Abstract
Background:
Despite modern imaging methods, a long symptom-to-diagnosis interval can be observed in patients with primary brain tumors.
Objective:
The study evaluated the initial and subsequent clinical presentation of patients with brain tumors in the context of time to diagnosis, localization, histology, and tumor grading.
Methods:
In this retrospective analysis of 85 consecutive patients with primary brain tumors, we assessed the presenting symptoms and signs. The analyses were based on entries from medical records at the Department of Neurology of Zurich University Hospital between 2005 and 2010.
Results:
A total of 54 men and 31 women with a mean age at diagnosis of 48 years were included. 60% of the patients present with a malignant tumor (World Health Organization grading III–IV), 24.7% with a benign tumor (I–II), and 15.3% were not classified. The interval between symptom onset and diagnosis varied from 1 day to 96 months (median: 39 days). High-grade tumors (III–IV) were diagnosed significantly earlier than low-grade tumors (II) after the first symptoms occurred (median: 26 vs. 138 days; z = −3.847, p < 0.001).
Conclusions:
Symptoms with a short symptom-to-diagnosis interval such as nausea/vomiting, seizures, as well as of personality change are assumed to contribute to a faster diagnosis in high-grade tumors. Visual disturbances and headaches, although occurring relatively seldom, did not contribute to a decrease in time to diagnosis and should therefore be considered for further diagnostic workup.
Introduction
Patients with primary brain tumors can present with focal or generalized symptoms and/or clinical findings. At an initial stage, especially in low-grade tumors, most symptoms are focal depending on the localization of the lesion. More generalized symptoms including headache, nausea, and vomiting develop when tumor size increases as hints for increased intracranial pressure. 1 However, some symptoms such as headaches, visual disturbances, and epileptic seizures can also be either a focal or a generalized sign. While the diagnostic guidelines for a first seizure suggest brain imaging, this cannot be recommended for headaches in general, but only for patients with “red flags” for secondary headache (thunderclap headaches, chronic headaches, neurological signs, general symptoms (e.g. fever or weakness), changes in the characteristics of a preexisting headache disorder, development of a new headache after the age of 50 years, the criteria for migraine or tension-type headache are not fulfilled, trigemino-autonomic cephalalgias, and headaches from the group of other primary headache disorders). 2– 4 In addition, the SNOOP criteria especially addressed red flags that require imaging and the SNNOOP10 list summarized orange and red flags with a need for a diagnostic workup. 2,5
Headache is frequent and can challenge primary care physicians but is even more challenging in the emergency setting. While prevalence estimates of secondary causes vary between 2% and 22%, depending on the setting, 6,7 most causes are not dangerous or life-threatening (e.g. half of the patients have a medication overuse headache). 8,9
Several studies on initial brain tumor symptoms in children and adolescents with brain tumors have been published. 10,11 In adults, one study reported the prevalence of symptoms preceding malignant cerebral gliomas. 12 Davies and Clarke reported in their interviews of treated patients with malignant cerebral gliomas that the clinical symptoms observed by the patients and their relatives differ compared to the medical records: Patients and relatives reported more personality changes (30 vs. 15%), cognitive loss (46 vs. 33%), and fatigue (48 vs. 14%). 12 However, the prevalence of the most common symptoms at diagnosis, weakness (60%), headaches (53%), and epilepsy (38%), did not differ between the records and the patients/relatives. They further reported a delay in the health-care management presumably due to these subtle clinical changes often only reported from the relatives. In contrast, at the emergency department, focal signs (60%) were observed more often than mental status alteration (25%), headaches (15%), and seizures (14%). 13 Here, the prevalence rates were 46% for glial tumors, 22% for meningeomas, and 17% for brain metastases.
While some brain tumors are diagnosed quickly, 14 the symptom-to-diagnosis period is at least 14 weeks in most studies. 15– 18 An investigation of medulloblastomas in children found that the median time to diagnosis was 65 days; these times have not been reduced in the Western world despite the development of modern imaging methods. 10 This underlines the difficulty to diagnose primary brain tumors after symptom onset within a reasonable period.
