Abstract
Given the surge in mpox outbreaks in 2022 and the advancements in domestic and international vaccine research, the effectiveness of smallpox vaccines in providing cross-protection against mpox remains crucial. Having learned from the COVID-19 pandemic, it is significant to continue evaluating existing vaccines to ensure their safety and efficacy. Developing new vaccines for widespread use against mpox and its emerging strains also serves as a preventive strategy in the ongoing battle against this dynamic infection. Here’s an opportunity to control human-to-human transmission, give short deadlines, and avoid vaccine disparity. Public health systems must take decisive action to prevent the global spread of mpox, particularly among vulnerable groups. This action should include strengthening global surveillance, improving vaccine access, and ensuring equitable distribution, particularly in resource-poor settings, to prevent future outbreaks. This review aims to assess recent advancements and barriers in mpox vaccine development, emphasizing cross-protection and equitable vaccine distribution in resource-poor settings.
Introduction
The public health system is increasingly concerned about the potential threat posed by Monkeypox, now named mpox, following a recent outbreak.1,2 The outbreak intensifies due to concerns about a novel transmission dynamic route, particularly sexual contact, and the emergence of more transmissible strains. The documented spillover of mpox from humans to animals complicates the situation, increasing the possibility of new animal reservoirs. 3 The mpox virus is a viral zoonosis caused by a double-stranded DNA virus belonging to the Orthopoxvirus genus of the Poxviridae family, which also includes variola, cowpox (CPX), and vaccinia viruses. 4 Mpox has historically been endemic in Western and Central Africa, especially in the Democratic Republic of the Congo (DRC); nevertheless, the recent occurrence of cases in countries where it is not endemic indicates previously unknown transmission and potential viral adaptation. 5 Mpox is divided into two genetic clades: the Congo Basin clade (Central African), which is more virulent and has a higher case fatality rate (>10%), and the West African clade, which has a lower-case fatality rate (<1%). 5 The latter is primarily linked to the 2022 outbreak in nonendemic regions and has raised urgent questions about global response, particularly in vaccine development, distribution, and accessibility. Recent findings revealed that the 2022 mpox virus genome sequences originated from the West African clade (lineage B.1), categorized as Clade IIb. 6 Global MPXV clade IIb sequences, dominated by the B.1 lineage, showed community transmission within and across regions due to importations. Detailed, since 2022, the World Health Organization (WHO) European Region has been experiencing an outbreak of mpox, primarily affecting males who engage in sexual activity with men (MSM). Following that, case numbers decreased quickly and persisted at low levels until a slight rise began in the summer of 2023, which has continued to the present. Low-level cases of sexual transmission among MSM continue in the summer of 2023, with a slight shift toward younger age demographics. The decrease and revival may be caused by behavioral changes, immunity acquired through vaccination, or previous infections in vulnerable populations. Additional research is required to guide effective national preparedness and control actions in the future. 7 The sequences in the European Region belong to clade IIb, but there is no evidence of significant transmission of clade I viruses. Concerns arise about the geographical expansion and sexual transmission of clade I in endemic countries. 8 The recent multinational outbreak in nonendemic regions received more attention, particularly because of the evolving characteristics of the disease. This review assesses the current state of mpox vaccine development, highlighting recent advancements, challenges in deployment, and future research directions to improve vaccine efficacy, safety, and accessibility globally. Numerous studies on mpox vaccines have been conducted, and this review emphasizes the most critical findings to highlight key areas for progress and action.
