Abstract
Lactate has been shown to have beneficial effect both in experimental ischemia–reperfusion models and in human acute brain injury patients. To further investigate lactate’s neuroprotective action in experimental in vivo ischemic stroke models prior to its use in clinics, we tested (1) the outcome of lactate administration on permanent ischemia and (2) its compatibility with the only currently approved drug for the treatment of acute ischemic stroke, recombinant tissue plasminogen activator (rtPA), after ischemia–reperfusion. We intravenously injected mice with 1 µmol/g sodium
Introduction
Acute ischemic stroke, the most frequent type of stroke, happens when blood supply to the brain is blocked. The primary therapeutic strategy is timely recanalization of the initial occlusion, preserving maximal brain functionality. The two currently approved strategies for recanalization are thrombolysis with recombinant tissue plasminogen activator (rtPA), the only approved drug for acute ischemic stroke treatment and a typical time window of 4.5 h, 1 and mechanical thrombectomy, which allows substantial reperfusion when performed within 7.3 h from ischemia onset. 2 Recent trials have provided evidence that careful patient selection based on imaging properties of the ischemic brain leads to effective reperfusion treatment in extended time windows (9 h for thrombolysis 3 and 24 h for thrombectomy 4 ) and unknown-onset stroke patients. 5,6 Nevertheless, rtPA remains the standard of care as, even if thrombectomy is considered, rtPA-eligible patients should receive the drug if the inclusion criteria are fulfilled. 7
The monocarboxylate lactate is obtained from glucose through glycolysis and is a source of metabolic energy as well as an important signaling molecule involved in a plethora of physiological and pathological conditions (for review, see in the literature
8,9
). Evidence from in vitro and in vivo studies has shown that the healthy brain uses lactate as an efficient energy substrate that may even be preferred to glucose to maintain synaptic transmission.
10
–12
Interestingly, lactate may also be the preferred energy substrate for recovery of neuronal function after insults like prolonged oxygen deprivation
13,14
such as in stroke, which likely plays a role in the brain’s attempt at self-healing. We and others have shown that
Further exploring the potential field of application of lactate prior to clinical testing, we investigated its effect in a permanent ischemia model, a situation with severe and permanent blood flow reduction, representing patients who cannot benefit from acute recanalization. Additionally, we examined whether lactate-induced neuroprotection is affected by the concomitant presence of rtPA in a model of ischemia–reperfusion. We tested the combined treatment in a suture-induced middle cerebral artery occlusion (MCAO) model with promptly restored blood flow. This model, previously used to test other neuroprotectants, 23,24 mimics the situation of patients who underwent thrombolysis with successful return of cerebral blood flow and who are eligible for potential neuroprotective therapy in the presence of residual rtPA, a situation that will be increasingly likely to happen.
Methods
Permanent MCAO (pMCAO)
Male C57BL/6J mice (19–30 g, Charles River, France) were housed under standard conditions with unlimited access to food and water. Mice were anesthetized and kept in 1.5–2% isoflurane in 30% oxygen - 70% nitrous oxide. Regional cerebral blood flow (rCBF) was monitored throughout surgery by laser-Doppler flowmetry (Perimed, Sweden) with a flexible probe fixed on the skull. Rectal temperature was maintained at 37 ± 0.5°C. Permanent ischemia was induced by inserting a silicone-coated filament (Doccol Corp., Sharon, MA, USA) through the left common carotid artery into the internal carotid.
25
Mice with successful ischemia (rCBF < 20% of baseline, stable for 15 min after induction) received, following randomization, a tail vein injection of 1 µmol/g sodium
Transient MCAO (tMCAO)
Male C57BL/6J mice (18–28 g, Charles River, France) were subjected to 35 min transient ischemia using the filament technique described above. Mice with satisfactory ischemia (rCBF < 20% of baseline) and reperfusion (rCBF > 50% of baseline) received, following randomization, a single intravenous (iv) injection of vehicle (5 µL/g Phosphate Buffered Saline (PBS), pH 7.4), single intracerebroventricular (icv) injection (0.9 mm laterally, 0.1 mm posteriorly, 3.1 mm deep from bregma
26
) of 2 µL of 100 mmol/L sodium
Ischemic lesion volume determination
Twenty-four hour after pMCAO induction, mice were euthanized and 2-mm thick fresh coronal brain slices were cut on a brain matrix, stained with 2% of 2,3,5-triphenyltetrazolium chloride for 15 min at room temperature in the dark and stored overnight at 4°C in paraformaldehyde 4%. Stained sections were scanned (Epson Perfection V500, Epson Deutschland Gmbh, Germany) and evaluated by a blinded examiner using ImageJ (NIH, Bethesda, MD, USA). Unstained areas (pale color) were defined as ischemic lesions.