In order to determine clinically relevant symptoms and to understand their relation with different factors such as dignity and localization, this study retrospectively assessed initial clinical presentation and further symptoms with respect to time to diagnosis, histology, and tumor grading.
Methods
We performed a retrospective analysis of patients with brain tumors based on entries from medical records. Information required to identify initial symptoms and findings was collected and anonymized for further analysis.
Patients and definitions
The study included all consecutive patients with primary brain tumors who were treated at the Department of Neurology of the Zurich University Hospital between January 2005 and December 2010.
The following variables were taken from the charts: age at the time of the diagnosis, sex, tumor type (according to the World Health Organization (WHO) classification of 2007 19 ), histology, WHO tumor grading, localization, all initial presenting symptoms, subsequent clinical findings, as well as the symptom-to-diagnosis interval. All symptoms were classified accordingly: Sensory disturbances included symptoms such as paresthesia and deafness, visual disturbances included diplopia, blurred vision, and visual field deficits; the causes of such symptoms were cranial nerve paralysis, optic nerve atrophy due to papilledema, or disruption of the visual pathways. Nausea and/or vomiting not related to food intake. Speech problems included all types of aphasia. Paresis ranging from minimal paresis of a single limb to complete hemiparesis. Symptoms were further subdivided into focal symptoms, symptoms of increased intracranial pressure (headaches and nausea/vomiting), and into seizures (here considered as either a focal or a generalized sign). They were separated into “first presenting sign/symptom” and further “symptoms developing over time” before diagnosis. In addition, the interval from the first symptom to diagnosis was analyzed for low- and high-grade tumors separately (grade II vs. III–IV).
Ethical approval
The study was approved by the Zurich Cantonal Ethics Commission (KEK-ZH no. 2011-0427) and Zurich University Hospital management; informed consent was not required owing to retrospective design of the study.
Statistics
SPSS 20.0 (IBM Corp, Armonk, New York, USA) was used for statistical analyses. Normal distribution was calculated by using the Kolmogorov–Smirnov test. Since the parameter symptom-to-diagnosis interval did not show a normal distribution, nonparametric testing was performed; mean and interquartile range (IQR) was given; for values with normal distribution, mean and standard deviation were given; a p-value >0.05 was considered significant.
Results
Patient characteristics
The data of 85 patients (54 male and 31 female) were recorded. The average age at diagnosis was 48 ± 16 years and ranged from 6 years to 79 years. A biopsy with subsequent histological diagnosis was performed in 82 patients. A WHO grading was obtained in 72 patients.
Tumor characteristics
A total of 60% patients presented with a malignant tumor (WHO III–IV), 24.7% with a benign tumor (WHO I–II), and 15.3% were not classified. Out of the classified tumors, astrocytic tumors were most commonly diagnosed, with glioblastoma accounting for the largest proportion (42.4%) (Table 1). Further astrocytic tumors diagnosed included anaplastic astrocytoma (grade III, 9.4%), diffuse astrocytoma (grade II, 7.1%), and pilocytic astrocytoma (grade I, 2.4%). Oligodendroglial tumors were the second most common overall and included oligodendrogliomas (8.2%) and their anaplastic variants (4.7%). Embryonic tumors such as medulloblastomas comprised only a small proportion (4.7%).
Frequency of all brain tumors from the neuro-oncological outpatient clinic seen between 2005 and 2010.

Initial symptoms and subsequent clinical findings
In total, 165 signs and symptoms were found in the charts; of 51% (84) were present at presentation and 49% (81) occurred in the time until the diagnosis was made.