Current status of mpox vaccines
Approved vaccines, status of clinical trials, and vaccine platforms
The smallpox vaccine has undergone three generations of technological advancements, with only the second and third versions now licensed. Three orthopoxviral vaccines are available: ACAM2000, 9 JYNNEOS,10,11 and LC16m8. 12 The initial vaccine, ACAM2000, is replicating; conversely, the other two vaccines are either nonreplicating or exhibit minimal replication. 13
Early-generation vaccinia virus (VACV) immunizations effectively protected against smallpox and mpox, but they were associated with adverse side effects such as progressive vaccinia, eczema vaccinatum, generalized vaccinia, postvaccinal encephalitis, myopericarditis, inadvertent self-inoculation, transmission to close contacts, and mortality and contraindication in certain populations.14,15 Dryvax and Lister/Elstree were among the first-generation smallpox vaccines, which develop from live and nonattenuated viruses. Numerous studies demonstrate the effectiveness of Dryvax against mpox,10,16,17 but due to significant safety and reliability concerns, these vaccines are no longer in use, leading to the development of second-generation vaccines such as ACAM2000, CJ-50300, and APSV.13,18,19
ACAM2000, a second-generation live and replication-competent vaccine received approval from the FDA in 2007, 20 is authorized for use in preventing smallpox exposure during an outbreak or emergency. 21 ACAM2000 was extracted from a single-clone virus isolates of the first-generation Dryvax vaccine. A multicenter randomized controlled study tested ACAM2000 and concluded that it is effective and acceptable as a booster dose for previously vaccinated individuals. ACAM2000, while having comparable immunogenicity to the first-generation and fewer severe side effects, can still cause serious conditions, making it contraindicated for individuals with compromised immune systems. ACAM2000 is associated with certain severe adverse events, including myopericarditis, encephalitis, eczema vaccinatum, and progressive vaccinia. 22 The Food and Drug Administration (FDA)-approved ACAM2000 against mpox under the Expanded Access Investigational New Drug (EA-IND) protocol, but it requires informed consent and the completion of additional forms. However, the 2022 outbreak did not use ACAM2000.19,23,24
