Abstract
According to the World Health Organization (WHO), in the second half of 2022, there are about 606 million confirmed cases of COVID-19 and almost 6,500,000 deaths around the world. A pandemic was declared by the WHO in March 2020 when the new coronavirus spread around the world. The short time between the first cases in Wuhan and the declaration of a pandemic initiated the search for ways to stop the spread of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) or to attempt to cure the disease COVID-19. More than ever, research groups are developing vaccines, drugs, and immunobiological compounds, and they are even trying to repurpose drugs in an increasing number of clinical trials. There are great expectations regarding the vaccine’s effectiveness for the prevention of COVID-19. However, producing sufficient doses of vaccines for the entire population and SARS-CoV-2 variants are challenges for pharmaceutical industries. On the contrary, efforts have been made to create different vaccines with different approaches so that they can be used by the entire population. Here, we summarize about 8162 clinical trials, showing a greater number of drug clinical trials in Europe and the United States and less clinical trials in low-income countries. Promising results about the use of new drugs and drug repositioning, monoclonal antibodies, convalescent plasma, and mesenchymal stem cells to control viral infection/replication or the hyper-inflammatory response to the new coronavirus bring hope to treat the disease.
Background
In the last days of November 2019, the new coronavirus was described as the agent of a lower respiratory tract infection in the Hubei province in China.1,2 The coronavirus disease 2019 (COVID-19) caused by the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), officially named by the World Health Organization (WHO) in February 2020, 3 has caused a global pandemic with significant morbidity and mortality. Although most coronaviruses that infect humans cause mild diseases infecting upper respiratory tract, the SARS-CoV-2 infection can cause severe acute respiratory syndrome as the severe acute respiratory syndrome coronavirus (SARS-CoV) and the Middle East respiratory syndrome coronavirus (MERS-CoV), responsible for mortality rates of 10% and 37%, respectively. 4 Until now, SARS-CoV-2 has had a mortality of about 2.35%, but with higher transmission than SARS-CoV and MERS-CoV. 5 The higher transmissibility and faster spread of SARS-CoV-2 led the WHO to announce the pandemic status, with many countries presenting increasing number of COVID-19 cases from March 2020 to June 2021, mainly in Europe, where a so-called second wave of infection is decreasing now. In the United States, an uncontrolled increase in infection was observed with rapid decrease after vaccination, different from India and Brazil, comprising almost 50% of confirmed cases around the world. 