Forty-eight hours after tMCAO onset, mice were euthanized and brains were snap frozen in liquid nitrogen vapor. Cresyl violet-stained coronal cryostat sections (20-µm thick, 720-µm apart) were imaged under a light stereomicroscope (Nikon SMZ25, Nikon Instruments Europe BV, Netherlands). An examiner blinded to the treatment group determined lesion areas using ImageJ.
Direct infarct volume was calculated by multiplying the sum of the infarct areas on each section by the spacing distance. To avoid bias due to edema, “indirect” lesion size [contralateral volume − (ipsilateral volume − direct infarct volume)] 27 was calculated. Cerebral hemisphere swelling was measured as [(ipsilateral volume − contralateral volume)–volume contralateral].
Behavioral assessment
Neurological deficit was assessed immediately after surgery, 24 h after tMCAO or pMCAO, and 48 h after tMCAO and graded for severity by a blinded examiner using the neuroscore (0 = no observable deficit, 1 = failure to extend the forepaw, 2 = circling, 3 = loss of circling or righting reflex). 26 Mice undergoing tMCAO were subjected to the wire-hanging test, where they are trained to suspend their body by holding onto a single wire stretched between two posts 60 cm above a soft ground. 28 Mice were trained 1 day before intervention and scored for escaping (reaching the posts) and/or falling events at baseline, 24 h after tMCAO, and before euthanasia (48 h).
Metabolic evaluation of permanent ischemia
The overall metabolic responses in the ischemic core following an identical pMCAO procedure have been reported in a proton magnetic resonance (1H-MRS) study from our group. 25 In brief, metabolites including acetate, alanine, ascorbate, aspartate, creatine, γ-aminobutyric acid, glucose, glutamine, glutamate, glycine, glutathione, glycerylphosphorylcholine, lactate, macromolecule, myo-inositol, N-acetyl-aspartate, N-acetylaspartyl-glutamate, phosphorylcholine, phosphocreatine, phosphorylethanolamine and taurine were measured using high-field 1H-MRS and quantified referencing to the endogenous water.
Metabolic results from the two time points at which we applied lactate treatment in the present study, that is, 1 h (n = 5) and 3 h (n = 7) post-pMCAO, and results of sham-operated animals (n = 8) were taken from the aforementioned study and analyzed. 25
Statistical analysis
Comparisons between continuous variables were done with one-way analysis of variance (ANOVA; Gaussian distributions), Kruskal–Wallis (non-Gaussian distributions), or two-way ANOVA (analysis across time) tests. Comparisons between categorical variables were done with the chi-square (χ2) test. Statistical tests were done on GraphPad Prism 6.0 (GraphPad Software, San Diego, CA, USA). The line on the box-and-whisker plots shows the median and the whiskers correspond to maximum and minimum values. Values reported in Table 1 are mean ± SD. Significance was considered at p < 0.05.
Lesion size measurements.

pMCAO: permanent middle cerebral artery occlusion; rtPA: recombinant tissue plasminogen activator; tMCAO: transient middle cerebral artery occlusion.
ap < 0.05 compared to control.
bp < 0.05 compared to rtPA.
Partial least squares discriminant analysis (PLS-DA) has been shown as a valuable exploratory tool that allows separation of a data set into different groups. Using metabolomic data provides insight into the significant metabolites contributing to the causes of discrimination via weights and loadings. 29–31 The PLS-DA of the neurochemical profiles was performed with MetaboAnalyst. 30
Results
We first tested the effects of lactate administered after pMCAO (Figure 1). Mice injected intravenously with 1 µmol/g of lactate 1 h after pMCAO induction showed smaller direct and indirect lesions than control mice (Figure 1(a) and (b); Table 1) as well as better functional outcome, measured by neuroscore (Figure 1(d)). Mice treated at the later time point (3 h) did not show significant differences compared to controls. Lactate treatment did not affect brain swelling (Figure 1(c); Table 1).

