Abstract
To address the coronavirus disease 2019 (COVID-19) pandemic, several antiviral agents targeting severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) have been developed for clinical use. However, antivirals that can be administered irrespective of risk factors were lacking until the approval of ensitrelvir fumaric acid (hereafter, ensitrelvir) in Japan, which took effect in November 2022. Ensitrelvir is an oral SARS-CoV-2 3C-like protease inhibitor currently approved in Japan and Singapore. This narrative review summarizes preclinical, clinical trial, and real-world data on ensitrelvir. The efficacy and safety of ensitrelvir were assessed in a seamless, randomized, double-blind, placebo-controlled, phase II/III study conducted in Japan, South Korea, and Vietnam (Japan Registry of Clinical Trials identifier, jRCT2031210350). This study enrolled patients with mild-to-moderate COVID-19 symptoms or asymptomatic individuals irrespective of the presence of risk factors for severe illness. Overall, ensitrelvir demonstrated favorable antiviral efficacy and symptom improvement, with an acceptable safety profile. In the phase III part, the time to resolution of the composite of five typical COVID-19 symptoms showed a difference between the ensitrelvir 125 mg and placebo groups, and the difference in median was approximately 1 day when the patients were randomized in less than 72 h of disease onset. This study is one of the clinical trials that used patient symptoms as a clinical efficacy endpoint. Additional clinical trials are currently underway to investigate the efficacy and safety of ensitrelvir in various patient populations. Moreover, published evidence generally supports the effectiveness of ensitrelvir in routine clinical practice and its antiviral activity against various SARS-CoV-2 variants of concern. Further research is granted to establish ensitrelvir as a novel antiviral treatment. Royalty-free licensing agreements concluded between drug manufacturers and the Medicines Patent Pool will facilitate access to COVID-19 therapeutics, including ensitrelvir, in low- and middle-income countries.
Plain language summary
Several new antiviral drugs have become available for the treatment of coronavirus disease 2019 (COVID-19). These drugs are designed to block the replication of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the virus that causes COVID-19. However, few antiviral drugs can be used for patients not at risk for severe COVID-19. Ensitrelvir is an oral antiviral drug currently available in Japan and Singapore. This paper reviews the preclinical, clinical trial, and real-world data on the efficacy and safety of ensitrelvir. The efficacy and safety of ensitrelvir were assessed in a phase II/III clinical study conducted in Japan, South Korea, and Vietnam. Both patients with and without a risk for severe COVID-19 participated in this study. Ensitrelvir treatment reduced the viral titer and viral RNA of SARS-CoV-2, which are indicators for viral levels in patients with COVID-19, in the nasopharynx (the upper part of the throat behind the nose). The treatment improved the symptoms of COVID-19 without safety concerns. The time to resolution of any of the five typical COVID-19 symptoms was shortened by approximately 1 day with ensitrelvir treatment when the patients were assigned to the treatment in less than 72 h after disease onset. The drug manufacturer is conducting additional clinical trials to further test the efficacy and safety of ensitrelvir. Ensitrelvir treatment in clinical practice has also been evaluated in patients in Japan. The manufacturer and Medicines Patent Pool, a public health organization supported by the United Nations, have signed a contract to make new COVID-19 antiviral drugs, including ensitrelvir, available in low- and middle-income countries.