6 Due to the severe characteristics of the disease, the augmentation of intensive care of hospitalized patients, and the lack of specific drugs, the entire world has been searching for effective therapies for the treatment of COVID-19. According to ClinicalTrials.gov, about 8160 clinical trials involving the diagnosis, prevention, or treatment of COVID-19 were in progress until August 2022, and that number is increasing every day. Here, we intend to describe the clinical trials undertaken around the world for the treatment of COVID-19 and to point out the differences in the approaches used. Most of these clinical trials are being conducted in the countries of Europe and the United States, performing more than 50% of those when compared with total trials (Figure 1). As for the type of clinical trial, most involve drug evaluation, biologics (such as vaccines and convalescent plasma), and, in smaller numbers, cell therapies, mechanical ventilation, diagnostic tests, and telemedicine (Figure 2).

Clinical trials involving the diagnosis, prevention, or treatment of COVID-19 worldwide are concentrated in the United States and Europe. Circles represent the number of clinical trials in that region of the world proportionally. Red represents the clinical trials present in the region indicated in the map compared with the total clinical trials in other areas (gray).
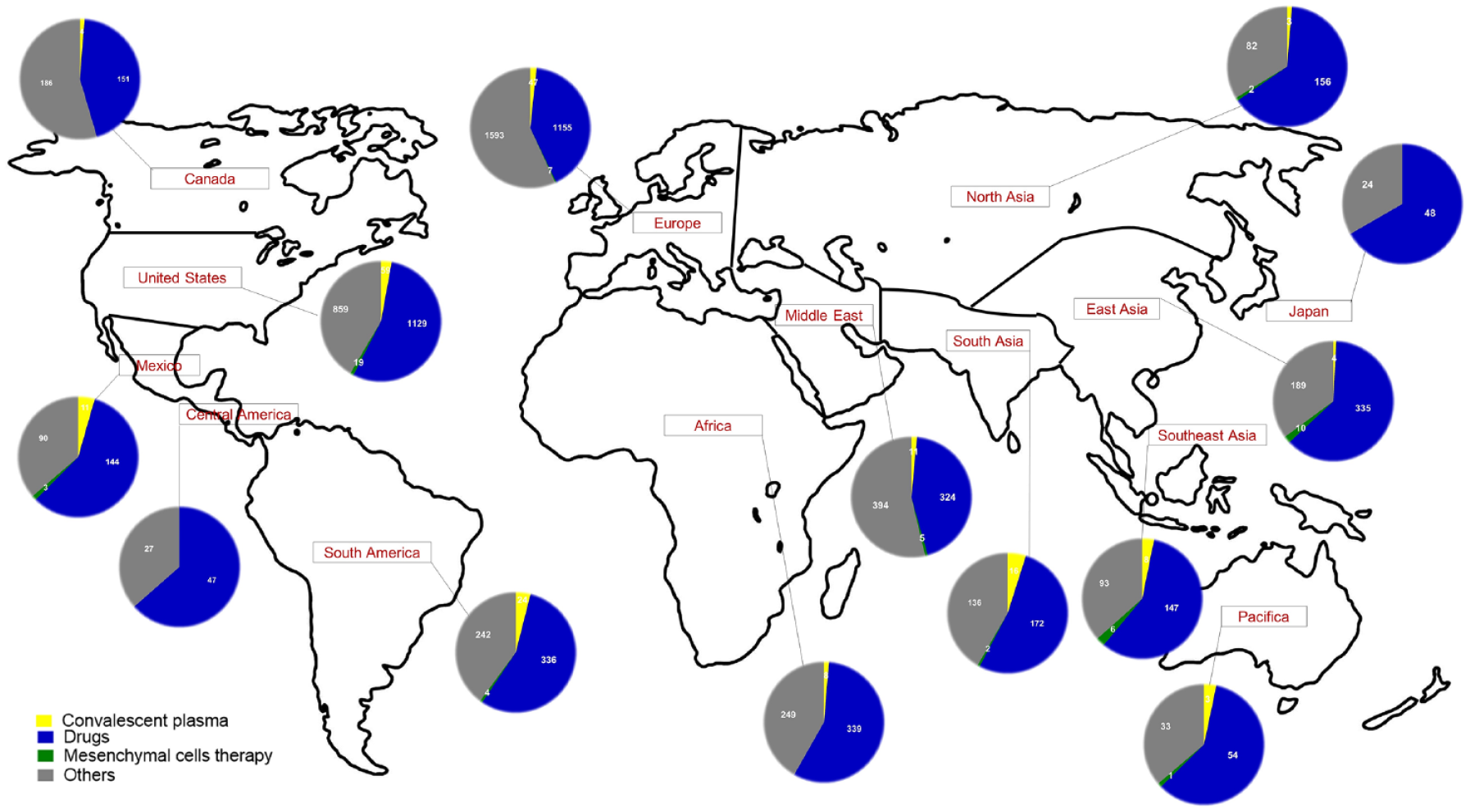
Clinical trials for COVID-19 worldwide regarding the different types of approaches. Circles represent different types of clinical trials in each part of the world. Colors indicate the use of convalescent plasma (yellow), drug evaluation (blue), use of mesenchymal cell therapy (green), and others (gray).
Methods
The present review was developed through the search for information on clinical trials involving the diagnosis, prevention, or treatment of COVID-19 worldwide. ClinicalTrials.gov was the clinical trials database used for this search. It took place in October 2020 and was updated in August 2022, because the number of clinical trials is increasing or updating every day. Articles on COVID-19, indexed in the databases Web of Science, PubMed, and Scholar Google, with perspectives on diagnosis, prevention, or treatment, were also included in the study. Articles that did not bring any new information were excluded after a full reading. The review is based on drugs (new drugs and drugs repositioning), monoclonal antibodies, convalescent plasma, and mesenchymal cell therapy to control the deleterious effect of COVID-19.
Drugs
In addition to developing new treatment options, such as new drugs, immunotherapies, and host-directed therapies, scientists worldwide continue to simultaneously explore the efficacy of existing drugs against SARS-CoV-2. From ClinicalTrials.gov, we found 4148 clinical trials that evaluated drugs in the treatment of COVID-19 until the month of August 2022. These drugs can be divided into two broad categories: those that can directly target the virus replication cycle and those based on immunotherapy approaches aimed at boosting innate antiviral immune responses or alleviating damage induced by dysregulated inflammatory responses.
New drugs for COVID-19
We considered new drugs for COVID-19 to be drugs that were not previously associated with studies of other diseases (Table 1). Some of the drugs with promising results are PF-07304814, DFV890, RBT-9, TD-0903, M5049, BLD-2660, and SNG001 (Figure 3).
New drugs developed to treatment COVID-19.
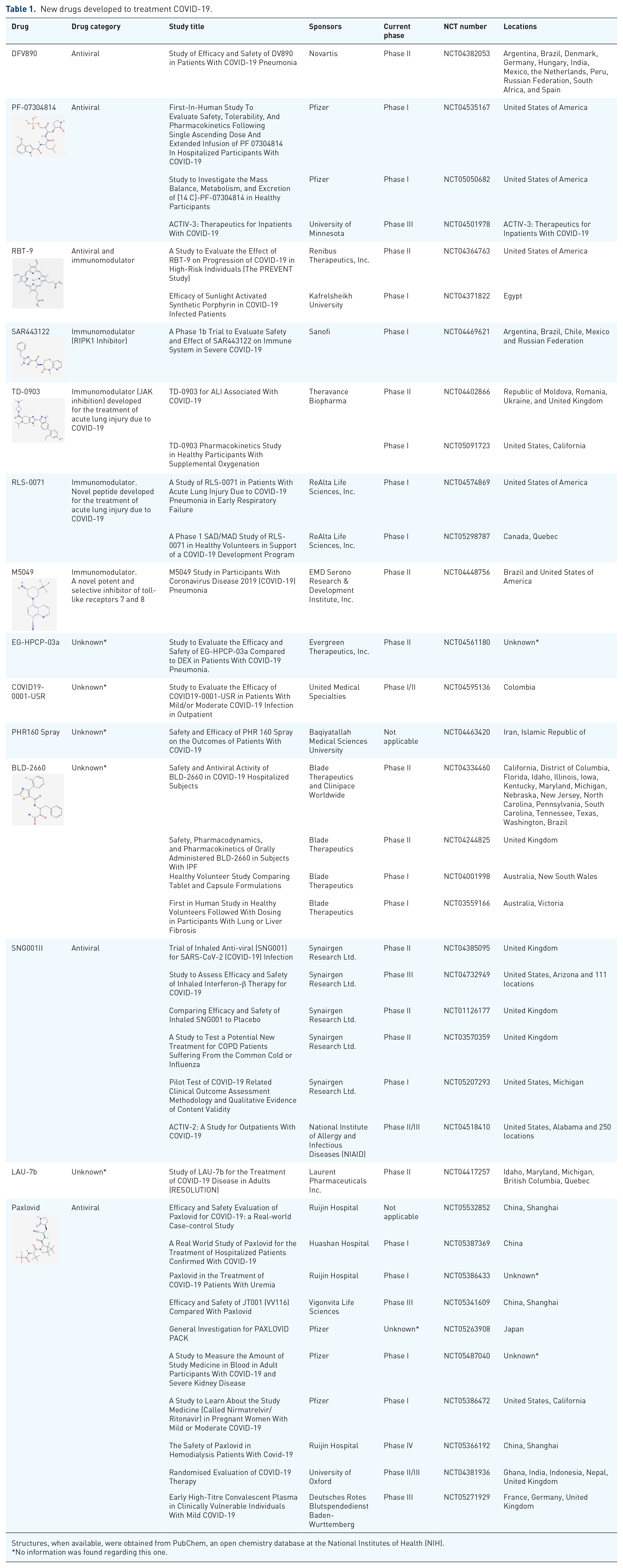
Structures, when available, were obtained from PubChem, an open chemistry database at the National Institutes of Health (NIH).
