Abstract
Vaccination in Africa faces significant challenges due to inadequate cold chain infrastructure, particularly in regions lacking reliable electricity. Approximately 80% of prequalified vaccines require cold chain systems to maintain their potency, which is often unfeasible in remote areas. Exposure to extreme temperatures can lead to a loss of vaccine potency, making it crucial to explore alternatives. Thermostable vaccines represent a technological advancement that addresses these challenges by eliminating the need for cold chain mechanisms during transport and storage. This narrative review analyzes trends in the uptake of thermostable vaccines and their impact across Africa. A total of 10 studies were reviewed, encompassing 14 African countries, revealing critical insights into the potential of thermostable vaccines to enhance immunization coverage in settings with limited access to traditional cold chain facilities. These vaccines, which maintain their efficacy even when exposed to higher temperatures for short periods, offer a promising solution to the logistical and economic hurdles of vaccine delivery in resource-limited settings.
Plain language summary
To ensure that the vaccine’s stability and viability are maintained, vaccines need to be stored at cooler temperatures. Exposing a vaccine to extreme freezing conditions and high temperatures may result in loss of its potency and viability. Therefore, the cold chain ensures that the potency of a vaccine is maintained. Thermostable vaccines come up to address the challenges faced while trying to maintain vaccines in a cold chain. Almost 80% of prequalified vaccines require cold chain systems to maintain their potency which is not feasible in settings that lack electricity connections. Thermostable vaccines however are technological advancements believed to solve this problem since they do not depend on cold chain mechanisms for vaccine transport and storage. This study involved a narrative review of published articles to assess the trends in the uptake of thermostable vaccines and their impact in Africa.
Background
In addition to being the second-largest continent, Africa is also ranked second in terms of population in the world, with a population of approximately 1.3 billion. It is estimated that one in four children in Africa does not always get access to vaccines, which are believed to be crucial. In addition, over 30 million children did not receive their essential vaccinations before the outbreak of the COVID-19 pandemic.
1
According to the World Health Organization
Introduction
Vaccination is one of the most effective public health interventions for preventing infectious diseases and reducing child mortality globally. One of the major obstacles to successful vaccination programs in Africa is the reliance on the cold chain—a system of storage and transportation that keeps vaccines at a constant low temperature from the point of manufacture to the point of administration. An estimated 80% of WHO-prequalified vaccines require continuous refrigeration between 2°C and 8°C to maintain their potency. 2
Studies have shown that up to 50% of vaccines are wasted due to cold chain failures, leading to significant financial losses and missed opportunities to protect vulnerable populations. The shortcomings of current vaccination storage include several critical factors that hinder effective immunization efforts. First, access to vaccines is limited in many regions due to inadequate healthcare services and infrastructure. Second, the availability of electricity poses significant challenges; frequent power outages disrupt the maintenance of cold chain systems that are essential for vaccine storage. In addition, insufficient storage equipment further complicates the ability to store vaccines properly. Overcoming these barriers is crucial, as it is estimated that improving vaccine availability could prevent approximately three million child deaths annually in developing regions. 3
Thermostable vaccines, which can withstand higher temperatures for a defined period without losing their effectiveness, offer a potential solution to overcome these cold chain challenges. These vaccines could significantly reduce the reliance on complex and expensive cold chain infrastructure, making it easier to reach remote populations and reduce vaccine wastage.
This narrative review aims to synthesize the available evidence on the uptake and impact of thermostable vaccines in Africa. Specifically, the review will:
Summarize the current state of thermostable vaccine development and implementation in Africa.
Identify the benefits and challenges associated with the use of thermostable vaccines.
Assess the impact of thermostable vaccines on vaccination coverage and disease incidence.
Highlight research and practice gaps related to thermostable vaccines in Africa.
Inform future research and policy decisions related to the adoption of thermostable vaccines.
Barriers to vaccination
The primary barriers to vaccination in Africa include several critical factors that hinder effective immunization efforts. First, access to vaccines is limited in many regions due to inadequate healthcare services and infrastructure. Second, the availability of electricity poses significant challenges; frequent power outages disrupt the maintenance of cold chain systems that are essential for vaccine storage. In addition, insufficient storage equipment further complicates the ability to store vaccines properly. Overcoming these barriers is crucial, as it is estimated that improving vaccine availability could prevent approximately three million child deaths annually in developing regions. 1 Thermostable vaccines are proposed as a potential solution to these challenges, particularly in remote areas where electricity is unreliable and vaccine storage is compromised.2,3
Overview of vaccines
Vaccines are substances that are used to elicit the production of antibodies. This will, in turn, lead to protection against a particular disease since your immunity will be boosted as a result.