Introduction
Coronavirus disease 2019 (COVID-19), a human respiratory disease caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has posed a significant burden on global health security. As of September 2024, more than 700 million confirmed cases and more than 7 million deaths have been reported worldwide. 1 However, these data likely underestimate the actual disease burden in many countries, particularly at the peak of the first wave of the pandemic that occurred in March 2020. 2 Moreover, several SARS-CoV-2 variants of interest have been observed globally.3,4 As of September 2024, SARS-CoV-2 BA.2.86 and JN.1 have been designated as variants of interest because of their increased ability to spread or cause serious disease, potentially posing an emerging risk on global public health. 4
Oral antiviral agents targeting SARS-CoV-2 have been developed to address this pandemic. The SARS-CoV-2 main protease (Mpro; alternative name, 3C-like protease (3CLpro)) inhibitor nirmatrelvir, when co-administered with the pharmacokinetic booster ritonavir, demonstrated efficacy among unvaccinated, nonhospitalized adults with COVID-19, 5 as demonstrated in the phase II–III EPIC-HR (Evaluation of Protease Inhibition for COVID-19 in High-Risk Patients) study. 5 The phase III component of the MOVe-OUT study demonstrated the efficacy of the SARS-CoV-2 viral RNA polymerase inhibitor molnupiravir in unvaccinated, nonhospitalized adults with COVID-19. 6 Moreover, monoclonal antibody-based treatment options, such as a SARS-CoV-2 neutralizing antibody sotrovimab 7 and a SARS-CoV-2 neutralizing antibody cocktail casirivimab and imdevimab, 8 were also developed for clinical use. These pivotal clinical trials for nirmatrelvir, molnupiravir, and monoclonal antibodies were conducted during the SARS-CoV-2 Delta-predominant period.5–8 These antiviral options initially targeted patients with risk factors for severe illness.5–8 However, antivirals that can be administered irrespective of risk factors were lacking until the approval of ensitrelvir fumaric acid (hereafter, ensitrelvir) in Japan, which took effect in November 2022. 9
Ensitrelvir is an oral SARS-CoV-2 3CLpro inhibitor that has demonstrated efficacy in patients with mild-to-moderate COVID-19 symptoms irrespective of the presence of risk factors for severe illness. 10 In Japan, ensitrelvir (125 mg tablets) obtained emergency use approval in November 2022, followed by standard approval in March 2024, for the treatment of SARS-CoV-2 infection. 9 The drug has also been approved in Singapore for the treatment of SARS-CoV-2 infection under the Special Access Route application. 9 Ensitrelvir oral administration should be initiated within 72 h of the onset of COVID-19, and the recommended dose is 375 mg on day 1 and 125 mg on days 2–5, once daily. 9 The efficacy and safety of ensitrelvir were assessed in a seamless phase II/III study conducted in Japan, South Korea, and Vietnam from September 2021 to August 2022 (Japan Registry of Clinical Trials identifier, jRCT2031210350).10–14
This narrative review summarizes preclinical, clinical trial, and real-world data on ensitrelvir to discuss its efficacy and safety in patients with mild-to-moderate COVID-19 symptoms. We also discuss the potential role of ensitrelvir in the current treatment landscape based on the published literature.
Ensitrelvir: Discovery, mechanism of action, and preclinical evidence
Ensitrelvir was discovered through a university-industry research collaboration of Hokkaido University, Japan, and Shionogi & Co., Ltd. 15 A structure-based drug design strategy consisting of docking-based virtual screening and biological screening using an in-house compound library was employed. 15
SARS-CoV-2 3CLpro plays a key role in viral replication and has served as a target for small-molecule oral therapeutics for COVID-19 treatment. 15 Ensitrelvir is characterized by its noncovalent binding to the active substrate-binding site of 3CLpro, thereby inhibiting SARS-CoV-2 viral replication.15,16 This molecule has shown in vitro and in vivo antiviral efficacy against a wide range of SARS-CoV-2 variants, including Alpha, Beta, Gamma, Delta, and Omicron.15,17–19 Furthermore, in vitro antiviral activity against the Omicron XBB.1.5 variant has been demonstrated for ensitrelvir and other antivirals (remdesivir, molnupiravir, and nirmatrelvir). 20 In addition, the results of an in vivo study using aged BALB/c mice suggested the potential efficacy of ensitrelvir in pre-exposure prophylactic settings. 21 Another in vivo study showed the efficacy of ensitrelvir combined with methylprednisolone in improving lung pathology and host inflammatory responses in a COVID-19 hamster model. 22
Summary of the seamless phase II/III study
Study design and overview
A randomized, double-blind, placebo-controlled, phase II/III study was conducted as a pivotal trial to assess the efficacy and safety of ensitrelvir (Figure 1(a)). To maximize patient enrollment during the rapidly changing COVID-19 pandemic, this study employed a seamless design consisting of dose-finding (phase IIa and phase IIb), confirmatory (phase III, SCORPIO-SR study), and exploratory (phase IIb/III) parts.10–14 The study was conducted during the SARS-CoV-2 Delta-predominant and Omicron BA.1/BA-2-predominant periods.10–14 The phase IIa part was conducted in Japan, 10 and the phase IIb part was conducted in Japan and South Korea. 11 The phase III and phase IIb/III parts enrolled participants from Japan, South Korea, and Vietnam.10,14 This phase II/III study aimed to enroll all individuals infected with SARS-CoV-2 regardless of the presence of risk factors for severe COVID-19. Only patients with an awake oxygen saturation of >93% (room air) were included. Patients with mild-to-moderate COVID-19 were defined as those who have had at least one moderate or severe symptom among the 12 COVID-19 symptoms, assessed in accordance with the U.S. Food and Drug Administration (FDA) guidance, September 2020 version. 23 The severity of these symptoms was determined based on the symptom scores (none, mild, moderate, or severe) at enrollment. The phase IIa part enrolled participants with asymptomatic infection or mild-to-moderate COVID-19 symptoms. 11 Participants with mild-to-moderate COVID-19 symptoms were enrolled in the phase IIb or phase III part,10,12 and those with asymptomatic to mild COVID-19 symptoms were enrolled in the phase IIb/III part. 14

Study design. (a) Overview of the seamless phase II/III study. (b) Study scheme. (c) Fourteen acute-phase COVID-19 symptoms.