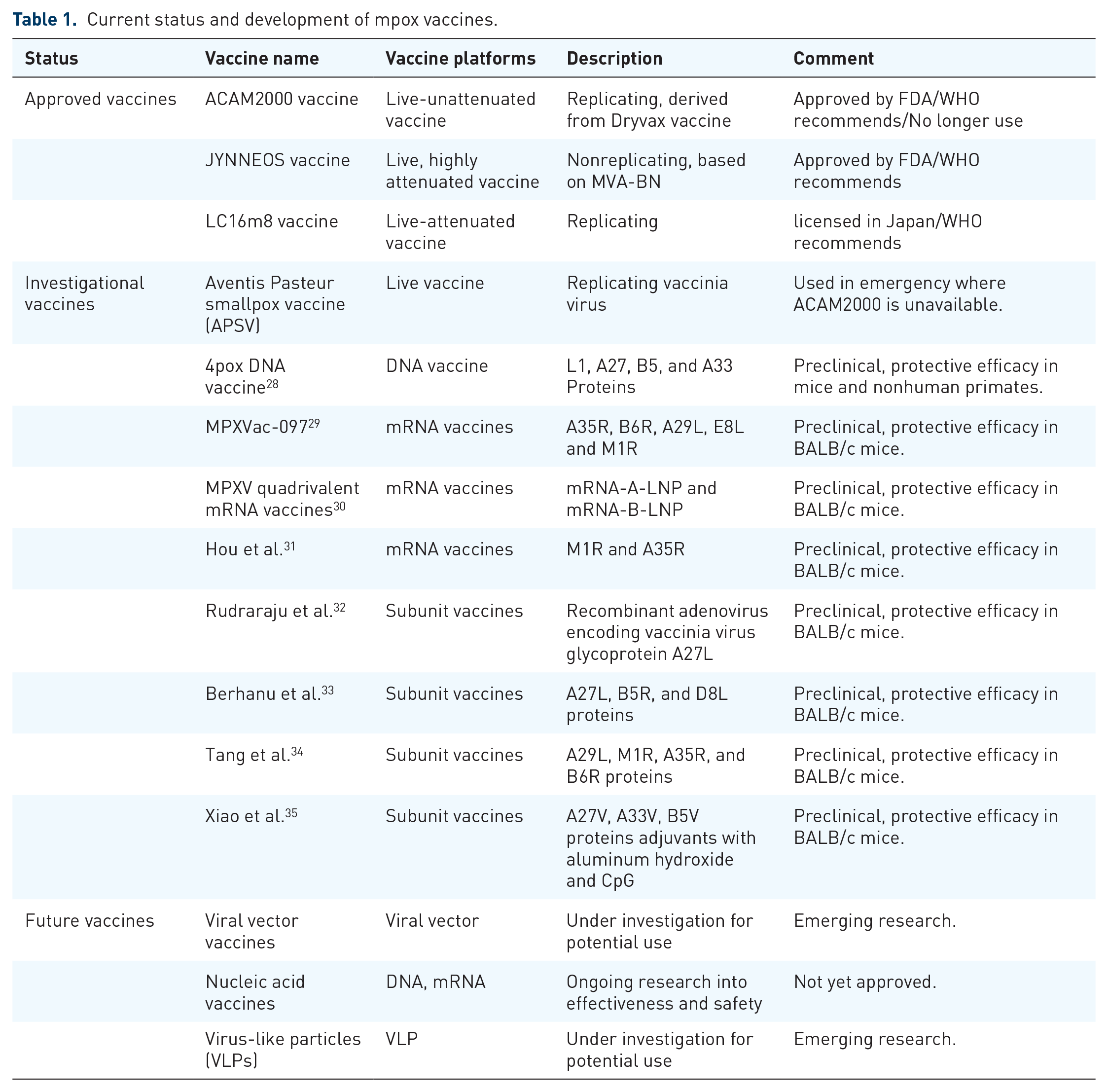
The third-generation vaccines, Modified Vaccinia Ankara-Bavarian Nordic (licensed by Bavarian Nordic in Hellerup company, Denmark, MVA-BN) and LC16m8 (licensed by KM Biologics company in Kumamoto, Japan), have reduced virulence and replication ability by multiple passages. The LC16m8 vaccine, a live attenuated formulation, demonstrates potential in animal models but necessitates additional research on its long-term protection. 15 Twenty-two clinical trials were submitted to support the effectiveness and safety of the MVA-BN vaccine. The FDA initially approved MVA-BN, a third-generation, live-attenuated, nonreplicating Ankara vaccine known as Modified Vaccinia Ankara,25,26 branded as JYNNEOS in the United States, Imvamune in Canada, or Imvanex in Europe, as a smallpox vaccine. On August 24, 2019, the FDA also approved JYNNEOS against mpox.18,24 In individuals participating in smallpox clinical studies, the effectiveness of JYNNEOS in preventing monkeypox was assessed. Studies in nonhuman primates also support JYNNEOS’s efficiency. Additionally, in more than 7800 individuals, JYNNEOS safety was evaluated. 23 Furthermore, a sequence of animal trials sanctioned JYNNEOS for application. Trials have established its strong efficacy and safety, allowing it to protect persons of various age groups from mpox infection.10,11 When the others are unavailable, the WHO recommends using the JYNNEOS, LC16, or ACAM2000 vaccines. In the United States, ACAM2000 and JYNNEOS are the only vaccines approved for preventing both smallpox and mpox. MVA, while genetically similar to variola and mpox, exhibits reduced pathogenicity. Due to its nonreplicating nature, MVA effectively elicits both humoral and cellular immune responses against orthopoxviruses 27 with about 66% efficacy after two doses in the 2022 outbreak. It has been reported that these two smallpox vaccines, which are based on the VACV, provide cross-protection against the mpox; however, as previously mentioned, a recent study indicates that smallpox vaccination does not provide complete protection against mpox during the current outbreak, 17 and ACAM2000 has been associated with significant adverse events. 22 Table 1 summarizes the various vaccines developed for mpox prevention, dividing them into approved and investigational stages.
Current status and development of mpox vaccines.