Vaccines were first introduced by Edward Jenner in 1796. He inoculated a small boy aged 8 years with a matter obtained from a maid who had fresh cowpox lesions. This came as a result of the tales he had initially heard about some of the dairymaids who had cowpox and were immune to smallpox. The boy later developed immunity against smallpox. He then decided to name the procedure vaccination, which was derived from “vacca,” which is a Latin word meaning a cow.
He later studied the procedure again in more people, a study that included all genders and ages. He inoculated them with the matter obtained from the lesion. The children who were not exposed to the disease but who were inoculated gained immunity to smallpox.
His efforts later initiated many vaccines to be developed and promoted advancements in the vaccine sector. However, various technologies have come up recently to improve vaccines since their development. These technologies and advancements are considered a ray of hope since they will help reduce the morbidity and mortality rates caused by preventable infectious diseases. These technologies include the use of live attenuated virus, which tends to isolate and allow the mild growth of virus and bacteria in vivo, the use of inactivated vaccines, the use of genomic-based approaches to help in the identification of the antigen, the use of thermostable vaccines, among others.
Cold chain mechanisms
This was first developed by Frederick McKinley Jones. The instability nature of vaccines, since they are prone to physical and chemical degradation, makes their shelf lives inadequate. 4 The rate of potency loss is dependent on temperature. This led to the application of cold chain mechanisms in vaccine logistics to maintain vaccine potency within a specified range. To achieve an effective immunization program, thermostability of the vaccines has to be considered since it largely affects the quality and nature of the vaccine.
Vaccine thermostability is achieved by maintaining the minimum required viral potency within specific temperature ranges. This is highly dependent on many factors such as the given vaccine type, mode of administration of a given vaccine, and its Active Pharmaceutical Ingredient (API). This has, in turn, led to the adoption of a cold chain mechanism for every vaccine. Cold chain mechanisms involve handling, transporting, and storing the vaccine within a given temperature (e.g., 2°C–8°C). This mechanism should begin from the manufacturer to the consumer, as shown in Figure 1.
Cold chain mechanism process from manufacturer to consumer, illustrating temperature requirements at each stage.
The adoption of cold chain mechanisms, however, has posed various challenges all over the world. This resulted in the introduction of vaccines that do not necessarily require refrigeration. This new intervention is being adopted to try and ensure more vaccines reach populations in remote and isolated localities.
Challenges with cold chain systems in Africa
Many case studies, however, have been conducted regarding the challenges posed by using this cold chain system method in vaccine logistics. In a study done in Ethiopia, Kenya, Malawi, Tanzania, Nigeria, Mozambique, and Cameroon, among the challenges raised were a lack of proper cold chain capacity, a lack of the latest technology, and an inadequate system for monitoring temperatures accurately. 5
The challenges were a bit similar in most studies, However, a study done in Tanzania in a remote area pointed out some health centers lacked basic equipment essential for vaccine storage, such as refrigerators, vaccine carriers, a cold box, and maximum and minimum thermometers essential for monitoring temperatures. Other health centers had the equipment, but they were ineffective. To achieve effective immunization, such factors should be considered.
Another challenge posed is that when doing an intensive immunization campaign, the use of the cold chain is labor-intensive. Logistical challenges exist in the process. This is not limited to frequent electric power failures, malfunctioning, or missing equipment. 6 Hence, approximately 44% of infants fail to receive an immunization. 7
In places where the average daily temperatures are >30°C, maintaining the cold chain always poses a serious challenge. The challenges are exacerbated during immunization campaigns where cold chain systems and logistics are required before the vaccine reaches a wide target population. 8
Cold chain systems have also been proven to be expensive in terms of transportation costs, especially in emergency campaigns when you have to resupply vaccines, cold boxes, and enough ice packs to ensure the given vaccine potency is maintained. The transportation costs may include fuel consumption costs and travel allowances offered to the workers. It is worth noting that the cost incurred in purchasing cold chain equipment, such as refrigerators and backup generators, at the national and district levels is high. Moreover, more costs are incurred when training staff on Cold Chain Logistics(CCL) since the number of personnel trained to handle vaccines is low.8,9
mRNA-based vaccines, such as the BioNTech/Pfizer COVID-19 vaccine, require storage at temperatures of −80°C with a shelf life of up to 6 months. By contrast, the Moderna COVID-19 vaccine necessitates storage at −20°C. These deep freezing requirements have complicated distribution in resource-poor settings, presenting significant challenges for vaccination campaigns in areas that lack reliable electricity and infrastructure. 10
According to WHO data on vaccine wastage, almost 50% of the vaccines are rendered unsafe for use during the cold chain process, thus affecting the vaccination process. This increases costs for vaccination programs. 11 This may be due to exposure to accidental freezing and high temperatures. Accidental freezing always occurs when vaccines are kept very close to the walls of refrigerators lined with ice. Ice packs should be allowed to melt (preconditioned) before they are used. Failure to allow it to melt may further expose the vaccine to freezing temperatures during logistics. This damages the given vaccine. In vivo and in vitro laboratory-based studies have shown that freezing of vaccines containing aluminum salt adjuvants tends to damage the vaccine. Although aluminum adjuvants are always unaffected by high temperatures, exposure of the vaccine to high temperatures may cause interference with their protein structure.