A common study scheme was employed across study components (Figure 1(b)). Patients aged 12 to <70 years who tested positive for SARS-CoV-2 within 120 h of randomization were enrolled. Patients were eligible for participation regardless of their COVID-19 vaccination history or presence of risk factors for severe illness. A questionnaire developed based on the U.S. FDA guidance, September 2020 version, 23 was used to define the severity of COVID-19 symptoms based on patients’ self-assessment.10–14 The questionnaire consisted of 14 typical acute-phase COVID-19 symptoms (Figure 1(c)). Eligible patients were randomized (1:1:1) to receive ensitrelvir 125 mg (375 mg as a loading dose on day 1), ensitrelvir 250 mg (750 mg as a loading dose on day 1), or placebo once daily for 5 days. The follow-up period was set to 28 days.10–14
Throughout the study, antiviral efficacy (SARS-CoV-2 viral RNA and/or viral titer levels), clinical efficacy (improvement in COVID-19 symptoms and development/worsening of COVID-19 symptoms or fever), development of severe illness, and safety were evaluated according to the endpoints specified before database lock and unblinding for each part. SARS-CoV-2 viral RNA and viral titers were measured using nasopharyngeal swabs, and COVID-19 symptoms were self-recorded by each patient using an electronic diary (Figure 1(b)). Among the 14 acute-phase COVID-19 symptoms (Figure 1(c)), 12 symptoms, excluding anosmia and dysgeusia, were used to assess one of the co-primary endpoints in the phase IIb part. 12 A composite of five symptoms (stuffy or runny nose, sore throat, cough, feeling hot or feverish, and low energy or tiredness) was used as the primary endpoint in the phase III part as they were most commonly observed in patients infected with the Omicron variant. 10
Characteristics of study participants
Throughout the study, baseline demographics and clinical characteristics were generally balanced across the treatment groups.10–12,14 More than 80% of the participants had received at least one dose of COVID-19 vaccines before enrollment,10–12,14 which reflects the real-world scenario at the time of the study. The mean patient age in each treatment group was approximately 35–40 years.10–12,14 A majority (72.2%) of participants in the phase III part of the study did not have any risk factors for severe disease. 10
Antiviral efficacy
