association described for HIV and EPTB persisted, while active smoking, DM, COPD, the other pulmonary diseases group, and the use of any immunosuppressive medication (OR 0.44; 95% CI 0.2–0.96) had a negative association with EPTB.
Factors associated with extrapulmonary presentation of tuberculosis.
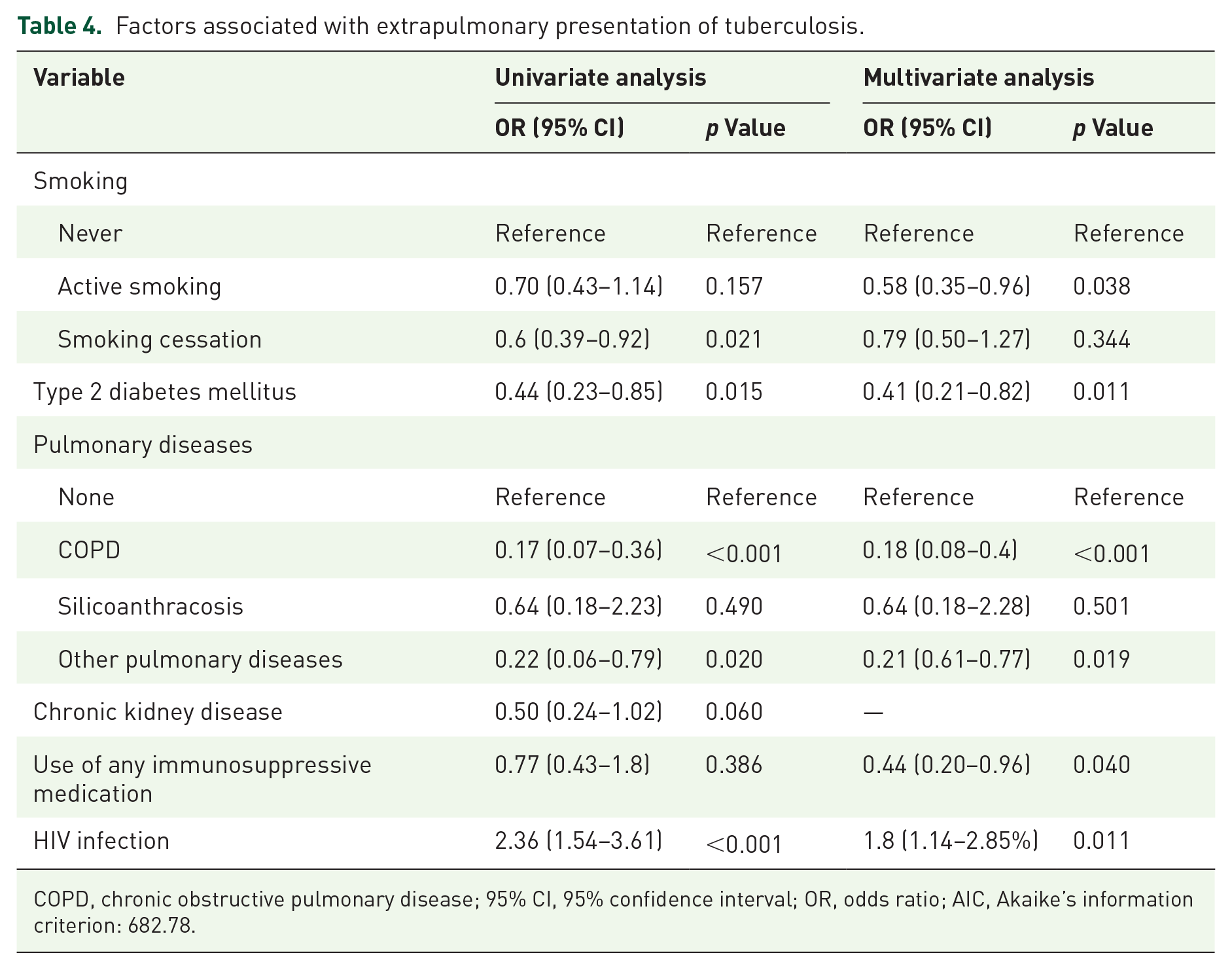
COPD, chronic obstructive pulmonary disease; 95% CI, 95% confidence interval; OR, odds ratio; AIC, Akaike’s information criterion: 682.78.