No information was found regarding this one.

New drugs mechanism of action against SARS-CoV-2 or COVID-19. Promising results of new drugs against new coronavirus or the disease. PF-07304814 acts blocking the 3CL protease from SARS-CoV-2 avoiding the cell infection. RBT-9 is an antiviral drug blocking virus replication. TD-0903 blocks the JAK-STAT signaling mediated by the cytokine storm avoiding the damages caused by the exacerbation of pro-inflammatory response in the lung. M5049 is an antagonist of TLR7/8 that can recognize the viral RNA and stimulates the innate immune response with higher expression of pro-inflammatory cytokine genes, as IL-6. The use of M5049 helps the pro-inflammatory cytokine storm avoiding the inflammatory damage caused by the infection. BLD-2660 blocks the IL-6 action during the cytokine storm caused by the new coronavirus.
PF-07304814: The designed phosphate prodrug PF-07304814 is metabolized to PF-00835231, which is a potent inhibitor in vitro of the SARS-CoV-2 3CL protease in preclinical studies.7,8 PF-00835231 exhibits potent in vitro antiviral activity against SARS-CoV-2 as a single agent. It is additive/synergistic in combination with remdesivir and can be more potent than remdesivir.7,8 This drug was developed by Pfizer, and it is being evaluated in phase I clinical trials (clinical trial number NCT04535167) in hospitals in the United States.
DFV890: Developed by Novartis, and the efficacy and safety of DFV890 for the treatment of COVID-19 patients with pneumonia and impaired respiratory function, it is being assessed in phase II clinical trials (NCT04382053). The clinical trials involve patients from 12 different countries: Argentina, Brazil, Denmark, Germany, Hungary, India, Mexico, the Netherlands, Peru, the Russian Federation, South Africa, and Spain (recruitment status completed).
RBT-9: This drug is being developed by Renibus Therapeutics as an antiviral with organ-protective characteristics. Renibus Therapeutics will conduct phase II trials of this compound with antiviral and immune-modulating activity to evaluate the efficacy, tolerability, and safety of a single dose of RBT-9 versus placebo in COVID-19 infection in non-critically ill adults who are at high risk of progression (NCT04364763).
TD-0903: It is a lung-selective, nebulized pan-JAK inhibitor that was discovered and developed by Theravance Biopharma for the treatment of acute lung injury associated with COVID-19. Through its mechanism, TD-0903 has the potential to broadly inhibit Janus kinase/signal transducers and activators of transcription (JAK-STAT) signaling in airways, blocking the release of cytokines and chemokines that may be associated with acute lung injury and the initiation of a cytokine storm syndrome. Preclinical studies suggest that TD-0903 has a very high lung-to-plasma ratio and rapid metabolic clearance, resulting in low systemic exposure, which is compatible with its lung selectivity. TD-0903 is administered via a nebulised inhalation solution, which further enhances its lung selectivity. Preclinical pharmacodynamic studies indicate that TD-0903 has an extended duration of action that should enable once or twice daily dosing in humans [two studies, both with recruitment status completed: one in phase I (NCT04350736) and the other in phase II (NCT04402866)].
M5049: First-in-class small molecule blocks the activation of Toll-like receptor (TLR) 7 and TLR8, which are two innate immune sensors that detect single-stranded RNA from viruses such as the SARS-CoV-2 genome. The activation of TLR7/8 leads to immune cell activation and inflammation, which, when not properly controlled, can cause severe immunopathology. Merck will initiate a phase II randomized controlled clinical study evaluating the safety and efficacy of M5049, aiming to prevent or ameliorate the hyper-inflammatory response and to prevent progression to cytokine storm syndrome in COVID-19 patients.
BLD-2660: Novel, synthetic, orally active small molecule inhibitor of calpain 1, 2, and 9 is selective over the cathepsins as well as other protease families. It displays good metabolic stability and permeability, oral bioavailability, and low cytochrome P450 inhibition. It is under development for the treatment of patients with COVID-19 in phase II clinical trials in the US and Brazil hospitals (NCT04334460).
SNG001: This is an inhaled drug that contains an antiviral protein called interferon beta (IFN-β). Many viruses inhibit IFN-β as part of their strategy to evade the immune system. IFN-β protects cells against the MERS and SARS coronaviruses. SNG001 is in phase II clinical trials for COVID-19 (NCT04385095) and chronic obstructive pulmonary disease (COPD) (NCT03570359) and two more studies, NCT04732949 in phase III and NCT04518410 in phase II/III.