Effect of lactate administration after pMCAO. (a) TTC staining of brains from mice treated with lactate (1 µmol/g, iv injection) 1 h or 3 h post-induction of pMCAO. The brains were obtained 24 h after pMCAO induction. Pale color shows damaged tissue, whereas bold color shows healthy tissue. (b) Lesion volume measurements show the direct lesion volume (left) and the indirect lesion volume (right). One-way ANOVA with Tukey’s post hoc test. (c) Total brain swelling 24 h after pMCAO in control mice and mice treated with lactate 1 h or 3 h after pMCAO induction. One-way ANOVA with Tukey’s post hoc test. (d) Neurological deficit scores (0 = no deficit, 1 = failure to extend right forepaw, 2 = circling, 2.5 = circling with partial loss of righting reflex, 3 = complete loss of righting reflex). χ2 test. Filled dots represent individual animals. *p < 0.05; **p < 0.01; ***p < 0.001. TTC: 2, 3, 5-triphenyltetrazolium chloride; iv: intravenous; pMCAO: permanent middle cerebral artery occlusion; ANOVA: analysis of variance; Lac: lactate.
We investigated brain metabolite concentrations of mice subjected to 1 h or 3 h of permanent ischemia by 1H-MRS using data taken from our previously published data. 25 Overall, analysis showed that the metabolic profile evolves with time, in particular between 1 h and 3 h. Whereas energy-related substrates like glucose or energy buffers like phosphocreatine show a decrease after permanent ischemia, a strong increase is observed in endogenous lactate that shows significantly different levels between 1 h and 3 h after permanent ischemia (Supplementary Figure 1). On the other hand, there is a time-dependent decrease in glutamine and in the excitatory neurotransmitter glutamate concentrations, while the inhibitory neurotransmitters γ-aminobutyric acid and glycine increase, with higher levels at 3 h. There is a strong increase in acetate 3 h after pMCAO that is practically undetected in sham and at 1 h, as well as a near-depletion of glutathione at 3 h (no change at 1 h), reflecting either a greater vulnerability or a higher exposure to reactive oxygen species. Further analysis of the neurochemical profiles of sham-operated, 1 h post-pMCAO, and 3 h post-pMCAO mice using the PLS-DA showed three clearly distinct profiles with the two different ischemia time points both well separated from control animals (Figure 2(a)). Further evaluation of the corresponding weights and loadings of each neurochemical component (i.e. loading factors; Figure 2(b)) revealed substantial contributions of the selected metabolites described above to the final PLS-DA separations.

PLS-DA of pMCAO neurochemical profiles. PLS-DA scores of the overall neurochemical profiles (a) and selected metabolites (c) of sham-operated mice (black dots), mice 1 h after pMCAO start (green diamonds), and 3 h after pMCAO start (red squares). Mice 1 h after pMCAO are clearly separated from 3 h after pMCAO and sham-operated mice both using the overall neurochemical profile (a) as well as selected metabolites (c). The corresponding loading factors (b and d) of PC1 (black triangles) and PC2 (black squares) show the contribution of each metabolite to the final separation of the three groups. All metabolic data are taken from Berthet et al., 25 which are summarized in Supplementary Figure 1. Ace: acetate; Ala: alanine; Asc: ascorbate; Asp: aspartate; Cr: creatine; myo-Ins: myo-inositol; GABA: γ-aminobutyric acid; Glc: glucose; Gln: glutamine; Glu: glutamate; Gly: glycine; GPC: glycerophosphocholine; GSH: glutathione; Lac: lactate; Mac: macromolecule; NAA: N-acetyl-aspartate; NAAG: N-acetyl-aspartyl-glutamate; PCho: phosphocholine; PCr: phosphocreatine; PE: phosphorylethanolamine; Tau: taurine; PLS-DA: partial least squares discriminant analysis; pMCAO: permanent middle cerebral artery occlusion; PC1: principal component 1; PC2: principal component 2.