The initial symptoms and subsequent clinical findings are listed in descending order of their frequency in Figure 1 and with respect to the tumor type in Figure 2. Epileptic seizures were the most common initial symptom (33%), followed by headaches (19%), sensory disturbances (10%), cognitive impairment (10%), ataxia (6%), visual disturbances (6%), and nausea/vomiting (5%). During the time course until diagnosis the most common symptoms were headaches (28%), cognitive loss/personality change (28%), sensory disturbances (27%), and nausea/vomiting (25%), followed by epileptic seizures (20%), visual disturbances (18%), ataxia (17%), fatigue (15%), aphasia (7%), motor weakness (6%), and appetite loss (4%).

Frequency of initial and subsequent signs and/or symptoms of all brain tumors.
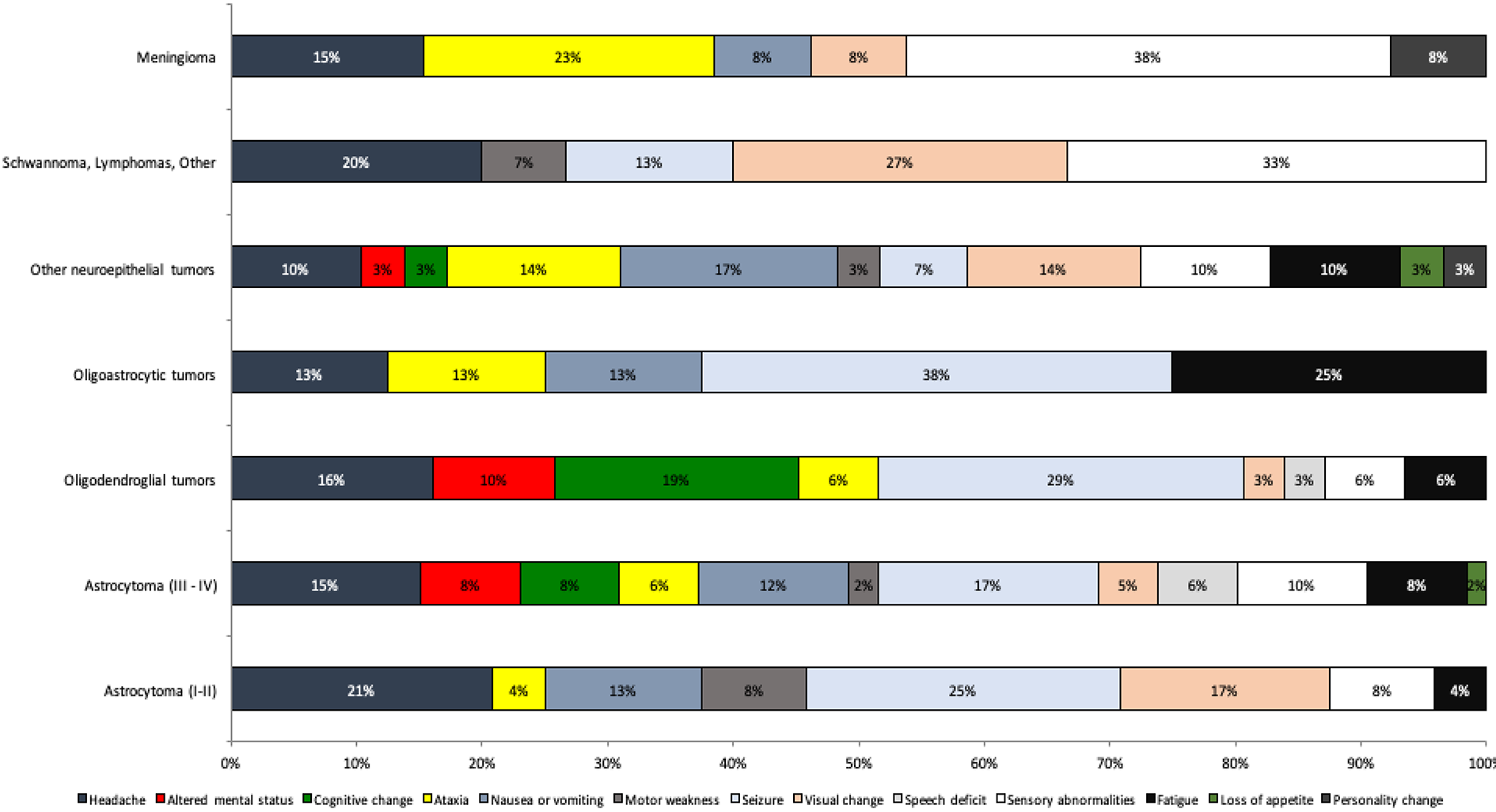
Initial and subsequent signs and/or symptoms with respect to tumor type.