Challenges in mpox vaccine development
Cross-protective immunity and duration of protection
The increase in mpox cases in nonendemic countries is probably attributable to a lack of immunity to orthopoxviruses. Orthopoxviruses exhibit analogous genetic and antigenic characteristics. There exists significant genetic homology between smallpox and mpox, with numerous shared immunological epitopes and markers. Because of this resemblance, multiple studies indicate that smallpox vaccines confer effective cross-immunity against mpox. 18 Proposals indicate that an increasingly immunologically naive population, due to the discontinuation of smallpox vaccinations, is accountable for the increase in mpox. 36 Evidence indicates that infection with any of these species may provide significant protection against other species of orthopoxvirus.37,38 Smallpox immunization may cause an mpox reaction, potentially protecting people from infection by up to 85%.17,36,39 Period 1 had a higher proportion of cases vaccinated against smallpox, which was attributed to older cases. Conversely, in Period 2, more members of high-risk populations received vaccinations against mpox owing to enhanced vaccine accessibility and knowledge. Evidence indicates that immunization for smallpox provides protection against mpox.7,17,40 Vaccination is essential for the mpox response; yet, a substantial proportion of cases are unvaccinated, underscoring the necessity for further initiatives. Smallpox vaccination, particularly in older populations, marked Period 1, while recent mpox vaccination efforts, specifically targeting high-risk groups, marked Period 2. Although smallpox vaccines encourage cross-protective immunity, the duration of this protection is a significant issue. Findings indicate that the protection conferred by smallpox vaccinations may diminish over time, rendering previously vaccinated persons vulnerable to mpox infection as global immunity decreases with age. People who received smallpox vaccinations decades ago should be especially concerned, as the duration of this cross-protection remains unclear. 41 JYNNEOS is recommended for high-risk persons exposed to mpox; however, its safety in pregnant women and individuals under 18 remains uncertain due to insufficient data. Research indicates that immunity diminishes with time, requiring booster vaccinations. A significant number of at-risk individuals, particularly from racial and ethnic minority groups, have failed to complete the advised two-dose regimen for postexposure prophylaxis, despite its clearance. 15
Due to the high homology (96%–99%) among orthopoxviruses, all three smallpox vaccinations are effective in protecting mpox. Studies, on the other hand, show that JYNNEOS produces low levels of antibodies that can fight mpox, especially the new strain (Clade IIb). Despite the improvement in JYNNEOS’s efficiency being between 36% and 86%, the emergence of breakthrough infections after two doses highlights the need for a more effective vaccine. In the Clade IIb mpox circulating from 2022 to 2023, many single nucleotide polymorphisms have been detected, exhibiting an uncommon mutational pattern indicative of prolonged human-to-human transmission. These changes include amino acid mutations in critical proteins, suggesting possible viral adaptation. Proteins such as M1R, A29L, B6R, H3L, and A35R, linked to immune responses in poxviruses, are seen as prospective targets for forthcoming vaccine development.42–44 For example, the A29 and M1 antigens are promising candidates for orthologous orthopoxvirus vaccines because of their strong cross-neutralizing immune responses against mpox. The mpox has genes capable of differentiating between mpox and vaccinia infections, which may facilitate the development of diagnostic techniques. 15 A quantitative postadsorption ELISA test has demonstrated 100% sensitivity and specificity in detecting clinically manifest monkeypox infections. 45 The human mpox vaccination must focus on the circulating strain and elicit enduring, mucosal-specific antibody responses against mpox. Employing novel vaccination platforms, administering immunizations at various locations, or combining certain adjuvants can facilitate this. 42
Emerging mpox strains can evade immune responses, potentially diminishing vaccine efficacy. Additional research is required to assess vaccine efficacy against these variants and determine the need for booster doses. Achieving equilibrium between safety and immunogenicity is a challenge, particularly for high-risk populations. 46 ACAM2000, while effective, presents significant adverse effects, particularly in immunocompromised individuals, underscoring the necessity for safer vaccine alternatives for high-risk groups, including individuals with HIV, organ transplant recipients, and those receiving immunosuppressive treatments.
Personalized vaccines designed for at-risk populations, accounting for genetic features, immunological profiles, regional variations, and viral strains, could enhance vaccine efficacy and diminish transmission rates, providing a more targeted approach to prevention, particularly for high-risk populations such as healthcare professionals and MSM.