The numerous studies conducted across Ethiopia, Kenya, Malawi, Tanzania, Nigeria, Mozambique, and Cameroon have identified several challenges associated with cold chain systems for vaccine storage and distribution. Key issues include inadequate capacity in many health centers, which often lack the necessary cold chain equipment. In addition, logistical difficulties arise from frequent power failures and malfunctioning equipment, which hinder effective vaccine distribution. The high costs associated with maintaining cold chain systems also pose a significant barrier for many healthcare facilities. These challenges underscore the urgent need for alternative solutions that can ensure vaccine stability without relying on traditional cold chain logistics.
Controlled temperature chain
This is a mechanism that allows vaccines to be stored out of the cold chain. Normally, a cold chain allows exposure to temperatures ranging from +2°C to +8°C for cooled vaccines from the manufacturer to the various health centers being delivered. This mechanism, however, allows exposure to temperatures until 40°C for many days just before it is administered. In this system, the risk of exposing the given vaccine to freezing temperatures is eliminated.
As part of its efforts to ensure the quality of the vaccine, WHO has also come up with guidelines that form a basis for vaccine acceptance globally. It also states that it is important to define the stability characteristics of a vaccine and emphasize the role of national regulatory authorities in overall vaccine evaluation.
The vaccines should be used in a campaign setting instead of an immunization setting or a strategy instead of an immunization setup.
Temperatures up to 40°C have to be well tolerated by the vaccines. Each vial should have a system that allows accurate monitoring of temperatures.
Vaccines must have acquired a license for them to be included in the Controlled Temperature Chain (CTC) by the relevant bodies. The conditions to be endured during the CTC have to be clearly labeled. 12
Currently, two vaccines have been pre-qualified by WHO to be used for use under CTC. These are as follows:
MenAfrivac—It was prequalified by WHO. It is the first vaccine to be used under CTC in a mass campaign. It was developed by the Meningitis Vaccine Project and manufactured by the Serum Institute of India. It can be handled in CTC at 40°C for 4 days before its administration (WHO, CONTROLLED TEMPERATURE CHAIN Strategic Roadmap for priority vaccines 2017–2020, and 2017). 13
Gardasil—It is also known as the Human Papillomavirus 9—valent vaccine.
Out of the cold chain
As opposed to CTC, they do not necessarily possess a comprehensible description and monitoring regulations. The term Out of Cold Chain (OCC) implies the act of removing a vaccine out of the cold chain because the given vaccine is thermostable, without necessarily requiring approval from the regulatory bodies and without necessarily receiving prequalification from the WHO.
It is regarded to be “off label” since it denotes that it no longer follows the established vaccine manufacturer handling guidelines and the Expanded Program on Immunization (EPI) policies.
The role of thermostable vaccines
Thermostable vaccines present a promising solution to these challenges. Unlike traditional vaccines that require strict cold chain management, thermostable vaccines can be stored at higher temperatures without losing potency. This innovation is particularly beneficial for remote and isolated areas where electricity is unreliable.4,5
Trends in thermostable vaccines development
Since the introduction of vaccines, most have been presented in liquid form. Even as more vaccines were being introduced, the production of liquid vaccines continued. Production of freeze-dried vaccines was only done to achieve vaccine stability. 14 However, new ways have emerged to enhance vaccine stability to make it thermostable.
Thermostable vaccines are vaccines that can be stored at high temperatures, normally up to 40°C, without their potency being affected immediately before their administration. Methods commonly used include drying techniques, the addition of adjuvants in liquid formulations containing aluminum salts, minimizing proton exchange reactions in aqueous solutions, and the use of delivery technologies such as MN patch needles. The stabilization process in vaccines is normally achieved by enhancing the vaccine matrix composition, that is, the vaccine formulation. This is normally achieved through the removal of water and thus preventing the degradation of the vaccine.