Drugs repositioning for COVID-19 treatment
Pandemics, such as the COVID-19 pandemic, create unique challenges for health professionals, such as the selection of appropriate treatments and the lack of time available for drug discovery. Most of the clinical trials evaluate drug repositioning to control viral infection using antivirals or other drugs of similar effect, such as antiparasitic and antibacterial drugs. In addition, there are several clinical trials that are evaluating anti-inflammatory drugs, antineoplastics, antioxidants, food supplements, and medications related to cardiovascular diseases, glycemia, and lipid diseases (Table 2).
Repositioned drugs for treatment of COVID-19 being evaluated within 4148 clinical trials at ClinicalTrial.gov.
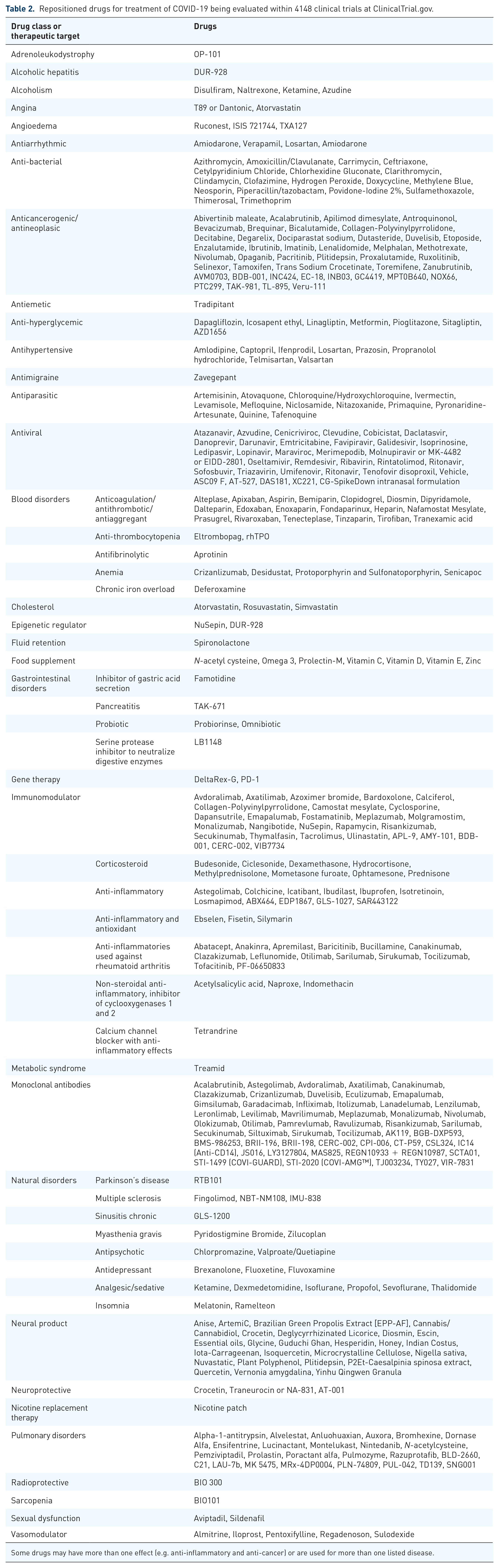
Some drugs may have more than one effect (e.g. anti-inflammatory and anti-cancer) or are used for more than one listed disease.
Antiviral drugs to treat COVID-19
We found 653 drugs developed for viral infections, such as hepatitis, HIV, Ebola, influenza, yellow fever, Zika, and Chikungunya, which are being tested to treat patients with COVID-19 (Table 2). Some drugs can inhibit viral replication (e.g. favipiravir, remdesivir, and ribavirin) or inhibit viral entry (e.g. lopinavir–ritonavir and umifenovir). 9
Molnupiravir is a small-molecule antiviral prodrug that is converted in plasma to NHC [N(4)-Hydroxycytidine] and then to the 5′-triphosphate form by host cell kinases. 10 In phase II clinical trials (NCT04405570 and NCT04405739), the drug showed efficacy against infections of the Wuhan strains (B.1.1.7 and B.1.351), with the potential to combat the worrisome variants of SARS-CoV-2. 11 In phase II studies (NCT04939428), with 202 unvaccinated participants, with confirmed SARS-CoV-2 infection, receiving 200–800 mg of molnupiravir or placebo, orally twice a day for 5 days, showed accelerated clearance of SARS-CoV-2 RNA and clearance of the infectious virus. 10 In phase III studies (NCT04575597), the drug has been shown to provide early treatment decreasing the risk of hospitalization or death in at-risk and unvaccinated adults. 12 Until now, the results in clinical trials suggest that molnupiravir works to reduce the chances of the SARS-CoV-2 virus multiplying in the body, making it possible to treat COVID-19 in adults who do not require supplemental oxygen and who are at increased risk of disease progression.
Remdesivir was developed for the treatment of RNA-based viruses and showed efficient against Ebola virus and Coronaviridae family viruses exemplified by MERS and SARS.13–15 This drug can be incorporated into nascent viral RNA and further inhibit the RNA-dependent RNA polymerase, inducing premature termination of viral RNA transcription. 13 Recent in vitro studies have shown that remdesivir is a potent SARS-CoV-2 inhibitor, which can justify the ongoing clinical trials for the treatment of COVID-19.8,16,17 Grein et al. 18 observed clinical improvement in 68% of patients hospitalized for severe COVID-19 who were treated with remdesivir. Another study showed clinical improvement using remdesivir, with a median recovery time of 10 days, as compared with 15 days among those who received placebo. Furthermore, the mortality was lower for the remdesivir-treated patients when compared with those who received placebo. 19
Favipiravir effectively inhibits the RNA-dependent RNA polymerase and viral replication. 17 The effectiveness in vitro against influenza viruses, Ebola virus, and SARS-CoV-2 was described.15,16 In 67 clinical trials, favipiravir is being investigated alone or in combination with other drugs to treat COVID-19. Favipiravir was shown to partially control inflammatory mediators in COVID-19 patients. However, the inflammation and cytokine storm continued after favipiravir administration that could be controlled with steroids. 20 Cai et al. 21 conducted an open-label control study where favipiravir + IFN-α exhibited faster viral clearance and radiological improvement compared with the lopinavir/ritonavir + IFN-α group.
