Abstract
Background:
Needle pain due to routine vaccination is an important factor contributing to low vaccine adherence and immunization coverage. Prophylactic oral analgesics can address this important issue of needle pain related to vaccination. Paracetamol and mefenamic acid are commonly used nonsteroidal anti-inflammatory drugs for pain relief, but there is little published literature on whether the same can be used for needle pain related to vaccination.
Objectives:
This study was planned to compare the efficacy of oral mefenamic acid and paracetamol over placebo as a prophylactic analgesic during vaccination and prophylactic antipyretic during the post-vaccination period.
Designs:
Three-arm, triple-blind, randomized controlled trial.
Methods:
This study was conducted at the outpatient department of a tertiary-level medical college in South India from January 2021 to June 2022. In this three-arm interventional trial, each arm had either a single dose of placebo or mefenamic acid (4 mg/kg/dose) or paracetamol (10 mg/kg/dose). These medicines were administered orally 30 min before vaccination to reduce needle pain.
Main outcome and measures:
Outcome was measured with the change of FLACC (Face, Leg, Activity, Cry, Consolability) scoring at the time of vaccination, subsequently at 15 and 30 min of vaccination in all three groups. Appearance of fever, grade of fever, and need for antipyretics 24 h after vaccination were also noted.
Results:
There was a significant difference in FLACC scores at the time of administration (p = 0.010) and at 15 min (p = 0.014) with mefenamic acid compared to placebo. Although the paracetamol group showed a difference when compared to the placebo, it was not significant at the time of administration (p = 0.401), at 15 min (p = 0.451), or 30 min (p = 0.892) post-vaccination. The appearance of fever, grade of fever, and use of antipyretic up to 24 h post-vaccination had no significant difference among any of the three groups.
Conclusion:
Mefenamic acid was more potent than placebo for pre-vaccination pain prophylaxis in children. There was no difference in the appearance of fever and its grade among the three groups. The promising results from this trial warrant further large-scale studies to recommend a single oral dose of mefenamic acid to tackle needle pain related to vaccination in children to improve vaccine adherence and coverage.
Trial registration:
CTRI (Clinical trials registry-India) (CTRI/2021/01/030239). [Date of Commencement: 13 Jan 2021, Date of last recruitment: 30 June 2022 (now closed for new participants)]
Introduction
Vaccination is the most crucial and cost-effective preventive step for various childhood infections. Pain and discomfort caused by needle prick during vaccination are significant adverse effects that lead to vaccine hesitancy. Pain during vaccination is the most common concern in parents of children getting vaccinated. 1 To achieve the best vaccine coverage for preventable diseases, the issue of vaccine hesitancy due to needle pain has to be addressed. The ‘needle pain’ during vaccination has been an area of interest for the last few decades, and multiple modalities of intervention tried to alleviate this, but none could be recommended for routine practice due to lack of evidence. 2 To have a good immunization practice globally, the WHO (World Health Organization) has prompted the need to have interventions to tackle needle pain during vaccination. 3 Various analgesics have been studied to reduce needle pain associated with vaccination, like EMLA cream (Eutectic mixture of local anesthetics), paracetamol, or ibuprofen, but lack of evidence has limited its use. Topical EMLA was found to reduce pain scores during vaccination, and vapor coolant spray or 25% sucrose/glucose solutions have been found to reduce crying time in the pain assessment scores, but the quality of evidence was low or very low, which opens the scope for further research. 4 Other multi-modalities like combination therapies, including sucrose solution plus breastfeeding or EMLA plus distraction or vapor coolant plus distraction, and coolant plus vibration, have also been tried, but the quality of evidence is low for combination therapies too. 5
Breastfeeding at the time of vaccination has been studied widely, but the problem lies in the generalization of this method as many children are not exclusively breastfeeding since birth or not breastfeeding anymore, or they are not accompanied by their mother to the hospital for vaccination.6–9 Sucrose and glucose solutions have also been tried due to their analgesic effect in infants but with limited success.10,11 One study reported the use of non-pharmacological measures by 97% of pediatricians and 92% of mothers during vaccination. The reported use of pharmacological measures like topical or oral analgesic as pre-vaccination measures in 25% of parents, whereas 33% used it in the post-vaccination phase on their own. 12 However, there is no published research advocating the use of prophylactic oral analgesics during vaccination. Oral analgesics are most frequently used in treating pain associated with vaccination. 12 Among NSAIDs, paracetamol and ibuprofen have been used in the management of pain and fever in vaccinated children. One Cochrane review suggested mefenamic acid use as an acute analgesic in adults after surgical procedures. Post-surgery mefenamic acid is used effectively in acute pain management in many studies as a single dose, like post-orthopedic surgeries, dental procedures, and episiotomies. 13 It is a widely used and effective drug in the pediatric age group for the treatment of pain.14,15 Mefenamic acid is twice as potent as analgesics as compared to paracetamol. 16 Most of the analgesics for needle pain are prescribed after the administration of vaccination. It is not clear if prophylactic analgesics can alleviate needle pain during vaccination. Fever is the most commonly reported adverse event following vaccination. Antipyretic medicines have been advised by healthcare workers for the treatment of fever as well as for prophylaxis. The commonly used antipyretics include paracetamol and, less commonly, ibuprofen. 17
Hence, this study was planned to know the prophylactic role of oral analgesics paracetamol and mefenamic acid in alleviating needle pain during vaccination and their effect on reducing fever during the post-vaccination period.