Common drying processes to produce thermostable vaccines
Drying is a common method that is used to stabilize various vaccines. Drying involves three procedures. First, an energy force is exerted on the medium through an external source. The next phase involves evaporation or a sublimation technique, depending on the nature of the product. The final procedure involves vapor transfer, which is obtained from the overall procedure. The process can be done by using a mechanism that involves evaporation, such as vacuum or foam drying. It can also be done through a mechanism involving a combination of evaporation and atomization, like spray drying. Other mechanisms that can be used include spray freeze drying (FD) and freeze-drying mechanism, which employs sublimation mechanisms, and the supercritical fluid drying method, which is commonly done using a mechanism that involves precipitation. 15
Freeze drying
This involves three phases: These processes include freezing, primary drying, and secondary drying. Table 1 shows examples of vaccines that have been successfully freeze-dried and their stability results.
Examples of vaccines that have been successfully freeze-dried and their stability results.

Freezing process
The freezing process involves allowing the material to freeze. This is made possible by putting the material inside an FD flask and rotating it inside a bath, which is normally referred to as a shell freezer. The process of cooling is made possible by methanol, dry ice, and refrigeration. This will later make sublimation occur in the next steps. The duration of the procedure depends on the size of the objects. Larger objects tend to take a longer duration. 16
Primary drying
This procedure involves lowering pressure and supplying heat, which is enough for it to sublimate. At this step, nearly 95% of the water sublimates. A vacuum is used in the process to speed up the sublimation process.15,16
Secondary drying
This process aims to remove the unfrozen water, which was left behind after primary drying. In this process, the temperature is raised, and then the pressure is lowered. Water content that tends to remain behind is less than 5%.15,16
Spray drying
This method is normally used in the stabilization of liquid vaccines. The procedure involves the production of a dry powder using hot gas. The procedures used do not need freezing at any point. Unlike other traditional procedures, which tend to take 3 days to be manufactured, this procedure takes less time. It takes seconds, and the process continues until enough vaccines are made. 17 The first procedure was executed in 1901 by Robert Stauf. 18 Despite the results from various spray-dried vaccines demonstrating encouraging and promising results, this is one of the new fields when it comes to vaccine thermostability and delivery. Examples of vaccines that have been successfully freeze-dried include influenza vaccine,19,20 tetanus, diphtheria, 21 hepatitis B vaccine, anthrax vaccine, 22 plague vaccine, 23 spray-dried measles, and TB vaccine 24 .
The advantage of the spray drying technique, in contrast to freeze-drying, is that it requires no freezing. Due to this, less energy is consumed in the process since it is a one-time process. This has, in turn, been associated with reduced costs of operations. 25 The procedure also ends up in the production of fine-textured particles in the form of powder compared to freeze-drying. This will, in turn, lead to handling and administration of powder without the need for reconstitution. The drawback associated with this mode of delivery is that the antigen may be exposed to very high temperatures and stress during the drying process. A secondary phase of drying might be required if the lower water content is desired in the final product. 26
Spray drying procedure
It is a procedure that involves the production of non-hydrous powder from a liquid medium. The first procedure involves the atomization of the liquid medium. In this method, the liquid medium and nebulizing agent are mixed and made to exit through a nozzle. This procedure results in the liquid becoming atomized, which is then converted to spray-like droplets. The droplets are then introduced into an environment that contains hot gas. It is important that in this phase, a co-current flow is considered if the vaccine is heat sensitive. In this process, the wettest spray-like droplets are exposed to high temperatures while the least wet droplets come into contact with the lowest temperatures. Apart from co-current flow, other kinds of flow available include mixed flow and counter-current flow. The dried gas used in this process is then filtered, and water is removed from it in a process called dehumidification. It is then reintroduced to the drying chamber. Such systems are said to be closed. In an open system, the gas is released to the outside, but it has to undergo filtration first. The final dried particles obtained in this process are always separated from the stream by the use of baghouse filters or cyclone separators.26,27
Super critical fluid
This has resulted in the production of thermostable dry powdered vaccines. An example of a supercritical fluid commonly employed is supercritical CO2. A given fluid is regarded as a critical fluid if its temperature and pressure have surpassed the given critical threshold. When the temperature rises above this given level, it cannot be changed into a liquid even when the pressure is increased. In addition to this, at this point, they tend to be unique and flexible, which makes them highly soluble and cosolvents added easily. This property has enabled it to be used in the drying process to produce vaccines. Examples of vaccines that have been produced using this process include measles vaccines, which can stay up to 1 week at 37°C. 28
Development of delivery technologies
The use of delivery technologies such as microneedle patches and biodegradable implants is also believed to solve problems caused by the cold chain.