Treatment with ensitrelvir showed favorable antiviral efficacy throughout the study. Phase IIa and IIb parts of the study used the change from baseline in SARS-CoV-2 viral titer as the primary virologic endpoint.11,12 In the phase IIa part, the change from baseline in SARS-CoV-2 viral titer on day 4 was greater with ensitrelvir 125 mg (
The antiviral efficacy of ensitrelvir also extended to phase III and phase IIb/III parts of the study.10,14 In the phase III part, ensitrelvir 125 mg treatment demonstrated a significant difference versus placebo in the least-squares mean change from baseline in the SARS-CoV-2 RNA level on day 4: The least-squares mean (standard error) change from baseline to day 4 was –2.48 (0.08) log10 copies/mL and –1.01 (0.08) log10 copies/mL in the ensitrelvir 125 mg and placebo groups, respectively (
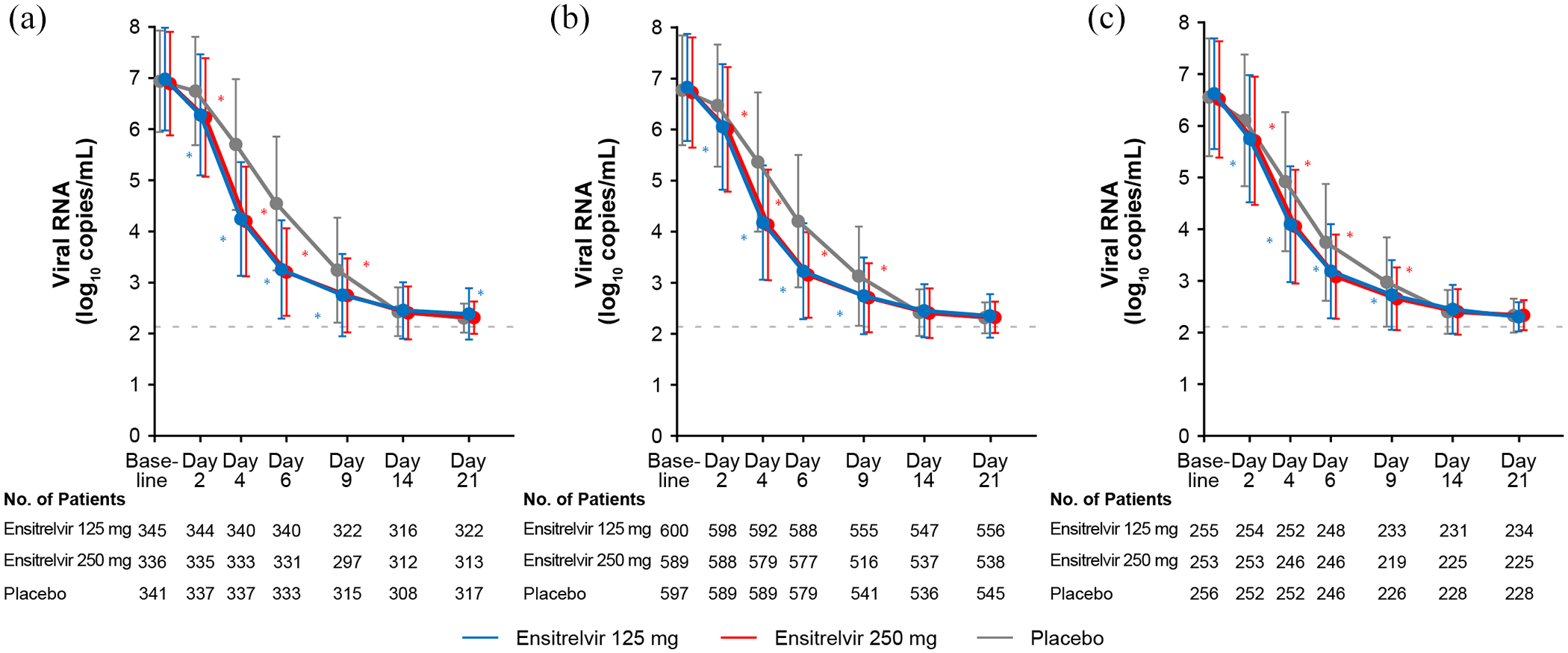
Findings of the phase III part: SARS-CoV-2 viral RNA levels up to day 21 in (a) Patients randomized in less than 72 h of disease onset. (b) Patients randomized within 120 h of disease onset. (c) Patients randomized within 72–120 h of disease onset.
Clinical efficacy
In the phase IIa part of the study, symptom improvement was assessed as a change from baseline in the total COVID-19 symptom score. 11 The mean change from baseline in the total score of the 12 COVID-19 symptoms was numerically greater in the ensitrelvir 125 and 250 mg groups than in the placebo group. 11 Similarly, the phase IIb part evaluated a time-weighted average change from baseline up to 120 h in the total score of the 12 COVID-19 symptoms as one of the co-primary endpoints. 12 The results showed no significant differences between the ensitrelvir and placebo groups. 12 In a post hoc analysis of the phase IIb part, wherein the 12 symptoms were divided into subcategories, the time-weighted average change from baseline up to 120 h was nominally and significantly greater with ensitrelvir versus placebo for several subcategory scores, including respiratory symptoms. 12
The phase III part of the study defined the primary endpoint as the time to resolution of the composite of five COVID-19 symptoms (stuffy or runny nose, sore throat, cough, feeling hot or feverish, and low energy or tiredness).10,13 The target dose for the efficacy evaluation was set to 125 mg (375 mg as a loading dose on day 1), and patients randomized in less than 72 h of disease onset were included in the primary efficacy analysis in view of the results of the phase IIb part.10,13 This phase III part used a Peto-Prentice generalized Wilcoxon test stratified by SARS-CoV-2 vaccination status to compare the survival distributions of the primary endpoint between ensitrelvir and placebo.