Astrocytic tumors (42.4%)
Symptomatic epilepsy was the most common manifestation among patients with diffuse astrocytomas. Anaplastic astrocytomas and glioblastomas also presented often with seizures. Headaches and personality changes, as well as changes in cognition and mental status occurred frequently.
Oligodendrogliomas and mixed gliomas (12.9%)
Oligodendrogliomas and oligoastrocytomas (II) and their anaplastic variants (III) likewise usually presented in conjunction with epileptic seizures. Fatigue was also common symptom in patients with oligodendrogliomas. However, this was rarely the case in anaplastic variants. Mixed glioma patients often complained of headache (23%).
Embryonic tumors (4.7%)
Headaches (22%) and vomiting/nausea (20%) were very common in the present population.
Meningiomas (4.7%)
Epileptic seizures were the most common symptom affecting meningioma patients (35%). Motor dysfunctions, aphasia, anosmia, visual disturbances, partial seizures, and paresis were other commonly observed symptoms.
Symptom-to-diagnosis interval
We calculated the symptom-to-diagnosis interval in 82 patients, which ranged between 1 day and 96 months with a median of 39 days (IQR, 126 days). The interval to diagnosis was shortest for nausea/vomiting (11 days, N = 4), followed by cognitive loss (24.5 days, N = 8), seizures (33 days, N = 27), ataxia (37 days, N = 5), sensory disturbances (40 days, N = 8), and headaches (59 days, N = 16). Visual disturbances led to the longest intervals (132 days, N = 5). Aphasia, paresis, fatigue, and personality changes occurred only seldom as first symptoms (N = 3 = 2 = 1 = 1).
Symptom-to-diagnosis interval—Differences between low- and high-grade tumors
Tumor grading was obtained in 72 patients. Grade I tumors present with the longest symptom-to-diagnosis interval (N = 6) with a mean of 232 days followed by grade II tumors with 138 days (N = 15). The symptom-to-diagnosis intervals of grade III (N = 14) and grade IV (N = 37) were similar (median: 26.5 vs. 26 days; z = −0.443, p = 0.657). Grade III and/or grade IV were diagnosed faster than grade II tumors (median: 26 vs. 138 days; z = −3.847, p < 0.001).
Nausea/vomiting occurred more often initially (8 vs. 0%) in the group of high-grade tumors, while headaches (16 vs. 24%) and seizures (31 vs. 38%) were observed less often as initial symptom in high-grade tumors compared to their low-grade counterparts (I–II).
Until diagnosis, the group of high-grade tumors showed similar frequencies in headaches (28 vs. 33%) and nausea/vomiting (24 vs. 24%), fatigue (18 vs. 19%), and paresis (6 vs. 5%) compared to low-grade tumors. However, seizures occurred with higher frequency (28 vs. 14%) in high-grade tumors. Also, aphasia (12 vs. 0%), personality change (22 vs. 14%), and appetite loss (6 vs. 0%) were more frequent. Cognitive loss (10 vs. 14%), ataxia (12 vs. 29%), visual disturbances (12 vs. 29%), and sensory abnormalities (18 vs. 29%) were more frequent in low-grade tumors.