Safety, efficacy, manufacturing, and regulatory barriers
During a global public health crisis, rivalry for vaccine production and distribution could lead to shortages in low- to middle-income countries. In the present mpox context, high-income countries ought to cooperate with low-income countries to effectively address the issue. Collaboration with clinicians, healthcare professionals, and researchers is essential for a systematic response strategy. In light of the COVID-19 pandemic, high-income countries should work together to ensure equitable resource allocation and management of resources.47,48 Even though there has been a lot of progress in developing the mpox vaccine, it is still not possible to prove its safety and effectiveness in strong human trials. Some data gaps make it challenging to fully understand how well the vaccine will protect against new strains of mpox in the long term. Improving the production and distribution of vaccines faces ethical, financial, regulatory, and logistical problems, especially in areas where mpox is not common. This is because different countries have different approval processes and fewer resources, which make it harder for everyone to get a vaccine. Resolving these interrelated difficulties is critical for mpox’s global management.49,50
Broader implications and preparedness for future outbreak
Additional diseases that can be prevented by vaccination should benefit from the knowledge learned from developing mpox vaccines. Meanwhile, shifts in social behaviors and population dynamics, including growing urbanization, globalization, and changing sexual health patterns, highlight the need for adaptable vaccination platforms to combat the infectious diseases.16,17 Collaboration between governments and manufacturers is essential for the lightning-fast deployment of vaccines during outbreaks.7,51 This necessitates robust manufacturing infrastructure, effective cold chain capabilities, and equitable distribution routes, as evidenced by emergency use authorizations during the COVID-19 and mpox pandemics. 15
Vaccine hesitancy often occurs when considering recommended vaccines. The 2022 mpox outbreak primarily affected men who have sex with men, leading to increased stigma and vaccine hesitancy. Agroia et al. 52 reviewed several studies that identified several key factors in vaccine hesitancy, including fear of stigma, inadequate knowledge of the disease, concerns about vaccine safety, limited availability, and access to the vaccine, and stigma associated with receiving it.
We recommend tackling these obstacles by implementing the following strategies: (1) initiatives focused on increasing public awareness campaigns regarding vaccine safety and efficacy through clear and stigma-free communication; (2) engaging with communities to improve vaccine uptake among at-risk populations; (3) improving access to vaccines in a manner that is nondiscriminatory and respects confidentiality; and (4) ongoing monitoring and research to evaluate public concerns and enhance strategies.53,54
Global landscape of mpox vaccine access
Vaccine equity: Disparities in access between developed and developing countries
Immunological and logistical obstacles impede global use of the mpox vaccine, resulting in limited availability in nonendemic regions. The global accessibility of vaccines, especially in nonendemic areas, is still restricted, and equitable distribution is a significant concern.51,53,55 Vaccine equity is critical to the fight against mpox, and it necessitates true collaboration and unity. In 2024, African countries, particularly the DRC, will account for more than 95% of all cases and deaths due to mpox; however, few vaccines are available for them. 56 In contrast, higher-income countries have stockpiled millions of doses of vaccines. To promote global health and reduce the recurrence of mpox, public health organizations must take action to promote vaccination before the crisis invades wealthy nations. 57 To facilitate resources and coordinate response efforts, the WHO declared the mpox outbreak a public health emergency of international concern on August 14, 2024. 58 However, several challenges impede effective vaccination efforts in African countries, including the high cost of vaccines, the absence of a centralized regulatory body for vaccine authorization across the continent, and inadequate infrastructure for vaccine delivery and distribution.59,60 Despite these obstacles, numerous countries and organizations have committed to donating mpox vaccines to African nations, as detailed in Table 2.
Donations of mpox vaccines to African countries.

Vaccine equity is crucial in preventing mpox outbreaks, with disparities in access between developed and developing countries. Addressing logistical, regulatory, and financial challenges in nonendemic regions is essential for global health. Global surveillance systems and swift international collaboration facilitate the integration of genomic sequencing and real-time monitoring, which is crucial for the early detection of pox outbreaks. They also promote rapid vaccine deployment and containment efforts. 50
Regional responses to mpox outbreaks and vaccination efforts