The analysis of associated factors to pleural and lymph node tuberculosis development is summarized in Tables S1 and S2. For the first outcome, patients with moderate anemia (OR 0.46, 95% CI 0.22–0.95,
Discussion
This study explored the differences between associated factors for the development of PTB and EPTB in HIV-positive and HIV-negative patients. The results identified HIV infection as an associated factor for EPTB, while patients with smoking, DM, COPD, and other lung diseases and with the use of any immunosuppressive medication, had a lower frequency of EPTB compared to PTB.
HIV infection has been described as one of the most important risk factors for TB, with a reported 20-fold increased risk 24 and a higher frequency of extrapulmonary presentation, 25 consistent with the results of this study. Several meta-analyses have analyzed the specific association of HIV and EPTB, finding a positive association but with marked heterogeneity in the studies.26,27 Among these, Naing et al. 27 found an increased risk in the subgroup of patients with CD4 counts less than 100. The most widely accepted explanation is impaired cellular immunity due to CD4 T-cell deficiency, which compromises the normal immune response to M tuberculosis infection and facilitates extrapulmonary dissemination. 25 In our study, no differences were found between the different CD4 counts, but there was a very low proportion of patients with CD4 greater than 200, limiting this analysis. Although HIV has been described as a risk factor for all extrapulmonary forms, some studies have reported a specific association with certain forms such as meningeal and miliary, while reporting lower risk for other forms, such as pleural TB. 28 This is consistent with the results of our study, as the regression model for specific forms of EPTB showed that patients with HIV had a lower risk of pleural TB but a higher risk of lymph node TB.
The association of DM with a lower risk of EPTB compared to PTB has already been described in observational studies in China, Brazil, Mexico, and Taiwan.8,12,29 –32 Of these, Leung et al. found an increased risk of PTB specifically in patients with uncontrolled DM. 31 In our study, glycosylated hemoglobin data were unavailable for all patients, so it was impossible to assess differences in disease control. It has been proposed that the mechanism by which patients with DM have a higher risk of PTB is due to a hyper-reactivity of the cellular immune response to the bacillus that causes an insufficient response to contain its growth at the pulmonary level but does not allow its extrapulmonary dissemination.32 –34 On the contrary, other studies have found an increased risk of EPTB in patients with DM and have explained it by their impaired cellular immunity. 35 Discordance may be attributed to a confounding factor, specifically the coexistence of DM with CKD, which has also been identified as a risk factor for EPTB.
The association of a history of smoking with a lower frequency of EPTB compared to PTB had already been described in other observational studies,36 –38 possibly explained by its specific pro-inflammatory effect at the pulmonary level.31,39,40 Similarly, the association between COPD and TB has been documented in multiple studies, although most of them evaluate the history of TB as a risk factor for airway obstruction and few mention COPD as a risk factor for TB. 41 We also found no studies comparing the frequency of presentation of PTB and EPTB in patients with COPD, but it is to be expected that pulmonary involvement is more frequent in these patients due to their anatomical involvement and altered local defense mechanisms, consistent with the findings of this study. However, it is notable that the pulmonary disease most frequently associated with TB risk is silicosis.42,43 In our study this group of patients did not differ between PTB and EPTB presentation. This may be limited by the low frequency of patients with silicoanthracosis included. Evidence for other lung diseases is scarce, with few studies analyzing the frequency or association with TB, 41 and none addressing differences between PTB and EPTB.