Development of thermostable liquid formulations
This is normally achieved by the use of a high screening output method. These are normally done by assays. These assays are used to monitor changes in the vaccine’s proteins when subjected to different conditions. The conditions include salts, buffers, PH, and temperature. The results obtained will assist in creating a profile of a vaccine’s stability. This assay can also be used to screen excipients so that the stability of the vaccine is enhanced even when exposed to various conditions. This method has been used in the malaria vaccine by screening the 32 GRAS compounds’ ability to stabilize the given vaccine at 45°C. This approach has also been applied to the Clostridium vaccine and the measles vaccine. 14
Stabilization by optimization of vaccines containing aluminum adjuvants
Aluminum in liquid vaccines tends to boost the immune response. However, exposure to freezing temperatures tends to damage the given vaccine by causing the proteins to unfold. This is due to depletion of the water molecule, resulting in disruption of the hydration shell that surrounds the antigen. Enhancing the stability of these vaccines requires one to be aware of how freeze-sensitive the adjuvant is, the heat and freeze sensitivity of the antigen, and ways in which it can be formulated to solve the instability issue. 29 The addition of excipients has proven to be effective. 14
By reducing nucleon exchange reactions
The uninterrupted proton exchange reactions that occur between the liquid environment and the protein itself always lead to denaturation. This can be controlled by the addition of particular buffers.
Methods
This study involved a narrative review of published articles to assess the trends in the uptake of thermostable vaccines and their impact in Africa. The search process was conducted independently by two authors. We searched through various databases, including ScienceDirect, Web of Science, PubMed, ClinicalTrials.gov, and Embase. The terms used to search for various publications for this study included “Thermostable vaccines” AND “Africa,” “vaccine*” AND “cold chain.” Information was also sought from key immunization stakeholders and references for the articles. The selection of studies and extraction of data were done by two authors, and they were filled out using a standard data extraction form. The result was reported through a narrative synthesis.
Results
Study selection
In all, 5130 articles were found during the literature search, 1099 of them were found to be duplicates and were eliminated, and 3500 articles were eliminated after making the search process more specific. The abstracts and titles of the remaining articles were then reviewed, and out of that, 200 were considered irrelevant to the topic. After reading the full articles, 321 studies did not meet the inclusion criteria and were removed. Finally, we considered 10 articles appropriate for the narrative synthesis as illustrated in Figure 2.
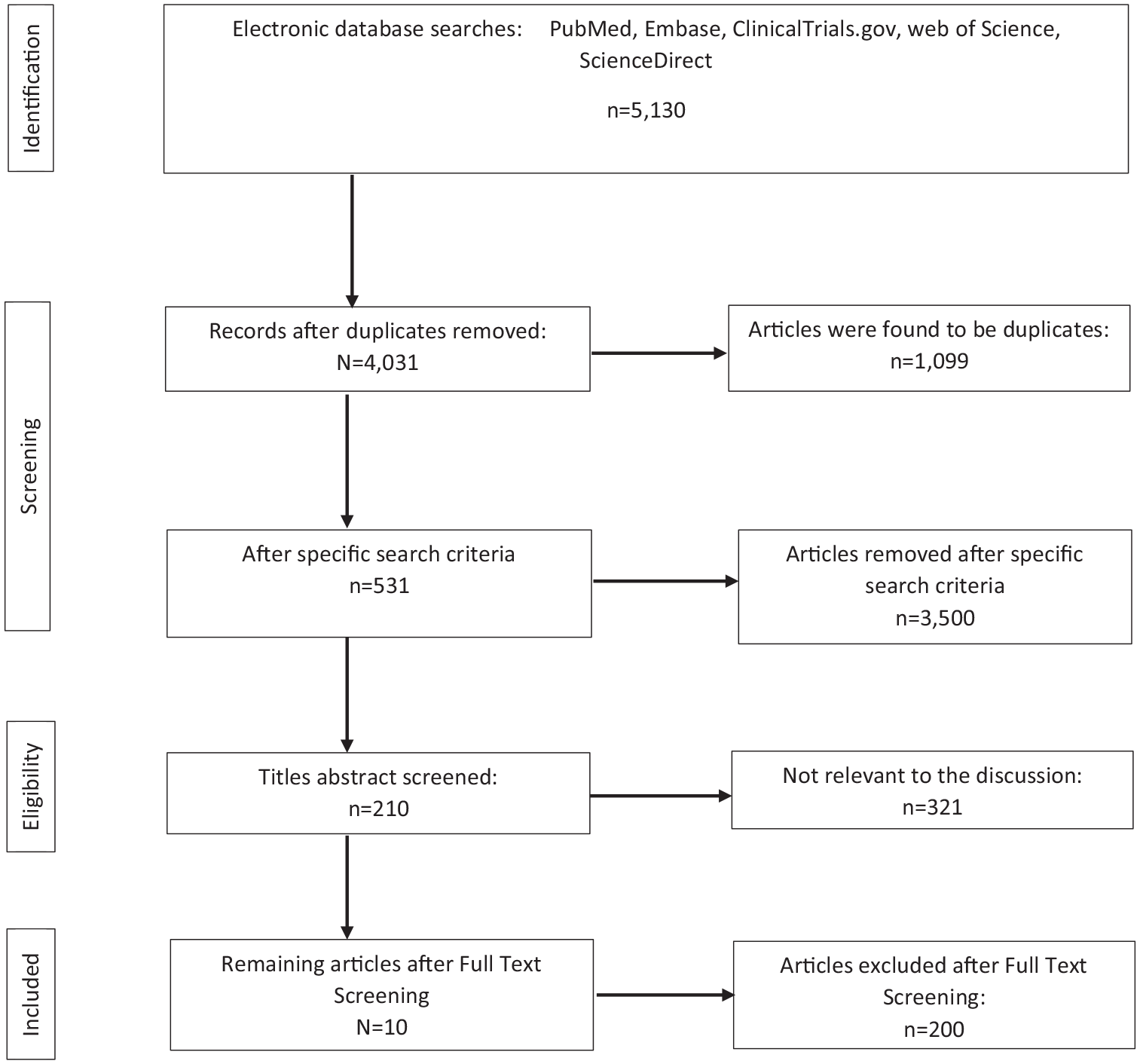
Study selection.