Materials and methods
Objective: To assess the effect of oral mefenamic acid, paracetamol, compared to placebo as a prophylactic analgesic in pain management in children during vaccination using the FLACC (Face, Leg, Activity, Cry, Consolability) scale. This study also assessed the effect of mefenamic acid, paracetamol, and placebo on reducing fever after vaccination.
Study design: This study was a prospective, triple-blind, three-arm parallel, randomized placebo-controlled trial done from 13 January 2021 to 30 June 2022 at a tertiary-level care teaching hospital in South India after obtaining institute ethical clearance and required CTRI registration prospectively. The protocol of this trial, including detailed methodology, has been published. 18 The CONSORT flow diagram is given in Figure 1. 19

Study CONSORT flow diagram. 19
Participants: All children attending the pediatric outpatient department for immunization in the age group 6 weeks to 7 years of age.
Randomization: Block randomization (block size-9) was done through computer automatic randomization table generation. Sequence generation was done through randomization software.
Allocation concealment: All the randomization sequences were individually concealed in a sequentially sealed opaque envelope. The size of blocks was not disclosed to the researcher involved in recruiting participants to avoid bias in predicting the next sequence of allocation.
Blinding: All medicines were coded alphabetically as either A/B or C. Decoding of this allocation was not shared with any of the investigators of the study till the statistical analysis was completed. The triple blinding protocol was followed where neither the participants nor the person administrating medicine/vaccine or assessing the FLACC score or the statistician were aware of the medicines coded as A/B or C. The physical characteristics such as color, consistency, flavor, or volume of administration were matched for all medications and placebo used in the study.
Intervention: Oral analgesic, paracetamol or mefenamic acid, and placebo were given as a prophylaxis for procedural pain during routine vaccination.
Procedure: We recruited participants whose parents/legal guardians had given written informed consent before enrolment. Children with known liver or kidney diseases, coagulation disorders, and a history of any allergic reactions were excluded. We have included 45 participants in each of the three groups, thus having a total of 135 sample sizes for this study. After obtaining written informed consent, allocation concealment envelopes were opened serially, and accordingly, as per coding, participants were enrolled into groups A, B, or C. Each participant received a drug (A, B, or C) as per allocation and weight band. After 30 min of drug administration, the child received vaccination as per the National Immunization Schedule. FLACC scoring was done at the time of administration of vaccination, then at 15 min and 30 min. After 24 h, the participants are asked about the presence of fever, grade, and the need for antipyretics post-vaccination either telephonically or through physical Out Patient Department (OPD) follow-up. The fever was documented if temperature elevation to >38°C (100.4°F) was measured at any site using any validated device. 17
All data were entered into an Excel sheet and analyzed by a statistician. After the result analysis had been completed, the decoding envelope was opened to reveal the drugs labeled as A/B or C.
Drugs dosage: Paracetamol syrup used in the study was 250 mg/5 ml and the dose at 10 mg/kg/dose. The mefenamic acid syrup used in the study is 100 mg/5 ml and the dose at 4 mg/kg/dose. Placebo was chosen to be a flavored, viscous, sunset yellow solution to match the physical characteristics of other drugs. The amount of drugs given was as per weight band such participants with weight from 3 to 5 kg were given 1 ml, 6 to 10 kg were given 2 ml, 11 to 15 kg were given 3 ml, 16 to 20 kg were given 4 ml, 21 to 25 kg were given 5 ml, 26 to 30 kg were given 6 ml, 31 to 35 kg were given 7 ml of drug A/B/C.