The use of tumor necrosis factor-alpha (TNFα) inhibitors has been associated with an increased risk of TB compared to the general population.44 –46 Similarly, several observational studies and case reports have shown a higher frequency of extrapulmonary involvement in this group of patients.47 –49 Our study found no difference in the frequency of exposure to TNFα inhibitors between PTB and EPTB, although the number of patients was small. It is important to note that the use of these drugs was less frequent in the first years evaluated in the study, as the approved indications for their use increased in the last 3 years. When analyzed separately for exposure to steroids, TNFα inhibitors, or other immunosuppressants, no associations were found in the logistic regression model. When analyzed under a single group as exposure to any immunosuppressive drug, these patients were found to have a lower frequency of extrapulmonary presentation compared to PTB. Few studies discuss the specific risk of EPTB for other immunosuppressants. Among these, Qian et al. 35 reported that immunosuppression is a risk factor for EPTB but did not clarify which patients they included in the definition of immunosuppression for the study. No other study has reported a lower frequency of EPTB in patients on immunosuppressive drugs.
Although HIV is the most important risk factor for EPTB identified, there is a high frequency of extrapulmonary presentation of TB in the population without this coinfection, corresponding to 75.85% of patients with EPTB in this study. Moreover, some forms of EPTB were more frequent in patients without HIV than in those with this coinfection. No other associated factors with the development of EPTB were found in the model. The results of other studies are heterogeneous. Some have found an association between female sex and EPTB,12,37,50 –52 arguing possible differences in risk factors due to this population’s lower frequency of tobacco consumption. 10 However, other studies found a higher frequency of male sex 53 and others found no differences in sex, as our study did.
The other most described risk factor in the literature is age, with a higher risk of EPTB in younger patients.12,50,51 In our study, in the initial analysis we found that patients with EPTB were significantly younger. However, when only HIV-negative patients were analyzed, there was no age difference between the two groups, so the higher frequency of EPTB in younger patients may be because HIV infection is more common in this age group. Most studies that showed this association did not take HIV infection into account as a confounding variable. Elderly patients are recognized to have a higher risk of mortality and complications arising from the diagnosis of tuberculosis and represent a diagnostic challenge, as clinical scenarios tend to be atypical. 1 However, some studies have reported conflicting data, indicating a higher incidence of complications in patients younger than 65. 54 This discrepancy may be attributed to the increased frequency of vulnerable scenarios in younger patients, which also heightens the risk of extrapulmonary tuberculosis (EPTB).
Another factor associated with EPTB is CKD due to alterations in cellular immunity.8,35,55 –58 Our study found a higher proportion of patients with CKD among patients with PTB but without a significant association in the logistic regression model. A possible explanation for the difference is that the studies reporting the association between CKD and EPTB refer specifically to advanced kidney disease, 59 and in our population most of the patients were in stage 3.
Other studies have found a relationship between peritoneal TB and cirrhosis, 9 however, in our population, only two patients with cirrhosis were included, limiting the analysis. Similarly, others have found an association between a history of TB and an increased risk of EPTB. 60 In our population there was a very low frequency of reported history of TB in all patients, which could correspond to a reporting bias due to the retrospective study design. An association of EPTB has also been documented with demographic variables such as rural housing, level of development, educational level, or access to drinking water sources, 61 however, we did not have this information from our patients, so it was impossible to explore.
In the analysis for specific forms of PTB, patients with HIV and anemia had a negative association with pleural TB compared to all other extrapulmonary forms, while HIV was associated with lymph node TB. Other studies have described specific risk factors for each site of presentation of EPTB, and even HIV is more strongly associated with specific forms such as meningeal or miliary. 27 These findings suggest that the most relevant risk factors may be specific to each presentation site, rather than the whole EPTB population. As for anemia, the association is noteworthy, but it is not possible to determine whether this is a cause or a consequence of the disease, as no hemoglobin data were available for the patients prior to admission.