As a collaboration between the U.S. government and Nigeria, the first-ever mpox vaccines delivered to Africa outside of trials, about 10,000 doses, arrived in Nigeria on August 27. 66 Following that, the Africa Centers for Disease Control and Prevention (Africa CDC), the United Nations Children’s Fund, the Health Emergency Preparedness and Response Authority, and the Bavarian Nordic Company collaborated to deliver a total of 200,000 MVA-BN doses to the Democratic Republic of Congo in September. 67 Africa CDC and WHO have launched the “mpox Continental Preparedness and Response Plan for Africa,” a 6-month project that began in September 2024 and will run through February 2025. This project will cover 29 African countries with an estimated budget of US$600 million, aiming to accelerate response to the mpox outbreak. 68 The Africa CDC estimates that a total of 10 million doses of vaccine are required by 2025 to fight mpox in Africa, and it has pledged to deliver them by 2025. 69
Past experiences in fighting mpox and COVID-19 will provide useful lessons to tackle the current mpox outbreak. For example, from May 2022 to May 2023, the U.S. Centers for Disease Control and Prevention (U.S. CDC) launched a domestic response plan to manage the mpox outbreak. This plan included active surveillance of high-risk populations, running diagnostic tests, administering 1.2 million MVA-BN vaccine doses (covering 37% of the estimated at-risk population with a single dose and 23% with double doses), providing therapeutics (such as tecovirimat and antiviral medications) for patients, and promoting research and communication. Notably, SARS-CoV-2 wastewater surveillance systems were used to identify the geographical distribution of mpox, serving as an early warning system. 70 Experiences from the 2022 mpox outbreaks in the Netherlands and Greece also demonstrate the importance of accurately characterizing high-risk populations and understanding behavioral patterns that lead to higher transmission rates. These experiences highlight the need for screening, vaccination, and education of the population.71–73 Studies into the 2022 mpox outbreak in Italy underline the effect of behavioral changes on the decline of the outbreak. The reduction in sexual contact between MSM accounts for the rapid decline in mpox transmission. Research also indicates that vaccinating 23% of MSM with the highest sexual activity could entirely prevent the resurgence of mpox. 54 Ensuring fair access to vaccines, especially for those at the highest risk, is essential for breaking the global transmission chain and achieving long-term control of mpox. 74
Surveillance systems have always been important for managing pandemic outbreaks because they provide valuable insights into disease prevalence, population risk, and vaccine prioritization. Mapping the spread of a disease—in this case, the mpox outbreak—is a priority. Using integrated surveillance systems such as contact tracing, travel history tracing, wastewater sampling, real-time epidemiological data, and genomic sequencing together helps identify high-risk areas and allocate resources.7,75 These systems should be beneficial to the mpox vaccination program. Vaccine manufacturers can use surveillance data to prioritize vaccine delivery in areas close to the outbreak. This strategy ensures active immunization efforts before large-scale outbreaks in neighboring regions and shortens the delay in vaccine availability, which in turn increases resource sharing and regulatory approvals for cross-border vaccination. Therefore, we can conclude that addressing these challenges requires a global health security agenda.7,76
Future directions for Monkeypox vaccine development
Next-generation vaccines: mRNA, DNA, and novel delivery platforms
The developing novel mpox vaccines are essential, requiring enhanced potency, durability, and safety profiles. Furthermore, these vaccines should be readily scalable for rapid manufacturing and global distribution, ensuring broad access. Both mature virions (MVs) and extracellular virions (EVs) have distinct surface proteins. Some protection can be gained by targeting a single antigen, but studies on animals using subunit vaccines and monoclonal antibodies show that it is better to combine antibodies against multiple antigens from both MVs and EVs.16,38,77 In the past two decades, many studies have been conducted to develop new investigational vaccines based on DNA vaccines, mRNA vaccines, and subunit vaccines, which all carry no risk of causing infection. DNA vaccines contain a gene-encoded antigen plasmid, transcribed and translated into cells, triggering an immune response, and can be easily adapted to new mutants by changing the plasmid’s DNA sequence.78,79 There are two primary forms of mpox: the extracellular enveloped virus (EEV) and the intracellular mature virus (IMV). A ruptured cell releases IMV, which has one membrane, while an infected cell sheds EEV, which has two membranes. 80 In contrast to DNA vaccines, mRNA vaccines use messenger the mpox DNA vaccine is made up of two IMV genes (L1R and A27L) and two EEV genes (A33R and B5R). Studies have demonstrated significant protection against mpox infection in macaques from both the mpox DNA vaccine and another multivalent smallpox DNA vaccine, which consists of eight DNA-encoded antigens.13,18,28,81