10
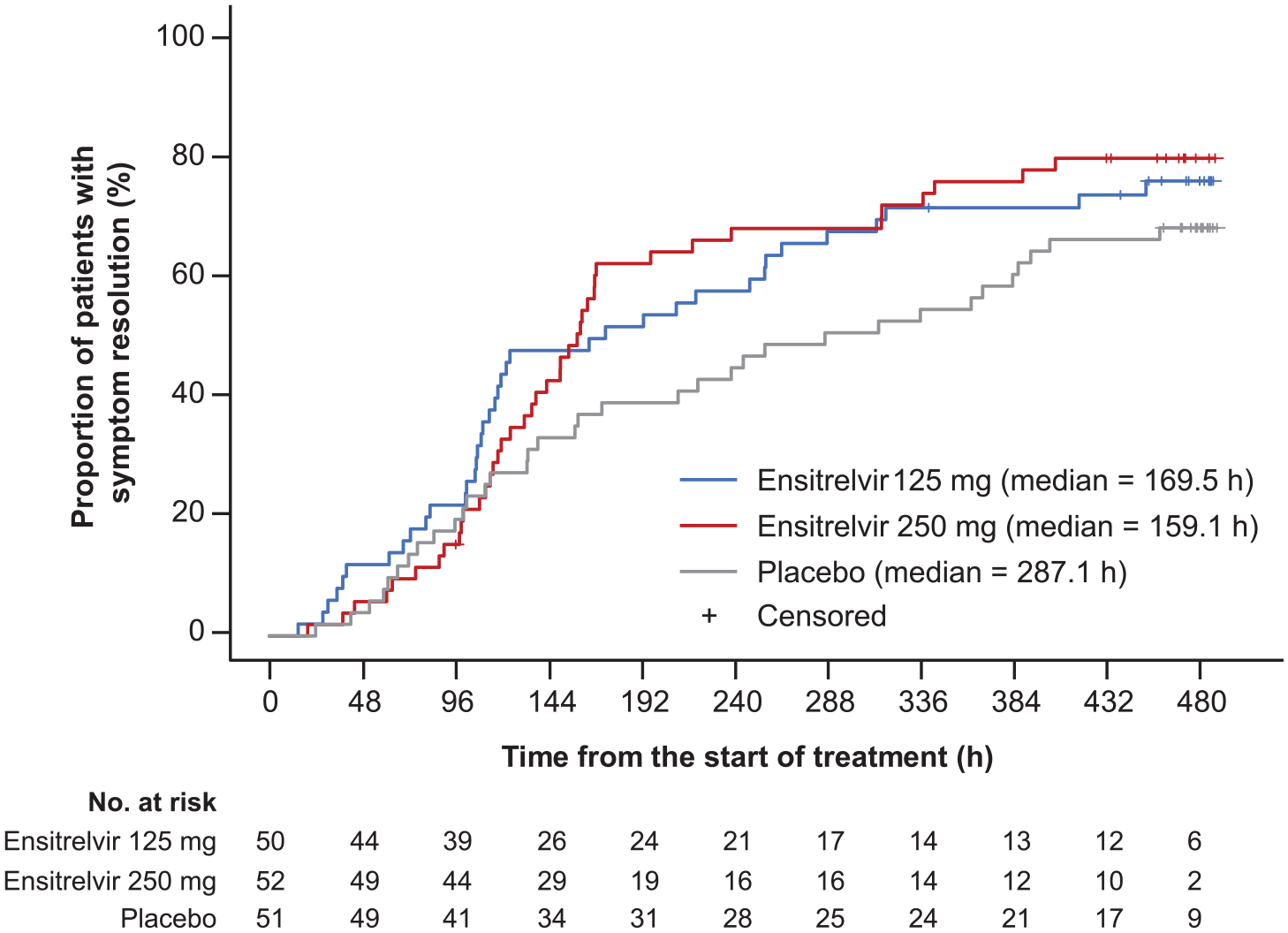
The time to resolution of the composite of five COVID-19 symptoms showed a difference between the ensitrelvir 125 mg and placebo groups (

Findings of the phase III part: time to resolution of the composite of five COVID-19 symptoms (patients randomized in less than 72 h of disease onset).

Findings of the phase IIb part: time to resolution of the composite of five COVID-19 symptoms (patients randomized in less than 72 h of disease onset).
Findings of the phase IIb part: time to resolution of the 14 acute-phase COVID-19 symptoms (patients randomized in less than 72 h of disease onset).