We also tested the compatibility between lactate and rtPA in an ischemia-reperfusion model. Lesion volume quantification (Figure 3(b); Table 1) showed a significant decrease in the direct lesion size of lactate-treated mice compared to rtPA-treated mice with no significant difference for the combined treatment. No significant reduction was observed in the indirect infarct volume. Brain swelling assessment (Figure 3(c); Table 1) showed differences between lactate-treated and rtPA-treated mice, with a trend toward smaller swelling for the combined treatment. As posterior brain structures seemed be damaged to a lesser extent when animals were treated with lactate (Figure 3(a)), we analyzed the lesion areas on individual coronal sections (Figure 3(d)). The analysis showed significant lesion size reduction on posterior slices in lactate-treated animals compared to PBS- and rtPA-treated mice. The lesion size of rtPA+lactate-treated animals falls in between the larger lesions of control and rtPA-treated mice and those of lactate-treated mice (Figure 3(d)). Analysis of the different brain areas (Figure 3(e) to (h)) showed smaller corticostriatal lesions in lactate-treated mice than in rtPA-treated mice, whereas no significant changes were observed in the hippocampus or the midbrain.
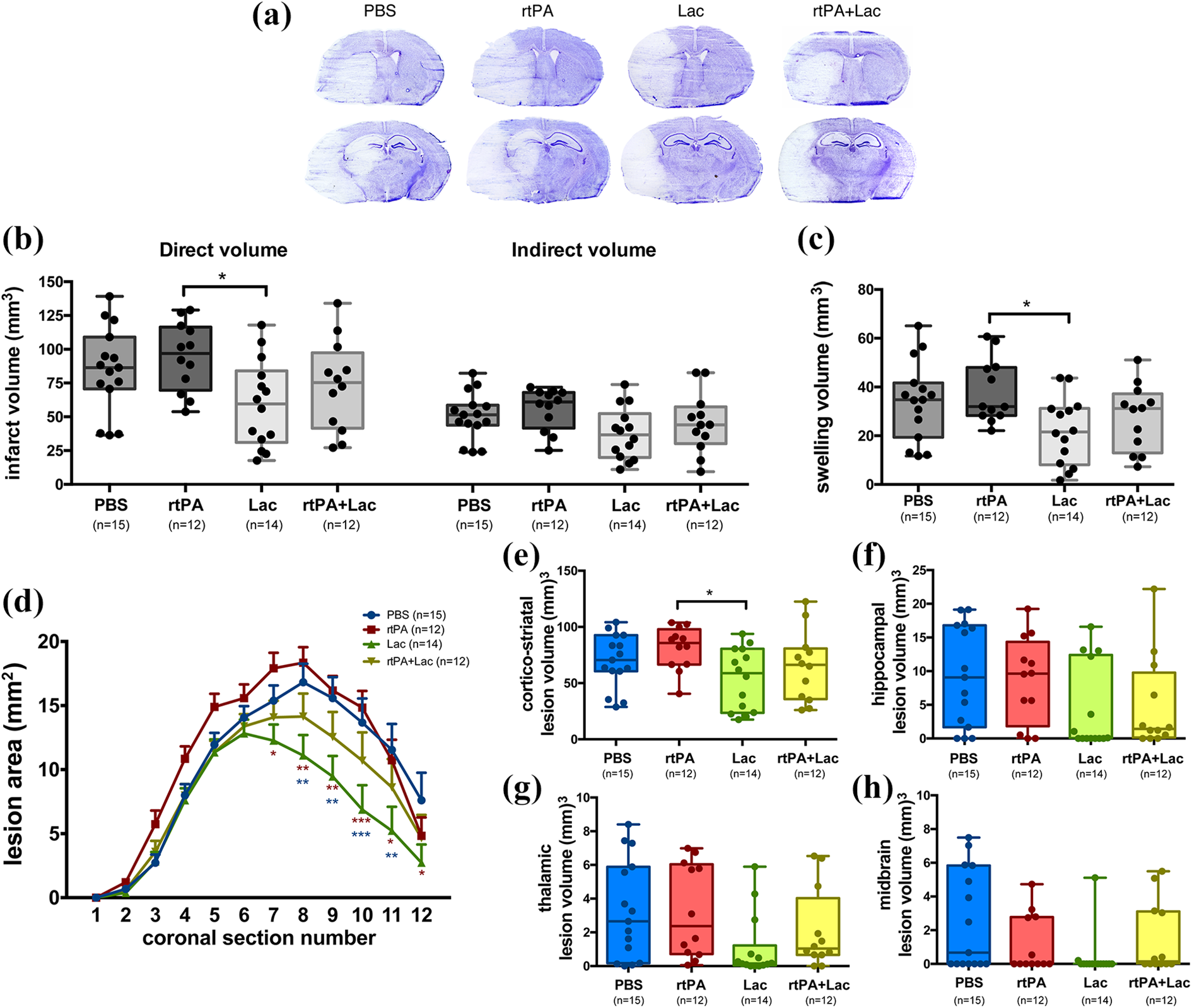
Effect of rtPA and/or lactate after tMCAO. (a) Representative images of cresyl violet–stained mouse brain coronal sections obtained 48 h after 35 min tMCAO and following treatment with PBS (control), rtPA (0.9 mg/kg, iv injection), lactate (2 µL of 100 mmol/L, icv injection), or rtPA+lactate (0.9 mg/kg iv injection + 2 µL of 100 mmol/L icv injection). Two sets of images are provided, to better appreciate the damage in anterior (top) as well as