Our review included 10 studies conducted across 14 African countries (Table 2). Figures 2 and 3 illustrate the uptake of thermostable vaccines across Africa. The studies evaluated the use of thermostable vaccines for meningitis (MenAfriVac), measles, and polio. The primary outcome measures were vaccination coverage rates, vaccine efficacy, and cost-effectiveness.
Uptake of thermostable vaccines in Africa.

OCC, out of cold chain.

The uptake of thermostable vaccines in African countries.
MenAfrivac, Rotasiil, Gardasil, ND12, and NOBIVAC are among the thermostable vaccines that have been used and stored under OCC. Table 2 gives a good picture of the year of uptake, countries in Africa where they were used, and their storage temperatures.
Clinical trial on ROTASIIL, Nigeria
This study was published in the NEJM in 2017. The study aimed at comparing the efficacy of ROTASIIL, which is thermostable, with regards to placebo. The temperature of storage of the vaccines when they were being shipped to the health facilities was stated. On reaching the facility, the temperature was up to 20°C and ambient when transferred from the facility. The randomization process was ideal since the intervention assignment was computer-generated and the researchers, as well as participants, were blinded. This action reduced performance bias.
The authors concluded that ROTASIIL was safe and efficacious in regards to the placebo. According to the data, severe gastroenteritis was reported in 31 infants who took ROTASIIL compared to 87 in the placebo group, resulting in an overall vaccine efficacy of 66.7% at a 95% confidence interval in the per-protocol group. On the other hand, in the intention-to-treat group, gastroenteritis was observed in 35 infants in the ROTASIIL compared to 125 infants in the placebo. The overall efficacy was 69.1% at 95% CI. 30 The author highlighted the advantages, saying that the overall costs were reduced and the logistic process was eased 26 and further gave evidence that this can be used practically in the field. ROTASIIL stability features are based on the fact that it is lyophilized and possesses a stability agent. It can be kept at temperatures up to 37°C for 20 months, 45°C for 7 months, and 5°C for more than 3 years. 32 ROTASIIL has been licensed but has not been introduced on a national scale. 37
Emergence of Newcastle disease thermotolerant vaccine (ND12), Ethiopia
Newcastle disease has been a serious problem affecting mostly village flocks. This led to the production of thermotolerant 1-2 and V4 master seeds. These were produced by Panvac Ethiopia. Various laboratory and field-based studies have been done in Tanzania, Zambia, and Mozambique. In Tanzania, the field study was done in the southern regions, that is, Lindi and Mtwara. The study aimed to find out whether when chickens were vaccinated with the thermotolerant ND 12, it would enhance their immunity, which later led to the production of IgG antibodies after they were vaccinated.
The results clearly show that the use of ND12 induced a significant response of IgG in the chickens. 73.3% of the sampled chickens were found to be immune to Newcastle disease. The vaccine was found to be potent for Newcastle disease control and was very flexible for use in a rural setting. The study was done in a remote area. 35 Many countries have implemented the use of the ND12 vaccine. The countries include Malawi, Mozambique, and Tanzania. They are now at the lead in the control of Newcastle disease in animals and are producing and using increased amounts of this vaccine. 38
Field trial on Tetanus, Chad
This article was published on the Elsevier website on 9th September 2011. The trial aimed to compare the delivery of TT when administered CTC compared to that administered in the cold chain. It was a non-inferiority type of study demonstrating the non-inferiority of this vaccine when kept under two different conditions. 36
It was a cluster-randomized field trial that took place in the Moissala district in Chad. Allocation to the two groups was performed at the cluster level. In all, 2128 female participants were recruited for this study, where CTC had 1068 women and Standard Cold Chain (SCC) had 1060 women.