The frequency of symptoms did not differ between the groups considering the different group sizes (high vs. low grade (51 vs. 21 patients); 95 vs. 41 symptoms). When symptoms of increased intracranial pressure, focal signs, and seizures were compared for the different tumor gradings at presentation, data showed a similar distribution: intracranial pressure 24 in high grade versus 24% in low grade, focal sign 45 versus 38%, and seizure 31 versus 38%. However, when all grades were considered separately, 67% of grading I tumors showed focal signs (N = 6).
Symptoms with respect to localization
Of all tumors 21.2% were localized frontally, 17.6% were temporal, 10.6% were parieto-occipital, and 12.9% were localized in the brainstem and/or cerebellum (Figure 3). The majority of tumors were in the right brain hemisphere (49.4%); 35.3% were left-sided while 15.3% were bilateral. Low-grade tumors (N = 21) were localized more often in the brainstem/cerebellum (24%), and frontally (19%), whereas high-grade tumors (N = 51) were localized preferentially frontal (24%), temporal (20%), and parieto-occipital (16%). Brainstem tumors (N = 11) rather were associated with headache (27%), frontal tumors (N = 18) with seizures (44%) and headache (22%), and temporal tumors (N = 12) mainly were associated with seizures (50%).
Distribution of tumor localizations.
Headaches
In the 16 patients with headaches as a first symptom (19%), the main localizations were brainstem/cerebellum (19%), frontal (25%), and suprasellar (13%). In the majority of the cases, it was a malignant tumor (III–IV, 56%).
A similar picture was observed when all patients (N = 24) were considered that develop headaches over time until diagnosis (brainstem/cerebellum (17%), frontal (17%), and suprasellar (21%)). Again, most had a high-grade astrocytomas (III–IV, 42%), followed by oligodendroglial tumors (17%), low-grade astrocytomas (I–II, 13%), and meningeomas (13%).
Discussion
The present data showed the frequency of initial and subsequent symptoms of primary brain tumors until diagnosis in relation with time to diagnosis and tumor pathology in all patients treated at the Department of Neurology at the University Hospital in Zurich from 2005 to 2010.
It showed the preferred localizations (frontal or temporal with a preference of the right hemisphere (presumably due to broader therapeutic options)) as well as the tumor grading with respect to the clinical symptoms until diagnosis. A relatively high prevalence of malignant mainly astrocytic tumors of 60% can be explained by the specialized center. Headaches were not seen frequently as first symptom (19%) but often associated with malignant tumors with frontal, brainstem/cerebellar, and suprasellar localization. In general, frontal and temporal tumors in the present data are/were mostly associated with seizures, whereas headaches are/were more frequent in brainstem/cerebellar tumors.
An interesting finding was the dependence of the diagnostic interval on the grading of the brain tumor, and furthermore the differences in clinical symptoms and localizations between benign and malignant tumors. The occurrence of nausea/vomiting with a short symptom-to-diagnosis interval only in high-grade tumors explains part of the variance. Additionally, the higher frequency of seizures as well as of personality change—again a symptom with a short symptom-to-diagnosis interval—contributed to a faster diagnosis in high-grade tumors. This is maybe explained by the fact that also relatives observe such symptoms more frequently. 12 On the other hand, visual disturbances that occur frequently in low-grade tumors were rather associated with a later diagnosis. The occurrence of headaches did not differ between low- and high-grade tumors leading rather to a delay of final diagnosis. Since these factors explain only part of the variance, we suggest that also symptom severity as well as urgency for intervention contribute to a shorter symptom-to-diagnosis interval in high-grade tumors.
In addition, subtle personality or cognitive changes as well as fatigue should be considered as primary symptom, since these symptoms were often not mentioned in the medical records but in interviews of patients and caregivers. 12 A psychological study showed that subtle changes were often misinterpreted by the patients and that a faster decision-making was rather obtained within a social context. 20 The relatively long time to diagnosis interval in brain tumors should lead to a reconsideration of certain symptoms, especially of visual disturbances. Visual disturbances especially of blurred and/or double vision might be an indicator for increased intracranial pressure and should therefore lead a more intense diagnostic workup.