posterior brain structures (bottom). (b) Lesion volume measurements showing the direct lesion volume (left) and the indirect lesion volume (right). One-way ANOVA with Tukey’s post hoc test. (c) Brain swelling volume for each treatment. One-way ANOVA with Tukey’s post hoc test. (d) Infarcted areas on individual coronal sections for the different treatments. Two-way ANOVA with Tukey’s post hoc test. (e–h) Lesion volumes measured in different brain areas: (e) cortex and striatum. One-way ANOVA with Tukey’s post hoc test. (f) Hippocampus. Kruskal–Wallis with Dunn’s post hoc test. (g) Thalamus. Kruskal–Wallis with Dunn’s post hoc test. (h) Midbrain. Kruskal–Wallis with Dunn’s post hoc test. *p < 0.05. rtPA: recombinant tissue plasminogen activator; tMCAO: transient middle cerebral artery occlusion; iv: intravenous; Lac: lactate; icv: intracerebroventricular; ANOVA: analysis of variance.
Twenty-four hours after tMCAO, lactate-treated mice showed better functional outcome, measured by neuroscore, than control or rtPA-treated mice. Mice that received double treatment scored better than rtPA-treated mice and similar to lactate-treated animals (Figure 4(a)). At 48 h, all groups showed an overall improvement, with a significantly better outcome for mice that received lactate alone or in combination with rtPA compared to controls. To evaluate grip strength and balance, we subjected mice to the wire-hanging test (Figure 4(b)). Considering only falling from the wire as the worst outcome and only escaping as best, lactate-treated mice performed significantly better than the rest 24 h after intervention. Mice that received double treatment performed better than controls. At 48 h, performance improved in all groups, with lactate-treated mice showing again the best outcome.