Lopinavir, a HIV type 1 aspartate protease inhibitor, showed SARS-CoV-2 inhibitory activity in vitro.17,22 Ritonavir is combined with lopinavir to increase its plasma half-life through the inhibition of cytochrome P450. 17 Lopinavir/ritonavir is being investigated in 74 clinical trials. In their study with hospitalized adult patients with severe COVID-19, Cao et al. 23 concluded that no benefit was observed with lopinavir/ritonavir treatment. In another open-label randomized phase II trial by Hung et al., 24 a triple combination of IFN-β-1b and lopinavir/ritonavir was proven to be safe and superior to lopinavir/ritonavir, with patients showing alleviation of symptoms, shortened duration of viral shedding, and shortened hospital stay.
In monotherapy, umifenovir, which inhibits membrane fusion of the influenza virus, was shown to be superior to lopinavir/ritonavir in treating COVID-19. No viral load was detected in the umifenovir group after 14 days of admission, but the viral load was found in 44.1% of patients treated with lopinavir/ritonavir. 25 Also, favipiravir significantly improved the latency to relief for pyrexia and cough. 26 The combination of umifenovir + lopinavir/ritonavir indicates a faster reduction in viral load, preventing overstimulation of the immune system, hence diminishing the severity, duration, and infectiveness of the illness when compared with lopinavir/ritonavir treatment. 27
Ribavirin, which is usually used for hepatitis C treatment and is activated by adenosine kinase to ribavirin triphosphate (RTP), binds to the nucleotide-binding site and prevents further nucleotide addition and acts disrupting 5′ CAP addition to viral mRNA.28,29 Ribavirin is being investigated in 18 clinical trials to treat COVID-19. Tong et al. 30 conducted a trial with severe COVID-19 patients, showing no association with negative conversion time for real-time polymerase chain reaction (PCR) for SARS-CoV-2 test and the ribavirin therapy is not associated with improved mortality rate.
Other anti-hepatitis C virus drugs, such as daclatasvir and sofosbuvir, showed in vitro antiviral activity against SARS-CoV-2. Both antivirals prevented virus-induced neuronal apoptosis and the release of cytokine storm-related inflammatory mediators, respectively. Daclatasvir also targeted secondary RNA structures in the SARS-CoV-2 genome. 31 Sofosbuvir is being investigated in 12 clinical trials, and daclatasvir is being investigated in 10 clinical trials for the treatment of COVID-19. Eslami et al. conducted a trial where severe COVID-19 patients treated with sofosbuvir/daclatasvir had lower mortality than only ribavirin-treated patients. 29 Other studies have suggested that sofosbuvir/daclatasvir improves survival and clinical recovery in patients with moderate-to-severe COVID-19.32,33
Some experimental drugs developed for other viral diseases that have not yet been approved by regulatory agencies are also being evaluated for the treatment of COVID-19, such as ASC09 F (HIV), AT-527 (hepatitis C), DAS181 (parainfluenza), XC221 (influenza), and galidesivir. In preclinical studies, galidesivir (BCX4430) has been shown to act as a non-obligate RNA chain terminator, which can inhibit viral RNA polymerases of a wide array of RNA viruses, including flaviviruses (yellow fever and Zika virus), filoviruses (Ebola virus and Marburg virus), and coronaviruses (SARS-CoV and MERS-CoV).34,35 Target-based virtual screening and molecular docking results have shown that the antiviral galidesivir has promise against SARS-CoV-2 since it tightly binds to the RNA-dependent RNA polymerase.36,37 Currently, human trials are completed in Brazilian hospitals to evaluate the pharmacokinetics, safety, and antiviral activity of galidesivir in subjects with yellow fever and COVID-19 (NCT03891420).
Antiparasitic and antibacterial drugs applied to treat COVID-19
Some drugs normally used as antiparasitic drugs have been evaluated in the treatment of COVID-19, such as artemisinin, atovaquone, chloroquine/hydroxychloroquine, ivermectin, levamisole, mefloquine, niclosamide, nitazoxanide, primaquine, pyronaridine-artesunate, quinine, and fafenoquine (Table 2). Ivermectin is a Food and Drug Administration (FDA)-approved antiparasitic agent, with demonstrated antiviral effects against HIV, Zika virus, and dengue virus.38,39 Vero/hSLAM cells infected with SARS-CoV-2 were exposed to 5 µM ivermectin, with 93% reduction in viral RNA present in the supernatant (indicative of released virions) of samples treated with ivermectin after 24 h, and a 5000-fold reduction in viral RNA compared with the control was found after 48 h. 40 The authors proposed that the drug may have antiviral effects by inhibiting the importin (IMP) α/β receptor, which is responsible for transmitting viral proteins into the host cell nucleus. 40 We found 88 clinical trials evaluating this drug until August 2022. Although randomized controlled trials are needed to confirm the effects of ivermectin for the treatment of COVID-19, a study involving 280 patients showed lower mortality during treatment of COVID-19, especially in patients with severe pulmonary involvement. 38 Another study compared the viral clearance in 400 COVID-19 patients treated with ivermectin/doxycycline or hydroxychloroquine/azithromycin. The clearance was 132 (66%) on day 5 and 167 (83.5%) on day 6 in the group treated with ivermectin, whereas there was 154 (77%) viral clearance on the 11th day and 163 (81.5%) viral clearance on 12th day of hydroxychloroquine ingestion. The authors concluded that ivermectin and doxycycline is a safe combination that was found to be effective in terms of early viral clearance in patients with mild-to-moderate COVID-19. 41 A clinical trial of ivermectin plus doxycycline for the treatment of COVID-19 showed lower risk of mortality in patients treated with these drugs (NCT04523831). It is important to remember that FDA has not approved ivermectin for use in treating or preventing COVID-19. According to The COVID-19 Treatment Guidelines by National Institutes of Health (NIH), there are insufficient data for recommendation either for or against the use of ivermectin for the treatment of COVID-19 and more studies are needed.