In the phase IIb/III part, the efficacy of ensitrelvir was evaluated in an exploratory manner. In this part, ensitrelvir 125 mg treatment showed a 77% reduction in the risk of developing any of the 14 acute-phase COVID-19 symptoms or fever versus placebo among asymptomatic individuals, while the results were statistically nonsignificant. Similarly, a 29% reduction in the risk of worsening of any of the 14 acute-phase COVID-19 symptoms or fever was observed among patients with mild COVID-19 symptoms (statistically nonsignificant). 14
Severe illness development
In the phase IIa and phase IIb parts of the study, the development of severe illness was evaluated using an eight-point ordinal scale (asymptomatic = 0; symptomatic, no limitation of activities = 1; symptomatic, limitation of activities = 2; hospitalized, no oxygen therapy = 3; hospitalized, with oxygen therapy (<5 L/min) = 4; hospitalized, with oxygen therapy (⩾5 L/min) = 5; hospitalized, with ventilation = 6; death = 7).11,12 A score of ⩾3 was regarded as severe illness requiring COVID-19-related hospitalization. The number of patients experiencing severe illness development was low across the study components. In the phase IIa part, 2 of 14 patients in the placebo group recorded ⩾3 on the 8-point ordinal scale, whereas none of the patients in the ensitrelvir groups experienced severe illness. 11 In the phase IIb part, only one patient in the ensitrelvir 250 mg group recorded a score of ⩾3 during the treatment and follow-up periods, whereas none of the patients in the ensitrelvir 125 mg or placebo group had severe illness (Shionogi & Co., Ltd., data on file). None of the patients in the phase III part required mechanical ventilation or died. Only one patient in the ensitrelvir 125 mg group and one in the placebo group experienced COVID-19-related hospitalization or equivalent recuperation during the study period. 10 The evaluation of severe illness development was not performed for the phase IIb/III part, which was conducted during the Omicron-predominant period. 14
Safety
Safety was assessed using treatment-emergent adverse events (TEAEs), treatment-related adverse events (AEs), and laboratory findings.10–12,14 The safety findings, including TEAEs and laboratory data, were consistent across the study components, and no notable safety concerns were identified. Most TEAEs were mild to moderate in severity.10–12,14 A decrease in high-density lipoprotein levels was the most frequently reported treatment-related AE in the ensitrelvir 125 mg group throughout the study components (Table 1).10–12,14 The phase IIa, phase IIb, and phase III parts observed transient and asymptomatic changes in high-density lipoprotein cholesterol, triglycerides, total bilirubin, and iron levels after treatment initiation with ensitrelvir, which resolved without additional treatment.10–12 Laboratory parameter changes were not assessed in the phase IIb/III part. 14
Findings of the phase IIa, phase IIb, phase III, and phase IIb/III parts: most common (⩾5% in any study group) TEAEs and treatment-related AEs.

AE, adverse event; TEAE, treatment-emergent adverse event.
Clinical studies of ensitrelvir in other patient populations
Additional clinical trials are currently underway to investigate the efficacy and safety of ensitrelvir in various patient populations. The global phase III SCORPIO-HR study (NCT05305547) was designed to assess the efficacy and safety of ensitrelvir initiated within 3 days of symptom onset. This study included adult patients with mild-to-moderate COVID-19 who were at standard or high risk for developing severe disease from both Asian and non-Asian countries. 24 According to the topline results, ensitrelvir showed a numerical but nonsignificant reduction in the time to sustained symptom resolution compared with placebo; of note, the SCORPIO-HR study used 15 common COVID-19-related symptoms as the primary endpoint, which are different from the symptoms used for the primary endpoint in the SCORPIO-SR study. The treatment demonstrated antiviral efficacy without new safety concerns. 25 In addition, the Strategies and Treatments for Respiratory Infections & Viral Emergencies study in hospitalized patients (NCT05605093), 26 a phase III study in children (jRCT2031230140), 27 and a phase III prophylaxis study (SCORPIO-PEP, jRCT2031230124) 28 are being conducted. Further clinical trial evidence is warranted to determine whether the efficacy and safety of ensitrelvir demonstrated in Asian patients with mild-to-moderate COVID-19 symptoms extend to other patient populations.