In the population studied, 41.83% corresponded to EPTB, which differs from what was reported at the national level (15.6%)2,3 but is similar to the findings in other studies in Colombia. 16 This difference may be due to selection bias, as all the studies were conducted in referral hospitals. Furthermore, the most frequent forms were pleural, lymph node, and miliary, as reported in other studies.15 –17
In general, patients with PTB were diagnosed earlier, mostly by culture and smear microscopy, which is to be expected considering the paucibacillary nature of EPTB and, thus, with a more difficult diagnostic process, with lower test yields and a need for scheduled sampling procedures. In our center, no differences were found in mortality between the two groups and the frequency of drug-resistant TB was low (
Among the strengths of this study is that it was conducted in a referral hospital with a wide range of patients in a country with a high disease prevalence. HIV infection status was known in all patients, unlike most previous studies that looked at-risk factors for EPTB. Among the limitations are those inherent to the study’s retrospective nature, with the possibility of bias in relation to the information available. In addition, the diagnostic difficulty of EPTB implies the possibility of underdiagnosis, which limits the availability of patients to analyze risk factors.
Considering that TB remains a global public health problem with particular importance in high-prevalence countries, it is important to expand studies of vulnerable populations to identify at-risk patients better and facilitate timely diagnosis. This is the first study of its kind in the country, and no risk factors for EPTB were found in addition to HIV infection, which increases the risk for any form of TB. Those previously identified in the literature have heterogeneous results and many have biases that limit their interpretation. Considering the evidence that risk factors may be specific to each form of EPTB, it would be important to conduct separate studies for each presentation to characterize their populations better.
New diagnostic methods that utilize molecular analysis should be implemented to improve the accuracy of diagnoses. Similarly, there is evidence of alterations in the immune system and its receptors that could explain a higher frequency of EPTB. 62 However, in vulnerable countries where tuberculosis is more frequent, there is less availability of advanced diagnostic aids that can identify them. Therefore, it is important to continue searching for clinical variables associated with its presentation, thus facilitating diagnosis in countries with few resources.
Conclusion
The principal factor associated with EPTB identified in this study is HIV infection, especially for the lymph node form while smoking, DM, COPD, lung disease, and immunosuppressant use appear to have a lower risk of EPTB compared to PTB. Information about HIV-negative patients remains limited, and it is not known which factors predispose to EPTB in this population, so studies should be expanded, including some specific to each form of EPTB presentation.
Supplemental Material
sj-docx-1-tai-10.1177_20499361251330802 – Supplemental material for Factors associated with extrapulmonary tuberculosis in comparison to pulmonary tuberculosis in patients with and without HIV in Bogotá, Colombia: an observational study
Supplemental material, sj-docx-1-tai-10.1177_20499361251330802 for Factors associated with extrapulmonary tuberculosis in comparison to pulmonary tuberculosis in patients with and without HIV in Bogotá, Colombia: an observational study by Andrea Vejarano-Pombo, Sandra B. Amado-Garzón, Javier I. Lasso-Apráez, Sebastián Moreno-Mercado, Samuel Martínez-Vernaza, Yulieth García-Pardo and Santiago Carvajal-Leonel in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-2-tai-10.1177_20499361251330802 – Supplemental material for Factors associated with extrapulmonary tuberculosis in comparison to pulmonary tuberculosis in patients with and without HIV in Bogotá, Colombia: an observational study
Supplemental material, sj-docx-2-tai-10.1177_20499361251330802 for Factors associated with extrapulmonary tuberculosis in comparison to pulmonary tuberculosis in patients with and without HIV in Bogotá, Colombia: an observational study by Andrea Vejarano-Pombo, Sandra B. Amado-Garzón, Javier I. Lasso-Apráez, Sebastián Moreno-Mercado, Samuel Martínez-Vernaza, Yulieth García-Pardo and Santiago Carvajal-Leonel in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