Outcome: Assessment of pain among children receiving vaccination was done with the help of the FLACC score. It is among the most commonly used non-verbal scales among children, comprising five parameters face, leg, activity, cry, and controllability, where each parameter is scored objectively from 0 to 2. The total score is expressed out of 10; the higher score signifies a more painful experience for the child. It has been used in this study considering its objectivity and ease of administrating in the younger age group children.20–22 In this study, it was done by a trained nursing staff observing the procedure during vaccination at 15 min and 30 min thereafter. The differences in scores at the time of administration, 15 min or 30 min after vaccination, were compared among all three medicine groups to know the most efficacious intervention among all three groups. All participants were followed up for 24 h for the appearance of fever and need for antipyretics.
Sample size: Previous studies done on vaccine-related pain from India showed the mean (SD) FLACC score after immunization to be 6.2 (1.14). 23 Expecting at least a 10% reduction in FLACC score with 5% alpha error and 80% power using Open-Epi software version 3.01, (Dean AG, Sullivan KM, Soe MM. Open Source Epidemiologic Statistics for Public Health) the sample size was calculated to be 42 in each group, adjusting for block randomization (block size 9). The final sample size rounded off to 45 episodes of vaccination in each group.
Statistical analysis
Continuous variables like age and FLACC score were summarized as mean, and standard deviation, and categorical variables like gender and study group were summarized as frequency and percentage. The association between continuous variables and categorical variables was analyzed using an independent t-test and ANOVA (analysis of variance). The association between categorical variables and categorical variables was analyzed using the chi-square test. The intention to treat the analysis method was used. p Value <0.05 was considered as a statistical significance. The analysis was done using Epi-info and R-software.
Results
A total of 135 participants were included in the study, with 45 in each group. The majority of study participants were infants and toddlers (Table 1). There was no statistically significant difference in basic demographic characteristics among the study groups. The total mean FLACC score during administering the vaccine was lowest in the mefenamic acid group (6.16), followed by the paracetamol group (7.11) and the placebo group (7.51). Similar results were found in the individual domains of FLACC. The difference in FLACC score among the study group was statistically significant (p-Value 0.029), and the post hoc Bonferroni showed a statistically significant difference between mefenamic acid and placebo groups. The comparison of FLACC scores at 15 and 30 min after administering the vaccine between the groups was also made. The total mean (SD) FLACC scores after 15 min of administering the vaccine among the placebo group, mefenamic acid group, and paracetamol group were 3.07(1.96), 1.98(2.14), and 2.73(2.21), respectively. The total mean (SD) FLACC score after 30 min of administering the vaccine among the placebo, mefenamic acid, and paracetamol was 0.87(1.49), 0.47(1.14), and 0.82(1.60). The trends of FLACC score over time have been plotted, which showed lower FLACC scores at all three points, for example, at administration, 15 min, and 30 min of administrating the vaccine in a mefenamic acid group compared to paracetamol or placebo (Figure 2). The repeated measure ANOVA showed the trends of FLACC score over the time between the study groups were statistically lower in mefenamic acid compared to other groups. The comparison between two groups out of placebo/mefenamic acid or paracetamol during administration, 15 min and 30 min after vaccination was done to know the difference among the FLACC scores (Table 2–4). The mefenamic acid group, in comparison to placebo, showed a significant difference in FLACC scoring during the time of vaccine administration (p = 0.010) and at 15 min (p = 0.040).
Basic demography of the study participants.


The trend of FLACC Score among the study group.
Comparison of FLACC Score among mefenamic acid and placebo.

FLACC, Face, Leg, Activity, Cry, Consolability.
Comparison of FLACC Score among mefenamic acid and paracetamol.