Headaches were observed with a relatively low prevalence of 19% when compared to the study of Davis and Clarke that reported a prevalence of 53%. 12 It indicates rather a malignant brain tumor situated frontal or in the brainstem/cerebellum. Compared to seizures and nausea, the time to diagnosis interval remains relatively long for headaches as first symptom (59 days) presumably due to the unspecific character of this symptom as shown previously. One recent study assessed the preceding clinical symptoms but the time-to-see a specialist without considering tumor type, as well as the time to diagnosis. 21 It showed that headache only (not together with further symptoms) or memory complaints led to a considerable delay in the time-to-see a specialist, which has also been suggested by the general practitioner. The delay of the diagnosis for headaches can be further explained by a similar presentation as of primary headaches (mostly tension-type headaches 22 ) in patients with a predisposition and that unique brain tumor headache (localized, progressive, worse in the morning, and aggravated by coughing and bending forward (see IHS definition 23 )) occurs only seldom. 24 A detailed headache history is missing and therefore we cannot conclude which “red flags” for symptomatic headaches occurred and whether they were considered for further diagnostics. The relatively low prevalence of headaches in primary brain tumors as a first symptom together with the high prevalence of primary headaches in the community suggests to perform a brain imaging only in patients with a red flag for a symptomatic headache and/or with additional neurological sometimes only subtle cognitive symptoms. Studies support this procedure since the frequency of pathological findings in brain imaging was similar in typical migraine headache compared to the general population (0.2–0.4%) but higher in patients with unclassified headaches and/or neurological symptoms. 25,26 By assessing red flags summarized in the SNOOP criteria, a secondary headache disorder that requires further imaging can be detected (SNOOP criteria: S: systemic symptoms and secondary risk factors; N: neurological symptoms; O: abrupt onset, thunderclap headache, age over 50 years; P: positional headaches, changing of prior headaches, papilledema, precipitated by coughing). 2 In addition, they propose imaging in trigemino-autonomic cephalalgias in general, and in migraine when pathological neurological findings or an atypical aura occur. In traumatic headaches, imaging should be performed if the headache mechanism is unclear or the neurological exam is abnormal.
Limitations
One limitation of the study is that it does not distinguish between visual symptoms caused by increased intracranial pressure and visual symptoms due to an affection of the visual pathway. Furthermore, headache description, symptom severity and the kind of seizure (focal vs. generalized), was mostly missing in the charts. A further limitation of a retrospective study is that it was not possible to analyze if all the presenting symptoms were causal and not coincidental. However, the population of the sample was typical for a specialized tertiary center but without the consideration of secondary brain tumors. Since the patients recordings refer to the period between 2005 and 2010, the tumor types refer to the WHO classification of 2007 19 and not to the recent one from 2016. 27
Conclusion
In times with medico-economic pressure not every new symptom may be investigated with a brain scan. Thorough history taking and clinical examination are not only important for neurologists but also for general practitioners. With a focus on typical presenting symptoms of primary brain tumors such as epileptic seizures and focal signs, time to diagnosis may be shortened. Visual symptoms require attention as they may suggest increased intracerebral pressure. Red flags for headaches as well as symptoms of secondary headaches and brain tumors (i.e. the SNOOP/SNNOOP10 criteria) should be considered for further diagnostic workup.
Footnotes
Acknowledgments
In memoriam: The co-authors, colleagues, and patients thank Benedict Alther for his great contribution to the project and his care for his patients.
Author contributions
MW and ARG researched literature and conceived the study. BA, ARG, and MW were involved in protocol development, gaining ethical approval, data recruitment, and data analysis. VM was involved in data analyses and wrote the first draft of the manuscript. All authors but BA reviewed and edited the manuscript and approved the final version of the manuscript.
Availability of data
The data sets generated and/or analyzed during the current study are available from the corresponding author by request from a qualified researcher.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