Chloroquine and hydroxychloroquine, which are used in the treatment of malaria and autoimmune disorders, have been the focus of great public attention.42,43 Some preliminary studies have shown that hydroxychloroquine is effective at inhibiting SARS-CoV-2 infection in vitro, with less toxicity, and it is more potent than chloroquine.44,45 However, large-scale randomized controlled trials have failed to show any survival benefit of such drug therapy in this disease.46–48 Despite all this information, these drugs are being evaluated in 249 clinical trials, even in comparison or in combination with other drugs. Seventy-one of these studies analyze the combination of hydroxychloroquine and azithromycin. On 28 March 2020, the US FDA has issued the United States of America (USA) for the inclusion of chloroquine and hydroxychloroquine in the treatment of COVID-19. Subsequently, on 15 June 2020, the US FDA revoked the emergency use authorization based on its ongoing analysis. The FDA further stated that both of these drugs show no benefit on mortality or in speeding recovery and hence are unlikely to be effective in treating COVID-19 patients. On 27 May 2021, NIH published The COVID-19 Treatment Guidelines based on an assessment of the collective evidence from several clinical studies that recommends against the use of these drugs for COVID-19 prophylaxis or treatment because there were no differences between treated groups and placebo. In addition, there were reports of adverse events after using these drugs (https://www.covid19treatmentguidelines.nih.gov/). A review study on these drugs emphasizes the need for evidence-based treatment approaches from large randomized clinical trials to confront the ongoing COVID-19 pandemic and not the mere observational study that mislead the public healthcare system, which paralyzes the entire world. 49
Another broad-spectrum antiparasitic, nitazoxanide, has shown antiviral activity against MERS-CoV with inhibition of the production of pro-inflammatory cytokines tumor necrosis factor α (TNF-α), IL-2, IL-6, and IL-8. 50 A recent review evaluated nine nitazoxanide clinical trials, concluding that this drug demonstrates a good safety profile at the approved doses; however, further evidence is required regarding hepatorenal and cardiovascular effects as well as teratogenicity. There were 31 nitazoxanide clinical trials underway worldwide up to August 2022. A recent study showed that 78% and 57% of patients in the nitazoxanide and placebo groups, respectively, reported complete resolution of symptoms at the 1-week follow-up. The viral load was also reduced after treatment with nitazoxanide compared with placebo, and no serious adverse events were observed. 51
Anti-cancer drugs repositioned to COVID-19 treatment
Originally developed for cancer treatment, opaganib, a sphingosine kinase-2 (SK2) selective inhibitor, is thought to be an anti-inflammatory and potential antiviral drug for COVID-19. Opaganib demonstrated potent anti-SARS-CoV-2 activity, achieving complete blockage of viral replication in an in vitro model of human lung tissue. In a small cohort of severe COVID-19 patients, opaganib was safe and well tolerated, with demonstrated improvement in both clinical and laboratory parameters in all treated patients. 52 We found four clinical trials evaluating this drug in the treatment of COVID-19, three complete and one withdrawn.
According to investigators, DeltaRex-G, another anti-cancer drug, is the first and, so far, only disease-targeted gene therapy that has the potential to effectively treat COVID-19-induced cytokine storm and respiratory distress syndrome. The hypothesis is that DeltaRex-G can hasten recovery from pneumonia and prevent chronic lung disease by killing the activated immune cells that cause acute respiratory distress syndrome and the proliferative fibroblasts that cause chronic lung disease. DeltaRex-G has been investigated in a phase I/II clinical trial (NCT04378244).
COVID-19 treatment based on immunomodulatory drugs
These drugs are based on immunotherapy approaches aimed at either boosting innate antiviral immune responses or alleviating damage induced by dysregulated inflammatory responses. Corticosteroids, anti-inflammatory drugs, antineoplastic agents, monoclonal antibodies, and even food supplements and products of natural origin are being evaluated for the treatment of COVID-19 (Table 2).
The use of systemic glucocorticoids has been discussed in SARS-CoV-2 infection since they may prolong viral clearance. However, it is also known that the underlying pathogenesis of COVID-19 pneumonia is composed of both the direct damage caused by the virus and the excessive immune response from the host. Thus, the administration of drugs such as methylprednisolone would help suppress the unwanted immune reactions. 53 A study indicated that single-dose pulse methylprednisolone (40–500 mg) had no apparent negative impact on SARS-CoV-2 removal or the production of specific IgG, while effectively stopping the inflammatory cascade. 54 Another study also concluded that methylprednisolone could lower the mortality rate in more severe forms of the condition, 55 and it is being evaluated in 74 clinical trials, some of them in phase III.
A prospective meta-analysis of clinical trials of critically patients with COVID-19 showed lower 28-day all-cause mortality after administration of systemic corticosteroids when compared with usual care or placebo. 56 Another anti-inflammatory corticosteroid drug, dexamethasone, has been recently suggested to treat severe COVID-19 patients. Patients on mechanical ventilation or oxygen support who received dexamethasone exhibited lower mortality rates compared with those receiving standard care alone. 57 Currently, 110 clinical trials are underway to assess the risk versus benefit for the use of glucocorticoids in the treatment of COVID-19.