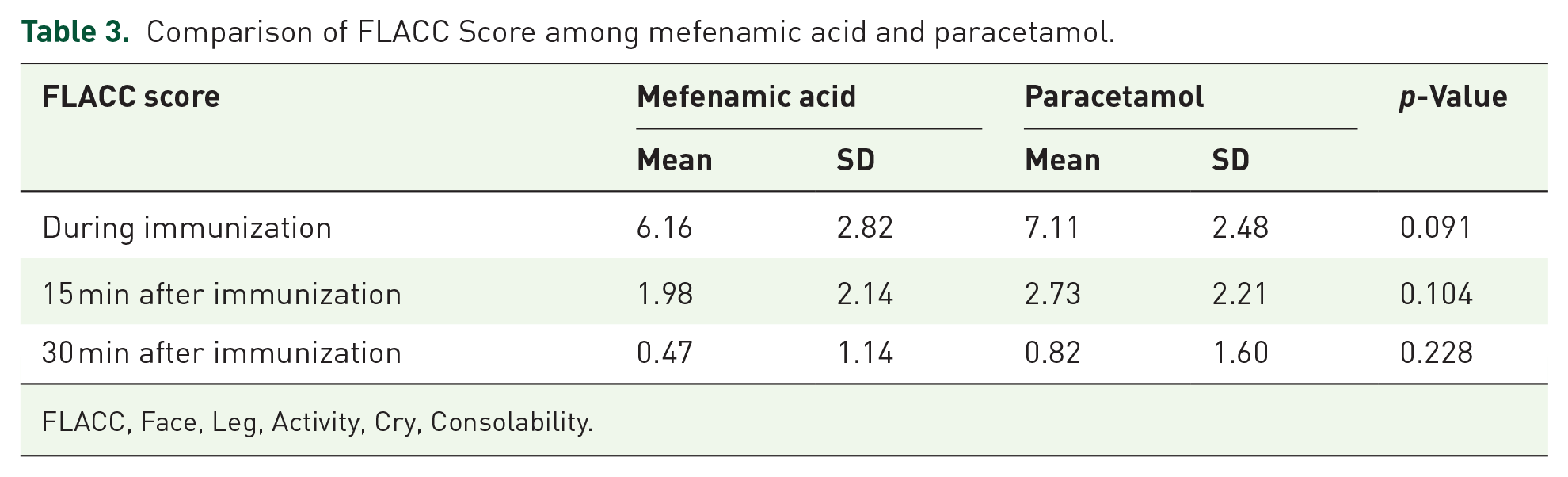
FLACC, Face, Leg, Activity, Cry, Consolability.
Comparison of FLACC Score among paracetamol and placebo.

FLACC, Face, Leg, Activity, Cry, Consolability.
Sixty percent of study participants in all the study groups had a fever within 24 h. In the placebo and paracetamol groups, 66% had a fever within 24 h, but in the mefenamic acid group, only 54.5% of participants had a fever. This difference was not statistically significant (p = 0.405). Among the participants who had fever majority of them had mild fever. The percentage of participants with high fever was lower in the mefenamic acid group (4.4%) compared to the placebo group (6.7%) and paracetamol (6.7%). The mean (SD) time taken for the next dose of antipyretic for the study group of a placebo, mefenamic acid, and paracetamol were 6.74 (3.34), 6.35(3.14), and 6.48(3.02), respectively. There was no significant difference in the time taken for antipyretic dose among the study groups (p = 0.904). (Table 5).
Comparison of appearance fever and need for antipyretic among the study groups for 24 h post-vaccination period.

Adverse effects and events: No adverse events were noted during the trial period in any of the participants of this study.
Lost to follow-up: There was no loss to follow-up participants in the study. All children who were enrolled were asked to stay for observation after vaccination for at least 30 min at the outpatient department of our institute as a routine practice post-vaccination and hence monitored. After 24 hours, the participants are called telephonically or asked for physical OPD follow-up. All enrolled participants responded in either way.
Discussion
Our study included the age group from 6 weeks to 7 years, whereas most studies in the past have used narrow age groups of infants, toddlers, or adolescents as study participants.5,10 Many studies have targeted needle pain against a particular vaccine or way of administrating vaccines, whereas we have included all routine vaccinations with their different mode of administrating to children as per the national immunization schedule.5,24 Thus, most routine vaccinations are given to younger age groups starting from 6 weeks of age. Our study has shown the effectiveness of using mefenamic acid as a prophylactic analgesic for routine vaccinations under a national immunization program.
One study has compared the analgesic effect of paracetamol or mefenamic on the intensity of headaches due to migraine in adults. They found that mefenamic acid is more potent and almost twice as effective as paracetamol in migraine as an analgesic. 16 This finding is similar to our study, where we also found that mefenamic acid is more effective as an analgesic as compared to placebo, whereas paracetamol had an equivocal effect as compared to placebo. We also found that the analgesic effect is statistically significant during the time of vaccination and, also, 15 min after vaccination with the use of mefenamic acid. Our study also showed that there is no statistically significant local analgesic effect of paracetamol as compared to placebo at any point of vaccine administration.