In contrast to DNA vaccines, mRNA vaccines use messenger RNA to express antigens that trigger an immune response. These vaccines are highly promising due to their high efficiency, rapid production, and low cost. 82 The MPXVac-097 vaccine targets five different antigens from EEV (A35R and B6R) and IMV (A29L, E8L, and M1R). The mpox quadrivalent mRNA vaccine is another mRNA vaccine. The vaccine consists of mRNA-A-LNP and mRNA-B-LNP, specifically targeting antigens A29L, A35R, M1R, and B6R. This vaccine generates antibodies and T-cell-mediated immunity in mice without causing severe adverse reactions.29,30,83
Subunit vaccines use a specific immunogenic protein from a pathogen to stimulate the immune system. They are safe, but compared to live-attenuated vaccines, they may not efficiently trigger an immune response. 84 Several subunit and recombinant vaccines have been developed for mpox. Some subunit vaccines were developed using antigens such as A33, B5, L1, and A27, along with adjuvants like CpG. Multiepitope vaccines have been created using bioinformatics and machine learning techniques, which target B and T-cell epitopes, and also recombinant adenovirus vaccines, which target six poxvirus glycoproteins, were developed. All of them have shown promising immune responses and protection in preclinical trials, but they are still at the laboratory stage.13,18
New types of vaccines, such as viral vector vaccines, nucleic acid vaccines, and virus-like particles, are being tested to see if they can make people more resistant to getting mpox. 13
To address the need for a highly effective and accessible vaccine, Zuiani et al. designed the BNT166 vaccine, aiming to create a next-generation vaccine that is highly immunogenic, safe, scalable, and capable of protecting against mpox virus and related orthopoxviruses. 14 In this preclinical study and to address the presence of multiple viral forms and broaden the immune response, two multivalent mRNA vaccine candidates were evaluated preclinically: a quadrivalent vaccine (BNT166a; encoding MPXV antigens A35, B6, M1, and H3) and a trivalent vaccine (BNT166c; excluding H3), and their phase I/II clinical study is now underway (NCT05988203) in a dose-escalation. Phase I/II trial is evaluating the safety, tolerability, reactogenicity and immunogenicity of the investigational RNA-based multivalent vaccine candidate BNT166a for active immunization against mpox. In the preclinical study, both trivalent and tetravalent vaccine candidates induced vigorous T cell responses and IgG antibodies in mice, including neutralizing antibodies against both MPXV and VACV. Furthermore, in a challenge model, both vaccine candidates provided complete protection against vaccinia, clade I, and clade IIb mpox. The study demonstrated 100% protection from death and lesion suppression in a lethal clade I mpox virus challenge in cynomolgus macaques. These preclinical results support the ongoing clinical evaluation of BNT166 (NCT05988203).
Sang et al. conducted an animal study in mice to evaluate two mpox quadrivalent mRNA vaccines, named mRNA-A-LNP and mRNA-B-LNP. These vaccines were designed to target four specific MPXV proteins: A29L and M1R, which are intracellular mature virus-specific, and A35R and B6R, which are extracellular enveloped virus-specific. 30 In this animal study on mice, both mRNA-A-LNP and mRNA-B-LNP, administered intramuscularly in two doses, successfully induced MPXV-specific IgG antibodies and potent neutralizing antibodies against VACV. Furthermore, these vaccines elicited robust mpox-specific Th1-biased cellular immunity and durable effector memory T cell and germinal center B cell responses. Importantly, mice are protected against VACV challenges with two doses of either vaccine. Passive serum transfer from mRNA-A-LNP and mRNA-B-LNP-immunized mice also conferred protection against VACV challenge in nude mice. These findings suggest that mRNA-A-LNP and mRNA-B-LNP are promising vaccine candidates for monkeypox outbreaks and may offer protection against other orthopoxviruses, including smallpox. The authors conducted safety evaluations in animals and found no local skin reactions or inflammation at the injection site. Monitoring of various organ function parameters (heart, liver, and kidney) in the study mice revealed no abnormalities. These findings contribute to the conclusion that the evaluated mRNA vaccine exhibits an acceptable safety profile.
Mucker, Eric M. et al. tested the efficacy of mRNA-1769, an mRNA-lipid nanoparticle (LNP) vaccine that expresses mpox surface proteins, against a lethal mpox challenge in nonhuman primates. The study compared the effectiveness of mRNA-1769 to that of modified vaccinia Ankara vaccination. 85 The animal study found that mRNA-1769, like MVA, offered protection against mpox challenge, lessening symptoms and shortening the duration of the disease. Analysis of antibody profiles revealed a collaborative effort between neutralizing and Fc-functional EV-specific antibodies in controlling viral spread. Furthermore, the study identified opsonophagocytic and cytotoxic antibody functions as crucial factors in preventing lesion formation. The study found that mRNA-1769 was better at stopping viruses and lowering the severity of disease than MVA. This shows that mRNA vaccines could be used to effectively deal with future pandemic threats.