Several natural products and food supplements are also being evaluated for the treatment of patients with COVID-19 because many of these drugs have immunomodulatory effects (Table 2). Colchicine is an alkaloid derived from autumn crocus, which has been used to treat several inflammatory diseases, such as gout, for many years, and it is being investigated in 35 clinical trials for the treatment of COVID-19. The results of a clinical study conducted by Brazilian researchers showed that colchicine can help combat lung inflammation with faster recovery for patients with moderate-to-severe COVID-19. 58
A nanoparticle formulation for spray administration, called ArtemiC (comprising artemisinin, curcumin, Boswellia, and vitamin C), has been proposed as a treatment for COVID-19. ArtemiC has well-established immunomodulatory activities as demonstrated in vitro and in vivo, and published over the years, diminishing the activity of TNF-α and IL-6 levels what can be relevant to the pathophysiology processes involved in the progressive form of COVID-19. In addition, the active agents have prominent antioxidant, anti-inflammatory, as well as anti-aggregating and anti-microbial activities.59–61 In this context, natural compounds, such as curcumin, have shown beneficial effects that make them a promising target in the adjuvant treatment of COVID-19 as recently reviewed.62,63 A phase II controlled clinical study designed to evaluate the effect of ArtemiC in patients diagnosed with COVID-19 is being done in hospitals in Israel and India (NCT04382040).
Angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) to treat COVID-19
Hypertension and cardiovascular disease are risk factors for death in COVID-19. Angiotensin-converting enzyme 2 (ACE2), an important component of the renin–angiotensin system, serves as the binding site of SARS-CoV-2 [spike (S) protein] and facilitates host cell entry in the lungs. In experimental models, 64 angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) have been shown to increase ACE2 expression in several organs, potentially promoting viral cell invasion, although these findings are not consistent across studies. 65 Alternatively, ACEIs and ARBs may actually improve mechanisms of host defense or hyperinflammation, ultimately reducing organ injury. Finally, ACEIs and ARBs may have direct renal, pulmonary, and cardiac protective benefits in the setting of COVID-19. Therefore, it is unclear whether ACEIs and ARBs may be beneficial or harmful in patients with COVID-19. Given the high prevalence of hypertension and cardiovascular and renal diseases in the world, the high prevalence of ACEIs or ARBs in these conditions, and the clinical equipoise regarding the continuation versus discontinuation of ACEIs/ARBs in the setting of COVID, a randomized trial is urgently needed. The aim of the NCT04338009 trial with 152 participants was to assess the clinical impact ACE inhibitors and angiotensin receptor blockers on outcomes in patients hospitalized with COVID-19. According to this study, compared with discontinuation of renin–angiotensin system inhibitors, continuation had no effect on the global rank score. 66 Another study with 611 patients with COVID-19 showed that ACEI exposure significantly reduced the risk of severe disease [odds ratio (OR) = 0.37, 95% confidence interval (CI): 0.15–0.87, p = 0.03). ACEI exposure was associated with milder infiltrations seen on baseline computed tomography, lower C-reactive protein and ferritin, higher monocytes, shorter hospitalization, and less requirement for specific empirical treatments (favipiravir and meropenem). The data suggest that exposure to ACEI drugs may have favorable effects in the context of COVID-19 pneumonia. 67 According to The COVID-19 Treatment Guidelines by NIH, patients with COVID-19 who are receiving angiotensin-converting enzyme (ACE) inhibitors or angiotensin receptor blockers (ARBs) for cardiovascular disease (or other non-COVID-19 indications) should not discontinue these medications unless discontinuation is otherwise warranted by their clinical condition. The COVID-19 Treatment Guidelines recommends against the use of ACE inhibitors or ARBs for the treatment of COVID-19, except in a clinical trial. Additional investigations of the role of ACE inhibitors, ARBs, and recombinant human ACE2 in the management of COVID-19 are underway. Human recombinant soluble ACE2 (hrsACE2; APN01) can significantly block early SARS-CoV-2 infections by a factor of 1.000–5.000. 68 HrsACE2 has entered a placebo-controlled, double-blind, phase IIb trial in severe COVID-19 patients (NCT04335136), acting as a molecular decoy to block virus entry, and as a regulator of the renin–angiotensin system. Remdesivir has received authorization for COVID-19 and has been shown to improve outcomes; however, the dose of remdesivir is limited by hepatic and kidney toxicity. Monteil et al. 69 evaluated the therapy using remdesivir with recombinant soluble ACE2 targeting two different modalities of SARS-CoV-2 life cycle: cell entry via its receptor ACE2 and intracellular viral RNA replication. This combination treatment markedly improved their therapeutic windows against SARS-CoV-2 in vitro models. The data demonstrated that combining two therapeutic modalities with different targets exhibit strong additive effects at sub-toxic concentrations. Some clinical trials are evaluating the combination of recombinant bacterial ACE2 receptors and drugs (NCT04382950; NCT04375046).
Monoclonal antibodies as therapy against SARS-CoV-2 and to treat COVID-19
According to the Chinese Antibody Society, most of the antibodies that are candidates for use as therapy against SARS-CoV-2 have ‘Discovery’ status (33.3%) followed by ‘Preclinical’ status (25.8%).
Most of the studies uses monoclonal antibodies (78.3%), followed by single domain Ab (6.6%) and fusion protein (6.1%). The United States has the largest number of studies on the subject, followed by China and the United Kingdom.