Paracetamol is an inhibitor of prostaglandin synthesis; it acts both peripherally and centrally through COX-1 and COX-2 receptors, respectively. However, studies have proven its selective affinity for COX-2 receptors.25,26 The analgesic effect of paracetamol is of low potency to produce pharmacological effects through peripheral COX-1 blockage, which might explain its statistical insignificance as compared to placebo. The central analgesic effect of paracetamol is through its metabolites which finally act through periaqueductal gray.25–28 Mefenamic acid, on the other hand, has both peripheral and central action through the blockage of COX-1 and COX-2 receptors. It is rapidly absorbed through the oral route with a peak of plasma levels in 2–4 h and a half-life t1/2 of 2 h.13,29
The comparative effect of preoperative and postoperative use of analgesics like paracetamol, ibuprofen, and mefenamic acid has shown that preoperative use or before any painful procedures is more beneficial. The mechanism of preventive analgesia is due to the inhibition of the central or peripheral sensitization of noci-receptors before their stimulation, thus attenuating its excitability further.29–33
Recently, one systemic review was done for the use of paracetamol at the time of pneumococcal vaccination, which provides evidence for its use without affecting the optimum immune response. 34 One study had also been done to quantify the immune response by hemagglutination-inhibition titers and microneutralization titers post-H1N1 vaccine. They used paracetamol or ibuprofen as an antipyretic or analgesic 2 days before vaccination (pre-vaccination day and vaccination day), and they found that these medicines have no effect on immunogenicity after vaccination. 35 In our study, we used only one dose 30 min before vaccination. Thus, it has even fewer consequences on immunogenicity response after vaccination. Likewise, previous studies have shown single-dose analgesic use, such as mefenamic acid, as effective analgesia in surgical procedures such as dental, orthopedic, or obstetric surgeries.13,36 So, using single-dose analgesic medicines before vaccination, as in our study, analgesic effects can be achieved without the risk of reduced immunogenicity of vaccination. Different techniques of administration of vaccines have also been studied to find out which technique causes less pain. Aspiration before injecting the vaccine during intramuscular injections is the usual practice, but it has been postulated that aspiration causes more painful vaccination procedures. In that study, infants from 6 weeks to 6 months of age were included, they studied the technique of intramuscular injections for pentavalent vaccines. They recommended that a technique with rapid administration and no aspiration before injecting vaccination results in less pain in children. 37
The long-term effect of mefenamic acid, like appearance and grade of fever, is less than paracetamol or placebo. It has been proven in previous studies also that mefenamic acid is a better antipyretic with a longer duration of action as compared to paracetamol.17,38,39 However, the difference was not statistically significant in the present study.
The strength of this study is a randomized, triple-blind study; the findings have overcome the biases that are usually present in other study designs. The medicines used in this study, paracetamol or mefenamic acid, are easily available and are already being used in children for various other reasons as an analgesic. No published research has considered mefenamic acid for this indication despite being an important issue to tackle to address vaccine hesitancy under the national immunization program, and mefenamic acid is a more potent analgesic. This study is the first evidence in this direction. Large or multicentric randomized controlled trials (RCTs) are required, which further encourages our policymakers to consider this simple, easily feasible step before vaccination in national vaccination guidelines to increase coverage and adherence to vaccination.
Limitations
The main limitation of this study is that it was conducted in a single-center study, and further large-scale studies are warranted from other centers to generalize the results. We have included all vaccines in this study, but there may be a chance that different vaccines cause different degrees of pain and fever, and the effect of age on the amount of pain and fever also cannot be overcome.
Conclusion
Prophylactic mefenamic acid was associated with significantly lower FLACC scores when compared to placebo in children receiving vaccination. Prophylactic mefenamic acid is more effective than placebo in reducing needle pain related to vaccination in children, whereas paracetamol was equivocal. Prophylactic mefenamic acid or paracetamol did not reduce fever or the need for antipyretics when compared to placebo during the post-vaccination period. Single-dose prophylactic mefenamic acid should be considered to reduce needle pain related to vaccination in children to deal with vaccine hesitancy and to improve vaccine coverage.