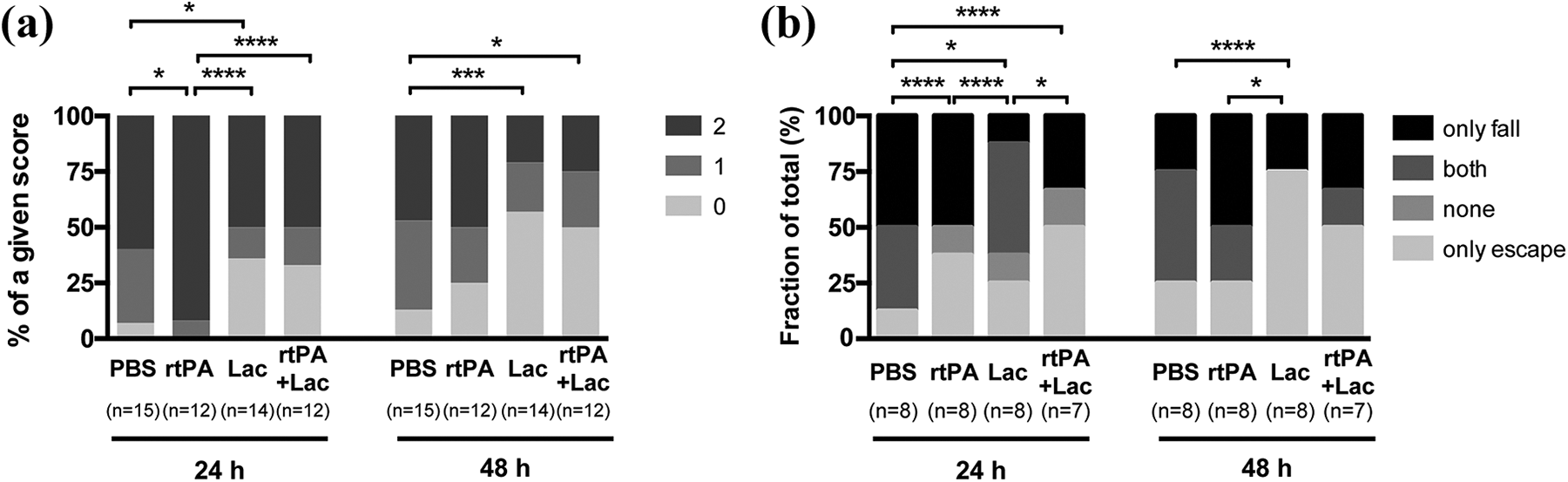
Effect of rtPA and/or lactate on behavior after tMCAO. (a) Neurological deficit scores (0 = no deficit, 1 = failure to extend right forepaw, 2 = circling) at 24 h or 48 h after 35 min of tMCAO and following treatment with PBS, rtPA, lactate, or rtPA+lactate. χ2 test. (b) Wire-hanging test results (only fall, escape and fall, neither escape nor fall, and only escape) at 24 h or 48 h after MCAO. χ2 test. Filled dots represent individual animals. *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001. rtPA: recombinant tissue plasminogen activator; MCAO: middle cerebral artery occlusion; tMCAO: transient middle cerebral artery occlusion; Lac: lactate.
Discussion
The present work brings insight for careful selection of patients who may be eligible for potential neuroprotective lactate treatment in future clinical studies. As expected, and consistently with previous results, 15,16 lactate-treated mice have smaller lesions and a better functional outcome. In the severe pMCAO model, protective effects of lactate are observed when administered shortly after ischemia onset. Lactate effects may relate to its contribution as energy supply, 14 possibly preventing the immediate demise of suffering neurons, its induction of plasticity genes, 32 and/or the functional modulation via lactate receptor interaction, which results in decreased neuronal activity 33 that could alleviate the effects of excess excitotoxic stimulation. It is interesting to note that in the lactate-treated groups, fewer animals had to be euthanized due to repeated seizures than controls. However, the beneficial effects of lactate seem to have a restricted time window as no evident protection was observed when administered 3 h after ischemia onset and in a previous report no protection was observed with very early (15 min) administration. 17 As has been suggested for other potential neuroprotective treatments, properly timed intervention, for example, before they begin to interfere with tissue recovery after stroke, may be key for achieving the full beneficial effects, as interventions that initially contribute to the rescue/protection of as many cells as possible may hamper the endogenous healing response of the brain. 34,35 In line with the possible relevance of the time of intervention, the PLS-DA on brain metabolites presented here clearly demonstrates very different conditions of the brain parenchyma in this relatively short lapse of time, with endogenous lactate, cell death–related metabolites, and reactive oxygen species–sensitive metabolites having a large impact on these differences. Interestingly, the differences correlate with the distinct response to lactate seen at 1 h and 3 h after ischemia onset (Figure 2), suggesting that the metabolic environment at 3 h could be related to the little benefit obtained by lactate administration at this time point. A good example reflecting these differences and related to lesion size is the composite score obtained by adding the concentrations of N-acetyl-aspartate, taurine, and glutamate (NAA+Tau+Glu), which we previously showed at 3 h after transient cerebral ischemia correlates with the later development of a lesion. 36 We went on to show here that the NAA+Tau+Glu score (Supplementary Figure 1) was lower at 3 h than at 1 h after permanent ischemia, reflecting the more advanced tissue demise 25 and more advanced cell death at the later time point. In a future study, it will be interesting to compare the effect of lactate treatment on the metabolic profiles at 1 h and 3 h after ischemia.