Real-world evidence
Postmarketing surveillance of ensitrelvir is currently underway in Japan as part of the legal and regulatory requirements in this country. According to the final analysis of 3760 patients, 25% of participants had risk factors for severe illness, and a majority (97.5%) had mild COVID-19 at baseline. More than 90% of the patients initiated ensitrelvir within 72 h of disease onset. The results showed a median time to fever resolution of 36.0 h and a median time to all symptom resolution of 156.0 h. Treatment-related AEs were observed in 271 patients (7.2%), and the most common events were diarrhea in 91 patients (2.4%), nausea in 43 patients (1.1%), and headache in 42 patients (1.1%). No additional safety concerns were identified.29,30
Additionally, research conducted in Japan assessed the effectiveness of ensitrelvir in routine clinical practice. One of the largest studies of this kind is a health insurance claims database study that retrospectively assessed the effectiveness of ensitrelvir in reducing the risk of hospitalization among outpatients aged ⩾18 years. 31 This study involved 5177 patients at high risk for severe illness who were treated with ensitrelvir and 162,133 patients at high risk who did not receive antiviral treatment. The evaluation period was set from 1 day after COVID-19 diagnosis (day 2) to day 28. After adjustment with the inverse probability of treatment weighting method, the incidence of all-cause hospitalization was significantly lower in patients treated with ensitrelvir than in patients without antiviral treatment (risk ratio, 0.629; 95% CI, 0.420–0.943). 31 In a single-center, retrospective chart review conducted in 32 patients hospitalized with SARS-CoV-2 infection, 32 most patients initiated ensitrelvir treatment after a failure of previous antiviral therapy with remdesivir, molnupiravir, casirivimab/imdevimab, or their combination. This study reported positive virological outcomes after ensitrelvir treatment. 32 Another single-center, retrospective, observational study compared the efficacy of remdesivir, molnupiravir, and ensitrelvir in 154 patients hospitalized with COVID-19. 33 This study reported lower SARS-CoV-2 antigen levels in patients treated with ensitrelvir than in patients receiving other antivirals, although the differences were not statistically significant. 33 The length of hospital stay was significantly shorter in the ensitrelvir group than in the molnupiravir group, but no significant differences were noted in the time to resolution of fever among the groups. 33
Ensitrelvir as a novel option for mild-to-moderate COVID-19 in diverse real-world settings
The results of this seamless phase II/III study demonstrated the efficacy of ensitrelvir without notable safety concerns.10–12,14 However, several points should be considered for its use in diverse real-world clinical scenarios.
First, as of August 2024, ensitrelvir is yet to be approved for clinical use in countries other than Japan and Singapore and thus is not part of the COVID-19 treatment recommendations outside these countries. In the current Japanese guidance for COVID-19 treatment, 34 ensitrelvir is recommended for patients with mild (percutaneous oxygen saturation (SpO2) ⩾96%) to moderate I (SpO2 >93% to <96%) COVID-19 irrespective of risk factors for severe illness (Table 2). In patients without risk factors, ensitrelvir can be considered when they have symptoms such as fever, sore throat, and cough that are severe. 34 However, the guidance also states that evidence to support the use of ensitrelvir in patients with risk factors for severe illness remains insufficient. 34 Although nearly 30% of patients involved in the phase III SCORPIO-SR trial had at least one risk factor, the most commonly observed factors were smoking, body mass index ⩾30 kg/m2, and dyslipidemia, with only <1% of patients being aged ⩾65 years. 10 Ensitrelvir is contraindicated for pregnant or possibly pregnant women, which is also applicable to molnupiravir (Table 2). Safe and effective treatment options for pregnant or possibly pregnant women without risk factors for severe illness are warranted.
Japanese recommendations on the use of oral antivirals for COVID-19 treatment in adult outpatients.

Source: The recommendations are the summary of the COVID-19 treatment guidance issued by the Ministry of Health, Labour and Welfare (version 10.1, April 2024). 34
COVID-19, coronavirus disease 2019; SpO2, percutaneous oxygen saturation.
Management of drug–drug interactions is crucial in COVID-19 antiviral treatment, especially for acutely ill patients and elderly individuals.35–37 For ensitrelvir, drug–drug interactions have been reported for CYP3A substrates in a healthy volunteer study. 38 Another in vitro and human study reported possible interactions between ensitrelvir and transporter substrates (P-glycoprotein (P-gp), breast cancer resistance protein (BCRP), organic anion transporting polypeptide (OATP) 1B1, and OATP1B3). 39 Of note, however, a nonclinical investigation suggested minimal involvement of P-gp and BCRP in drug oral absorption and a low risk of ensitrelvir for their inhibition in humans. 40 SARS-CoV-2 3CLpro mutations may be associated with reduced susceptibility to its inhibitors, such as ensitrelvir and nirmatrelvir. 41 Although the global prevalence of such mutations is currently limited to 0.5%, 41 further monitoring for the emergence of drug-resistant SARS-CoV-2 variants is crucial. 42
Finally, treatment costs and drug accessibility in low- and middle-income countries (LMICs) are additional potential barriers that may hinder the clinical use of ensitrelvir. 43 To facilitate access to COVID-19 therapeutics in LMICs, royalty-free licensing agreements have been concluded between the drug manufacturers and Medicines Patent Pool (MPP), a public health organization supported by the United Nations. 44 Ensitrelvir is a part of the MPP licensing agreements, and its agreement covers 117 LMICs worldwide. 44 This initiative is expected to expand patient access to novel COVID-19 therapeutics once they are approved for clinical use in these global regions.