The results demonstrated that using CTC retained adequate levels of potency. However, there were no differences between the two groups in regards to the immunological response.
A study on thermostable vaccine against PPR, Kenya, and Ethiopia
This study was published on 29th May 2017 on the Elsevier website. The major aim was to find a heat-stable vaccine against PPR, which affects ruminants, and to compare its thermostability aspects with others of the same kind. The authors clearly stated the reason why this study was done and stated that it was because the use of cold chain systems required refrigeration, and they needed a vaccine that would be easily used in the field without the need for refrigeration to fully eradicate the disease.
The existing PPR vaccines require the use of a cold chain, which makes them disadvantageous for use in developing countries, more so in rural areas where the spread of the disease is rampant. The authors addressed the methodology they used to come up with the vaccine and how they intend to come up with the analysis. It was found out that stabilization using sucrose together with lactalbumin enhanced the thermostability aspects of the RP vaccine derived from PPR via the lyophilization process, and it was superior when compared to those tested. 39
A similar study was conducted in ILRI Ethiopia. However, this study did not give enough evidence that a similar methodology that was used could be used in a practical setup. 35 The authors also highlighted that when this is applied, it would reduce the burden posed by the use of cold chain mechanisms. However, they did not list the limitations of the study.39,40
Field trial on Menafrivac, Benin
The vaccine obtained a label variation to be used OCC at temperatures up to 40°C up to 4 days in October 2012, and the first field use was demonstrated in Benin in December 2012 at a district called Banikoara. Benin was the first country to use it in a CTC in humans. The pilot study that was conducted in Benin at a rural district ended up with a 106% coverage. In all, 155,000 people in 150 villages ended up getting vaccinated. There were no cases reported of the disease. Only four vials were discarded. During the immunization campaign, another study was carried out concurrently to monitor adverse events in those under CTC and those using the cold chain. They wanted to find out if there was any presence of adverse events, which occurred as a result of immunization. This study was done to give a clear picture of the safety of MenAfrivac when delivered under CTC and when it is delivered after being stored in a cold chain. They selected one district to receive the vaccine through CTC and the other one on the cold chain.
Results showed that this vaccine is safe for use; its use did not increase adverse events. 41 Other countries that used MenAfrivac under CTC are Cote Ivoire, Mauritania, and Togo in 2014. The use of a CTC approach was associated with remarkable benefits in regards to logistics. 42
Pilot study on Gardasil (HPV4), Uganda
Gardasil is a vaccine that helps in safeguarding against strains of HPV. Gardasil is heat stable and can be kept 130 months or more at 25°C, 1.5 years at 37°C, and 3 months at 42°C. 43 Uganda was the only country in Africa chosen for the HPV pilot study to take place by Path. Other countries where the pilot studies took place include Vietnam, Peru, and India. 44 The average mean temperatures were between 22.9°C and 30°C. 45
A cross-sectional study was done at Lira District to find out the uptake of Gardasil. Data regarding the uptake were taken based on respondents’ memory alternatively vaccination cards. However, obtaining information depending on the respondent’s memory might likely lead to information bias. Some participants may choose to give the wrong information, while others may be suffering from amnesia, which may affect the overall study process. The target group was females aged 12–17 years. The study showed an 18% uptake of Gardasil in Uganda. 46
Impact
The clinical trial was done in Niger demonstrated that it can reduce the burden caused by the cold chain including ease of access to remote areas, reduction of vaccine wastage due to exposure to extreme temperatures, and reduction of financial costs. These impacts were similar across most of the studies.47–49 A cross-sectional study was done in Cote d’ Ivoire, and it aimed to find out their opinion on CTC, 98% of the participants were excited about the new intervention after the introduction of MenAfrivac. The advantages cited were that no ice packs are required, it saves more money, and the vaccine carriers used are not heavy. 50
A study aimed at finding the economic benefits of keeping vaccine OCC during the Meningitis campaign in Chad showed decreased transportation costs, decreased costs in the purchase of equipment which is needed to maintain the cold chain system, and decreased expenditure in terms of training human resource. 8 In Benin, among the benefits listed was that it was easy to administer the vaccines, especially in places where the use of CCL was impractical. 6 However, the limitations of adopting this new method are that more time and resources will be needed to make health workers know about this new intervention and to avoid confusion.