At present, the fibrinolytic agent rtPA can be used in stroke patients up to 4.5–9 h from the time of stroke onset 1,3 and can be or cannot be followed by mechanical thrombolysis. Despite its usefulness, rtPA is known to exert some detrimental effects when administered after stroke in human patients as well as in preclinical models of stroke, including MCAO. 23,37 –41 Therefore, it was interesting to test whether lactate would have a positive or a negative impact on these unwanted secondary effects of rtPA. When combined with rtPA, there is only a trend toward lesion size reduction. Hence, it is likely that lactate only partially compensates/counteracts the deleterious effects of rtPA, thus providing insufficient protection in terms of lesion size. Contrarily to permanent ischemia, the unchanged indirect brain lesion size after lactate treatment in the tMCAO model suggests that the decreased lesion size is mainly due to a decreased apparent volume of infarction related to brain swelling. At later stages of stroke, the blood–brain barrier is compromised, causing vasogenic edema, which leads to brain volume expansion with, in severe cases, increased intracranial pressure, herniation, and additional ischemic injuries, 42 effects susceptible to worsening by rtPA. Brain edema and increased intracranial pressure are often associated with occlusion of large intracranial arteries that could contribute to the extended damage observed in control and rtPA-treated mice compared to lactate-treated mice. Interestingly, mice that received the double treatment performed better than control and rtPA-treated mice (Figure 4). Patients with brain edema have a significantly higher degree of neurological deficit at admission and discharge 43 and a worse long-term prognosis. 42,44 Hence, brain swelling reduction in lactate-treated mice could underlie the improvement in neurological outcome. The trend in brain swelling reduction observed after the combined treatment could account for the positive effect on the neurological outcome, milder than with lactate alone, but still present. Notably, our results and the significant effect of lactate treatment on functional outcome 2 h after 60 min tMCAO and 1 week after 30 min tMCAO 15,16 highlight a general positive effect of lactate treatment on stroke. Considering a translation to clinical setting, an impact on functional outcome is what truly matters, as it is the main criterion in stroke patients and this, in our opinion, makes lactate a very promising candidate as therapeutic agent in stroke.
Accurate selection of patients undergoing a particular therapeutic intervention might greatly increase chances of success. Good examples are the extended time windows for rtPA thrombolysis or endovascular thrombectomy and the use of rtPA in wake-up stroke patients selected on the basis of advanced imaging criteria. 45 The results of our translational study bring awareness for the careful selection of patients and suggest that lactate could be tested for neuroprotection in acute ischemic stroke patients not treated with rtPA but with mechanical thrombectomy alone. Alternately, it could be tested in patients without recanalization therapy and persistent arterial occlusion.
Supplemental material
Supplementary_material_(1) - Extended preclinical investigation of lactate for neuroprotection after ischemic stroke
Supplementary_material_(1) for Extended preclinical investigation of lactate for neuroprotection after ischemic stroke by Lara Buscemi, Camille Blochet, Melanie Price, Pierre J Magistretti, Hongxia Lei and Lorenz Hirt in Clinical and Translational Neuroscience
Footnotes
Acknowledgments
The authors acknowledge Leonardo Restivo from the NeuroBAU behavioral platform at the Department of Fundamental Neurosciences (University of Lausanne) for his help with behavioral testing and analysis and also acknowledge the Cellular Imaging Facility (University of Lausanne) for technical support.
Author contributions
LB, MP, PJM, and LH contributed to study conception. LB and CB acquired the data, and LB and MP analyzed the data. HL performed and analyzed the magnetic resonance spectroscopy experiments. LB, MP, HL, and LH drafted the manuscript and figures.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funding from the Biaggi and Juchum Foundations, the Novartis Foundation for Medical Biological Research grant 18C170, and the Swiss Science Foundation grant 31003A_163465/1.
Ethics approval
All animal experiments were approved by the Veterinary Office of Canton de Vaud and were conducted according to Federal and local ethics guidelines. Animal reporting follows the Animal Research: Reporting of in vivo experiments (ARRIVE) guidelines.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