Clinical efficacy of antivirals against COVID-19: What are the valid and meaningful endpoints in the next era?
Historically, clinical trials of antiviral treatment have employed hospitalization or death as the primary efficacy endpoint.5–8 However, the severity of COVID-19 has decreased worldwide because of the rollout of SARS-CoV-2 vaccination, host immunity evoked by a history of SARS-CoV-2 infection, and transition of SARS-CoV-2 to the less virulent Omicron subvariants. For example, the frequency of hospitalization among Japanese outpatients with COVID-19 without risk factors was 20.3% during the first wave (January 1–June 13, 2020), which decreased to 0.3% during the seventh wave (June 25–September 26, 2022). Mortality within a month from disease onset decreased from 0.06% during the second wave (June 14–October 9, 2020) to 0.004% during the seventh wave. 45 Analyses of the U.S. Veterans Affairs population hospitalized for COVID-19 showed no significant difference in the mortality risk before and during the epidemic of the JN.1 variant. The adjusted mortality rate at 30 days was 5.46% and 5.82% before and during the JN.1-predominant period, respectively (adjusted hazard ratio, 1.07; 95% CI, 0.89–1.28). 46 Such a decrease in COVID-19 severity indicates that hospitalization or death may not be the best endpoints in evaluating the efficacy of antiviral treatment under these circumstances.
In response to the reduced COVID-19 severity, symptom recovery or improvement serves as a new efficacy endpoint in the clinical research of antivirals. Indeed, recent industry-sponsored clinical trials employed symptom-based assessments as the primary endpoint for novel COVID-19 therapeutics.47–50 The seamless phase II/III study of ensitrelvir also employed symptom-based endpoints. A questionnaire developed based on the U.S. FDA guidance 23 was used to define mild-to-moderate COVID-19 symptoms or asymptomatic SARS-CoV-2 infection and assess the clinical efficacy of the drug.10–14 Similarly, a phase II–III study of simnotrelvir plus ritonavir 51 and a phase III study of leritrelvir, 52 both of which have been conducted in China, used recovery from the 11 COVID-19–related symptoms based on the U.S. FDA guidance (stuffy or runny nose, sore throat, shortness of breath, cough, muscle or body aches, headache, chills, fever, nausea, vomiting, and diarrhea) as the primary endpoint. Both studies demonstrated the efficacy of the intervention in adult patients with mild-to-moderate COVID-19 without notable safety concerns.51,52 A secondary and post hoc analysis of the phase III component of the MOVe-OUT study also employed a patient diary developed based on the 2020 U.S. FDA guidance. This analysis reported symptom improvement with molnupiravir treatment in at-risk, unvaccinated outpatients with mild-to-moderate COVID-19. 53 In contrast, the phase II–III EPIC-SR (Evaluation of Protease Inhibition for COVID-19 in Standard-Risk Patients) study of nirmatrelvir, which also used patient-reported symptoms for efficacy among adult outpatients, did not show a significant difference in the time to sustained alleviation of all signs and symptoms of COVID-19 between the nirmatrelvir and placebo groups. 54
Post COVID-19 condition (PCC), or Long COVID, is another COVID-19–associated public health concern that equally affects persons living in different global regions. 55 In an exploratory analysis of the phase III SCORPIO-SR study, ensitrelvir treatment in the acute COVID-19 phase showed a trend toward risk reduction for PCC symptoms compared with placebo. 56 However, because of the exploratory nature of the results, the effect of ensitrelvir in preventing PCC warrants further investigation. Currently, the definition of PCC and methods to assess the risk of PCC vary among published studies, which may have led to mixed results regarding the efficacy of anti-SARS-CoV-2 antivirals.57–60 Further research will help fully establish the study design and endpoints for PCC assessments.
Conclusion
In this review, we summarized preclinical, clinical, and real-world data on ensitrelvir. Overall, the results support the efficacy of this antiviral treatment in patients with mild-to-moderate COVID-19 without notable safety concerns. Further research is necessary to establish the clinical efficacy and safety of ensitrelvir in a wider patient population.