Another promising vaccination strategy for mpox prevention, currently under investigation, is the use of DNA vaccines. Nucleic acid vaccines have garnered significant attention in recent years due to their superior efficacy, safety profile, cost-effectiveness, and faster development timelines than conventional vaccination platforms. 86 There is a DNA-based vaccine against SARS-CoV-2 in India, 87 and numerous clinical trials are currently underway to evaluate the efficacy of DNA vaccines for preventing various infectious diseases (NCT04591184, NCT01498718, NCT01487876, and NCT04445389). Hooper et al. demonstrated in a preclinical study that a DNA vaccine containing four VACV genes (L1R, A27L, A33R, and B5R) protected rhesus macaques from severe disease after a lethal mpox virus challenge. 80 Rcheulishvili et al. 86 conducted an in silico study that focused on designing a potentially universal DNA vaccine against the mpox virus. They retrieved and aligned the sequences of four antigen proteins (A5L, A15L, A35R, and B6R) and identified conserved sequences within these proteins. These conserved sequences were then used to predict B-cell, MHC-I, and MHC-II binding epitopes, which are crucial for generating a robust immune response. These four antigens were chosen for the reasons already mentioned, as well as the desire to make a combined vaccine that would allow testing of the immune response caused by all four antigens at the same time. The study concluded that the DNA vaccine possesses favorable physicochemical properties and elicits robust cellular and humoral immune responses. The immune simulation study showed that the multi-epitope vaccine candidate increases the production of HTLs and CTLs while also increasing the number of B cells, the production of antibodies, and the release of cytokines. However, further validation through in vitro and in vivo studies is recommended to confirm these findings.
Using novel platforms for delivering vaccines against orthopoxvirus strains has been proposed as a strategic approach to effectively manage the current mpox outbreak and prepare for future public health emergencies. Freyn et al. 88 conducted a study to explore the efficacy of an mRNA-lipid nanoparticle vaccine encoding four highly conserved mpox virus surface proteins involved in viral attachment, entry, and transmission. The study revealed that the nanoparticle-based mRNA vaccine successfully elicited mpox-specific immunity, which, in turn, provided protection against a lethal challenge with the VACV. The mRNA-based mpox vaccine demonstrated enhanced neutralizing activity against mpox and more effectively inhibited viral spread between cells than the current MVA vaccine. Furthermore, the mRNA vaccine elicited stronger Fc effector TH1-biased humoral immunity to the four encoded mpox antigens. The mpox virus mRNA-lipid nanoparticle (mRNA-LNP) vaccine in animal studies was also evaluated in another study by Xia et al. 89 They found that mRNA-LNP targets A27, L1, A33, and B5 of mpox virus can induce potent humoral immunity in mice. They also found the four antigenic mRNA-LNPs protect mice from the VACV challenge with a good safety profile in mice. The researchers concluded that the antigenic mRNA-LNP vaccine candidates demonstrate both safety and efficacy against mpox and hold potential for combating diseases caused by other orthopoxviruses.
Table 3 provides an in-depth overview of mpox vaccines, highlighting their development, target populations, regulatory status, immunological responses, and global manufacturing capacities, highlighting challenges and distribution barriers for equitable global access.
Comprehensive overview of mpox vaccines: Development, immunological responses, and global manufacturing capacity.

Conclusion
WHO declared a global public health emergency in light of the significant challenges mpox poses to African public health systems and global health security. Reduced protection from smallpox vaccinations, viral evolution, COVID-19 disruptions, and insufficient global investment have all contributed to the reemergence of mpox, which was previously limited to Central Africa. It is critical to use AI-based monitoring technology, understand sociocultural factors, and adjust public health efforts and preventive actions. Healthcare staff should receive case management, diagnosis, and surveillance. Cooperation between African countries, world leaders, and the pharmaceutical sector is essential to produce vaccines and allocate resources.