Discussion
The findings of this review align with previous research on the use of thermostable medications in resource-limited settings, which have demonstrated that reducing the reliance on the cold chain can significantly improve access to essential medicines. 51
The emerging development of cold chain mechanisms demonstrates how many challenges and limitations can be solved through the use of technological advancements. This is not limited to the development of vaccine vial monitors due to the need to monitor the exposure of vaccines. The need to store vaccines during CCL has also led to the introduction of refrigerators, cold boxes, and vaccine carriers. To overcome the various barriers that the cold chain mechanisms present, various vaccine manufacturers have started venturing into the production of thermostable vaccines.
The use of thermostable vaccines results in cost savings in various ways. The vaccine supply bottlenecks in remote areas were eliminated since additional trips needed to supply cooled vaccines were eliminated. The logistics involved while handling and distributing thermostable vaccines were also made easier and more cost-effective. Therefore, as more vaccines reached the targeted population, there was an overall decrease in costs and vaccine wastage. These findings help support the positive impact of thermostable vaccines, hence encouraging their development.
Currently, little has been done to encourage the research and development of thermostable vaccines. This is attributed to the fact that, as long as other vaccines are still required to be maintained in a cold chain, then thermostable vaccines will have limited value. One assumption was that thermostable vaccines would completely obviate the use of refrigerators and freezers. However, there are still additional benefits even when these cold chain mechanisms remain. The thermostable vaccines will still help eliminate constraints in the vaccine supply chain by making more cold space available for other vaccines.
A study conducted in 2007 assessed the effects of introducing four thermostable vaccines in Ghana: Measles, Yellow Fever, Bacille Calmette-Guérin (BCG), and Diphtheria–Tetanus–Pertussis–Hepatitis B. Utilizing a cost-effectiveness model, the research estimated both the health and programmatic cost impacts of these new vaccines. The findings indicated that the implementation of thermostable vaccines could potentially reduce the disease burden in Ghana by 16%–48%, depending on the specific antigen involved. 51
There is high feasibility and general acceptability of thermostable vaccines by healthcare workers regardless of their area of specialization or experience. This indicates that professionals, vaccine manufacturers, governments, and other stakeholders need to place more emphasis on thermostable vaccines. This will result in improved vaccine delivery and logistics, widened immunization coverage, and increased vaccine efficiency. Ultimately, this will result in decreased mortality associated with preventable diseases.
The studies also demonstrate that thermostable vaccines are safe and stable and offer remarkable benefits to both animals and humans. Being an era of one health, we thought of including the uptake of thermostable vaccines in humans and animals.
Thermostable vaccines have demonstrated that they can be successfully used in Africa, especially in places with inadequate electricity supply and in remote areas and places where the temperatures are elevated. The use of thermostable vaccines has increased vaccination coverage, with some areas covering more than the expected threshold. It has also led to reduced transportation costs, especially in immunization campaigns where vaccines and icepacks need to be replaced, and a reduction of costs incurred due to training human resources on CCL. Moreover, there has been a reduction in the costs incurred on purchasing storage equipment at the national level and the health facility level.
This study covered the majority of thermostable vaccines under CTC, but they can still be administered under OCC, but not fully under OCC. Since it is a new intervention, we found very few interventions in this field in Africa. The majority of the trials and studies were being conducted outside Africa. However, this study has critical positive impacts on the uptake of thermostable vaccines in Africa.
Limitations
This review is subject to several limitations. The number of studies evaluating the uptake and impact of thermostable vaccines in Africa remains limited. Furthermore, the included studies were primarily conducted in limited countries in Africa, which may limit the generalizability of the findings to other areas. There may also be publication bias, as studies with positive results are more likely to be published.
Conclusion
The study found that although the majority of the healthcare personnel had experience in the supply chain of cooled vaccines, they had poor knowledge about thermostable vaccines. The general feasibility and the acceptability of thermostable vaccines by healthcare workers were generally high. This denotes that healthcare professionals are aware of the various challenges faced in the vaccine supply chain and are constantly seeking various ways of overcoming them. It is therefore concluded that to optimize the vaccine supply chain in remote and marginalized areas, the development and use of thermostable vaccines should be encouraged. This sets the stage for governments, manufacturers, and other stakeholders to consider investing more resources into researching and developing thermostable vaccines. Cold chain systems should be complemented with thermostable vaccines and be included in the routine immunization process and campaigns to achieve a successful immunization program that will tend to cover a larger area, especially in pandemics such as COVID-19. This will enable more health centers to have access to these vaccines. Manufacturers should also incorporate methods of producing thermostable vaccines so that the use of cold chain is minimized, if not eliminated. More clinical trials should also be performed in Africa to give a clear picture of the importance of thermostable vaccines in Africa. Thermostable vaccines should be considered, especially when developing vaccines, particularly during an outbreak, especially in Africa, to reach a wider coverage. WHO should consider this data and put in place measures and guidelines that are needed in the implementation of OCC practice.