Monoclonal antibodies are also classified as ‘Drug Interventions’ according to ClinicalTrials.gov, and some of these antibodies have been developed especially for the treatment of COVID-19, such as LY3819253 (LY-CoV555 or bamlanivimab), LY3832479 (LY-CoV016), and BAT2020. A phase I/II study to assess the safety, pharmacokinetics, immunogenicity, and efficacy of BAT2020 is being carried out in patients hospitalized with COVID-19 (NCT04432766). LY-CoV555 is a potent, neutralizing IgG1 monoclonal antibody (mAb) directed against the spike protein of SARS-CoV-2, and it was identified from a blood sample taken from one of the first US patients who recovered from COVID-19. The LY-CoV555 clinical trial has successfully completed enrollment and primary safety assessments in a phase I study of hospitalized patients with COVID-19 (NCT04411628), and long-term follow-up is ongoing as a phase II/III study in people recently diagnosed with COVID-19 in the ambulatory setting (NCT04427501). Recently, the phase III study of LY-CoV555 for the prevention of COVID-19 in residents and staff at long-term care facilities has begun (NCT04497987). This drug received emergency use authorization from the FDA on 10 November 2020. 8 In addition, LY-CoV555 is being tested in combination with LY-CoV016, which binds a different epitope in the SARS-CoV-2 spike region. The trial is currently enrolling a larger confirmatory cohort of higher risk patients and is testing the ability of the antibody combination to reduce the number of patients with persistently high viral load and to reduce COVID-related hospitalizations.
The monoclonal antibody that most presents clinical trials use is tocilizumab, which is a recombinant humanized anti-human IL-6 receptor. IL-6 takes center stage in COVID-19 because it acts as a cytokine storm amplifier, inducing the production of more pro-inflammatory cytokines and the production of acute phase proteins by the liver, weakening the papillary muscle and leading to myocardial dysfunction.6,7 Seventy randomized trials with tocilizumab are ongoing, and some of these are in phases III and IV. Tocilizumab might be useful in the treatment of COVID-19 patients with a hyperinflammatory state, presenting lower hazards of intubation or death, but the efficacy is under discussion.14–16,70
Baricitinib, an oral JAK1/JAK2 inhibitor, has been proven to be promising for lowering airway inflammation in SARS-CoV-2-infected rhesus macaques, leading to fast and potent suppression of the alveolar macrophage-derived production of cytokines and chemokines responsible for inflammation and neutrophil recruitment. 16 Eighteen clinical trials are ongoing, including in combination with remdesivir (NCT04401579).
Several other monoclonal antibodies are being evaluated for COVID-19 (Table 2). Most of them target S protein, GM-CSF or GM-CSFR, IL-6, or IL-6R. Among them, sarilumab, an anti-IL6R, is present in 12 clinical trials at different stages; canakinumab, an inhibitor of IL-1beta, is present in 6 clinical trials; and clazakizumab (targets IL-6) and mavrilimumab (targets GM-CSFRα).
Convalescent plasma from patients who have recovered from COVID-19
Convalescent plasma (CP) therapy is another procedure being tested for COVID-19. Patients who have recovered from COVID-19 should have an elevated number of polyclonal antibodies produced by the immune system to prevent new circles of infection by SARS-CoV-2.71,72 Clinical trials have provided robust evidence that the transfusion of CP is safe in hospitalized patients with COVID-19, supporting the notion that earlier administration of plasma within the clinical course of COVID-19 is more likely to reduce mortality.73–75 One limitation of this therapy is the variability in potency found in the sera of recovered patients in neutralizing the antigen. 76 A study in the United States (NCT04343261) demonstrated that the early administration of CP in the group of patients with severe illness resulted in improved outcomes. 77
The United States has the largest number of studies addressing this theme, totaling 184 clinical trials, and the second region with the largest number of studies is Europe (especially Italy and Spain). South America has a significant number of studies when compared with certain regions, such as North and East Asia (Figure 2).
There are 84 studies with completed status. Most of the studies are in recruitment status (a total of 73) or not yet recruiting status (a total of 18), and 17 of the studies were classified as observational.
Mesenchymal cell therapy to treat COVID-19
We found 70 clinical trials evaluating mesenchymal stem cells (MSCs) for the treatment of patients with COVID-19 (Figure 2). MSCs are non-specialized multipotent cells that have self-renewal and the capacity to differentiate into diverse cell types. MSCs can be isolated from peripheral blood, bone marrow, the umbilical cord, or the placenta, and after expansion, they can be administered to patients with cytokine storm and sepsis because of their powerful anti-inflammatory and immunomodulatory abilities. 78 Some clinical studies have suggested that transplantation of MSCs can improve the outcome of COVID-19 patients, possibly by regulating the inflammatory response and promoting the recovery of antiviral immune cells and organs.78–80
Conclusion
The whole world has come together in the search for effective therapies for the treatment or prevention of COVID-19. Difficulties in terms of higher dose production, storage, and transportation may be challenges for pharmaceutical industries during vaccine distribution. On the contrary, efforts have been made to create different vaccines with different approaches so that they can be used by the entire population. Moreover, the search for new drugs or the repositioning of available drugs can be useful for treating COVID-19 until the virus is eradicated. Although there has been a great effort by researchers around the world, the higher number of clinical trials in Europe and the United States suggests the need for public and private investments in the development of cutting-edge research globally. Nevertheless, the different types of studies and clinical trials around the world have the same goal of discovering effective methods of treatment and prevention of COVID-19. Here, we summarized about 4.148 clinical trials that evaluated new drugs or drug repositioning in the treatment of COVID-19. Each study brings the pros or cons related to the use of drugs with important results of antiviral new drugs that are in phase II of the study. Monoclonal antibodies have been presenting good results targeting SARS-CoV-2 neutralizing the virus, as remdesivir, and the monoclonal antibodies targeting pro-inflammatory cytokines or signaling pathways to avoid the cytokine storm what can help patients suffering from the clinical manifestations of COVID-19.
